Abstract
Introduction:
The differential diagnosis of pleural effusion is difficult, and studies have reported on the potential role of adenosine deaminase (ADA) in the differential diagnosis of undiagnosed pleural effusion. This retrospective study aimed to investigate the diagnostic role of ADA in pleural effusion.
Methods:
266 patients with pleural effusion from three centers were enrolled. The concentrations of ADA and lactate dehydrogenase (LDH) were measured in pleural fluids and serum samples of the patients. The diagnostic performance of ADA-based measurement for tuberculous pleural effusion (TPE), malignant pleural effusion (MPE), and parapneumonic effusion (PPE) was examined by receiver operating characteristic (ROC) curve analysis.
Results:
An area under the ROC curve (AUC) value of 0.909 was obtained using the pleural ADA values as the indicator for TPE identification (sensitivity: 87.50%, specificity: 87.82%). The ratio of serum LDH to pleural ADA (cancer ratio) provided the predictive capacity with an AUC of 0.879 for MPE diagnosis (sensitivity: 95.04%, specificity: 67.06%). At a cut-off value of 14.29, the pleural ADA/LDH ratio showed a sensitivity and specificity of 81.13% and 83.67%, respectively, and a high AUC value of 0.888 for the differential diagnosis of PPE from TPE.
Conclusion:
ADA-based measurement is helpful for the differential diagnosis of pleural effusion. Further studies should be performed to validate these results.
Keywords
Introduction
Pleural effusion is a commonly clinic manifestation associated with more than 50 recognized diseases and disorders. In the clinic, malignant pleural effusion (MPE), tuberculous pleural effusion (TPE), and parapneumonic effusion (PPE) are the most likely causes of exudative effusions. 1 Approximately one-third of patients with tuberculosis develop extra-pulmonary tuberculosis, while a quarter of them have TPE. 2 However, traditional microbiology and molecular biology methods show poor performance when pleural fluids are used to diagnose TPE, especially in an acute setting.3–5 MPE is distinct from TPE and PPE in that it has worse prognosis and a median survival time of only 3–12 months. 6 When pleural biopsy is used, cytological examination has a low rate of MPE detection because of the poor preservation of tumor cells and small sample volume.7,8 Thoracoscopic biopsy is an efficient diagnostic method for both TPE and MPE, but its invasiveness limits clinical application.9–11 Furthermore, the performances of tumor biomarkers such as vascular endothelial growth factor, carcinoembryonic antigen (CEA), carbohydrate antigen (CA) 125, CA 15-3, CA 19-9, and CYFRA 21-1 (a fragment of cytokeratin 19) in the diagnosis of MPE are also limited.12–14 Therefore, it is important to identify invasive and effective methods to differentiate subtypes of exudative effusions.
Adenosine deaminase (ADA), an enzyme secreted by mononuclear cells, lymphocytes, neutrophils, and red blood cells (RBCs), 15 plays an important role in purine nucleoside metabolism15,16 and is related to intracellular infection such as tuberculosis. An increased total pleural fluid ADA level usually helps to discriminate TPE from PPE.17,18 Although a meta-analysis revealed that ADA can diagnose TPE with a high sensitivity and specificity of 92% and 90%, respectively, 17 a similar or even higher level of total pleural fluid ADA has occasionally been reported in PPE, especially in patients with pyothorax,15,19,20 which may be due to the high pleural level of ADA in the case of empyema.21,22 Thus, it remains a challenge to distinguish TPE from PPE on the basis of elevated pleural fluid ADA. In addition, a decreased level of ADA was reported in patients with MPE, 15 but it remains unclear if pleural fluid ADA alone can help to diagnose MPE.
Lactate dehydrogenase (LDH) is a ubiquitous enzyme present in a variety of tissues including liver, kidney, myocardium, skeletal muscles, and RBCs.
