Abstract
Background:
Fractional exhaled nitric oxide (FeNO) measured at multiple exhalation flow rates can be used as a biomarker to differentiate central and peripheral airway inflammation. However, the role of alveolar nitric oxide (CaNO) indicating peripheral airway inflammation remains unclear in gastroesophageal reflux-associated cough (GERC).
Objectives:
We aimed to characterize the changes in alveolar nitric oxide (CaNO) and determine its clinical implication in GERC.
Design:
This is a single-center prospective observational study.
Methods:
FeNOs at exhalation flow rates of 50 and 200 ml/s were measured in 102 patients with GERC and 134 patients with other causes of chronic cough (non-GERC). CaNO was calculated based on a two-compartment model and the factors associated with CaNO were analyzed. The effect of anti-reflux therapy on CaNO was examined in 26 GERC patients with elevated CaNO.
Results:
CaNO was significantly elevated in GERC compared with that in non-GERC (4.6 ± 4.4 ppb versus 2.8 ± 2.3 ppb, p < 0.001). GERC patients with high CaNO (>5 ppb) had more proximal reflux events (24 ± 15 versus 9 ± 9 episodes, p = 0.001) and a higher level of pepsin (984.8 ± 492.5 versus 634.5 ± 626.4 pg/ml, p = 0.002) in sputum supernatant than those with normal CaNO. More GERC patients with high CaNO required intensified anti-reflux therapy (χ2 = 3.963, p = 0.046), as predicted by a sensitivity of 41.7% and specificity of 83.3%. Cough relief paralleled a significant improvement in CaNO (8.3 ± 3.0 versus 4.8 ± 2.6 ppb, p < 0.001).
Conclusion:
Peripheral airway inflammation can be assessed by CaNO measurement in GERC. High CaNO indicates potential micro-aspiration and may predict a necessity for intensified anti-reflux therapy.
Plain language summary
Keywords
Introduction
Gastroesophageal reflux-associated cough (GERC), a special phenotype of gastroesophageal reflux disease with a predominant cough, is one of the most common causes of chronic cough (CC). 1 Epithelial damage and airway inflammation have been found in patients with GERC.2,3 Micro-aspiration (reflux theory) and the esophago-tracheobronchial reflex mediated by the distal esophageal vagal afferent nerves (reflex theory) are likely to play a key role.1,4 The measurable technique of airway inflammation for GERC patients usually involves cytology of induced sputum, bronchoalveolar lavage fluid (BALF), bronchial mucosal biopsies, and other tests to identify several available inflammatory markers, such as substance P and histamine.2,3,5 However, due to their time-consuming and invasive nature, these methods have been poorly practiced in the clinical setting.
Fractional exhaled nitric oxide (FeNO) detection is a noninvasive tool that is widely used as a biomarker to monitor eosinophilic airway inflammation and guide individualized care for asthma. 6 FeNO50 refers to FeNO measured at the flow rate of 50 ml/s, reflecting NO levels in large airways but not sensitive to inflammatory changes in the small airways.7–9 FeNO200 was measured at the flow of 200 ml/s, indicating the inflammation in peripheral airways. Using a two-compartment model, alveolar nitric oxide (CaNO) can be calculated at multiple expiratory flow rates. 10 CaNO is similar in origin to FeNO200, both being alveolar sources. 7
GERC can be understood as a chronic airway inflammatory disease. However, there is currently no demonstrated evidence regarding the usefulness of FeNO50 for GERC assessment. 11 Elevated eosinophil counts have been reported in BALF but not in induced sputum from GERC patients.2,12 These findings may suggest a peripheral eosinophil-related airway inflammation in GERC. We hypothesized that FeNO levels from distal airspaces (CaNO and FeNO200) may reflect the airway inflammatory status of GERC. However, whether the two parameters are translated into meaningful use in mapping the pathophysiology of GERC remains unknown. Thus, a prospective study was conducted to address this question in GERC by measuring three different models of FeNO (FeNO50, FeNO200, and CaNO) in GERC and non-GERC patients (other causes of CC). Their clinical usefulness in assessing the therapeutic efficacy of anti-reflux therapy in GERC patients was also evaluated in this study.
