Abstract
Background:
Liver injury is the hallmark adverse reaction of endothelin receptor antagonist (ERA). Since the first drug, bosentan has been widely used in clinical practice, hepatotoxicity has been accompanied by the history of ERA. The new ERA has been proven to have a lower liver risk but the current research findings are inconsistent. ERA-based targeted drug combinations are commonly used in the treatment of pulmonary arterial hypertension, where the risk of liver injury is difficult to estimate.
Objectives:
This study aimed to compare the correlation between ERA and different ERA combination regimens with liver injury in the real world.
Design:
This is a retrospective study using data from the Adverse Event Reporting System (Food and Drug Administration AERS, FAERS).
Methods:
The study used proportional imbalance and Bayesian analysis to mine FAERS data from January 2004 to December 2022 to determine the association of three ERAs with liver injury and to further mine the risk of liver injury due to the combination of ERAs with other targeted drugs. In addition, we analyzed the onset time, mortality, and hospitalization rate of liver injury caused by different ERA combination regimens.
Results:
We screened out 3581 ERA-related liver injury events, of which bosentan (59.82%) had the largest number of cases. The patients with liver injury were mainly female (60.63%), and the age was concentrated between 61 and 75 years (26.75%). According to different signal mining methods, reporting odds ratio (ROR; 3.38, 95% confidence interval = 3.23–3.53), proportional reporting ratio (PRR; 3.22, χ2 = 37.84), Bayesian confidence propagation neural network (BCPNN; 1.68, 95% confidence interval = 1.61), multi-item gamma Poisson shrinker (MGPS; 3.21, 95% confidence interval = 3.09), bosentan had the strongest association with liver injury compared to ambrisentan and macitentan. Furthermore, bosentan + sildenafil [ROR (2.52, 95% confidence interval = 2.23–2.84), PRR (2.44, χ2 = 15.92), BCPNN (1.29, 95% confidence interval = 1.14), MGPS (2.44, 95% confidence interval = 2.21)], bosentan + epoprostenol [ROR (5.39, 95% confidence interval = 4.29–6.77), PRR (4.94, χ2 = 65.18), BCPNN (2.30, 95% confidence interval = 1.83), MGPS (4.94, 95% confidence interval = 4.08)], bosentan + iloprost [ROR (2.70, 95% confidence interval = 2.11–3.45), PRR (2.61, χ2 = 31.03), BCPNN (1.38, 95% confidence interval = 1.08), MGPS (2.61, 95% confidence interval = 2.12)] had a higher risk of liver injury caused by the three ERA combination regimens. The median time to onset of hepatotoxicity associated with all ERA combination regimens was 259 days (interquartile range: 58–716.5 days). Finally, the hospitalization rate for patients experiencing hepatotoxicity with ERA combination regimens was 47.86% and the mortality rate was 12.67%.
Conclusion:
By mining the FAERS, we analyzed and compared the risk of liver injury related to different ERA and ERA combination regimens, and the onset time and adverse reaction outcomes of all ERA combination regimens. According to the results of the study, bosentan had the highest risk of liver injury and the combination regimens bosentan + sildenafil, bosentan + epoprostenol, and bosentan + iloprost had a stronger risk of liver injury. From the early stages of treatment, we need to regularly monitor the liver function of patients, especially for females and the elderly, and discontinue the suspected drug as soon as the liver injury occurs.
Keywords
Introduction
Pulmonary hypertension (PH) is a fatal, chronic, progressive disease with a complex pathogenesis, which can be caused by cardiopulmonary disease or other systemic diseases. Depending on the underlying cause, PH can be classified as pulmonary arterial hypertension (PAH), PH caused by left heart disease, PH caused by lung diseases, hypoxia, or both, chronic thromboembolic PH, and PH caused by pulmonary artery obstructions, and PH caused by unclear or multifactorial mechanisms, 1 with PAH considered to be one of the most aggressive types of PH. 2 Accurate diagnosis is often a prerequisite for the development of rationalized therapeutic management. Accurate diagnosis is often a prerequisite for the development of rationalized therapeutic management. PAH is a pan-vascular lesion involving the pulmonary vascular system, characterized by elevated resistance in the precapillary vascular tree, 3 which, in turn, leads to increased pulmonary artery pressure and subsequent development of right heart failure or even death. Before the availability of specific therapeutic agents, the natural prognosis for PAH was poor, with a median survival of only 2.8 years and survival rates of 68%, 48%, and 34% at 1, 3, and 5 years, respectively. 4 And with the advent of the era of targeted drugs, the prognosis for PAH has improved significantly. To date, a total of 14 drugs, including those targeting the nitric oxide, endothelin, and prostaglandin pathways for the treatment of PAH, have been approved for clinical use by the Food and Drug Administration (FDA). 5 Endothelin plays an important role in the pathogenesis of PAH. Endothelin-1 is involved in the development of PAH by binding to endothelin receptors A and B in pulmonary vascular smooth muscle cells, causing vasoconstriction and promoting mitosis. Several pieces of evidence have shown that endothelin receptor antagonist (ERA) is not only effective in increasing the 6-min walking distance and improving exercise tolerance in patients with PAH but also significantly reduces the incidence of clinical deterioration events and improves hemodynamic parameters and time to clinical deterioration in patients.6–9 ERAs currently approved for marketing by the FDA include the dual endothelin receptor A and B antagonists, bosentan and macitentan, and the highly selective endothelin A (ETA) receptor antagonist, ambrisentan.
