Abstract
Background:
Lung cancer is the most common cause of malignant pleural effusion (MPE). Serum human epididymis secretory protein 4 (HE4) is a useful diagnostic marker for lung cancer.
Objective:
This study aimed to evaluate the diagnostic accuracy of pleural fluid HE4 for MPE.
Design:
A prospective, double-blind diagnostic test accuracy study.
Methods:
Patients with undiagnosed pleural effusion were enrolled in two cohorts (Hohhot and Changshu). Electrochemiluminescence immunoassay was used to detect pleural fluid HE4. The diagnostic accuracy of HE4 was evaluated by a receiver operating characteristic (ROC) curve, and the net benefit of HE4 was assessed by a decision curve analysis (DCA).
Results:
A total of 66 MPEs and 86 benign pleural effusions (BPEs) were enrolled in the Hohhot cohort. In the Changshu cohort, 26 MPEs and 32 BPEs were enrolled. In both cohorts, MPEs had significantly higher pleural fluid HE4 than BPEs. The area under the ROC curve (AUC) of HE4 was 0.73 (95% CI: 0.64–0.81) in the Hohhot cohort and 0.79 (95% CI: 0.67–0.91) in the Changshu cohort. At a threshold of 1300 pmol/L, HE4 had sensitivities of 0.44 (95% CI: 0.33–0.56) in the Hohhot cohort and 0.54 (95% CI: 0.35–0.73) in the Changshu cohort. The corresponding specificities were 0.90 (95% CI: 0.83–0.95) in the Hohhot cohort and 0.94 (95% CI: 0.84–1.00) in the Changshu cohort. In subgroup analyses, HE4 had an AUC (95% CI) of 0.78 (0.71–0.85) in exudates and an AUC of 0.69 (0.57–0.81) in patients with negative effusion cytology. The DCA revealed that HE4 determination had a net benefit in both cohorts.
Conclusion:
Pleural fluid HE4 has moderate diagnostic accuracy for MPE and has net benefit in pleural effusion patients with unknown etiology.
Introduction
Pleural effusion is a common sign associated with various disorders. Cancer, tuberculosis, pneumonia, and heart failure (HF) are frequent causes of pleural effusion.1,2 Pleural effusion caused by primary or secondary cancer of the pleura is classified as malignant pleural effusion (MPE), while caused by other disorders is classified as benign pleural effusion (BPE). The most common cancer in MPE is lung cancer, followed by breast cancer, lymphoma, gynecologic cancers, and malignant pleural mesothelioma. 1 MPE is an indicator of advanced tumor stage, and the prognosis of MPE patients is generally poor, with a median survival of <1 year.3,4 Timely and accurate diagnosis is a prerequisite for MPE management. 5
The gold standards for MPE are effusion cytology and closed or thoracoscopic pleural biopsy.5–7 However, these diagnostic tools have some limitations. The specificity of cytology is 1.00, but its sensitivity is only around 0.50.8,9 Closed pleural biopsy has high diagnostic accuracy for MPE but it is invasive and can cause operation-related complications (e.g. infection and bleeding) in some patients.10,11 In addition, the diagnostic accuracy of pleural biopsy is primarily affected by sample error and pathologists’ experiences. Thoracoscopic pleural biopsy has high diagnostic accuracy for MPE 12 but it is an invasive tool, and special training is needed. These limitations limit its applications in remote areas. Tumor markers in pleural fluid represent an easy-to-perform, rapid, and objective tool for diagnosing MPE. 13 Although it is widely accepted that tumor markers are insufficient to confirm or rule out MPE when used alone, they are indicators for a more invasive procedure to diagnose MPE. 14 Many studies have investigated the diagnostic accuracy of conventional tumor markers for MPE, such as neuron-specific enolase (NSE), carcinoembryonic antigen (CEA), carbohydrate antigen 125 (CA125), and a fragment of cytokeratin 19 (CYFRA 21-1). Systematic reviews and meta-analyses reveal that these tumor markers have high specificity (>0.90) for MPE but their sensitivity is only around 0.50.13,15–18 Therefore, developing novel tumor markers to improve the diagnostic accuracy for MPE remains valuable.