23
Verma
Patients and methods
Study design and subjects
This was a retrospective, multi-center study conducted across West China Hospital (Cohort 1, between June 2020 and January 2021), Chengdu Fifth People’s Hospital (Cohort 2, between June 2020 and December 2020), and Hospital of Chengdu University of Traditional Chinese Medicine (Cohort 3, between January 2018 and December 2020). The study participants were patients aged >18 years with accumulation of pleural fluid through chest ultrasonic examinations. Those who had inconclusive final diagnosis, incomplete data, a coexisting systemic disease, immunodeficiency, autoimmune disease, or hemothorax were excluded from this study. This study was designed according to the requirements of the Chinese Guidelines for Good Clinical Practice and was approved by the Ethics Committee of West China Hospital. The need for written informed consent was waived given the retrospective nature of the study.
Diagnostic criteria and data extraction
Based on previous study,
19
this study only included patients with exudates and focused on patients with MPE, TPE, and PPE. Further, the diagnostic criterion for MPE was based on the presence of malignant cells in pleural effusion or pleural biopsy specimens.
7
The inclusion criteria for patients with TPE were chronic granulomas in the pleural tissue, clinical response to anti-tuberculosis treatment, or acid-fast bacteria found in pleural fluid or sputum. PPE was identified as exudative effusions associated with bacterial pneumonia, lung abscesses, or bronchiectasis, absence of
Sample collection and quantification of ADA and LDH levels
A volume of 3 ml of venous blood was collected and centrifuged at 4000 r/min for 10 min to separate serum. The pleural fluid samples were collected via thoracentesis and centrifuged at 3000 g for 10 min. The levels of pleural ADA, LDH, protein, glucose and serum LDH, albumin were measured on the automated chemistry analyzer in clinical laboratory. The operation was performed in strict accordance with the SOP (standard operating procedure). All experiments were performed in accordance with the manufacturers’ protocols.
Statistical analysis
After a normality test, the data were summarized as the mean and standard deviation or median and interquartile range. When comparing two groups, an independent samples
Results
Baseline characteristics of patients
Data for a total of 266 patients suffering from pleural effusion were collected and reviewed. There were 102, 109, and 55 patients enrolled in Cohorts 1, 2, and 3, respectively. Of the patients with benign pleural effusion (BPE), 58 and 62 had TPE and PPE, respectively. The mean ages of the MPE and BPE groups were 69.00(60.00−76.00) and 64.00(46.25−75.75) years, respectively (
Pleural levels of ADA and LDH and serum LDH levels
The clinical and laboratory findings of patients with MPE and BPE are summarized in Table 1. Compared with the BPE group, the MPE group had a significantly higher serum LDH ((217.50(179.50−299.00)
Pleural fluid parameters of patients with MPE, TPE, or PPE in three cohorts.

ADA, adenosine deaminase; Cancer ratio, serum LDH/ pleural ADA; MPE, malignant pleural effusion; LDH, lactate dehydrogenase; PPE, parapneumonic pleural effusion; TPE, tuberculous pleural effusion.
Data are presented as the mean ± standard deviation or as the median (interquartile range). Statistical significance was set at
Patients with TPE had a significantly elevated level of pleural ADA ((35.00(21.00−47.00)
Diagnostic accuracy of pleural ADA for TPE
The ability of pleural fluid ADA level to diagnose TPE from other causes of pleural effusion was explored by ROC curve analysis in Figure 1 and Table 2. In Cohort 2, the pleural fluid ADA level provided an AUC of 0.970, along with a sensitivity of 100.00%, a specificity of 90.59% and a PLR value of 10.63 at a cut-off value of 18.50 U/L to differentiate patients with TPE (Table 2, Figure 1(b)). At a cut-off value of 16.50 U/L, pleural fluid ADA level had an AUC value of 0.909, along with a sensitivity value of 87.50%, a specificity value of 87.82%, a PLR value of 7.18 and a NLR value of 0.14 in all subjects (Table 2, Figure 1(d)). It was found that pleural protein (OR = 1.029, 95% CI: 1.005–1.053) and pleural ADA (OR = 1.115, 95% CI: 1.083–1.148) were significantly associated with the diagnosis of patients with TPE (all

Receiver operating characteristic curve for pleural ADA to diagnose patients with TPE. Receiver operating characteristic curves showing the performances of the pleural ADA level for diagnosing TPE in cohort 1 (a), cohort 2 (b), cohort 3 (c), and all subjects (d). ADA, adenosine deaminase; TPE, tuberculous pleural effusion.