With the widespread use of ERA in the clinic, patients continue to express concerns about the adverse effects of this class of drugs, despite the superior efficacy demonstrated. Since 2005, when the first ERA, bosentan, was introduced into clinical, adverse events such as abnormal liver function, gastrointestinal reactions, peripheral edema, and anemia have always accompanied the history of ERA administration. 10 Hepatotoxicity is a rare but potentially life-threatening adverse reaction, and drug-induced liver injury (DILI) is an important cause of cases of acute liver failure in Western countries. 11 Long-term results from the EARLY study of bosentan showed that the most common cause of discontinuation due to adverse events was abnormal liver function, with 29 (16.8%) patients experiencing alanine aminotransferase (ALT) and/or aspartate aminotransferase (AST) > 3 × upper limits of normal (ULN) over the 5 years of the study. Although clinical trials with ambrisentan have confirmed that patients were not found to have transaminase elevations up to three times the normal upper line during dosing, 12 this does not prove that ERA is safe. Sitaxentan is an ETA receptor antagonist. Initially, sitaxentan was considered to have a low correlation with hepatotoxicity and was used as an alternative to bosentan in the treatment of patients with hepatic dysfunction. However, the potential of sitaxentan to cause severe hepatotoxicity proved to be underestimated. In December 2010, sitaxentan was urgently withdrawn from the global market due to lethal hepatotoxicity.13,14 At present, the safety of the other three ERAs for PAH is considered to be relatively reliable.
In general, the initial treatment of PAH is a combination therapy, 15 and ERA, one of the three cornerstones of PAH-specific therapy, is often used in combination with other drugs. In the event of an adverse event of hepatotoxicity, we tend to associate it directly with ERA and underestimate the risk of co-administration. The combination of ERA and other PAH-targeted drugs may increase the risk of liver injury. 16 Pharmacovigilance information on ERA is mostly derived from clinical trials or case reports, and only a few studies have explored its safety in the real world.17,18 No studies have systematically evaluated the clinical characteristics and risk of liver injury due to ERA, and the risk of liver injury from ERA in combination with other drugs is unknown. More pharmacovigilance studies are needed to evaluate the inconsistency in the risk of liver injury for the three drugs about their adverse effects. Therefore, we will explore the correlation between ERA and liver injury by tapping into the drug safety database [FDA Adverse Event Reporting System (FAERS) was chosen for this study] and using real-world information on adverse drug events, and then compare the risk of occurrence of ERA in combination with different targeted drugs and analyze the clinical characteristics of adverse reactions to provide a basis for rational clinical use of drugs.
Materials and methods
Data source
FAERS (https://www.fda.gov/drugs/surveillance/questions-and-answers-fdas-adverse-event-reporting-system-faers) is an open database containing a large number of adverse event reports submitted to the FDA. The database is designed to support the FDA’s post-marketing safety surveillance program for drug and therapeutic biologic products. Consumers, pharmaceutical manufacturing enterprises, and professional medical personnel can spontaneously report adverse event information. FAERS makes safety reports of cases freely available to the public in a variety of ways to monitor the safety of products after FDA approval. The report information of FAERS consists of seven files, namely DEMO (patient demographic and administrative information), DRUG (drug/biologic information), REAC (all terms coded for the event), OUTC (patient outcomes), RPSR (report sources), THER (drug therapy start dates and end dates), and INDI (all terms coded for the indications). The study retrospectively analyzed all adverse events from January 2004 to December 2022, yielding a total of 20,061,664 reports. After removing duplicate reported information (the most recent FDA_DT was selected when the CASE_ID was the same as the FDA_DT, as recommended by the FDA), the final number of actual reports was 17,042,127. The screening is shown in Figure 1.

The process of screening endothelin receptor antagonist-induced liver injury cases from FAERS.
Data cleaning
The study used the Preferred Term in the Medical Dictionary for Regulatory Activities (MedDRA 26.0) as the name of adverse reactions and searched the database. The keywords related to the liver injury are as follows: acute hepatic failure, ALT abnormal, ALT increased, AST abnormal, AST increased, blood bilirubin unconjugated increased, hepatic enzyme abnormal, hepatic enzyme increased, hepatic failure, hepatic function abnormal, hepatic necrosis, hepatitis, hepatitis acute, hepatitis fulminant, hepatitis toxic, hepatocellular injury, hepatotoxicity, jaundice hepatocellular, liver function test abnormal, liver injury, mixed liver injury, subacute hepatic failure, transaminases abnormal, transaminases increased. As for the name of the medicine and the trade name (Table 1), we checked MICROMEDEX (Index Nominum) for confirmation.
Information summary of endothelin receptor antagonist.

Data mining
Signal detection is an important component of post-marketing safety monitoring of pharmaceuticals. 19 Among the data mining tools for signal detection, the proportional imbalance has been the most active in the field of application. Reporting odds ratio (ROR), and proportional reporting ratio (PRR) based on the emergence of proportional imbalance, is typical of the frequency count method, while Bayesian confidence propagation neural network (BCPNN) and multi-item gamma Poisson shrinker (MGPS) are proposed based on Bayesian theory. 20 Some studies have shown that Bayesian methods (such as MGPS and BCPNN) are superior to frequency count methods such as PRR, but MGPS and BCPNN also have low sensitivity and tend to detect less signal than ROR and PRR. 20 Therefore, it was decided in this study to use four methods to tap into the adverse effect signal (Supplemental Table 1),21–24 to compare the correlation between the three ERAs and liver injury. In addition, for the sake of research accuracy, we only included ERA’s single drug treatment regimen, that is, under the ROLECOD project of the DRUG file, only relevant records with the target drug being the primary prospect drug were screened, and other cases of ERA appearing in secondary supervised drug (SS) and concomitant drugs (C) were excluded. Furthermore, the risk of liver injury due to the combination of ERA with other targeted agents was explored and the timing of adverse reactions due to potentially threatening combinations was assessed [the time interval between the onset of dosing (START_DT) and the onset of the adverse event (EVENT_DT) was used as the specific time of occurrence of the adverse reaction]. The study excluded illogical records such as EVENT_DT earlier than START_DT. Finally, we analyzed the outcomes of potentially risky drug combinations, screening all records for reports with outcomes and calculating the probability of death and hospitalization, both serious conditions, in the overall adverse event. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology Statement. 25
Statistical analysis
The study used descriptive analysis to demonstrate the clinical characteristics of liver injury due to ERA. A nonparametric test (Mann–Whitney U test for two samples, Kruskal–Wallis test for multiple samples) was used to compare whether there was a difference in the time to liver injury with different combinations of ERA combined with targeted drugs. Fisher’s exact test or Pearson’s chi-square test is used to compare the results of different combinations of adverse reactions. p < 0.05 with 95% confidence intervals was considered statistically significant. The software used for statistical analysis and data mining is SAS, version 9.4 (SAS Institute Inc., Cary, USA).