Methods
Study population
Consecutive patients with GERC and non-GERC who visited our cough clinic between March 2021 and October 2022 were recruited into the study. A GERC diagnosis was considered when the following criteria were met: (I) the patient presented with a cough which had lasted for at least 2 months; (II) multichannel intraluminal impedance and pH monitoring (MII-pH) revealed abnormal gastroesophageal reflux: acid exposure time (AET) >6%, symptom association probability (SAP) ⩾ 95%, and reflux episodes >80 times in 24 h 13 ; and (III) cough responded to a stepwise anti-reflux treatment protocol,14–16 available in Supplemental Table 1. Patients with predominant abnormal acidic and non-acidic reflux were diagnosed with acid GERC and non-acid GERC, respectively. All enrolled patients with suspicious GERC were naïve to anti-reflux treatment. Diagnosis of non-GERC was made based on the positive etiological findings or successful therapeutic trials according to the previously described stepwise diagnostic algorithm. 14 All subjects had no history of respiratory infection or use of anti-inflammatory medications within 8 weeks. Smokers within the last 2 years and GERC combined with other cough etiologies were excluded from this study.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 17
Study design and procedures
This was a single-center observational study. When patients with CC attended specialist cough clinics, general information was collected and cough condition was assessed by cough symptom score, 18 Hull airway reflux questionnaire (HARQ), 19 and Leicester cough questionnaire (LCQ). 20 All subjects underwent routine blood tests, spirometry, histamine bronchial provocation, FeNO measurement, cough challenge test, 15 and sputum induction. MII-pH was performed 15 when suspicious GERC was considered. The patients were followed up every 2 weeks. Therapeutic responses were reported, and the cough symptom score was rated at each follow-up. The cough was considered to be resolved when the cough disappeared completely, or the cough symptom score decreased by ⩾50%. 15
FeNOs were measured using the Nano Coulomb Breath Analyzer (Sunvou-CA2122, Wuxi, China) by ATS/ERS. 21 Patients were required to avoid smoking, strenuous exercise, eating, and drinking for 1 h before testing. After a full inhalation of NO-free air, subjects immediately exhaled against a positive pressure, generating exhalation flow rates (V′E) of 50 and 200 ml/s, recorded as FeNO50 and FeNO200, respectively. For each V′E, the elimination rate of NO (V′NO) was calculated as a function of V′E (V′NO = V′E × FeNO). The V′E was kept constant by a biofeedback visual display on the computer. The flow-independent NO parameters, CaNO, and large-airway NO flux (J′awNO), were calculated by the device software based on a two-compartment model developed by Tsoukias et al. 10 : FeNO = CaNO + J′awNO/V′E. For each patient, a simplified method was used to assess the linear relationship between the two points (50 and 200 ml/s) of NO flux and flow. 8 The slope and intercept of the linear relationship were approximations of CaNO and J′awNO, respectively. Data were stored and analyzed using a computer, utilizing NO analysis software (Sunvou Detector 4.2.8.18, Wuxi, China). Previously established cutoff values for normal FeNO50, FeNO200, and CaNO were less than 25, 10, and 5 ppb (parts per billion), respectively.22,23 FeNO measurements were also repeated in GERC patients whose baseline CaNO was >5 ppb and cough was resolved by anti-reflux therapy if consent was given.
Spirometry was performed according to the guidelines developed by the ATS. 24 Parameters including FEV1, FVC (forced vital capacity), mean forced expiratory flow during the 75%, 50%, and 25% of the FVC (MEF75, MEF50, and MEF25), and maximum mid-expiratory flow (MMEF) were measured at baseline. Small airway dysfunction (SAD) was considered when at least two of MEF75, MEF50, and MMEF were <65% of the predicted value. 25
Induction of sputum was performed according to the protocol previously described. 2 Cell pellets were smeared onto slides and stained with hematoxylin and eosin for cell sorting and counting. The inflammatory cytokines in the sputum supernatant were measured by enzyme-linked immunosorbent assay according to the manufacturer’s instructions (R&D Systems, Minneapolis, MN, USA). The lower limit of detection for bradykinin, calcitonin gene-related peptide (CGRP), IL-4, IL-5, histamine, pepsin, substance P, IL-6, IL-8, TNF-α, MMP-9, and mucin was 16, 7.8, 0.3, 3.9, 0.78, 15, 39, 12.5, 1, 15.6, 31.2 pg/ml, and 2 ng/ml, respectively. The intra- and inter-assay variability over the range of measurable concentrations was 5% and 10%, respectively. These inflammatory mediators were reasonably selected in this study based on that histamine, bradykinin, substance P, and CGRP are tussive mediators and have demonstrated association with cough sensitivity 2 ; IL-4 and IL-5 are involved in eosinophilic inflammation 26 ; IL-6, IL-8, and TNF-α are common inflammatory cytokines from neutrophils or epithelial cells5,26; MMP-9 and mucin are key mediators in the regulation of airway mucus secretion 27 and sputum pepsin is a biomarker of aspiration and may function as an indirect sign of proximal reflux or airway reflux. 28
Statistical analysis
From our preliminary observations, the proportion of high CaNO in GERC and non-GERC was 37% and 17%, respectively. A minimum of 74 patients per group would be required to provide 80% power to detect a 20% difference between the two groups using a 5% two-sided test. Considering a 20% dropout or loss to follow-up rate, 93 patients were finally recruited in each group.
Normally distributed data were expressed as mean ± SD while skewed data were presented as median (interquartile range). The t-test and Mann–Whitney U test were used to compare data between two groups where applicable. Correlations were tested using Spearman’s rank method. The comparisons of categorical variables between groups were assessed using the chi-square test. Receiver operating characteristic (ROC) curve analysis was performed to predict a need for intensified anti-reflux therapy for GERC patients. SPSS 26.0 software (IBM Corp., Armonk, NY, USA) was used for all statistical analyses. Differences were considered significant at p < 0.05.
Results
General characteristics
A total of 102 patients with GERC (41 acid GERC and 61 non-acid GERC) and 134 patients with non-GERC were enrolled in this study. Non-GERC subjects were set as the control group, including cough variant asthma (CVA, n = 50), eosinophilic bronchitis (EB, n = 15), atopic cough (n = 25), post-infectious cough (n = 11), and upper airway cough syndrome (UACS, n = 33). The demographic and clinical characteristics are shown in Table 1. HARQ scores were significantly higher in GERC than that in non-GERC. By contrast, an elevated LCQ score, total serum IgE, and eosinophils in the peripheral blood and induced sputum were observed in non-GERC. The cough duration, cough threshold, cough symptom score, and spirometry parameters were comparable between the two groups.
