Abstract
Background:
Complex pleural effusion and empyema (CPPE) is treated with intrapleural fibrinolytic therapy (IPFT) using tissue plasminogen activator (tPA) and deoxyribonuclease (DNase) We present our single-center retrospective observational data using a simplified regimen of coadministering six divided doses of tPA and DNase over the course of 3 days.
Objective:
To study the safety, utility, and clinical outcomes of IPFT.
Design:
This is a single-center retrospective study of patients who received co-administration tPA/DNase for CPPE over a period of 5 years. The primary outcome was successful treatment without the need for surgery. Secondary outcomes were bleeding risk, post-procedural pain, treatment complications, and all-cause mortality at 30 days and 6 months. We have tested the clinical role RAPID score (Renal function measured as urea, Age, fluid Purulence, Infection source, Dietary status measured as albumin) to predict treatment success, and all-cause mortality at 6 months.
Results:
A total of (n = 55) patients were included in the study. The mean age of the population studied was 67 (Interquartile range 57–74), including 47.3% male and 52.7% Female. 92.7% of the population studied was Caucasian. Comorbidities including chronic obstructive pulmonary disease, congestive heart failure, and Diabetes mellitus were present in 41.8%, 41.8%, and 43.6.% respectively. Patients were treated with tube thoracostomy with 14 French percutaneous pigtail catheters in 47 (85.5%) or 28–32 French chest tubes in 8 (14.5%) patients. Twenty-nine percent (16) of patients had acceptable clinical and radiographic improvement and did not require additional surgical or radiological intervention. Seventy-one percent (39) of patients required additional surgical drainage; video-assisted thoracoscopic surgery in 37, and open thoracotomy in 2 patients. The discriminating ability of the RAPID score for treatment success after IPFT was found to be poor (AUC: 0.601, 95% CI: 0.429–0.773, p = 0.24). All-cause mortality at 6 months was 23.6% (13) of patients. The predictive ability of the RAPID score for mortality at 6 months was found to be poor (AUC: 0.640, 95% CI: 0.478–0.802, p = 0.13). The optimal cutoff for the RAPID score for mortality was ⩾4, with 84.6% sensitivity and 46.3% specificity.
Conclusion:
Results of our single-center study suggest that IPFT can be safely adopted by small and mid-sized clinical centers, as the risk of bleeding is low. The results of coadministering tPA and DNase are safe, and it reduces the need for surgical intervention in nearly one-third of patients.
Plain language summary
This study evaluated the use of fibrinolytic medications—commonly known as clot busters—to treat infections in the pleural space. It found that surgical intervention could be avoided in approximately one-third of patients. The treatment was shown to be safe and feasible for implementation in small to mid-sized clinical centers.
Introduction
Complex pleural effusions and empyema (CPEE) complicate approximately 5%–20% of cases of pneumonia.1 –3 In addition to early systemic antibiotic therapy, drainage of the infected pleural fluid is recommended. 4 Studies have shown that 15%–40% of patients with CPEE ultimately require surgical drainage of the pleural space.1,5,6 Administration of intrapleural fibrinolytic therapy (IPFT) to lyse fibrinous septations forming loculations within the pleural space has shown a reduction in the need for surgical intervention, lowering the morbidity and associated cost of care. Although the larger MIST-1 trial utilizing intrapleural streptokinase showed no difference in outcomes, the use of intrapleural tissue plasminogen activator (tPA) and Deoxyribonuclease (DNase) in the MIST-2 trial showed a reduced rate of surgical referrals and length of hospital stay in patients with pleural infections.7,8 A few observational reports of higher incidence of bleeding with the use of intrapleural tPA and DNase have limited their use at smaller clinical centers. The rates of intrapleural hemorrhage have been reported to be between 1.8% and 12%5,9,10 in observational studies and 6% in the tPA-DNase combination group in the MIST-2 trial. 8 The RAPID score (Renal function measured as urea, Age, fluid Purulence, Infection source, Dietary status measured as albumin) was initially derived from the cohort of patients in the MIST-1 trial to stratify patients with infected pleural effusions based on severity to support decision making regarding treatment strategies for infected pleural effusions. 11 However, the score is yet to be validated in larger trials and real-world data to be incorporated into routine practice. MIST-2 trial described the administration of 12 serial doses of tissue plasminogen activator (tPA) and deoxyribonuclease (DNase) separately. However, this regimen is labor intensive and requires trained personnel to open and close the chest tube circuit eight times a day for 3 days. We present our observational data using a simplified regimen of coadministering tPA and DNase. A simplified regimen of coadministering tPA-DNase has been previously described in the literature.12,13 We investigated clinical outcomes, adverse effects including pain and intrapleural bleeding, and the utility of the RAPID score in predicting treatment success rates with IPFT, and all-cause mortality at 6 months.