Results
Descriptive analysis
From January 2004 to December 2022, FAERS collected a total of 17,042,127 reports, including 199,665 reports related to ERA and 348,265 reports leading to liver injury. In the end, we obtained 3581 reports of liver injury after using ERA. ERA most suspected of causing liver injury was bosentan (n = 2142, 59.82%), while only 578 (16.14%) records indicated that macitentan may cause liver injury. These adverse events occurred mainly in North America (n = 2099, 58.61%), with a relative balance in other regions, and were predominantly reported by healthcare professionals (n = 2157, 60.24%). The patients with liver injury were more female than male (60.63% versus 20.78%), and the age was concentrated in 61–75 years (26.75%). The average age of males was 51.28
The specific clinical characteristics of ERA-induced liver injury were excavated from FAERS.
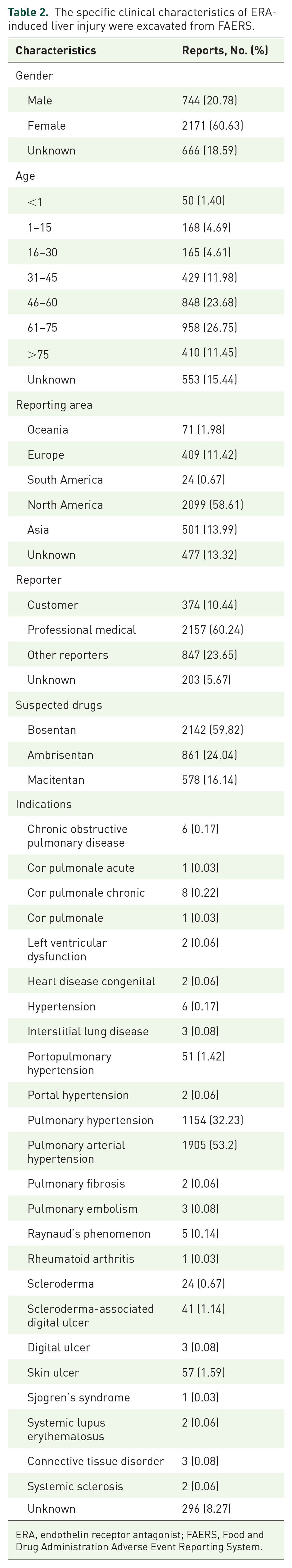
ERA, endothelin receptor antagonist; FAERS, Food and Drug Administration Adverse Event Reporting System.
Data mining results
In this study, four methods were used to process the target signal, and the results were detected concerning the criteria corresponding to the different algorithms. The specific situation is shown in Table 3. Bosentan had the highest risk of liver injury, and its results met the detection criteria of ROR, PRR, BCPNN, and MGPS, followed by macitentan, which was associated with liver injury according to the results of BCPNN. None of the results for ambrisentan met the test criteria, so ambrisentan did not present a risk of liver injury. On this basis, we further evaluated the relationship between bosentan and macitentan in combination with other targeted drug regimens and liver injury, the results of which are shown in Table 4. The results of all four methods confirmed that bosentan + sildenafil, bosentan + epoprostenol, and bosentan + iloprost had the strongest risk of liver injury, followed by macitentan + sildenafil, macitentan + epoprostenol, and the results of the ROR, PRR, and BCPNN showed an association between the two combination regimens and liver injury. Surprisingly, the combination regimens of macitentan all carry a risk of liver injury. Based on the results of the ROR and BCPNN, macitentan + tadalafil, macitentan + riociguat, macitentan + selexipag, and bosentan + treprostinil, all four combination regimens carry a risk of liver injury, while the regimens macitentan + iloprost, macitentan + treprostinil, and bosentan + tadalafil were also validated by the BCPNN to carry a risk of liver injury. As the results of bosentan + riociguat and bosentan + selexipag did not meet the test criteria, the risk of liver injury can be considered low.
The correlation between different ERAs and liver injury.
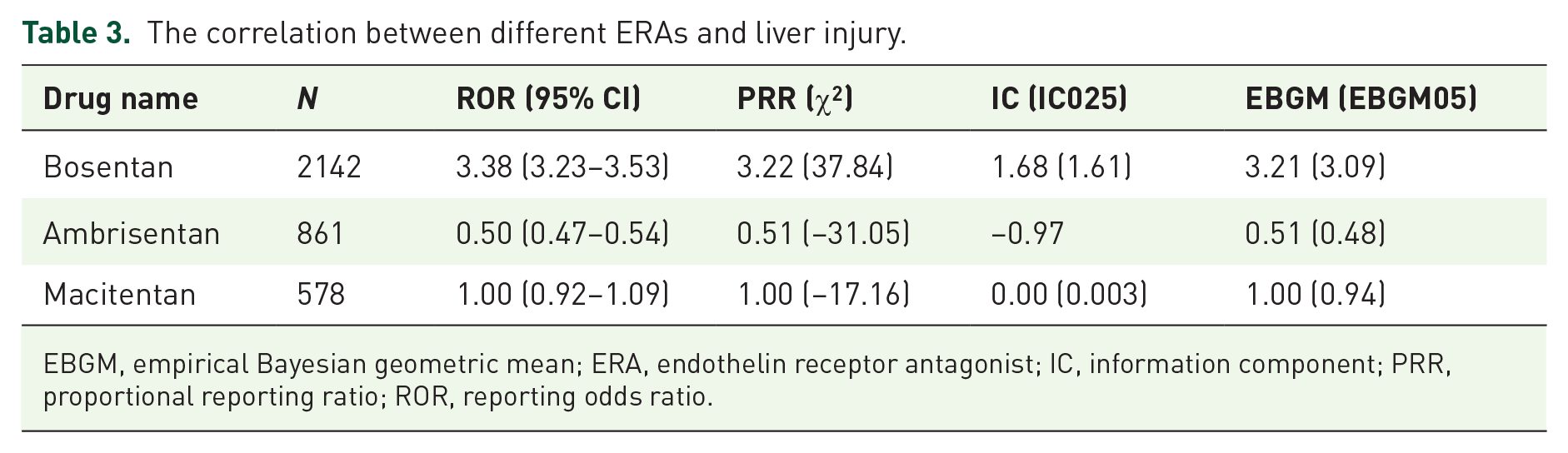
EBGM, empirical Bayesian geometric mean; ERA, endothelin receptor antagonist; IC, information component; PRR, proportional reporting ratio; ROR, reporting odds ratio.