Baseline characteristics of the study population.
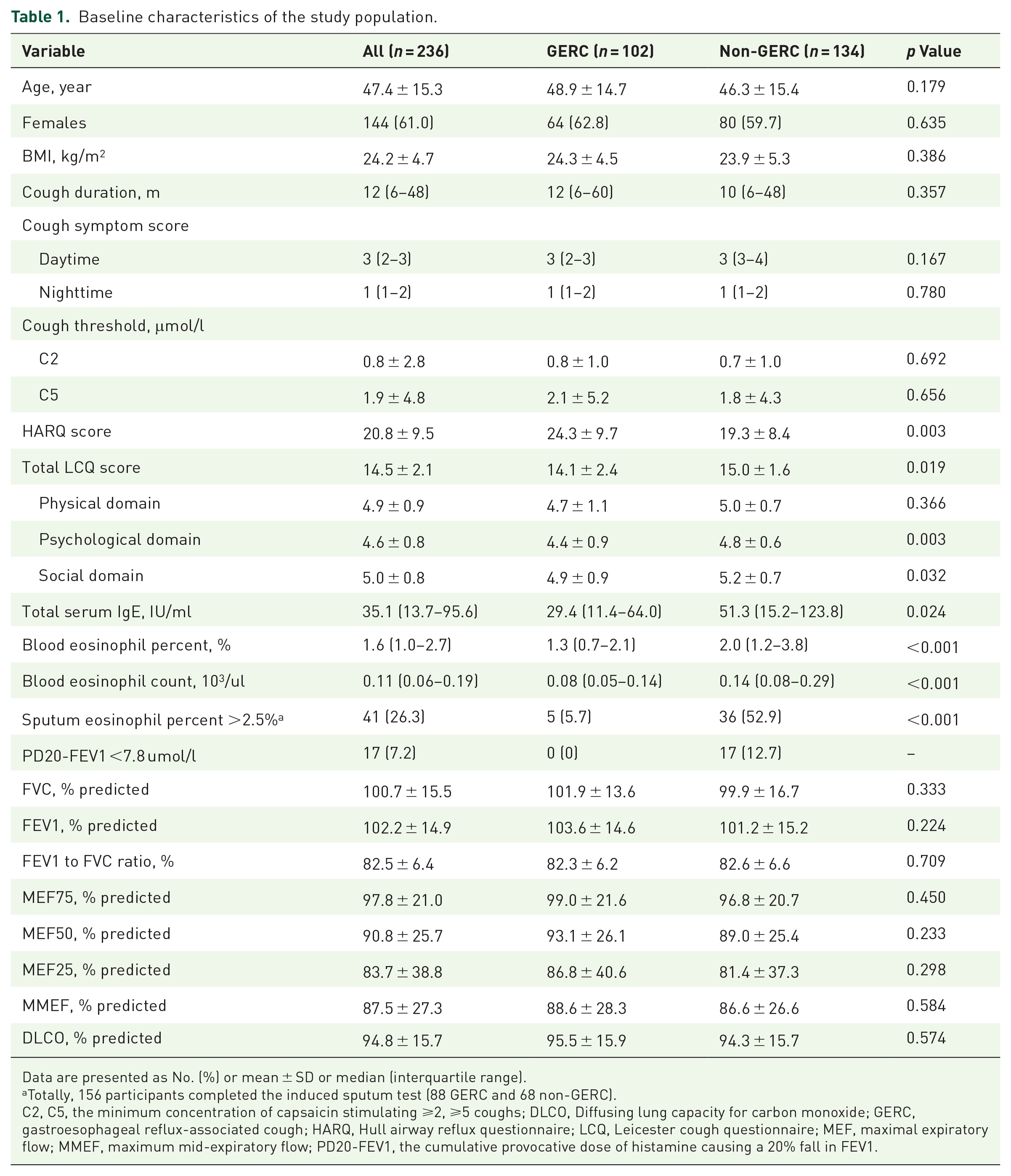
Data are presented as No. (%) or mean ± SD or median (interquartile range).
Totally, 156 participants completed the induced sputum test (88 GERC and 68 non-GERC).
C2, C5, the minimum concentration of capsaicin stimulating ⩾2, ⩾5 coughs; DLCO, Diffusing lung capacity for carbon monoxide; GERC, gastroesophageal reflux-associated cough; HARQ, Hull airway reflux questionnaire; LCQ, Leicester cough questionnaire; MEF, maximal expiratory flow; MMEF, maximum mid-expiratory flow; PD20-FEV1, the cumulative provocative dose of histamine causing a 20% fall in FEV1.
Comparison of FeNOs between GERC and non-GERC
Compared to non-GERC, GERC exhibited markedly higher CaNO (4.6 ± 4.4 versus 2.8 ± 2.3 ppb, p < 0.001) but lower FeNO50 (16.4 ± 9.3 versus 24.5 ± 27.1 ppb, p = 0.002), while FeNO200 was comparable between the two groups [8.2 ± 4.6 versus 9.4 ± 8.1 ppb, p > 0.05; Figure 1(a)–(c)]. Weak correlations were observed between CaNO and FeNO200 (r = 0.358, P < 0.001) [Figure 1(d)]. The contribution of CaNO/FeNO50 (55.0 ± 26.9% versus 41.5% ± 25.1%, p < 0.001) and CaNO/FeNO200 (35.2% ± 29.6% versus 24.2% ± 26.2%, p = 0.003) was much higher in GERC than that in non-GERC [Figure 1(e) and (f)].