The cut-off values, sensitivity, specificity, and AUC of the pleural ADA distinguishing TPE, cancer ratio detecting MPE, and pleural LDH/ADA ratio differentiating PPE from TPE.
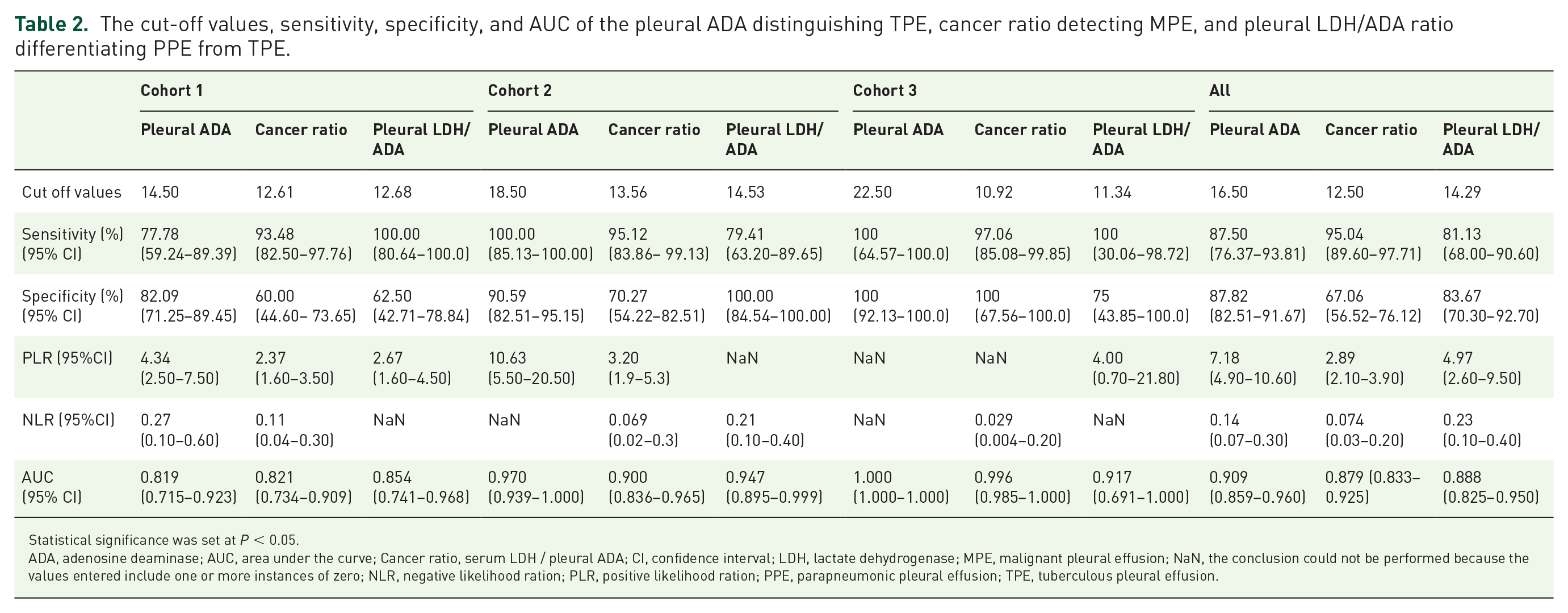
Statistical significance was set at
ADA, adenosine deaminase; AUC, area under the curve; Cancer ratio, serum LDH / pleural ADA; CI, confidence interval; LDH, lactate dehydrogenase; MPE, malignant pleural effusion; NaN, the conclusion could not be performed because the values entered include one or more instances of zero; NLR, negative likelihood ration; PLR, positive likelihood ration; PPE, parapneumonic pleural effusion; TPE, tuberculous pleural effusion.