The correlation between different combination regimens of bosentan/macitentan and liver injury.
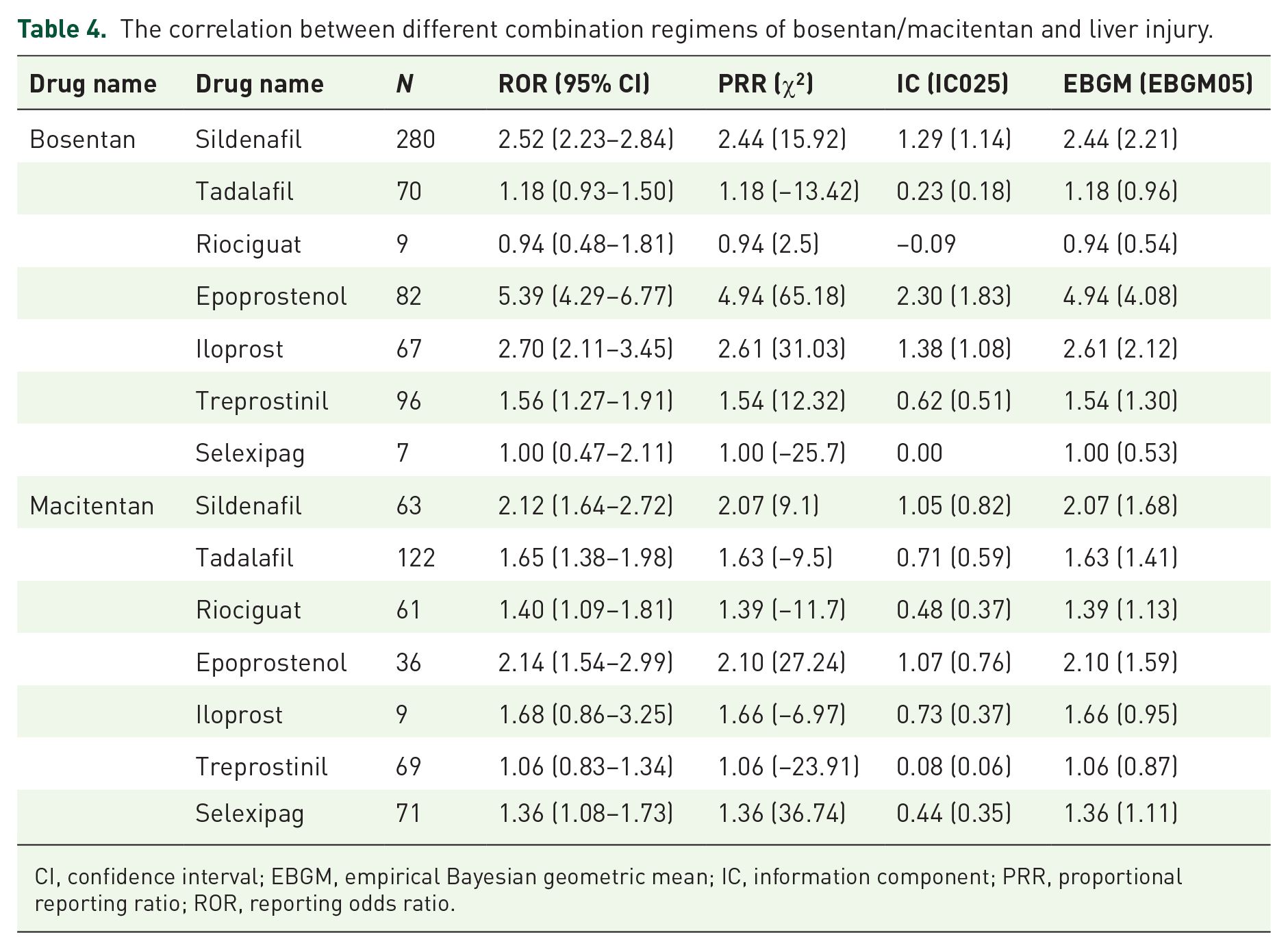
CI, confidence interval; EBGM, empirical Bayesian geometric mean; IC, information component; PRR, proportional reporting ratio; ROR, reporting odds ratio.