Comparison of FeNOs between GERC and non-GERC. (a) FeNO50, (b) FeNO200, (c) CaNO, (d) correlation analysis between FeNO200 and CaNO in all subjects. (e and f) The contribution of CaNO/FeNO50 (e) and CaNO/FeNO200 (f).
With CaNO > 5.0 ppb as the abnormal cutoff point, high CaNO was more common in GERC patients than in non-GERC patients (37.3% versus 17.1%, χ2 = 12.197, p < 0.001).
Difference in cough characteristics in GERC
More GERC patients with high CaNO presented a productive (p < 0.001) and nocturnal cough (p = 0.004), with accompanying throat clearing (p = 0.041), itchy throat (p = 0.004), and pharyngeal foreign body sensation (p = 0.002). In addition, higher HARQ (p = 0.015) and low LCQ (p = 0.004) were rated in the high CaNO cohort (Table 2).
Clinical characteristics in GERC with high versus normal CaNO.

Data are presented as No. (%) or mean ± SD or median (interquartile range).
CaNO, alveolar nitric oxide; C2, C5, the minimum concentration of capsaicin stimulating ⩾2, ⩾5 coughs; GERC, gastroesophageal reflux-associated cough; HARQ, Hull airway reflux questionnaire.
Findings of MII-pH in GERC
GERC patients with high CaNO demonstrated markedly higher non-acid SAP (p = 0.032), more frequent weakly acidic reflux (p = 0.024), proximal reflux events (p = 0.001), proximal acidic reflux (p = 0.027), proximal weakly acidic reflux (p = 0.044), and decreased upper esophageal sphincter pressure (p = 0.048), while those with normal CaNO presented higher AET (p = 0.048), DeMeester score (p = 0.047), and more acidic reflux events (p = 0.036; Table 3). A high CaNO was more frequently examined in non-acid GERC than in acid GERC (50.8% versus 17.1%; χ2 = 5.826, p = 0.017).
Results of MII-pH in GERC with high versus normal CaNO.

Data are presented as mean ± SD or median (interquartile range).
CaNO, alveolar nitric oxide; DCI, distal contractile integral; GERC, gastroesophageal reflux-associated cough; IRP4s, integrated relaxation pressure at 4 s; LESL, lower esophageal sphincter length; LESP, lower esophageal sphincter pressure; MII-pH, multichannel intraluminal impedance and pH monitoring; SAP, Symptom association probability; UESL, upper esophageal sphincter length; UESP, upper esophageal sphincter pressure.
Relationship between CaNO and inflammatory or physiological variables
GERC patients with high CaNO demonstrated comparable differential cell counts in sputum with normal CaNO group (all p > 0.05) but increased pepsin (p = 0.002), IL-8 (p = 0.045), MMP-9 (p = 0.016), and mucin (p = 0.013; Table 4). A marked linear correlation was found between pepsin and CaNO (r = 0.272, p = 0.010) or proximal reflux events (r = 0.485, p < 0.001) in GERC patients [Figure 2(a) and (b)]. No significant relation was found between CaNO and blood eosinophil count (r = −0.037, p = 0.625) while both FeNO50 (r = 0.543, p < 0.001) and FeNO200 (r = 0.519, p < 0.001) were moderately positively correlated with blood eosinophil count in all participants [Figure 2(c)–(e)].
Inflammatory characteristics in the sputum supernatant from patients with GERC.