Methods
This is a single-center retrospective observational study of patients who were treated with intrapleural tPA (Alteplase) and DNase co-administered for complex pleural effusion and pleural space infection from January 2018 to November 2022 at the Medical Center of Bowling Green, Kentucky. Informed consent was waived due to the retrospective nature of the study. This study was approved by the Institutional Review Board (IRB ID: 22-8-18-Sedh-IntraPleur-tPA). The study was registered on ResearchRegistry.com. We have abided by the STROBE checklist. 14 A copy of the STROBE checklist is included in the Supplemental Material. All the medical records were deidentified before the analysis. Electronic medical records were reviewed, and data pertaining to demographics, clinical presentation, pleural fluid characteristics, laboratory analysis, chest tube size, timing of administration, total duration of treatment, and clinical outcomes of interest were abstracted by physician abstractors.
Study cohort
Patients were identified using a search strategy of the pharmacy database of tPA and DNase recipients. Only the patients who received intrapleural tPA and DNase for the treatment of CPEE were included in the study. CPEE was defined based on the American Association of Thoracic Surgeons (AATS) consensus guidelines as pleural fluid with any of the following: (1) Exudative and loculated in a patient with community or hospital-acquired pneumonia, (2) Gram stain or culture positive, (3) Macroscopically purulent. 15 Pleural fluid pH is not readily measured at our institution and is not used as a criterion for chest tube insertion. Exclusion criteria were pleural space infections as a complication of a procedure or viscous perforation or age below 18 years. Patients were considered eligible for IPFT if they met one of the three AATS criteria for CPEE. However, the decision to administer IPFT was at the discretion of the treating physician and clinical team.
Intervention details
All the patients underwent radiologic evaluation of the thoracic cavity either with chest x-ray, computed tomography, and or ultrasonography. A percutaneous 14 Fr pigtail catheter or a 28–32 Fr chest tube was inserted at the discretion of the treating physician. The method of the 14 Fr pigtail catheter is described in the illustration in Figure 1. IPFT comprised a total of six divided doses of tPA and DNase, separated by 12 h for 3 days. Each dose included 10 mg of tPA and 5 mg of DNase. Medicine was administered into the intrapleural space through the chest tube. Each medicine was separately prepared by diluting in 50 mL of normal saline. In each dose, tPA was administered first, immediately followed by DNase. After administering both medicines, chest tubes were flushed with 60 mL of normal saline and clamped for 1 h. Routine chest tube flushes were not performed. The patient continued to receive concomitant medical therapy, including antibiotics. The type and duration of antibiotic therapy were at the discretion of the treating team of clinicians.

Illustration of ultrasound-guided percutaneous pigtail catheter insertion in the thoracic cavity and intrapleural fibrinolytic therapy description.
Clinical and radiological evaluation after initiating IPFT
Patients underwent daily clinical and radiological evaluations following the initiation of IPFT. The clinical team monitored the volume of fluid drained via the chest tube, along with relevant clinical and laboratory parameters. A daily chest radiograph was performed to assess the progression of pleural effusion resolution. In cases where there was insufficient resolution of pleural effusion or inadequate lung expansion, a computed tomography (CT) scan of the chest was conducted. CT imaging also aided in identifying pleural rind and loculated pockets when present. In addition, a point-of-care ultrasound (POCUS) of the chest was utilized daily as part of the physical examination.
Outcome of interest
We defined treatment success as clinical and radiological improvement of pleural space infection without the need for a formal surgical intervention, including but not limited to video-assisted thoracoscopic surgery (VATS) or a formal thoracotomy. Patients who could not achieve clinical or radiological improvement and who required surgical intervention were deemed as treatment failures. The designation of clinical improvement was determined at the discretion of the treating clinician.