CR for the diagnosis of MPE
ROC curves were created to analyze the diagnostic performance of CR as a marker for MPE from BPE in Figure 2 and Table 2. In Cohort 2, the CR provided an AUC of 0.900, along with a sensitivity of 95.12%, a specificity of 70.27%, a PLR of 3.20 and a NLR of 0.069 at a cut-off value of 13.56 to differentiate patients with MPE (Table 2, Figure 2(b)). In Figure 2(d), CR had an AUC of 0.879 at a cut-off value of 12.50, along with a sensitivity value of 95.04%, a specificity value of 67.06%, a PLR of 2.89 and a NLR of 0.074 in all subjects (Table 2, Figure 2(d)). Univariate analysis found that age (>65 years, OR = 0.490, 95% CI: 0.300–0.800), smoking status (smoker, OR = 0.597, 95% CI: 0.358–0.995), serum CA199(OR = 0.986, 95% CI: 0.974–0.998), serum CA153(OR = 0.974, 95% CI: 0.956–0.993), serum NSE (OR = 0.967, 95% CI: 0.937–0.997), serum CYFRA211 (OR = 0.959, 95% CI: 0.924–0.995) and cancer ratio (OR = 0.871,95% CI: 0.936–0.908) were associated with the diagnosis of patients with MPE (all

Receiver operating characteristic curve for cancer ratio to distinguish patients with MPE from BPE. Receiver operating characteristic curves showing the performances of the cancer ratio as the indicator for MPE identification in cohort 1 (a), cohort 2 (b), cohort 3 (c), and all subjects (d). BPE, benign pleural effusion; MPE, malignant pleural effusion.
Pleural fluid LDH/ADA ratio for differentiating PPE from TPE
Next, we evaluated the diagnostic performances of pleural fluid LDH/ADA ratios in identifying PPE from TPE in Figure 3 and Table 2. In Cohort 2, with an AUC of 0.947, the pleural fluid LDH/ADA ratio diagnosed PPE with a sensitivity of 79.41%, a specificity of 100.00% and a NLR of 0.21 at a cut-off value of 14.53 (Table 2, Figure 3(b)). In all subjects, the