Abstract
Background:
The prognosis of malignant pleural effusion (MPE) is poor. A timely and accurate diagnosis is the prerequisite for managing MPE patients. Carbohydrate antigen 72-4 (CA72-4) is a diagnostic tool for MPE.
Objective:
We aimed to evaluate the diagnostic accuracy of pleural fluid CA72-4 for MPE.
Design:
A prospective, preregistered, and double-blind diagnostic test accuracy study.
Methods:
We prospectively enrolled participants with undiagnosed pleural effusions from two centers in China (Hohhot and Changshu). CA72-4 concentration in pleural fluid was measured by electrochemiluminescence. Its diagnostic accuracy for MPE was evaluated by a receiver operating characteristic (ROC) curve. The net benefit of CA72-4 was determined by a decision curve analysis (DCA).
Results:
In all, 153 participants were enrolled in the Hohhot cohort, and 58 were enrolled in the Changshu cohort. In both cohorts, MPE patients had significantly higher CA72-4 levels than benign pleural effusion (BPE) patients. At a cutoff value of 8 U/mL, pleural fluid CA72-4 had a sensitivity, specificity, and area under the ROC curve (AUC) of 0.46, 1.00, and 0.79, respectively, in the Hohhot cohort. In the Changshu cohort, CA72-4 had a sensitivity, specificity, and AUC of 0.27, 0.94, and 0.86, respectively. DCA revealed the relatively high net benefit of CA72-4 determination. In patients with negative cytology, the AUC of CA72-4 was 0.67.
Conclusion:
Pleural fluid CA72-4 helps differentiate MPE and BPE in patients with undiagnosed pleural effusions.
Introduction
Malignant pleural effusion (MPE) is caused by primary or metastatic cancer to the pleura. 1 The most frequent cause of MPE is lung cancer, followed by breast cancer, ovarian cancer, and lymphoma. 2 The prognosis of MPE is poor, with a median survival of less than one year after diagnosis. 3 A timely and accurate diagnosis is the prerequisite for managing MPE patients. 4 Diagnosing MPE is challenging for clinicians because pleural effusion (PE) is not a specific sign of cancer. It can also be caused by benign disorders, such as tuberculosis, heart failure, pneumonia, and pulmonary embolism. 2 Therefore, further examinations are needed to determine whether the PE is caused by cancer.
Pleural fluid cytology, pleural biopsy, and thoracoscopy are gold standards for diagnosing MPE. 5 However, these diagnostic tools have limitations. Cytology has the advantages of low cost, short turn-around time (TAT), and high specificity, but its sensitivity is less than 60%, and its diagnostic accuracy is observer-dependent.6–8 Image-guided biopsy of pleural nodules and thoracoscopy have high sensitivity and specificity but they are invasive and can cause operation-related complications (e.g. infection and bleeding). In addition, special training is needed for thoracoscopy, limiting its use in remote areas. Tumor markers in serum and pleural fluid represent promising diagnostic tools for MPE because they are less invasive, objective, economical, and rapid. 9 Systematic reviews and meta-analyses indicated that tumor markers in pleural fluid [e.g. carcinoembryonic antigen (CEA), carbohydrate antigen 15-3 (CA15-3)] had specificities of >90% but their sensitivities were only approximately 50%.7,10–12 Therefore, developing novel and accurate tumor markers for MPE is a challenge.
Carbohydrate antigen 72-4 (CA72-4) is a tumor-associated polymorphic epithelial mucin highly expressed in several human adenocarcinomas. 13 Serum CA72-4 is increased in various cancers, such as gastric cancer, 14 ovarian cancer, 15 and colorectal cancer. 16 Some previous studies reported that serum CA72-4 has moderate diagnostic accuracy for lung cancer.17,18 Since more than half of MPE are caused by lung cancer, it is reasonable to hypothesize that CA72-4 could be a useful diagnostic marker for MPE. Indeed, some studies have investigated the diagnostic accuracy of pleural fluid CA72-4 for MPE, but their results varied.19–23 Therefore, the findings derived from previous studies need to be validated. In addition, the net benefit of CA72-4 determination in undiagnosed PE remains unknown. Here, we conducted a prospective and double-blind study to evaluate the diagnostic accuracy of pleural fluid CA72-4 for MPE.