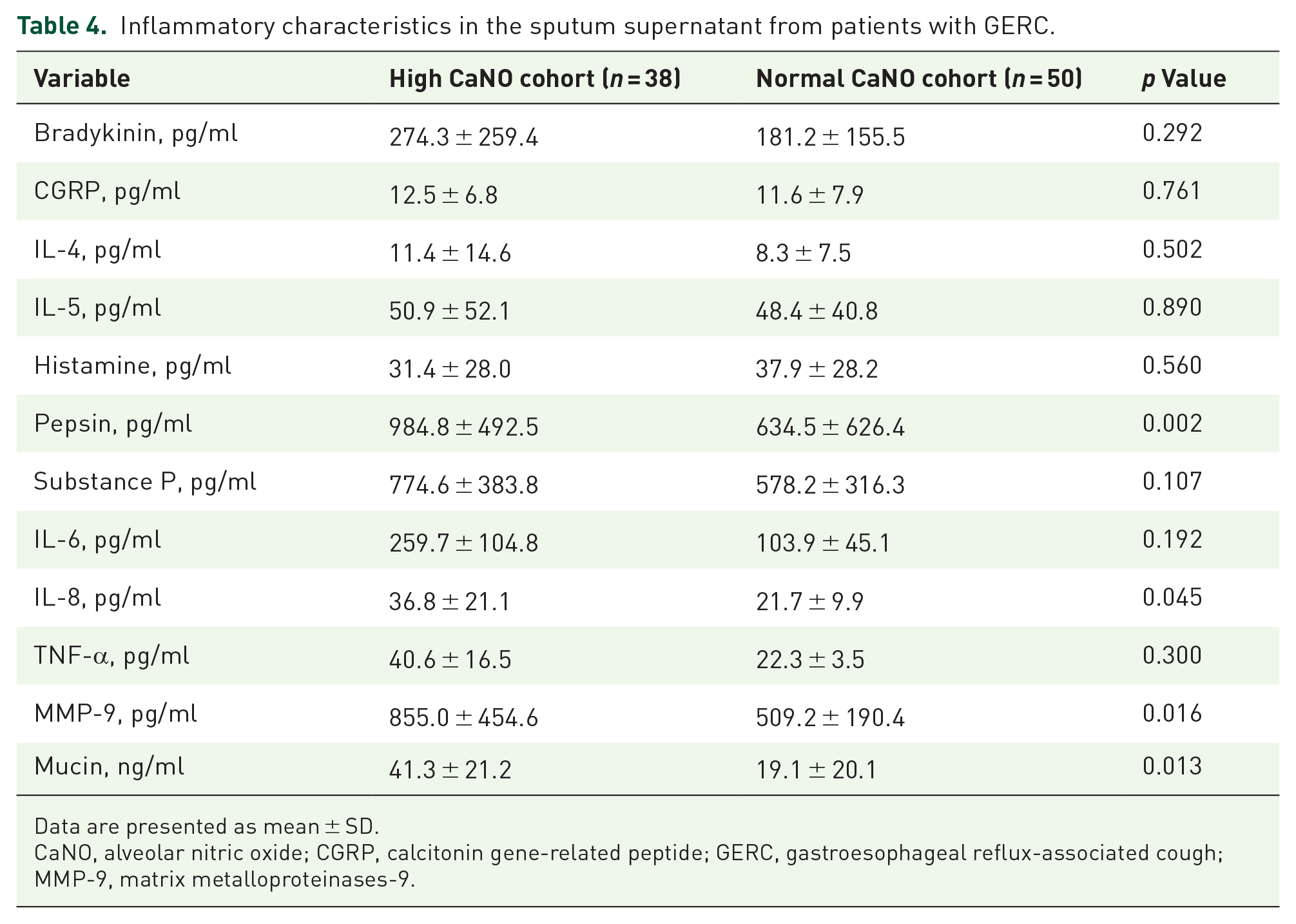
Data are presented as mean ± SD.
CaNO, alveolar nitric oxide; CGRP, calcitonin gene-related peptide; GERC, gastroesophageal reflux-associated cough; MMP-9, matrix metalloproteinases-9.

Correlation analysis between FeNOs and inflammation. (a) Pepsin and CaNO; (b) pepsin and proximal reflux events; (c–e) blood eosinophil count (103/ul) and FeNO50 (c) or FeNO200 (d) or CaNO (e).
SAD was found in 17.8% (42/236) of patients. CaNO was significantly higher in GERC patients with SAD than those without SAD (p = 0.002), whereas no difference in CaNO was found in non-GERC with and without SAD (p > 0.05; Figure 3). In addition, DLCO decreased in 12.7% (13/102) of GERC patients but was not correlated with CaNO levels (r = −0.148, p = 0.145). Although no significant difference between high and normal CaNO cohorts was observed, GERC patients with higher CaNO tended to have lower DLCO (92.8 ± 13.4 versus 96.0 ± 17.4, t = −0.897, p = 0.372).

CaNO comparison in patients with or without SAD. (a) GERC patients; (b) non-GERC patients.
Response to anti-reflux therapy in GERC patients
In all, 18 patients responded well to the standard anti-reflux treatment and 84 patients benefited from the subsequent intensified anti-reflux therapy, including an increase in omeprazole dosage (n = 10) and add-on neuromodulators with baclofen (n = 4), gabapentin (n = 50), and flupentixol/melitracen (n = 20). More GERC patients with high CaNO had experienced further intensified anti-reflux treatments than those with normal CaNO (92.1% versus 76.6%; χ2 = 3.963, p = 0.046) because of the poor response to the initial treatment. High CaNO demonstrated an appropriate ability to predict the need for intensified anti-reflux therapy, with a sensitivity of 41.7%, specificity of 83.3%, positive predictive value of 92.1%, and negative predictive value of 23.4%, respectively [Figure 4(a)].