AUC for distinguishing PPE from TPE was 0.888 at a cut-off value of 14.29, with a sensitivity of 81.13%, a specificity of 83.67%, a PLR of 4.97 and a NLR of 0.23 (Table 2, Figure 3(d)). It was revealed that pleural protein (OR = 0.950, 95% CI: 0.920–0.982) and pleural LDH/ADA ratio (OR = 1.238, 95% CI: 1.133–1.354) were associated with the diagnosis of patients with PPE from TPE (all
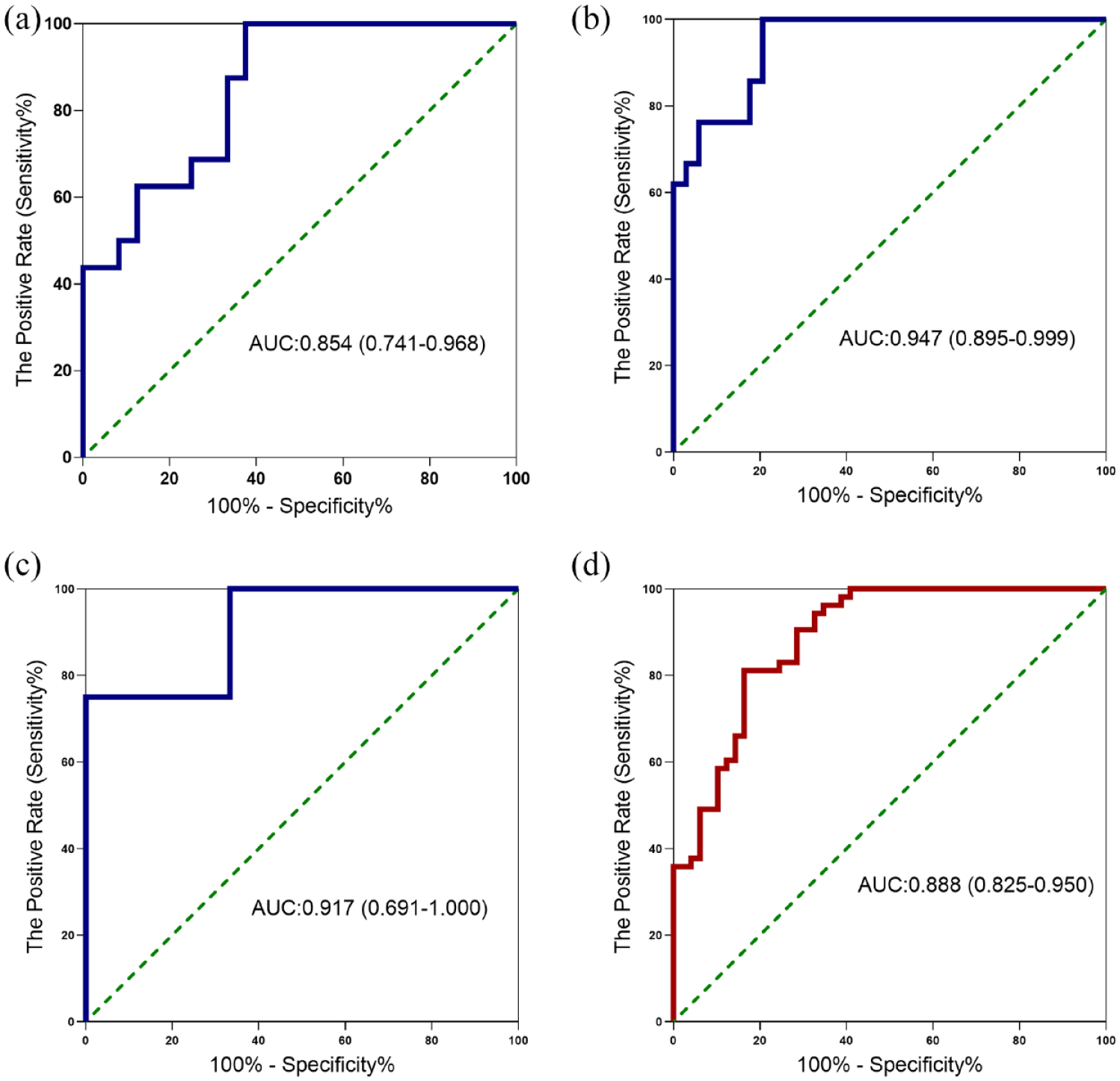
Receiver operating characteristic curve for pleural fluid LDH/ADA ratio to distinguish patients with PPE from TPE. Receiver operating characteristic curves showing the performances of the pleural fluid LDH/ADA ratio as the indicator for PPE differentiation from TPE in cohort 1 (a), cohort 2 (b), cohort 3 (c), and all subjects (d). ADA, adenosine deaminase; LDH, lactate dehydrogenase; PPE, parapneumonic effusion; TPE, tuberculous pleural effusion.
Discussion
It is necessary to identify noninvasive, quick, and effective biomarkers to differentiate multiple types of pleural fluid. For patients with undiagnosed pleural effusion, the conventional blood and pleural biochemical tests regarding pleural fluid ADA and LDH levels are usually performed without any additional costs. We conducted a retrospective study in multiple centers and aimed to explore the diagnostic performance of pleural ADA-based measurements in the differential diagnosis of pleural effusion. Overall, our results showed that the pleural fluid ADA can be an effective marker for the diagnosis of TPE with an AUC of 0.909. Moreover, CR is a useful biomarker in predicting MPE with an AUC of 0.879, and the pleural fluid LDH/ADA ratio is also meaningful to distinguish PPE and TPE, providing an AUC of 0.888, suggesting that pleural ADA plays a critical role in the diagnosis of TPE, MPE, and PPE.