Human epididymis secretory protein 4 (HE4) is encoded by the WFDC2 gene constitutively expressed in the ovary, pancreas, tracheas, kidney, and lungs. 19 The physiological role of HE4 remains largely unknown. Animal studies with HE4 knockout mice indicated that HE4 is associated with the development of the respiratory system. 19 HE4 is involved in the development of various cancers. Previous studies showed that HE4 could promote invasion,20,21 chemoresistance, 22 radiation resistance, 23 and cancer metastasis. 21 Serum HE4 is a widely used marker for diagnosing ovarian cancer. A previous systematic review and meta-analysis indicated that serum HE4 had a sensitivity of 0.81 and a specificity of 0.91 for ovarian cancer. 24 Recent studies also revealed that serum HE4 was a useful diagnostic marker for other types of cancer, such as lung cancer25,26 and endometrial cancer.27,28 In addition, serum HE4 increases in HF patients, and higher HE4 predicts worse outcomes in HF patients. 29 Notably, previous studies showed that both serum tumor markers and their pleural fluid counterparts helped diagnose MPE, 13 and the diagnostic accuracy of pleural fluid tumor markers was higher than their serum counterparts.13,30 Given that serum HE4 has diagnostic utility for lung cancer,25,26 and lung cancer is the most common cause of MPE, we hypothesized that pleural fluid HE4 was a useful diagnostic marker for MPE. In this study, we investigated the diagnostic accuracy of pleural fluid HE4 for MPE. We reported this study following the Standards for Reporting of Diagnostic Accuracy Studies (STARD) guidelines (Supplemental Material 1).31,32
Methods
Participants
This work is a diagnostic test accuracy study. The participants in this work were from the SIMPLE (a
This study was approved by the ethics committees of the AHIMMU (No: 2018011, 15 June 2018) and the ACHXMU (No: 2020-KY-009, 24 April 2020). Signed informed consent forms were obtained from all individual participants included in the study. All patient details were de-identified. This study was conducted following the Declaration of Helsinki.
Diagnostic criteria
The diagnostic criteria for MPE were effusion cytology and pleural biopsy. In some patients, MPE was defined by the presence of primary or metastatic cancer with the exclusion of BPE, and the clinical data are highly suggestive of MPE. Tuberculous pleural effusion (TPE) was defined as positive pleural fluid Mycobacterium tuberculosis culture, acid-fast staining, or pleural biopsy. Positive pleural biopsy was defined as granulomatous inflammation with the exclusion of other granulomatous diseases. In some patients, treatment response to antituberculosis was used to define TPE. The diagnostic criteria for parapneumonic pleural effusion (PPE) were bacterial culture, pleural biopsy (neutrophil infiltration), pus cell, and imaging characteristics (loculation). In patients with negative bacterial culture and clinical data were highly suggestive of PPE, antibiotic treatment was initiated, and treatment response was evaluated. Patients with response to antibiotics were categorized as PPE. The diagnostic criteria for pleural effusion caused by HF were transudate, ultrasound and chest X-ray characteristics (bilateral pleural effusion), biochemical tests (e.g. serum natriuretic peptides), echocardiographic characteristics (e.g. increased heart size, decreased left ventricular ejection fraction), and treatment response to anti-heart failure medications. The participants in the Hohhot cohort were diagnosed by two senior clinicians (Z-D H and L Y). The participants in the Changshu cohort were diagnosed by three senior clinicians (T-W J, J-H H, and H C). Any disagreements were resolved by consensus. Pleural fluid HE4 concentration was masked to clinicians who made the diagnosis.
Data collection and pleural fluid HE4 assay
All participants received thoracentesis after enrollment. About 10 mL of the pleural fluid specimen was collected into an anticoagulant-free tube. After centrifugation, the supernatant of the specimen was aliquoted and stored at −80°C to −70°C until analysis. Pleural fluid HE4 in both cohorts was measured by the ARCHITECT i2000SR analyzer. The intra-assay coefficient of variation is 4.65% at 148 pmol/L. The upper limit of this HE4 assay is 1500 pmol/L. Specimens containing HE4 above the upper limit were regarded as 1500 pmol/L. The participant’s diagnosis was masked by the laboratory technician who measured HE4.
Statistical analysis
Continuous variables were expressed as the median and quartile range. The Kolmogorov–Smirnov test was used to determine the normality of the continuous variable. The Mann–Whitney U or independent student’s t-test was used to compare continuous variables between two groups where appropriate. The Kruskal–Wallis H test was used to compare continuous variables among three or more groups. We used a receiver operating characteristic (ROC) curve to evaluate the diagnostic accuracy of HE4 for MPE. A decision curve analysis (DCA) was used to assess the net benefit of HE4 measurement. 35 All statistical analyses were performed using R (version 4.0.5). A p-value less than 0.05 was defined as statistically significant.