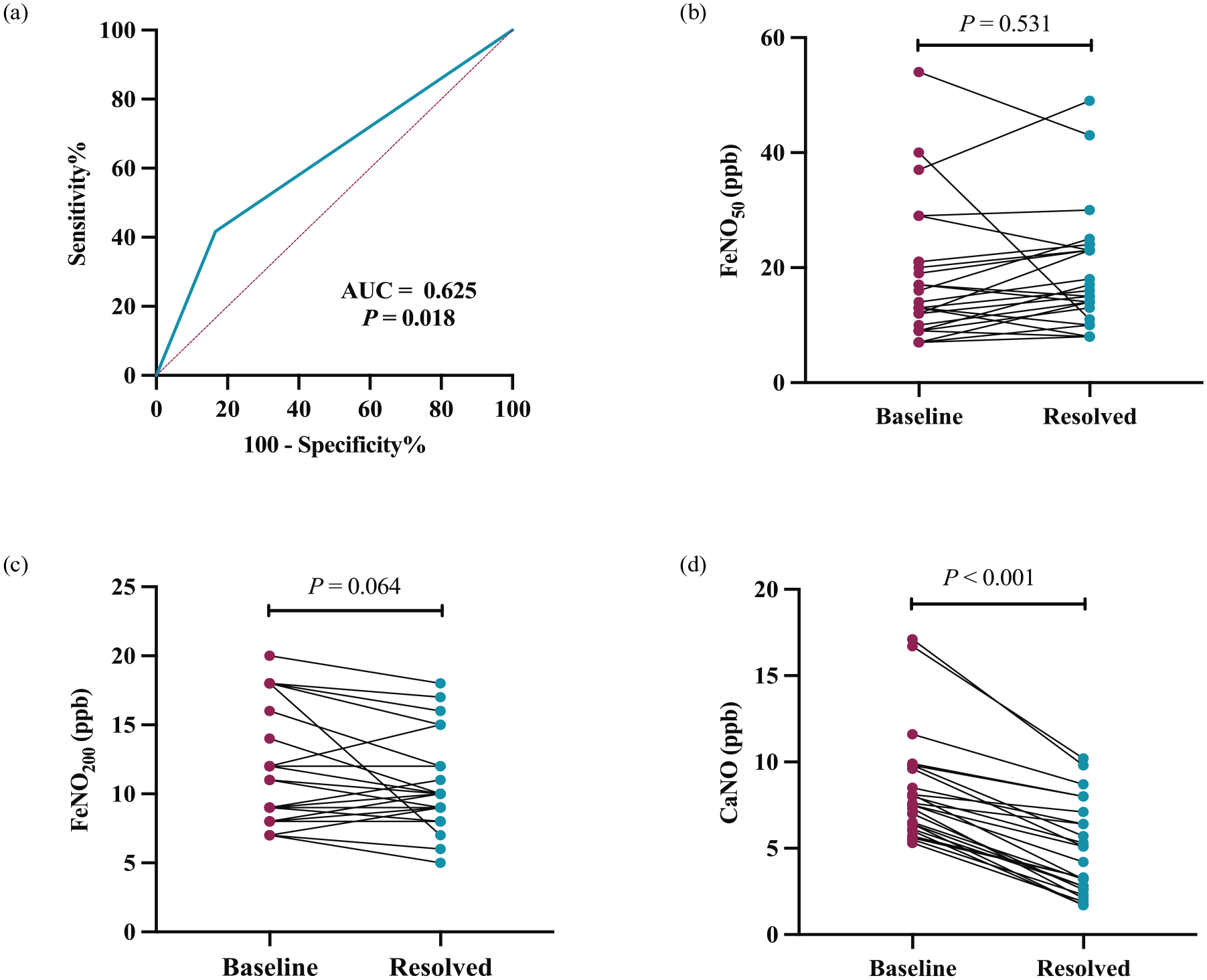
FeNOs in response to anti-reflux therapy in GERC patients. (a) ROC curve analysis of high CaNO for predicting the need for intensified anti-reflux therapy; (b–d) changes in FeNOs in GERC patients after therapy; (b) FeNO50; (c) FeNO200; and (d) CaNO.
In a subgroup of 26 GERC patients who had high CaNO and responded well to anti-reflux therapy, CaNO was significantly decreased from 8.3 ± 3.0 to 4.8 ± 2.6 ppb (p < 0.001). FeNO200 decreased in 15 out of 26 patients (11.5 ± 4.1–10.5 ± 3.3 ppb) but did not achieve statistical significance (p = 0.064). No distinct change was found in FeNO50 of post-treatment [19.3 ± 10.0 to 18.3 ± 11.4 ppb, p = 0.531; Figure 4(b)–(d)].
Discussion
In this study, we defined peripheral airway inflammation in patients with GERC using noninvasive CaNO detection. This is the first study to report the elevated CaNO in patients with GERC. A high CaNO (>5 ppb) was demonstrated to be positively associated with potential micro-aspiration, as indicated by higher pepsin levels in induced sputum. GERC patients with high CaNO were more likely to develop SAD. CaNO may predict the need to employ intensified anti-reflux therapy with high specificity and positive predictive value in GERC, given that more GERC patients with high CaNO required intensified anti-reflux therapy and the reversal of elevated CaNO in response to treatment.