Secondary outcomes of interest were the need for intravenous opiates during the first 48 h, pleural space bleeding, and adverse effects including hemoptysis, nausea, and vomiting. Other secondary outcomes of interest were the need for escalation of care to the ICU, duration of chest tube from insertion to removal, and length of hospitalization. Patients were followed up after discharge for 6 months. We analyzed all-cause mortality within 30 days and at 6 months. We calculated the utility of the RAPID score to predict treatment success and predict mortality at 6 months using receiver operating characteristics (ROC) curves.11,16,17
Statistical analysis
Clinical data points of interest were collected using a Research Electronic Data Capture (REDCap). Analysis was performed using SAS version 9.4 (SAS Inc., Cary, NC, USA). Continuous variables were expressed as median and interquartile range (IQR). Categorical data were presented as absolute values and percentages. Clinical and demographic parameters were compared in patients who had treatment success and failure between two arms. All-cause mortality was calculated at 30 days and 6 months and patients were classified into two groups based on mortality versus survival at 6 months. A comparison of categorical variables was done with Fisher’s exact tests. Differences in continuous variables expressed as median (IQR) between groups were tested using the Mann–Whitney U test. All hypothesis testing was 2-tailed, and p < 0.05 was considered statistically significant.
Results
Demographic summary
We identified a total of 55 patients who received intrapleural tPA and DNase for complex pleural effusion and empyema from January 2018 to November 2022. The mean age of the population studied was 67 years (IQR 57–74 years). There was even gender distribution. More than 90% of patients included in the study were white, and there was an under-representation of black and ethnic minorities. 27.2% of the patients in the study cohort were obese. A detailed demographic summary of the populations studied is summarized in Table 1.
Clinical and demographic characteristics of the patients who received tPA and DNase for complex pleural effusion and empyema.

CKD, Chronic kidney disease; COPD, Chronic obstructive pulmonary disease; DNase, deoxyribonuclease; eGFR, Estimated glomerular filtration rate; ESRD, End-stage renal disease; GERD, Gastroesophageal reflux disease; HFeEF, Heart failure with reduced ejection fraction; IQR, interquartile range; LVEF, Left ventricular ejection fraction; tPA, tissue plasminogen activator.
Clinical outcomes
Treatment success was defined as clinical and radiological recovery without needing surgical intervention, it was achieved in 16 (29.1%) of patients. Treatment failure was defined as the inability to achieve complete resolution of the pleural effusion, the development of a pleural rind, or failure of lung expansion, necessitating surgical intervention. Thirty-nine (70.9%) patients required were treated with additional surgical intervention; VATS in 37 (67.28%) patients and open thoracotomy in 2 (3.64%) patients. Demographic and clinical factors between the treatment success and failure groups were compared using Fisher’s exact test or Mann–Whitney U test. There was no statistically significant difference in the demographic characteristics and comorbidities between the two groups. Median RAPID score was 4 (IQR 2–5) in the treatment success group compared to the median RAPID score of 4 (IQR 3–5) in the treatment failure group (p = 0.23). Clinical outcomes of interest are summarized in Table 2.
Summary of clinical outcomes.

IQR, interquartile range; VATS, video-assisted thoracoscopic surgery.
Adverse effects
Observed post-procedural complications included pain; 16 (29.1%) patients required at least one dose of intravenous opiate analgesics during the first 48 h after initiation of IPFT. Six (10.9%) patients required an additional six doses of intravenous opiate analgesics during the first 48 hours. This suggests that IPFT is a painful intervention likely attributable to the enzyme-induced irritation of the inflamed pleura, given that the pleura has extensive neural innervation. Bleeding in the pleural space leading to a drop in hemoglobin by 2 gm/dL or requiring transfusion of packed red blood cells was seen in 8 (14.5%) of patients. There was no significant difference in the incidence of bleeding between patients with and without chronic anticoagulation (20.0% vs 13.6%, p = 0.63).
Secondary outcomes
Half of the study cohort had an escalation of medical care to the intensive care unit. However, upon review of patient-level data, none of these were secondary to the complications related to the intervention. Median duration of chest tube insertion was 8 days (IQR 4–12 days); range 2–59 days. Median duration of hospitalization was 15 days (IQR 10–19 days).