Results
Characteristics of the participants
Figure 1 shows a flowchart of the participant. A total of 170 participants were recruited in the Hohhot cohort, and 152 were included in the data analysis, including 66 MPEs and 86 BPEs. A total of 62 participants were recruited in the Changshu cohort, and 58 participants were included in the data analysis, including 26 MPEs and 32 BPEs. The characteristics of the participants are shown in Table 1. The pleural biochemistries of the Hohhot cohort and Changshu cohort were different. For example, the medium protein concentration of BPE in the Hohhot cohort was 30 g/L, while it was 46 g/L in the Changshu cohort. This inconsistency may be due to the differences between the disease profile (e.g. the proportion of HF) and the medium age of the two cohorts.

A flowchart of the participant selection process.
Characteristics of the participants in the pleural fluid.

Continuous data with skewed distribution were expressed as medium and interquartile and compared with the Mann–Whitney U test. Continuous data with normal distribution were expressed as mean and standard deviation and compared with the student’s t-test. Categorical data were expressed as absolute numbers and percentages.
ADA, adenosine deaminase; BPE, benign pleural effusion; MPE, malignant pleural effusion; LDH, lactate dehydrogenase; WBC, white blood cell.
Diagnostic accuracy of pleural fluid HE4
Because the Hohhot and the Changshu cohorts are independent, the participants in these cohorts were analyzed separately. As shown in Figure 2, the median (interquartile range) HE4 in the MPEs and BPEs of the Hohhot cohort were 1166 (605–1500) pmol/L and 602 (349–928) pmol/L, respectively (p < 0.001). In the Changshu cohort, the median (interquartile range) HE4 in the MPEs and BPEs were 1429 (675–1500) pmol/L and 606 (300–778) pmol/L, respectively (p < 0.001). In the combined cohort, the median (interquartile range) HE4 in the MPEs and BPEs were 1171 (619–1500) pmol/L and 602 (323–901) pmol/L, respectively (p < 0.001).

Violin plot depicting the pleural fluid HE4 levels in MPE and BPE from the Hohhot cohort (a), the Changshu cohort (b), and the pooled cohorts (c).
Next, we analyzed the association between HE4 and the causes of pleural effusion. As shown in Figure 3, HE4 significantly differed across all causes of pleural effusion (p < 0.001) in the Hohhot cohort [Figure 3(a)], Changshu cohort [Figure 3(b)], and their combination [Figure 3(c)]. Notably, HF patients had the highest HE4 among BPE patients. Although MPE patients had higher HE4 than HF, no statistical significance was observed in both cohorts and their combination [Figures 3(a)–(c)]. In patients with MPE, although lung cancer had higher HE4 than other cancers, the difference was not statistically significant in the Hohhot cohort [p = 0.15; Figure 3(a)] and Changshu cohort [p = 0.21; Figure 3(b)]. We pooled the two cohorts to increase the statistical power. In the pooled cohorts, mean HE4 levels in lung cancer and other cancers were 1096 pmol/L and 879 pmol/L, respectively, with a marginal statistical significance [p = 0.06; Figure 3(c)].

HE4 and causes of MPE and BPE in the Hohhot cohort (a), the Changshu cohort (b), and the pooled cohorts (c).
Figure 4 shows the ROC curves of HE4 in the two cohorts. The area under ROC curves (AUCs) of HE4 in the Hohhot and Changshu cohorts were 0.73 (95% CI: 0.64–0.81, p < 0.001) and 0.79 (95% CI: 0.67–0.91, p < 0.001), respectively. A previous study showed that data-driven selection of the optimal thresholds for continuous index tests using the Youden index might overestimate sensitivity and specificity, and a prespecified threshold was advocated to avoid this bias. 36 Therefore, we prespecified the specificity of HE4 at 0.90. The threshold of HE4 at this specificity was 1300 pmol/L. With this threshold, we calculated the sensitivity (95% CI) and specificity (95% CI) of HE4 in the Hohhot cohort, Changshu cohort, pooled cohort, exudates, and patients with negative effusion cytology. The results are summarized in Table 2.