The multiple exhalation flow rate method has greatly improved the measurement of FeNO since it allows the separate assessment of inflammatory activity in alveolar and bronchial sources. 9 In the study, increased CaNO was observed in GERC patients, while FeNO50 was normal, supporting the hypothesis that peripheral airway inflammation may be involved in the pathophysiology of GERC. FeNO50 has been reported to be normal in most GERC patients. 2 There is limited research looking at CaNO levels in CC, and this research reported somewhat conflicting results. No significant difference was observed among CVA, EB, UACS, and GERC over a small sample size of GERC patients. 11 One possible reason for this could be methodological issues, such as the use of higher flow rates.7,8
Gastroesophageal reflux may induce airway inflammation by either reflux or reflex mechanism. 4 In this study, high CaNO in GERC was related to proximal reflux episodes and pepsin levels in induced sputum. Reflux-related throat symptoms and a high HARQ score were also more common in these patients. As shown by the MII-pH, more non-acid reflux events and lower upper esophageal sphincter pressure can explain the more proximal reflux in patients with high CaNO. These findings indicated the importance of micro-aspirations or airway reflux in the production of CaNO. Evidence suggests that repeated micro-aspirations produce chronic damage to the terminal bronchioles and alveoli, thus progressively leading to widespread fibrosis. 28 The damage tends to coalesce locally in response to gravity during aspiration. When the gaseous non-acid mist is excessive or the airway is sensitized, airway reflux could lead to lower airway inflammation and fibrosis of the terminal airways. 29 The presence of lipid-laden alveolar macrophages in patients with gastroesophageal reflux 30 further confirmed the hypothesis that aspiration may readily reach the peripheral airways resulting in airway inflammation, though the disagreement is not absent. 31 In addition, reduced airway pH along with micro-aspiration may also be responsible for airway epithelial dysfunction, decreased ciliary motility, abnormal airway mucous secretion, and NO generation. 32
FeNO is mainly produced by airway epithelial cells, which control the intracellular upregulation of inducible NO synthase in response to various types of inflammation. Our results revealed that inflammatory changes are accompanied by micro-aspirations, as reflected by increased levels of IL-8, MMP-9, and mucin in induced sputum from GERC patients in the high CaNO cohort. It has been shown previously that pepsin can increase the levels of mucin via MMP-9 pathways in the lower respiratory epithelium under non-acidic conditions, promoting the hypersecretion of airway mucus and causing airway inflammation. 27 A higher number of GERC patients with elevated CaNO presenting with a productive cough further reinforced the observation of airway mucus hypersecretion. An increase in IL-8 suggests a neutrophilic activation in patients with high CaNO, which is in line with enhanced neutrophilic inflammation in GERC patients due to micro-aspiration. 33 In addition, pepsin may induce IL-8 release from bronchial epithelial cells, given its cytotoxic nature. 34 It is worth noting that lung injury is independent of acidity, and factors other than acid (i.e. gaseous mist and bile acids) may be involved in its pathogenesis. 35 Bile acid aspiration has been reported to be associated with increased neutrophils and IL-8 in BALF. 36 Moreover, airway acidification can attract and activate neutrophils and promote NO production. 37 Although there was no evidence of increased neutrophils in induced sputum from the high CaNO cohort, high levels of inflammatory mediators indicated these cells were activated. As a result, increased CaNO could be attributed to pepsin-induced alveolar epithelial injury secondary to micro-aspiration, which possibly involves the MMP-9/mucin pathway or IL-8-mediated neutrophil inflammation.
In asthma, FeNO is undoubtedly a biomarker of type 2 airway inflammation. GERC was found to be a risk factor for uncontrolled asthma, whereas GER (Gastroesophageal reflux) itself did not aggravate preexisting airway inflammation in asthmatics, as measured by induced sputum cell differentials and FeNO50.33,38 GER alone presents a neutrophilic inflammatory pattern, as determined by elevated inflammatory mediators (i.e. IL-6) in sputum. 33 In this study, type 1 inflammation rather than type 2 airway inflammation was evidenced in GERC patients with high CaNO. However, eosinophilia in BALF has been previously reported in GERC. 12 One can also not exclude the possibility that the profiles of inflammatory cells and mediators shown in induced sputum might not accurately reflect the nature of true type 2 airway inflammation in the peripheral airways, as CaNO did. Persistent inflammation in peripheral airway/alveolar sites may induce airway mucosal congestion, bronchoconstriction, and structural remodeling, leading to increased small airway resistance and further progression to SAD. 39 The involvement of proinflammatory factors (i.e. IL-8 and IL-6) may warn against the risk of SAD progressing to airway obstruction. 39 The thickening of the bronchial wall implies the presence of airway inflammation and remodeling. CaNO levels which reflect the peripheral airway inflammation have been reported to be positively correlated with a gradual increase in airway wall area toward the distal bronchi. Peripheral airway wall thickness may interfere with gaseous diffusion and contribute to the relationship between the airway wall area and CaNO levels. 40 In this study, there was a trend toward a decrease in DLCO in GERC patients with high CaNO, indicating the possibility of reduced diffusion capacity and small airway thickening. Furthermore, SAD is also accompanied by high CaNO, implying more severe airway inflammation in some patients with GERC. Since only 16.7% of GERC patients had SAD and 12.7% had reduced DLCO, airway inflammation may be mild in most patients with GERC. In addition, CC is characterized by a global hypersensitivity of the afferent vagus and is associated with neurogenic inflammation. Increased epithelial sensory nerve density was reported in chronic cough airways compared with healthy airways before. 41 In the present study, we did compare the levels of some biomarkers in sputum that may indicate cough hypersensitivity and neurogenic inflammation (i.e. substance P, Bradykinin, CGRP) between high and normal CaNO cohorts but did not find any significant differences. Based on the available evidence, we believe that the airway sensory nerve plasticity may not be a crucial contributor involved in the alveolar inflammation in GERC patients with higher CaNO.