Materials and methods
Participants
The participants in this study were from the SIMPLE (A Study Investigating Markers in PLeural Effusion), a prospective, preregistered (ChiCTR1800017449), and double-blind diagnostic test accuracy (DTA) study. The design details of the SIMPLE have been described previously. 24 Briefly, we prospectively recruited undiagnosed PE patients who visited the Department of Respiratory and Critical Care Medicine at the Affiliated Hospital of Inner Mongolia Medical University between September 2018 and July 2021 (Hohhot cohort). The inclusion criteria were as follows: (i) patients admitted with undiagnosed PE and (ii) patients who underwent diagnostic thoracentesis. The presence of PE was confirmed by chest X-ray, computed tomography, or ultrasound. The exclusion criteria were as follows: (i) patients with a PE history and known etiology during the past 3 months; (ii) <18 years old; (iii) pregnant women; (iv) patients with insufficient pleural fluid specimens; (v) patients who developed PE during hospitalization; and (vi) trauma- or surgery-induced PE.
In 2020, another center (Changshu, China) participated in the SIMPLE (Changshu cohort) study. The participants in the Changshu cohort were enrolled at the Affiliated Changshu Hospital of Nantong University between June 2020 and July 2021. The inclusion and exclusion criteria in the Hohhot and Changshu cohorts were identical. We reported this work following the Standards for Reporting of Diagnostic Accuracy Studies guidelines (Supplemental Table 1).25,26
Diagnostic criteria
MPE was confirmed by pleural biopsy or cytology. In some cases, MPE was diagnosed with primary cancer and the exclusion of a benign pleural effusion (BPE). Tuberculous pleural effusion was diagnosed with pleural fluid Mycobacterium tuberculosis (Mtb) culture, acid-fast staining, biopsy, and response to anti-tuberculosis treatment. Parapneumonic effusion was diagnosed with pleural fluid bacterial culture, biopsy, imaging characteristics, and antibiotic treatment response. The diagnosis of cardiac effusions was based on the clinical characteristics of the participants, including history, imaging features, laboratory tests (e.g. serum natriuretic peptides), and treatment response to diuretics. The results of pleural fluid CA72-4 were masked to clinicians who made the final diagnosis.
CA72-4 and biochemical markers
A pleural fluid specimen was collected into an anticoagulant-free tube at the time of patient admission. After centrifugation, the supernatant of pleural fluid was obtained and stored at −80°C to −70°C until analysis. In both cohorts, CA72-4 was measured by Roche Cobas e602 (Hohhot) and e801 (Changshu) immunoassay analyzers (Roche Diagnostic International Ltd, Rotkreuz, Switzerland) in November 2021. 27 Laboratory technicians who measured CA72-4 did not know the clinical details of the participants. In addition, we recorded the pleural fluid glucose, adenosine deaminase, white blood cell count, lactate dehydrogenase, and protein from the participant’s medical records.
Statistical analysis
Continuous data were expressed as median and quartile range. Categorical data were expressed as absolute numbers and percentages. We used the Kolmogorov–Smirnov method to test the normality of continuous data. 28 The Mann–Whitney U test was used for continuous data comparisons between groups. The chi-square test was used to compare categorical variables. We used a receiver operating characteristic (ROC) analysis to evaluate the diagnostic accuracy of CA72-4 for MPE. A decision curve analysis (DCA) was used to assess the net benefit of the CA72-4 measurement. All statistical analyses were performed with R (R Foundation for Statistical Computing, Vienna, Austria), and Stata SE 16 (StataCorp, TX, USA). p < 0.05 was considered statistically significant.
Results
Characteristics of the participants
In all, 153 participants were recruited in the Hohhot cohort and 58 in the Changshu cohort. Table 1 lists the participants’ baseline characteristics. MPE was diagnosed in 66 participants in the Hohhot cohort and 26 participants in the Changshu cohort.
Characteristics of the participants.

Data are presented as the median (interquartile range) or absolute number (percentage).
Numbers of patients in parentheses.
ADA, adenosine deaminase; BPE, benign pleural effusion; HF, heart failure; LDH, lactate dehydrogenase; MPE, malignant pleural effusion; PF, pleural fluid; PPE, parapneumonic effusion; TPE, tuberculous pleural effusion; WBC, white blood cell.