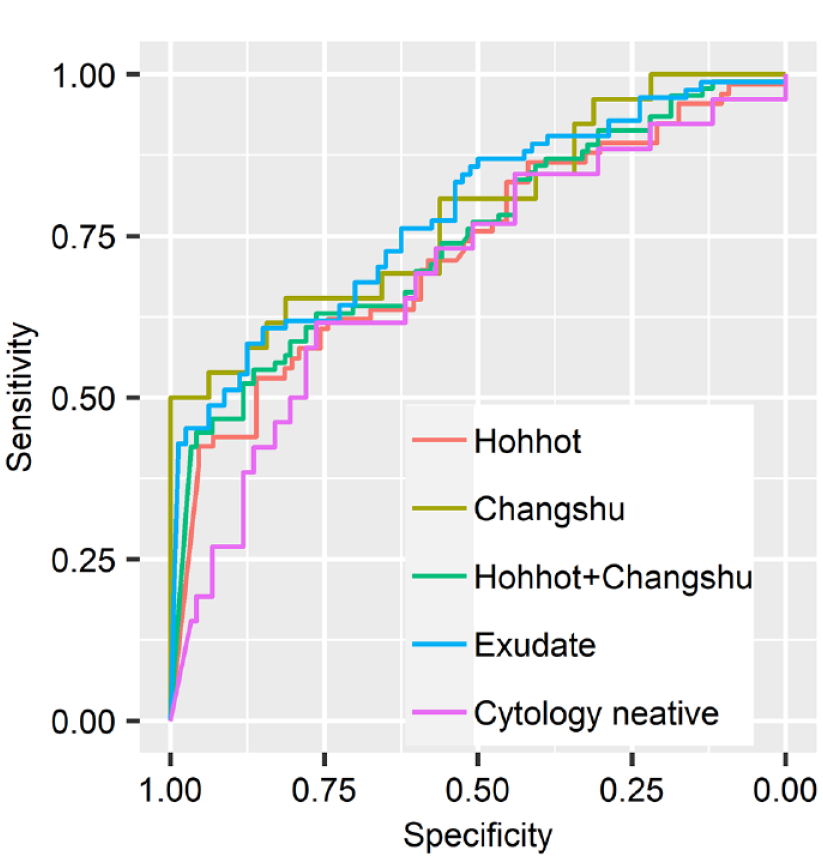
ROC curve of HE4 for MPE.
Diagnostic accuracy of HE4 for MPE at the threshold of 1300 pmol/L.

AUC, an area under curve; CI, confidence interval; HE4, human epididymis secretory protein 4; MPE, malignant pleural effusion.
Next, we used a DCA to estimate the net benefit of HE4 determination.35,37 In this study, net benefit represents the proportion of patients that correctly classified as MPE (true positive) minus the proportion of patients misclassified as MPE (false positive). 38 The DCA of HE4 in the two cohorts is shown in Figure 5. The decision curves of HE4 were above the reference lines, indicating that HE4 determination has net benefits in patients with undiagnosed pleural effusion.

A decision curve of HE4 for MPE. The net benefit of HE4 was assessed in participants from the Hohhot cohort (a), the Changshu cohort (b), and the pooled cohorts (c), with exudates (d) and with negative or unknown cytology (e).
Discussion
In this study, we evaluated the diagnostic accuracy of pleural fluid HE4 for MPE. We found that the median HE4 in MPE patients was significantly higher than in BPE patients. The AUC of HE4 for MPE was between 0.70 and 0.80. In BPE patients, HF patients had the highest HE4. In patients with exudates or negative cytology, HE4 also showed moderate diagnostic accuracy. These results suggest that pleural fluid HE4 has moderate diagnostic accuracy for MPE, and its measurement in patients with pleural effusion has a potential net benefit.
To our knowledge, three studies have evaluated the diagnostic accuracy of HE4 for MPE.39–41 Compared with these studies, our study has some strengths. First, we investigated the diagnostic accuracy of HE4 with two independent cohorts (n = 210), and similar results were observed in both cohorts, indicating our findings were robust. Second, we evaluated the net benefit of HE4 measurement by DCA and found that HE4 had a potential net benefit. By contrast, previous studies did not evaluate the net benefit of HE4 measurement. Third, we analyzed the association between HE4 and the causes of BPE and found that HF patients had the highest HE4 among BPEs. This finding was supported by a previous study which revealed that increased serum HE4 positively correlated with the New York Heart Association grade in HF patients. 29 In addition, increased serum HE4 is associated with a poor prognosis of HF. 42 We hypothesized that the increased serum HE4 could passively diffuse into the pleural cavity and lead to high pleural fluid HE4 in HF patients. This finding implies that increased HE4 is suggestive of HF. Therefore, HF should be considered when interpreting HE4. Because MPE patients did not show significantly higher HE4 than HF patients, it is reasonable to speculate that the proportion of HF patients may affect the diagnostic accuracy of HE4 for MPE. In other words, the diagnostic accuracy of HE4 may be impaired in clinical settings with a high prevalence of HF in undiagnosed pleural effusion. However, more than 70% of HF patients were transudate and can be verified by the Light’s criteria. After excluding transudate patients from our cohort, we found that HE4 had an AUC of 0.78, slightly higher than the AUC of the combined cohort (0.75). This result indicates that the prevalence of HF patients only slightly affected the diagnostic accuracy of HE4.