Protocol and deviation
The hospital protocol was adopted from the MIST 2 trial with subtle modifications 8 45 (81%) patients received a total of six doses of IPFT as described by the MIST 2 trial. Three (5.4%) patients received less than six doses, ranging from 2 to 5 doses, deviating from the protocol. Seven (12.72%) patients required more than six doses of IPFT. All seven patients who received higher than the recommended six doses of IPFT required surgical intervention. IPFT was administered with 14Fr percutaneous pigtail catheters (PCs) in 47 (85.5%) and 28–32Fr surgical chest tubes in 8 (14.5%) patients. Insertion of PCs and surgical chest tubes was performed by pulmonary disease and critical care medicine fellows in training.
Mortality
A total of 3 (5%) patients died within the first 30 days. We performed the causes and characteristics of death analysis of patients who died within 30 days. We assessed for sepsis and dysfunction of 8 organ systems during the 72 h prior to death as described by Stapleton et al.18,19 Primary cause of death in the patients who died within 30 days was sepsis. Ten (18.18%) patients died during the follow-up period between one and 6 months, accounting for an all-cause mortality at 6 months of 13 (23.63%). We compared the demographic and clinical characteristics of those who died within 6 months, the results are summarized in Tables 3 and 4.
Characteristics of treatment outcome and results of chi-squared test.

DM, Diabetes mellitus; IPFT, intrapleural fibrinolytic therapy; IQR, interquartile range; RAPID, Renal function measured as urea, Age, fluid Purulence, Infection source, Dietary status measured as albumin.
Clinical and demographic characteristics of patients who had all-cause mortality at 6 months in comparison to the survivor arm.

IQR, interquartile range; RAPID, Renal function measured as urea, Age, fluid Purulence, Infection source, Dietary status measured as albumin.
RAPID score
We compared the ability of the RAPID score to predict treatment success and all-cause mortality at 6 months. The discriminating ability of the RAPID score for treatment success after IPFT was found to be poor (AUC: 0.601, 95% CI: 0.429–0.773, p = 0.24) (Figure 2). Also, the discriminating ability of the RAPID score for mortality was found to be poor (AUC: 0.640, 95% CI: 0.478–0.802, p = 0.13). The optimal cutoff for the RAPID score for mortality was ⩾4, with 84.6% sensitivity and 46.3% specificity Figure 3.
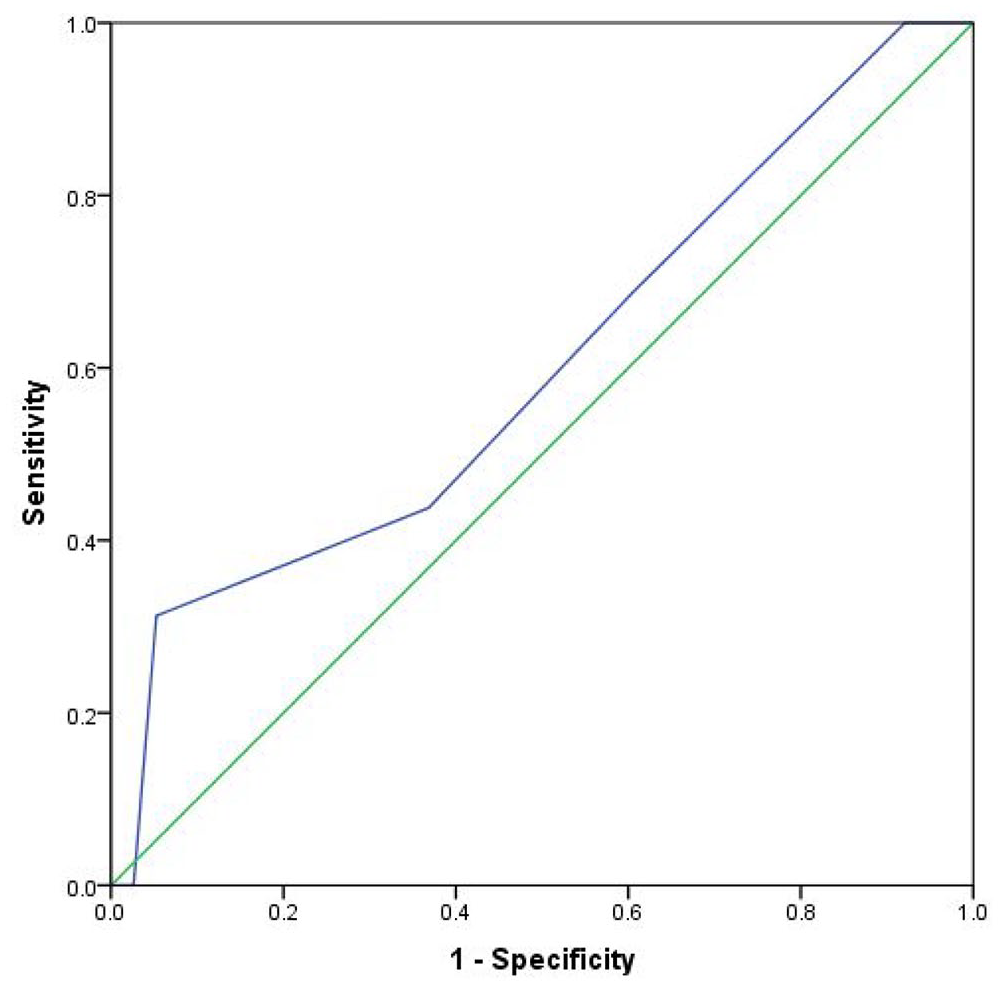
Receiver operating curve of RAPID score in predicting treatment success.
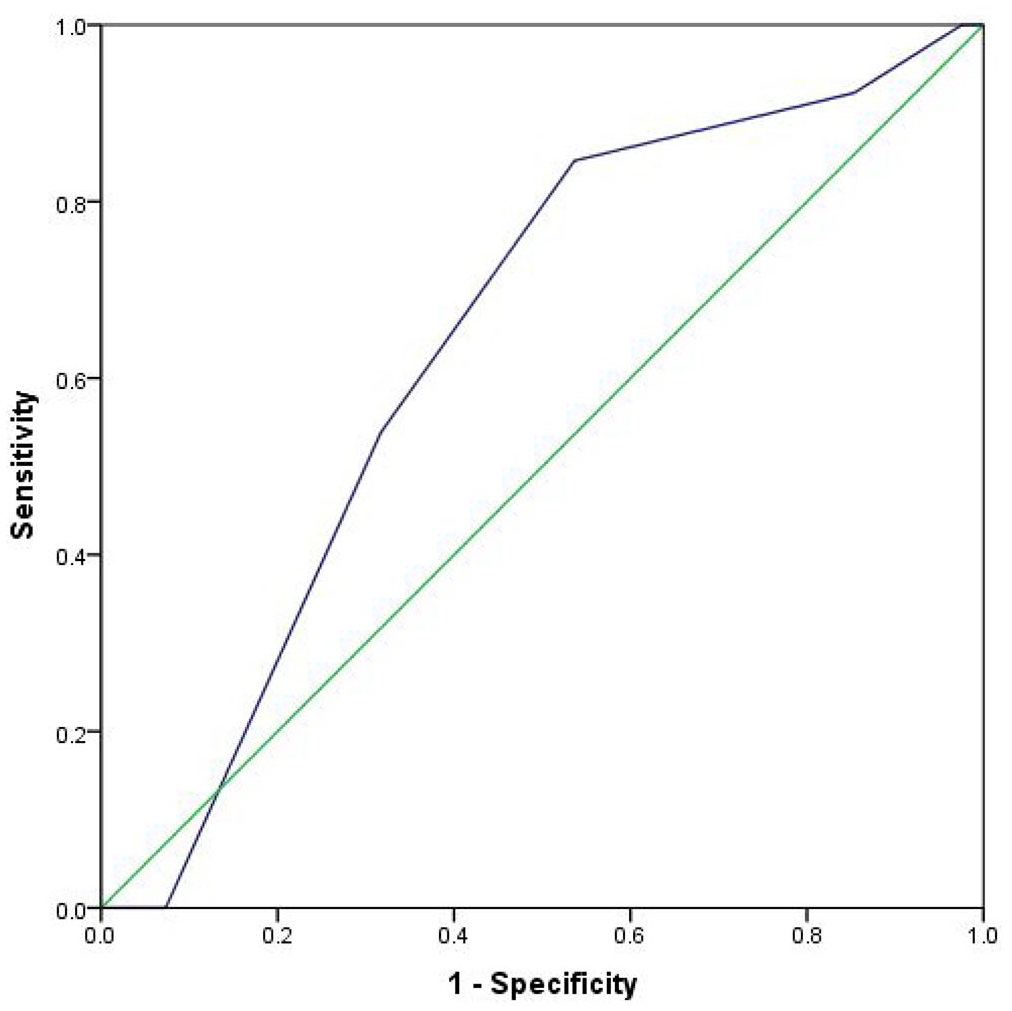
ROC curve of RAPID score in predicting all-cause mortality at 6 months.