CA72-4 in MPE and BPE
Figure 1 shows the pleural fluid CA72-4 levels in MPE and BPE patients. In the Hohhot cohort, the median (interquartile ranges) CA72-4 in MPE patients and BPE patients were 4.87 (1.19–27.77) and 0.93 (0.83–1.94) U/mL, respectively [Figure 1(a)]. In the Changshu cohort, the median CA72-4 (interquartile ranges) in MPE patients and BPE patients were 3.02 (1.90–9.41) and 1.06 (0.91–1.30) U/mL, respectively [Figure 1(a)]. In the pooled cohort, the median CA72-4 (interquartile ranges) in MPE patients and BPE patients were 4.28 (1.31–21.4) and 0.98 (0.85–1.60) U/mL, respectively. In all cohorts, MPE patients had significantly higher CA72-4 than BPE patients (p < 0.001) [Figure 1(a)]. In addition, we analyzed the association between CA72-4 and the pathological types of MPE. Since more than half of MPE were due to lung cancer and the sample size for gastric cancer and mesothelioma was small, we only compared CA72-4 levels between lung and non-lung cancers. Although significantly higher CA72-4 concentrations were observed in lung cancer patients from the Changshu cohort, this marker did not differ significantly between lung and non-lung cancer in the combined cohort [Figure 1(b)]. We also failed to observe any significant difference in the levels of CA72-4 among the different causes of BPE [Figure 1(c)].
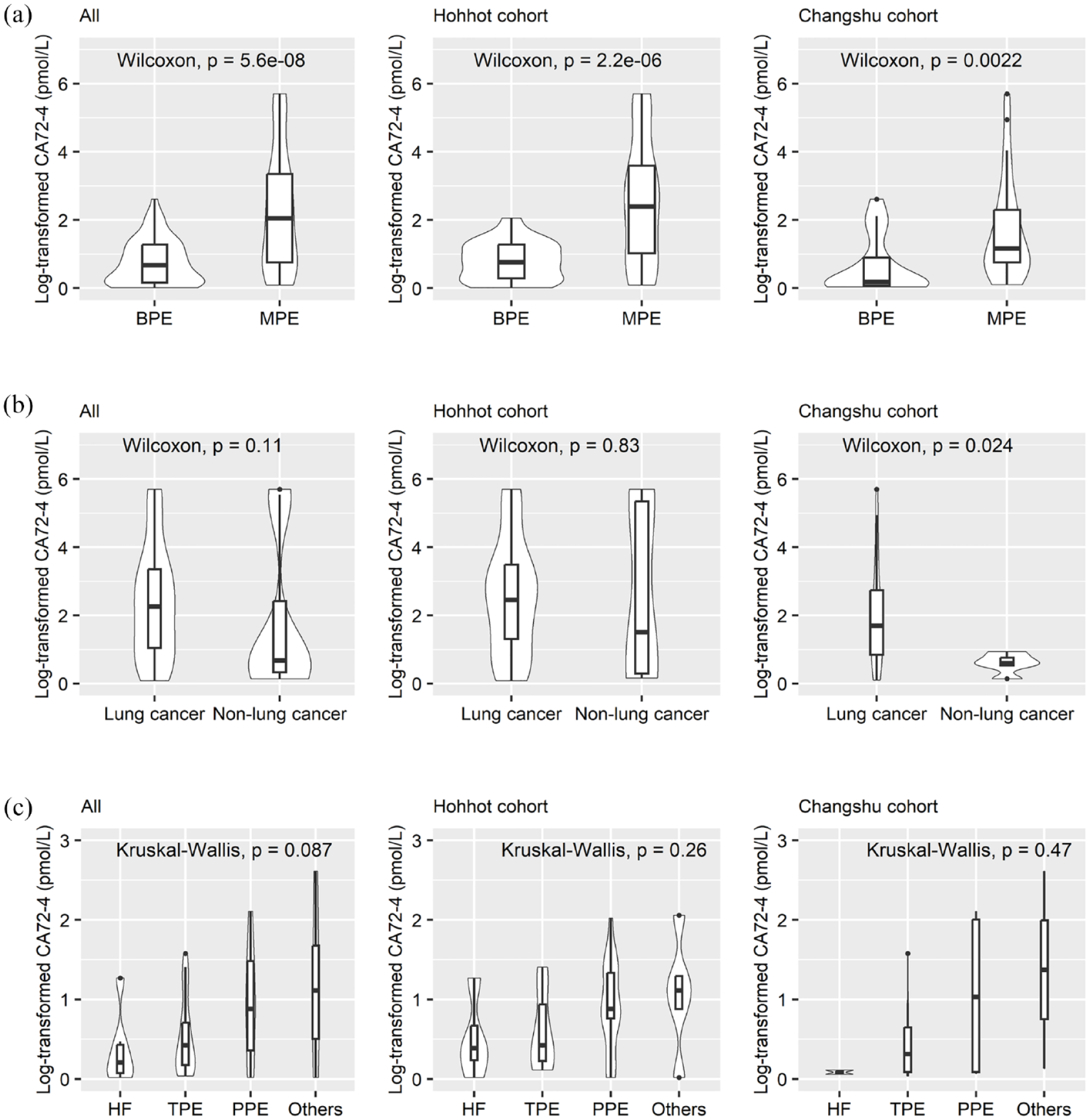
Comparison of log2-transformed CA72-4 between MPE and BPE. (a) CA72-4 in malignant and benign pleural effusions, (b) CA72-4 in MPE caused by lung cancer and other cancers, (c) CA72-4 in benign pleural effusions.