HE4 has an AUC of approximately 0.75, lower than that reported in previous studies.39–41 There are several possible explanations. First, previous studies only included lung cancer patients in MPE.40,41 Our study found that the mean HE4 in lung cancer (1096 pmol/L) was higher than in other cancers (879 pmol/L). The difference in HE4 was marginally significant (p = 0.06), which may be due to the small sample size of our study. This result implies that the studies that only included lung cancer in MPE may overestimate the diagnostic accuracy of HE4. Second, the median age of participants in our cohorts was all over 70 years, but the average ages in previous studies were between 60 and 65 years.39,40 Previous studies revealed that serum HE4 was affected by age.43–45 Therefore, the age of the participants may partially explain the difference in diagnostic accuracy between our study and previous studies.
Many studies have investigated the diagnostic accuracy of pleural fluid tumor markers for MPE. The specificities of these tumor markers are around 0.90, and the sensitivities are between 0.40 and 0.50.15,16,46 To compare the diagnostic accuracy of HE4 and conventional tumor markers, we set the specificity of HE4 at 0.90 and found that its sensitivities were 0.44 and 0.54 in the two cohorts. This result indicates that the diagnostic accuracy of HE4 and the conventional tumor markers (e.g. CEA, NSE, CA125) was comparable. Although the sensitivity of HE4 was only 47%, close to that of effusion cytology, it showed moderate diagnostic curacy for MPE in patients with negative cytology. Therefore, the combination of cytology and HE4 can be used simultaneously to improve the diagnostic accuracy of MPE. Notably, the threshold with a specificity of 1.00 has a particular significance because MPE can be confirmed without invasive procedures such as pleural biopsy or thoracoscopy. A previous study indicated that CEA > 45 ng/mL or CA 15-3 > 77 IU/mL had a specificity of 1.00 for MPE. 47 However, the specificity of HE4 in our study did not reach 1.00 even though the threshold was set at 1500 pmol/L, the upper limit of the assay. This result indicates that HE4 can only increase the post-test probability of MPE, and even extremely high HE4 cannot confirm the presence of MPE. HE4 value should be interpreted in parallel with the clinical characteristics of patients.
Our study has some limitations. First, sample size estimation was not performed before participant recruitment because this is a data-driven rather than a hypothesis-driven study. The sample size of the Changshu cohort was small, which may explain the lack of association between causes of pleural effusion and HE4 in the Changshu cohort. Second, we used the stored pleural fluid specimen to test HE4. It is unclear whether HE4 is stable at −80°C to −70°C for approximately 3 years. Third, due to the limited sample volume, we did not dilute the specimens with HE4 > 1500 pmol/L, which may affect the findings of our study. Fourth, the prognostic value of HE4 in MPE was not investigated in our study because the MPE patients’ outcomes were unknown. Nevertheless, this study provided new insight into the diagnostic accuracy and net benefit of pleural fluid HE4 for MPE.
Conclusion
In summary, pleural fluid HE4 has moderate diagnostic accuracy for MPE. HE4 also has potential net benefit in pleural effusion patients with unknown causes. Considering the limitations of our study, further studies are needed to validate our findings in the future.
Supplemental Material
sj-docx-1-tar-10.1177_17534666231216566 – Supplemental material for Value of human epididymis secretory protein 4 in differentiating malignant from benign pleural effusion: an analysis of two cohorts
Supplemental material, sj-docx-1-tar-10.1177_17534666231216566 for Value of human epididymis secretory protein 4 in differentiating malignant from benign pleural effusion: an analysis of two cohorts by Qian Yang, Yan Niu, Jian-Xun Wen, Dan-Ni Yang, Yu-Ling Han, Xu-Hui Wen, Li Yan, Jin-Hong Huang, Hong Chen, Wen-Qi Zheng, Ting-Wang Jiang and Zhi-De Hu in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