Onset time of liver injury related to ERA combination regimens
By counting the time to onset of liver injury due to ERA combination regimens, we found that the median time to an adverse reaction was 259 days [interquartile range (IQR): 58–716.5 days]. In addition, we also calculated the time to liver injury for different combinations separately. As shown in Figures 2 and 3, the median time for bosentan + sildenafil was 314.5 days (IQR: 49.25–774.5 days), the median time for bosentan + tadalafil was 231 days (IQR: 77.25–777.75 days), the median time for bosentan + epoprostenol had a median time of 204 days (IQR: 59–586.5 days), bosentan + iloprost had a median time of 313 days (IQR: 56.5–695 days), bosentan + treprostinil had a median time of 281 days (IQR: 82.25–609 days), while the median time for macitentan + sildenafil was 58 days (IQR: 6–318 days) and the median time for macitentan + tadalafil was 90 days (IQR: 32.5–381.25 days). The median time for macitentan + riociguat was 77 days (IQR: 21–201 days), the median time for macitentan + epoprostenol was 146 days (IQR: 16–382 days), the median time for macitentan + iloprost was 219.5 days (IQR: 26–648.5 days), the median time for macitentan + treprostinil was 145.5 days (IQR: 11.5–739.75 days), and the median time for macitentan + selexipag was 39 days (IQR: 5–250 days). There was a statistical difference in the time to liver injury between the different combination regimens mentioned above (Kruskal–Wallis test, p < 0.001). Patients treated with bosentan + sildenafil, versus those treated with macitentan + sildenafil (Mann–Whitney U test, p = 0.001), macitentan + tadalafil (Mann–Whitney U test, p = 0.038), macitentan + riociguat (Mann–Whitney U test, p = 0.021), and macitentan + selexipag (Mann–Whitney U test, p < 0.001) as treatment regimens had significant differences in the time to cause liver injury.

Distribution of the onset time of ERA combination regimens induced liver injury.
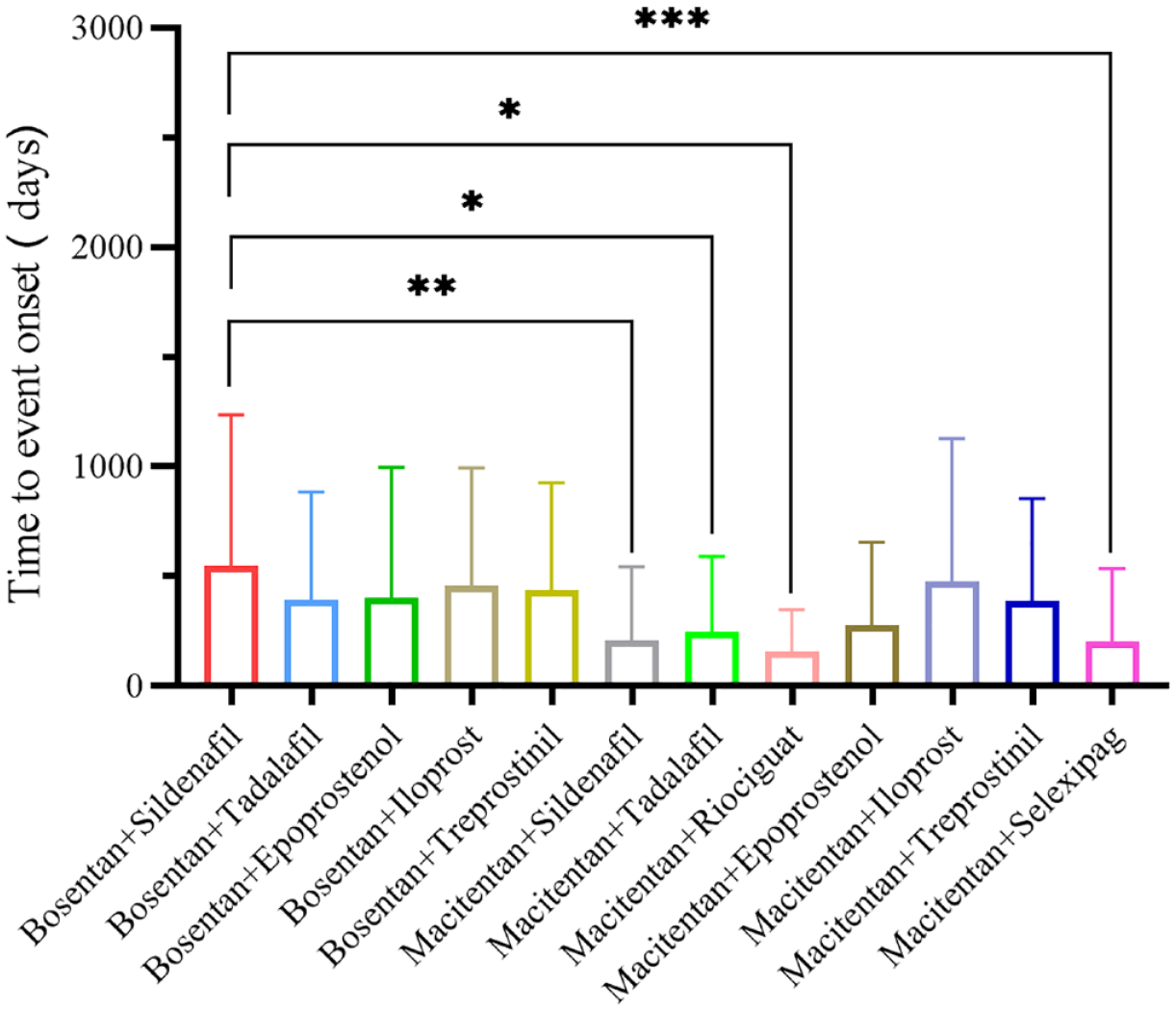
Onset time of liver injury exposed to ERA combination regimens.*Mann–Whitney U test p < 0.05, **Mann–Whitney U test p < 0.01, ***Mann–Whitney U test p < 0.001.