Some investigations during the past decade have addressed the potential role of pleural ADA level in the differential diagnosis of TPE with high sensitivity and specificity.17,27 However, inconsistent data were reported by Zaric
Although a higher pleural ADA level in pleural fluid strongly suggests TPE, PPE may also lead to a relatively high pleural ADA level (~40 U/L),15,29 and an extremely high ADA level is associated with a greater risk of empyema or lymphoma. 23 Conversely, pleural fluid LDH level might be a specific diagnostic indicator for empyema, 29 suggesting that pleural ADA or LDH level alone is limited in its ability to differentiate between clinical TPE and PPE. Thus, the two parameters were combined as a predictor of PPE, and the pleural fluid LDH/ADA ratio provided a sensitivity of 81.13% and a specificity of 83.67% yielded an AUC of 0.888. The reason why performance of pleural LDH/ADA ratio was inferior to that of previous studies might be due to lack of consideration of pleural fluid cellular predominance. However, the cut-off value of pleural fluid ADA for TPE diagnosis was controversial, further studies are required to validate these findings.
CR has been proposed as a predictor of MPE, and this ratio was found to be significantly higher in patients with MPE than in those with TPE or PPE. 24 The results of our study are consistent with previous research, 30 reporting an AUC of 0.879, along with high sensitivity of 95.04% and a relatively low specificity of 67.06%, suggesting that CR can be a sensitive biomarker of MPE. However, the specificity (67.06%) is lower than that reported in previous studies,24,31 because less patients with lung cancer (90.41%) were enrolled in our study (95% and 97.6%).24,31 The different subtypes of MPE should be considered in the clinical interpretation of CR results, given that LDH levels may be influenced by different types of tumors. Obviously, the relatively low specificity limited the diagnostic value of CR. The high sensitivity indicates that clinicians should exercise caution when patients have high CR value and conduct further examinations such as repeated cytologic test and invasive procedures such as medical thoracoscopy and pleural biopsy if needed. Although the optimal cut-off value of CR has not yet been determined, a prespecified threshold value is needed because a data-driven threshold can overstate indicator tests. 32
Among the three cohorts, the performance for ADA-based measurements of Cohort 3 was better than those of Cohorts 1 and 2. The main reason may be that there were only seven patients with TPE and five with PPE in Cohort 3, much less than the number of patients with TPE or PPE in Cohorts 1 and 2. It has been reported that the sample size can influence the diagnostic accuracy of the ROC curve. 33 Therefore, the pooled analysis of all three cohorts was representative when assessing the diagnostic performance of ADA-based measurements in differentiating MPF/TPE.
This study has some limitations. First, we conducted these analyses in a retrospective manner. The data obtained from the hospital’s medical records were limited and we were unable to further analyze the differences between the PPE subgroups. Second, the patients suffering from pleural effusions enrolled in our study were not representative of other diseases that could lead to pleural effusion, such as connective tissue diseases, 34 which may also have an increased pleural fluid ADA or LDH level.
Conclusion
Overall, our study has provided evidence that pleural fluid ADA-based measurements are effective biomarkers to discriminate multiple types of pleural effusion. Consequently, it may assist in the early diagnosis and treatment for patients with pleural effusion.
Supplemental Material
sj-docx-1-tar-10.1177_17534666231155747 – Supplemental material for Adenosine deaminase-based measurement in the differential diagnosis of pleural effusion: a multicenter retrospective study
Supplemental material, sj-docx-1-tar-10.1177_17534666231155747 for Adenosine deaminase-based measurement in the differential diagnosis of pleural effusion: a multicenter retrospective study by Lijuan Gao, Wujun Wang, Ying Zhang, Xueru Hu, Jing An, Yang Li, Mei Chen and Yongchun Shen in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