The findings in the study have the potential to be translated into meaningful clinical use. Although proton pump inhibitors decrease the acidity of refluxates, they cannot inhibit reflux. 15 Acid suppression therapy works poorly in patients with airway reflux or laryngopharyngeal reflux.29,42 GERC patients with high CaNO may have a greater need for intensified anti-reflux therapy due to the more proximal reflux and non-acid reflux. The decreased CaNO combined with cough relief also suggests the potential for CaNO to be used in predicting the response to anti-reflux treatment and being used as a treatment outcome indicator in real-world practice. Since relapse may occur upon withdrawal of treatment or reduction of dosage, 16 regular CaNO follow-ups may better guide the course of therapy. Currently, several MII-pH parameters such as SAP, AET, and mean nocturnal baseline impedance are used to predict GERC. 13 However, they have not been widely adopted in clinical settings due to the invasive nature of MII-pH. Therefore, CaNO may be a promising indicator to monitor peripheral airway inflammation and evaluate the therapeutic efficacy in patients with GERC.
FeNO200 is also considered to be a marker of peripheral airway inflammation and is strongly correlated with CaNO. 7 It does not appear to be vital for the assessment of GERC. This may stem from the fact that FeNO200 is related to eosinophilic inflammation. 22 In this study, FeNO50 and FeNO200 (but not CaNO) showed a significant correlation with blood eosinophil count. Although a moderate association was observed between FeNO200 and CaNO, FeNO200 partially reflects CaNO, rather than being fully reciprocal. Alternatively, a single exhalation at a constant expiratory flow rate is inherently nonspecific due to varying percentages of the J′awNO component included in FeNO50 and FeNO200, while CaNO helps identify the source of endogenous NO, offering a corrective measure for any axial NO back-diffusion. 43 Delclaux et al. reported that CaNO represented about 30% of FeNO50 and 60% of FeNO200 in healthy subjects. 7 However, the contribution of CaNO/FeNO50 and CaNO/FeNO200 in GERC was much higher than that in non-GERC, indicating that the J′awNO component is minimal in the contribution of FeNO50 and FeNO200 in GERC. This contributes to high CaNO occurrence even when FeNO200 is within the normal range and explains the non-parallel variation between CaNO and FeNO200 in detecting peripheral airway inflammation, as central airway inflammation might be mild in GERC patients. 3 Since lower levels of FeNO have been observed in the neutrophilic asthma phenotype than those in the eosinophilic phenotype, 44 the absence of elevated FeNO50 and FeNO200 in GERC does not preclude the presence of inflammation, but rather suggests that the pulmonary NO synthase pathway might not be involved. The above demonstrates that CaNO has the potential to assess peripheral NO concentrations more effectively and accurately in GERC.
In this study, the CaNO value was calculated at the single flow rate of 50 and 200 ml/s. Based on the conventional linear regression model, the estimation of CaNO requires at least three flow rates ranging from the lowest (100 ml/s) to the highest flow rates (350 or 400 ml/s). 45 However, it is difficult to perform these measurements in all patients in a clinical setting, especially at the highest rates. 22 To improve the success rate of measurement, a simplified CaNO estimation was developed 22 and recommended by the Chinese Expert Consensus. 46 This simplified CaNO estimation method has been widely used in previous studies to detect small airway inflammation and make diagnoses.22,39,47 Therefore, the validity of our results is unlikely to be affected.
Several limitations should be addressed in this study. First, potential mechanisms of NO production induced by aspiration were only evidenced by increased sputum mediators. Markers in induced sputum mainly provide information about inflammation in the large airways even though they include the components of bronchoalveolar space. 48 This study also indicated the association between CaNO and inflammatory variables in induced sputum. Second, sequential measurements of CaNO were not performed at every follow-up for all GERC subjects. It may influence the accuracy and generalizability of the research conclusion. Further validation work is needed. Third, the MII-pH test was not conducted in the non-GERC group. It had little effect on the reliability of the results because the non-GERC group was definitely diagnosed based on specific etiology treatments and a lower HARQ score indicated a lower likelihood of GER. 49 Finally, a normal CaNO value was adopted in this study based on the reported data of healthy people with no unified standard in the international guidelines. Thus, it should be evaluated extensively in the future.
Conclusion
In conclusion, this study reveals that the pathogenesis of GERC may involve small airway inflammation independent of the large airways. CaNO may have the potential to be used as a non-invasive biomarker for detecting peripheral airway inflammation in GERC. Increased CaNO may be associated with potential micro-aspiration and SAD and may indicate the need for intensified anti-reflux therapy in GERC.
Supplemental Material
sj-docx-1-tar-10.1177_17534666241231117 – Supplemental material for Role of alveolar nitric oxide in gastroesophageal reflux-associated cough: prospective observational study
Supplemental material, sj-docx-1-tar-10.1177_17534666241231117 for Role of alveolar nitric oxide in gastroesophageal reflux-associated cough: prospective observational study by Li Zhang, Mengru Zhang, Alimire Aierken, Ran Dong, Qiang Chen and Zhongmin Qiu in Therapeutic Advances in Respiratory Disease
Supplemental Material
sj-docx-2-tar-10.1177_17534666241231117 – Supplemental material for Role of alveolar nitric oxide in gastroesophageal reflux-associated cough: prospective observational study
Supplemental material, sj-docx-2-tar-10.1177_17534666241231117 for Role of alveolar nitric oxide in gastroesophageal reflux-associated cough: prospective observational study by Li Zhang, Mengru Zhang, Alimire Aierken, Ran Dong, Qiang Chen and Zhongmin Qiu in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