Diagnostic accuracy of CA72-4 for MPE
Figure 2 shows the ROC curves of CA72-4 for MPE in both cohorts. The AUCs [95% confidence intervals (CIs)] in the Hohhot and Changshu cohorts were 0.79 (0.72–0.87) and 0.86 (0.76–0.95), respectively. The AUC in the pooled cohort was 0.80 (0.74–0.86). The sensitivities and specificities of CA72-4 at the thresholds of 8 U/mL (specificity of 0.90) and 15 U/mL (specificity of 1.00), respectively, are summarized in Table 2.

ROC curves of CA72-4 and CEA for MPE.
Operating characteristics of pleural fluid CA72-4 for the identification of MPE.

AUC, area under the receiver operating characteristic curve; BPE, benign pleural effusion; CI, confidence interval; MPE, malignant pleural effusion; NLR, negative likelihood ratio; PLR, positive likelihood ratio.
In addition, we analyzed the diagnostic accuracy of CA72-4 for MPE in patients with negative pleural fluid cytology. The AUC (95% CI) of CA72-4 was 0.67 (0.55–0.79; Figure 2). The sensitivity and specificity of CA72-4 in these patients are summarized in Table 2. In the authors’ institution, only pleural CEA is routinely ordered by clinicians. The diagnostic accuracy of CEA for MPE has been published in our previous studies.29,30 In the current investigation, we also compared the diagnostic accuracy of CEA and CA72-4. The AUC of CA72-4 in the combined cohort was lower than that of CEA (p = 0.006, Table 2). We incorporated CEA and CA72-4 into a logistic regression model. The model can be expressed as follows:

The AUC of the model was 0.88 (95% CI: 0.83–0.93), comparable to that of CEA (p = 0.605, Table 2). Although the AUC of the model was close to that of CEA, using both markers resulted in a higher sensitivity than CEA alone (0.69 versus 0.48, Table 2), at the same specificity of 0.98.
The net benefit of pleural fluid CA72-4
Figure 3 shows the decision curves of CA72-4 in the Hohhot, Changshu, and the combined cohorts. In the decision curve plot, the x-axis is the risk threshold used to define positive CA72-4. It can be calculated by a logistic regression model and represents the probability of MPE for a given patient. The risk threshold represents the ‘exchange rate’ between missed and misdiagnoses. 31 For example, if a clinician believes that a patient with a risk threshold of >0.1 should receive a pleural biopsy, the corresponding odds is 1:9, indicating that missing an MPE is nine times worse than doing an unnecessary pleural biopsy in nine BPE patients. 32 The risk threshold depends on the clinical setting and the values of the caregiver and patient31,32; that is, how doctors value different outcomes for a given patient in a specific clinical setting. The y-axis is the net benefit of a CA72-4 value. The word ‘net’ means the overall benefit of asking MPE patients to receive pleural biopsy (true-positive) minus the overall harm of unnecessary pleural biopsy in BPE (false positive). The following equation can calculate it.