Discussion
The use of IPFT to break fibrinous adhesions in the pleural space to facilitate pleural drainage in the setting of CPEE is included in the 2023 British Thoracic Society (BTS) guidelines. 4 The strength of the recommendation was grade B based on small clinical trials.20 –23 BTS guidelines of 2023 were preceded by the MIST-1 and MIST-2 trials. MIST-1 trial sponsored by the United Kingdom Medical Research Council (UK MRC), which was published in 2005. It concluded that the intrapleural administration of streptokinase does not decrease mortality, rate of surgery, or the length of hospital stay in patients with pleural space infection. 7 These results were later supported by a meta-analysis of five clinical trials, including MIST-1 published in 2006. 7 Three of the five trials used streptokinase and two trials used urokinase. Following this, new studies in animal models and case series suggested the superiority of tPA and DNase. 24 This was followed by the MIST-2 trial, which was published in 2011 and used tPA and DNase as IPFT. 8 The MIST-2 trial demonstrated that intrapleural administration of tPA combined with DNase significantly improved pleural fluid drainage, reduced surgical referrals, and shortened hospital stays in patients with pleural infection, whereas neither agent alone was effective in preceding studies.
IPFT is still not utilized in smaller clinical centers due to a lack of institutional experience, resource constraints, and concerns for adverse effects like bleeding, especially in the absence of thoracic surgery support. The observed incidence of bleeding in our study was low. Bleeding was mostly self-limiting and non-life-threatening; results were consistent with prior studies.25,26 Also, there was no difference in the rate of bleeding among patients with or without chronic therapeutic anticoagulation. Pain was a common side effect observed in our study. Pain is reported in up to 11% of patients receiving IPFT. 26 Pain is likely secondary to the intricate neural innervation of the pleura and further irritation of an inflamed pleura by exogenously administered enzyme therapy. Intrapleural local anesthesia with lignocaine is used in malignant pleural effusion and during medical thoracoscopy.27,28 Further investigation is required for the utilization of local anesthetics and pain control during IPFT. IPFT in our study was mostly administered through a small caliber pigtail catheter, which points to the minimally invasive nature of the intervention. Thoracic surgical intervention can be avoided in nearly one-third of the patients, lowering resource utilization and associated cost of care. The result of our study suggests that IPFT can be adopted by small to mid-sized clinical centers with limited surgical sub-specialty support.
The MIST-2 trial described the administration of 12 serial doses of tissue plasminogen activator (tPA) and deoxyribonuclease (DNase) separately. However, this regimen is labor intensive, requiring trained personnel to open and close the chest tube circuit at least eight times a day for 3 days. A simplified regimen of simultaneous administration of tPA-DNase has been previously described in the literature by Bishwakarma et al. and Smith et al. suggesting its safety with similar efficacy.12,13 Results of the simplified regimen in our study aligns with the findings of prior studies although treatment failure rate in our study was much higher in comparison to MIST-2 study. It is important to note that our study was retrospective, and observation was not designed for a head-to-head comparison of two regimens. A more simplified approach with once-daily regimen is described in a single-center retrospective study by Goh et al. 29 Although the study by Goh et al. has several limitations including single-center retrospective design and limited statistical power (n = 87), it does suggest a simplified approach necessitating larger prospective studies.