Outcome of liver injury caused by ERA combination regimens
Liver injury often leads to serious adverse consequences. Therefore, to investigate the outcome of ERA combination regimens leading to liver injury, we calculated the mortality and hospitalization rates after adverse reactions occurred. We have compiled reports of adverse outcomes and found that after using ERA combination regimens, the probability of hospitalization due to liver injury in patients is 47.86%, and 12.67% of all patients die from liver injury. Through further exploration, we can find that even if liver injury occurs, the outcomes will be significantly different with different medication regimens. As shown in Figure 4, the mortality rate of bosentan + iloprost (25.37%) is the highest, followed by bosentan + epoprostenol (20.73%), bosentan + sildenafil (14.29%), macitentan + tadalafil (13.93%), bosentan + treprostinil (12.50%), macentan + iloprost (11.11%), bosentan + tadalafil (10.00%), macentan + sildenafil (9.52%), macentan + epoprostenol (8.33%), macitentan + selexipag (5.63%), macitentan + riociguat (4.92%), macitentan + treprostinil (1.45%), Bosentan + iloprost (Fisher’s exact test, p < 0.05), bosentan + epoprostenol (Fisher’s exact test, p < 0.05), and macitentan + treprostinil (Fisher’s exact test, p < 0.05), the mortality of these three regimens was different from that of other regimens. Interestingly, the high rate of hospitalization following adverse events did not represent a high mortality rate. The highest incidence of liver injury leading to hospitalization was seen with macitentan + iloprost (77.78%), with hospitalization rates in descending order of macitentan + treprostinil (59.42%), macitentan + epoprostenol (55.56%), bosentan + sildenafil (51.43%), bosentan + epoprostenol (48.78%), bosentan + tadalafil (48.57%), macitentan + selexipag (45.07%), macitentan + sildenafil (44.44%), macitentan + riociguat (44.26%), bosentan + treprostinil (43.75%), macitentan + tadalafil (36.89%), and bosentan + iloprost (34.33%). There were statistical differences in hospitalization rates among the above-mentioned regimens (Chi-square test, p < 0.05). After adjustment by Bonferroni, there was no statistical significance in the pairwise comparison of hospitalization rates after liver injury among different regimens (p > 0.05).
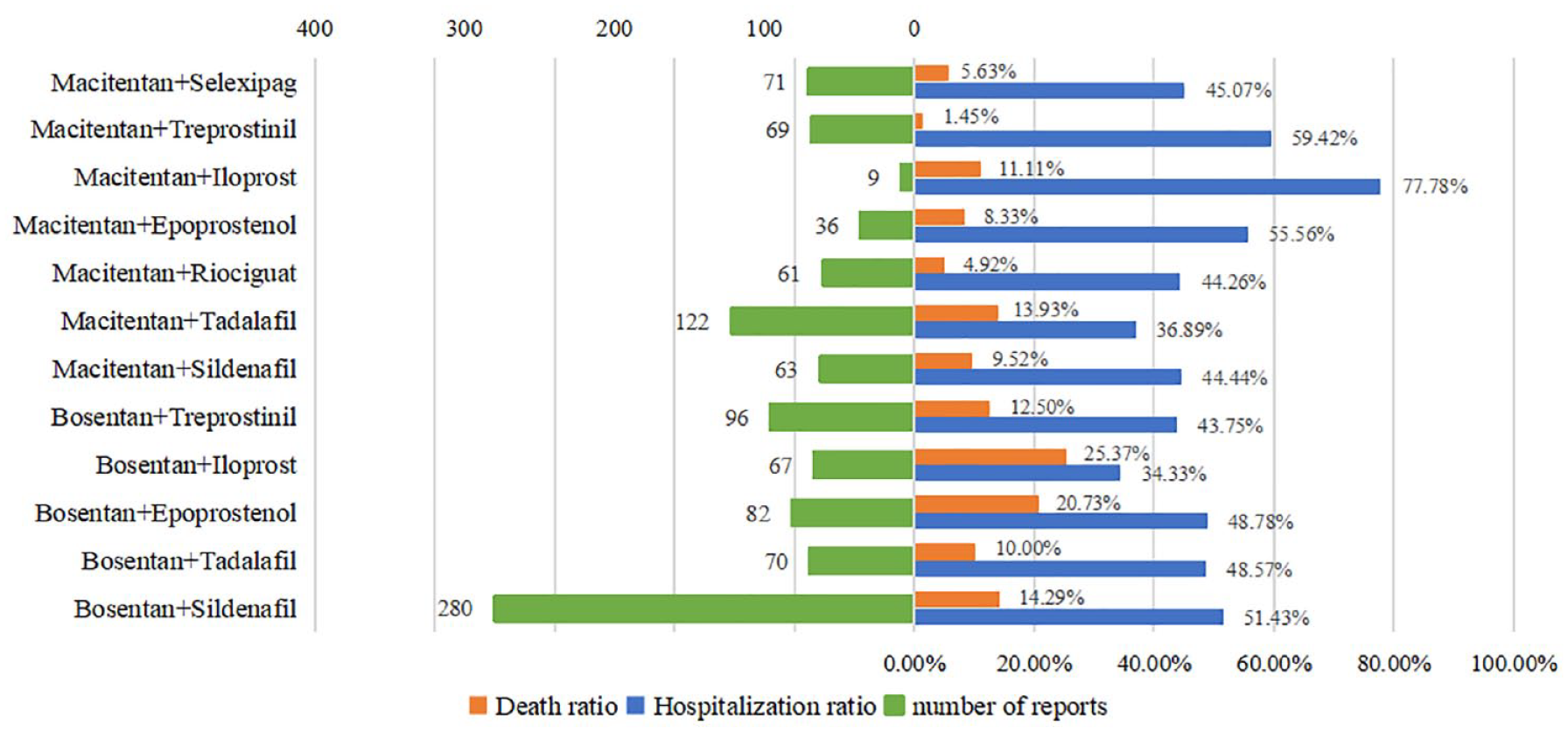
Mortality and hospitalization rate of liver injury caused by ERA combination regimens.
Discussion
Bosentan was launched in 2001 and was the first drug to be marketed in the ERA, followed by ambrisentan and macitentan. At present, ERA has been widely used in clinics and is an important therapeutic drug for patients with PAH. However, liver injury due to ERA exposure is always a problem that we have to face. To our knowledge, this study is the largest to date to analyze the risk of liver injury from different ERA and ERA combination regimens using the FAERS database, and on this basis, we have also compared the time to adverse effects and clinical outcomes of various ERA combination regimens. From the results, it is clear that bosentan and macitentan have a risk of causing liver injury, while their combination regimens with other targeted drugs, except bosentan + riociguat and bosentan + selexipag, have a risk of causing liver injury and show different clinical characteristics.