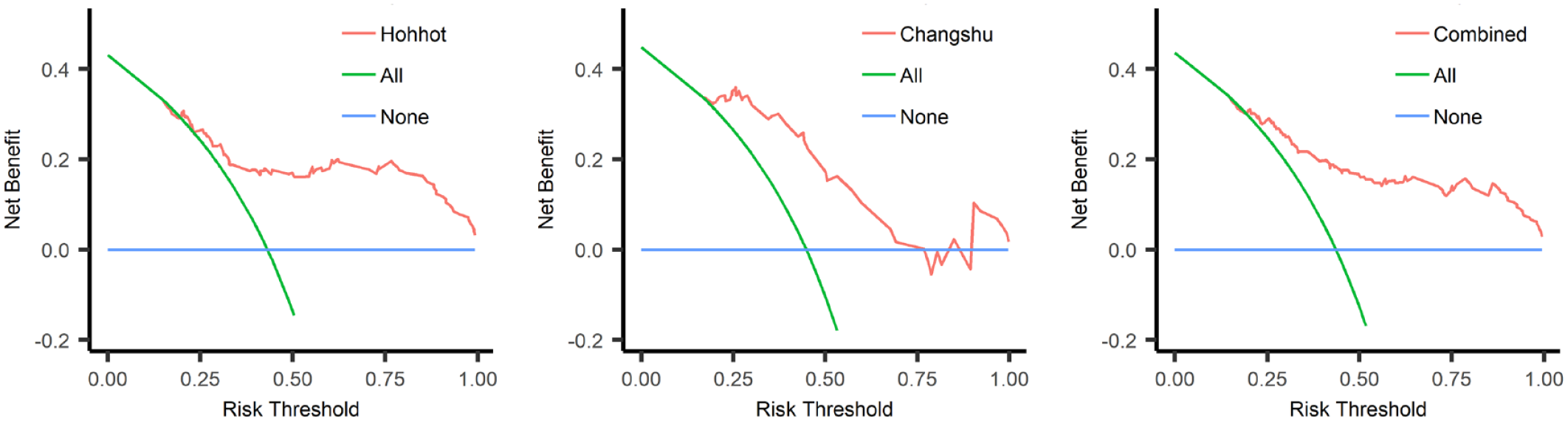
Decision curve analysis of CA72-4 for MPE. Red line: net benefit curve of CA72-4. Green line: assume all patients receive the CA72-4 test. Blue line: assume no patients receive the CA72-4 test.
True positive and true negative can be calculated by a two-by-two table at a given risk threshold. We assume that there is a cohort composed of 1000 undiagnosed PE patients (600 MPEs and 400 BPEs). At the threshold of 0.20, the sensitivity and specificity of CA72-4 are 0.80 and 0.80, respectively. The numbers of true-positive patients and false-positive patients were 480 and 80, respectively. The corresponding net benefit at this threshold can be calculated as follows:

In the above equation, 480/1000 represents the proportion of patients who will benefit from the CA72-4 test, while 80/1000 represents the proportion of patients harmed by the CA72-4 test (unnecessary pleural biopsy). In addition, 0.2/(1–0.2) means that the harm of four unnecessary pleural biopsies (false positive) equals the benefit of one necessary pleural biopsy (true positive).
In plain language, a net benefit of 0.46 can be explained as the following: at the risk threshold of 0.20, 46% of patients can be correctly identified as MPE without increasing the number of BPE patients who received an unnecessary pleural biopsy.
We assumed the clinically reasonable range of threshold probabilities was between 0.25 and 0.50. A clinically meaningful benefit can be observed in this threshold range (Figure 3).
Discussion
The primary findings of this study were as follows: (i) MPE patients had significantly higher pleural fluid concentrations of CA72-4 than BPE patients; (ii) CA 72-4 had moderate diagnostic accuracy for MPE; (iii) CA72-4 determination had a net benefit in patients with undiagnosed PEs; and (iv) CA72-4 >15 U/mL has a specificity of 1.00 for MPE.
To our knowledge, seven studies have assessed the diagnostic accuracy of CA72-4 for MPE.19–23,33 The characteristics and major findings of these studies and the current one are summarized in Table 3. Some studies revealed that CA72-4 had moderate diagnostic accuracy for MPE, with AUCs higher than 0.70.22,33 However, a study showed that the AUC of CA72-4 for MPE was only 0.60. 23 Notably, at the threshold of approximately 10 U/mL, the specificities in all studies were close to 1.00. Trapé et al. 20 reported that the specificity reached 1.00 when the threshold was set at 21 U/mL. Compared with these studies, one strength of our study is that we evaluated the diagnostic accuracy of CA72-4 in two prospective cohorts, and the results in these two cohorts were comparable, indicating that our findings are reliable and reproducible. The AUCs of CA72-4 in our cohorts were 0.79 (Hohhot cohort) and 0.86 (Changshu cohort), respectively, supporting the moderate diagnostic accuracy of CA72-4 for MPE. Another strength of our study is that we analyzed the diagnostic accuracy of CA72-4 in patients with negative pleural fluid cytology. The AUC of CA72-4 was 0.67 in patients with negative cytology, supporting the value of CA72-4 to improve the diagnostic accuracy of cytology. The third strength of our study is that we used DCA to assess the net benefit of CA72-4 determination in undiagnosed PE patients. In DTA studies, the ROC curve and its AUC are used to evaluate the diagnostic accuracy of the index test investigated.34,35 However, the ROC curve is the set of possible sensitivities and specificities obtained with different thresholds. Although the ROC curve is widely used in DTA studies, it has limitations. The primary limitation of the ROC curve is that it only considers the diagnostic accuracy of the investigated index test and does not incorporate information on consequences.36,37 Indeed, the improvement in diagnostic accuracy does not mean an improvement in patient outcomes. By contrast, DCA acts as a tool to address the potential effect of a marker or mathematic model on a patient’s clinical consequence, and thus, it entails high clinical relevance.36,38,39 In our study, the net benefit was observed in both cohorts, supporting the clinical utility of CA72-4 in undiagnosed PE patients.
Studies investigating the diagnostic accuracy of CA72-4 for MPE.

Several pleural tumor markers have been reported to be potentially useful diagnostic tools for MPE, such as CEA, CA15-3, and carbohydrate antigen 125 (CA125). 7 Systematic reviews of these tumor markers revealed that their sensitivities were approximately 0.50, and specificities were >0.90.7,11,12 In this study, CA72-4 had an AUC of approximately 0.80. At the threshold of 8 U/mL, its sensitivity was 0.40, and its specificity was 0.98. These results indicated that pleural CA72-4 had comparable diagnostic accuracy to conventional tumor markers (e.g. CEA, CA15-3, CA125). Our previous studies with the same cohort supported this conclusion, which found that the AUC of CEA for MPE was 0.89.29,30 Further studies are needed to investigate whether multiple marker strategies incorporating CA72-4 can improve the diagnostic yield of MPE.
A specificity of 1.00 has particular clinical significance because pleural biopsy, an invasive method, can be avoided in patients whose CA 72-4 was higher than the prespecified threshold.41,42 In our study, we found that the specificity of CA72-4 achieved 1.00 when the threshold was prespecified at 15 U/mL, indicating that in patients with CA72-4 >15 U/mL, a pleural biopsy may not be needed to support an MPE diagnosis. We reviewed the data in previously published works19–23,33 and found that only an extremely small portion of BPE patients had CA72-4 >15 U/mL.19,20,22,40 Therefore, we concluded that MPE could nearly be confirmed in patients with CA72-4 >15 U/mL.
Our study has some limitations. First, we only included patients with a definite etiology, and some participants with an unknown etiology or potential mixed causes were excluded from the final analysis. This design weakness may affect the study cohort’s representativeness and reduce our findings’ generalizability. Second, the sample size in our study was small, particularly in the Changshu cohort. Third, due to the cross-sectional design of this study, we could not investigate the prognostic value of CA72-4 in MPE patients. Fourth, we did not estimate the sample size before participant enrollment because this is a data-driven rather than a hypothesis-driven study. Therefore, further studies with larger sample sizes are needed to validate our findings. Finally, we used the frozen pleural fluid specimen to determine CA 72-4; but the long-term stability of CA 72-4 in the frozen specimen is unclear.
Conclusion
In summary, our study revealed that pleural fluid CA72-4 had moderate diagnostic accuracy for MPE, even in patients with negative cytology. In addition, CA72-4 determination showed a net benefit in patients with undiagnosed PE and thus should be considered as an adjunct tool for MPE. In patients with CA72-4 >15 U/mL, the diagnosis of MPE can be assumed without further testing (e.g. pleural biopsy), provided the patient is not a candidate for an active oncologic treatment.
Supplemental Material
sj-docx-1-tar-10.1177_17534666231222333 – Supplemental material for Pleural fluid carbohydrate antigen 72-4 and malignant pleural effusion: a diagnostic test accuracy study
Supplemental material, sj-docx-1-tar-10.1177_17534666231222333 for Pleural fluid carbohydrate antigen 72-4 and malignant pleural effusion: a diagnostic test accuracy study by Xi-Shan Cao, Li Yan, Ting-Wang Jiang, Jin-Hong Huang, Hong Chen, José M. Porcel, Wen-Qi Zheng and Zhi-De Hu in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