The mechanism by which bosentan causes liver injury is not yet clear. Available studies suggest that bosentan-induced liver injury may be mediated by inhibition of the bile salt export pump, which leads to intracellular accumulation of cytotoxic bile salts and bile salt-induced hepatocyte injury. 26 Despite the risk of hepatotoxicity, clinical trials attempting to explore the effects of bosentan in the treatment of various types of PH have not ceased, yet interestingly, the evaluation of bosentan-induced liver injury has been inconsistent across trials.6,27,28 Since the clinical application of ambrisentan and macitentan, the stereotype that ERA can cause liver injury seems to have been dispelled, and two drugs have shown excellent therapeutic effects and are not hepatotoxic.12,29 A meta-analysis of 24 randomized, double-blind, placebo-controlled clinical trials involving 4894 patients showed that bosentan increased the risk of liver dysfunction, while ambrisentan and macitentan were more associated with peripheral edema and anemia. 30 In the post-marketing study of 10,927 patients exposed to ambrisentan, only 72 patients (0.72%) were considered to be potentially related to liver events. 18 Based on this result, the FDA has removed the warning label for ambrisentan to monitor liver function monthly. However, the post-marketing status of macitentan is worrying. In recent years, there have been occasional case reports of liver injury caused by macitentan,16,31 including a case of explosive hepatitis caused by a combination of ERA and other targeted drugs.
It is well known that clinical trials are an essential step before drugs are marketed, which is the main source of information about the effectiveness and safety of new drugs. Unfortunately, however, clinical trials have limitations such as limited sample size and relatively limited drug observation period, which will undoubtedly affect the accuracy of research results. In addition, due to the relative lack of total samples, we cannot obtain the general characteristics of adverse reactions through clinical trials. Spontaneous reporting systems are an important tool for post-marketing drug safety monitoring, and the vast amount of drug data available can help us to determine the relationship between drugs and adverse reactions. Through data mining from FAERS, we can find that the risk of liver injury in females is significantly higher than that in males (60.63% versus 20.78%), with a concentration of ages ranging from 61 to 75 years old (26.75%). A large amount of epidemiological evidence indicates that due to the rarity and diagnostic complexity of PAH, the statistical methods and scope of PAH in different countries are inconsistent, leading to differences in survival rates and disease subtypes among patients in different regions. However, it is undeniable that females have always been the main group affected by the disease.32,33 Available studies have shown that females are risk factors for developing hepatocellular liver injury and acute liver failure.
34
The gender difference in ERA-related liver injury may be because females are the main group using drugs, and we need a more rigorous experimental design to validate this viewpoint. Interestingly, in 1981, The National Institutes of Health established the first registry for PH. At that time, the average age of diagnosis was 36
In our study, bosentan and macitentan were shown to be associated with liver injury, while ambrisentan did not induce hepatotoxicity. Interestingly, ambrisentan is a highly selective ETA receptor antagonist with an affinity of 260 times that of endothelin B (ETB), while bosentan and macitentan, as competitive dual antagonists for binding to ETA, have an affinity of only 20 and 50 times that of ETB receptors.30,37 Unlike the other two types of ERA, ambrisentan does not cause liver injury, which seems to prove that selectively blocking the ETB receptor increases the risk of liver injury. ETB receptors are widely distributed in the body, but compared to other peripheral tissues, the liver of normal individuals has the highest proportion. Ling et al. 38 found significant changes in the morphology of the liver by knocking out the ETB gene in mice, with contraction of the portal sinus and congestion of the large hepatic veins. Animal experiments have confirmed that inhibition of the ETB receptor may directly induce hepatotoxicity. At present, we still cannot determine the causal relationship between ERA-induced hepatotoxicity and direct inhibition of ETB. We look forward to more research to validate this hypothesis. The results obtained in this study are consistent with most previous studies that bosentan carries a risk of hepatotoxicity, but unlike the widely perceived results for bosentan, macitentan is generally not considered to be hepatotoxic. 29 A real-world study that included 5650 PAH patients newly receiving macitentan as a treatment regimen showed that 186/5650 (3.3%) patients had ALT or AST levels ⩾3 × ULN, 49/5650 (0.9%) patients experienced AST or ALT ⩾3 × ULN and TBIL ⩾2 × ULN. 17 At this stage, there is still a gap in large safety trials related to macitentan, which may explain the inconsistent findings, and we need to recruit more patients and a longer clinical observation period to validate the findings of this study. In addition, the prevailing view in the study of ERA causing hepatotoxicity focuses on drug interactions, including increased hepatic uptake and inhibition of cytochrome P450 enzyme activity. 16 Ambrisentan, whose primary metabolic pathway is hepatic glucuronidation, is at minimal risk of interaction with other drugs, whereas bosentan and macitentan, although both metabolized primarily by cytochrome P450 isozymes differ significantly in the extent to which they are affected. 39 Bosentan is not only metabolized by CYP3A4 and CYP2C9 but also participates in the liver uptake of human organic anion-transporting polymers. It is known to interact with various drugs, while macentan is mainly metabolized by CYP3A4, and is secondary metabolized by CYP2C8, CYP2C9, and CYP2C19. Although influenced by multiple metabolic enzymes, there was no significant increase in macitentan levels in combination with potent CYP3A4 inhibitors and no relevant change in exposure to its active metabolites. 40 Against the backdrop of such complex risks associated with ERA combination therapy, the risk of combining it with other targeted drugs is also our focus. According to the results of this study, bosentan + sildenafil, bosentan + epoprostenol, and bosentan + iloprost, the three combinations with the highest risk of inducing hepatotoxicity, were found. Several clinical studies in small samples have shown that the combination with sildenafil increases bosentan exposure due to the action of CYP3A4, leading to an increased incidence of adverse reactions. 40 We recommend enhanced pharmacovigilance monitoring for these three common combinations mentioned above. Surprisingly, all regimens of macitentan are associated with a risk of liver injury, with macitentan + sildenafil and macitentan + epoprostenol having the highest risk. The available evidence is insufficient to confirm that interactions are the most significant cause of liver injury from macitentan combination regimens, and the combination of macitentan with targeted drugs has been shown to have a low liver risk. 41 The current studies are, of course, characterized by low sample sizes. Caution should be exercised in the treatment of PAH with combinations containing macitentan, and we believe that more evidence is needed to evaluate its safety.
The time to onset of adverse reactions was also a key concern. The median time to liver injury with ERA combination regimens was 259 days (IQR: 58–716.5 days). There is no typical pattern in the onset of DILI; in general, the onset of idiopathic drug-induced liver injury (IDILI) is 1–3 months, but it can occur slightly earlier in some cases, as well as after 3 years of treatment, and in some cases, the onset of IDILI may even be more than 4 weeks after stopping treatment. 42 Therefore, IDILI is always a challenge for doctors and we cannot predict the pattern of its occurrence. The phenomenon is particularly evident in the case of ERA combination regimens. The time frame for the onset of liver injury can span from the day of administration to 7–8 years afterward, and the delay between the start of drug treatment and the appearance of signs of liver injury can make it difficult for physicians to determine a possible causal relationship. We recommend regular monitoring of liver function parameters from the start of drug treatment. In addition, macitentan-based combination regimens, mainly macitentan + sildenafil, macitentan + tadalafil, macitentan + riociguat, and macitentan + selexipag appear to be prone to hepatotoxicity at an earlier time than other treatment regimens, which warrants extra attention.
To further assess the severity of DILI, which undoubtedly poses a great threat to the health of patients, we compared mortality and hospitalization rates after liver injury with various ERA combination regimens. DILI is a cascade of events leading to cell death and inflammation, which has become a serious health problem due to its unpredictability and potential risk of death. 34 Unfortunately, according to the results of this study, 12.67% of the patients died due to liver injury, and 47.86% of the patients were hospitalized due to liver injury caused by ERA combination regimens, which indicates that most of the patients were seriously ill, but gradually improved after withdrawal of medication and active rescue measures. Notably, bosentan + iloprost (25.37%) and bosentan + epoprostenol (20.73%) showed surprisingly high mortality rates. In general, prostacyclin-based targeted agents are recommended for initial combination therapy in patients at high risk of death. 15 To exclude the presence of mortality due to underlying liver disease, we reconfirmed the reported information that all patients on both regimens did not register liver disease or medication cause denying portopulmonary hypertension. Although only 34.33% of patients were hospitalized due to hepatotoxicity with bosentan + iloprost, 25.37% of patients died due to hepatotoxicity. We suggest that doctors should pay special attention to the effects of this regimen on liver function, and more aggressive care and treatment should be carried out if adverse reactions occur. Fortunately, 59.42% of patients were hospitalized due to hepatotoxicity after using macitentan + treprostinil, while the dead patients only accounted for 1.45% of the total liver injury events caused by macitentan + treprostinil, indicating that the severity of liver injury caused by this regimen may be low, and timely treatment may contribute to the outcome of the event.
Hepatotoxicity is a vexing topic for both physicians and patients and with the widespread use of ERA-based combination regimens for PAH treatment, we have to be cautious about DILI. This study can be used for clinical decision-making, using a combination regimen containing bosentan and macitentan, especially high-risk medication regimens. It is necessary to be vigilant for patients with high liver risk and strengthen their monitoring. The results of the current study suggest that the foremost step in the therapeutic management of DILI is to discontinue the relevant medication and avoid re-exposure. 43 In most cases, spontaneous recovery from liver injury occurs without the need for further therapeutic measures. The results of this study seem to confirm this, although there is a higher mortality rate, the majority of patients have a better outcome after hospitalization or aggressive interventions. When ERA is used as a therapeutic drug, special attention should be paid to the symptoms and liver function indicators related to liver injury.
Limitations
Our research used real-world data and relied on data mining techniques to obtain adverse reaction signals, which has certain advantages compared with other related studies, but we cannot deny the existence of limitations. First, the spontaneous reporting system can only passively receive reported information on adverse events, and there may be under-reporting and duplicate reporting, which may affect data mining results. Second, there are instances where the reported information is incomplete, and although this relates to only a small number of cases, it can directly interfere with the accuracy of the results of the analysis. Finally, due to the shortcomings of the database, we are not able to retrieve the full range of underlying disease conditions of patients, nor do we have access to pre-drug liver function indicators, and we are only able to use the available information for analysis, which prevents us from uncovering the full range of risk factors for liver injury. Even with these shortcomings, the data mined in this study confirm the association of ERA and ERA combination regimens with liver injury, providing clues for further clinical studies and mechanistic exploration.
Conclusion
In this study, by mining the FAERS database, we analyzed and compared the risk of liver injury associated with different ERA and ERA combination regimens. According to the results of this study, bosentan and macitentan are associated with a risk of liver injury, and the combination regimens bosentan + sildenafil, bosentan + epoprostenol, and bosentan + iloprost have the highest risk. In addition, in the signal mining of combination regimens, it is of concern that all combination regimens of macitentan carry a risk of liver injury. Females and advanced age are risk factors for ERA-related liver injury, and we recommend regular monitoring of patients’ liver function for a high-risk combination regimen. Our study provides a reference for pharmacovigilance in the persistence of drugs for the treatment of PAH.
Supplemental Material
sj-pdf-1-tar-10.1177_17534666231223606 – Supplemental material for Liver injury due to endothelin receptor antagonists: a real-world study based on post-marketing drug monitoring data
Supplemental material, sj-pdf-1-tar-10.1177_17534666231223606 for Liver injury due to endothelin receptor antagonists: a real-world study based on post-marketing drug monitoring data by Shichao Dong, Xiaofei Guo, Huayu Wang and Chuan Sun in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
Thanks to all the staff and researchers who provided support for the study. Zipeng Zhang reviewed the code written for execution. Jinrong Zhang reviewed the pictures and proposed amendments.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