Although treatment success was achieved in 30% of patients, we acknowledge the 70% treatment failure rate necessitating surgical intervention. A higher rate of treatment failure in our study cohort could be attributed to the underlying comorbidities and overall state of health. While the judicious use of IPFT can reduce the need for surgical intervention, it may also delay surgery in patients who require it. Therefore, appropriate patient selection is crucial, and the clinical judgment of the treating physician becomes imperative in choosing the appropriate treatment modality. Early consideration for surgical intervention would be appropriate in the presence of thick pleural rind and extensively loculated effusions, with multiple pockets. Anatomically, a medially located fluid collection in relation to the lung may also necessitate an early surgical consultation as positioning a percutaneous or surgical chest tube medial to the lung would be technically difficult.
A relatively high all-cause mortality at 6 months was seen in our study population, likely due to multiple medical comorbidities and the overall state of health of the study cohort. Although a level of statistical significance could not be reached, the proportion of patients with pre-existing congestive heart failure, atrial fibrillation, and chronic anticoagulant use was higher in the patients who died within 6 months compared to those who did not. Although sepsis was the primary cause of death within 30 days, analysis to delineate causes and characteristics of death within 6 months could not be performed due to the retrospective nature of the study.
The RAPID score includes five clinical factors: Renal function measured as blood urea nitrogen (BUN), age, purulence, infection source, and dietary factors, and can potentially identify patients at increased risk for mortality.11,16,17 Herein, we sought to investigate the factors predicting treatment success and all-cause mortality within 6 months. The discriminating ability of the RAPID score for treatment success after IPFT was found to be poor (AUC: 0.601, 95% CI: 0.429–0.773, p = 0.24). Predictive ability of the RAPID score for mortality at 6 months was found to be poor (AUC: 0.640, 95% CI: 0.478–0.802, p = 0.13). The optimal cutoff for the RAPID score for mortality was ⩾ 4, with 84.6% sensitivity and 46.3% specificity. When applied to a diverse patient cohort, the RAPID score can be utilized to predict mortality up to 6 months.
Our study has several limitations. First, as a single-center study, it is possible that the results may lack generalizability. Second, clinical decisions in our study were made by the clinicians at their discretion, while there were protocol deviations, this likely represents real-world clinical scenarios. Given the lower number of outcome events in the cohort, the ability to estimate event rates could have been limited. We also acknowledge that it remains unclear if the concurrent use of tPA and DNase in IPFT somehow affects the activities of either agent. Although we were meticulous in our data collection, data was manually collected from the electronic health records, which is more prone to errors than data collected prospectively.
Conclusion
Our study suggests that IPFT can be safely adopted by small and mid-size clinical centers. Adverse clinical outcomes and the risk of bleeding were low, self-limiting, and non-life threatening. Intrapleural fibrinolytic therapy can be administered using a small caliber pigtail catheter and formal surgical intervention can be avoided in up to one-third of patients suggesting both the minimally invasive nature of therapy and the ability to reduce the associated cost of care.
Supplemental Material
sj-doc-1-tar-10.1177_17534666251343711 – Supplemental material for Intrapleural tissue plasminogen activator and deoxyribonuclease in complex pleural effusion and empyema, clinical outcomes, and predictors
Supplemental material, sj-doc-1-tar-10.1177_17534666251343711 for Intrapleural tissue plasminogen activator and deoxyribonuclease in complex pleural effusion and empyema, clinical outcomes, and predictors by Yub Raj Sedhai, Priyanka Bhat, Roshan Acharya, Nada Qaisar Qureshi, Fawaz Mohammed, Irfan Waheed, Tahir Muhammad Abdullah Khan, Muhammad Altaf Ahmed, Nisarfathima Kazimuddin, Akinchan Kafle, Rodney T. Steff and Karan Singh in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
Clinical illustration in Figure 1 is hand-drawn by Akinchan Kafle, MD. Dr. Kafle is a clinical anatomist and medical illustrator. Hand-drawn figures were collaged using Adobe Photoshop (Adobe Inc. San Jose, CA, USA).
Author’s note
The abstracts of this study were previously presented at the American College of Chest Physicians (ACCP) Annual Meeting 2023- CHEST 2023 in Honolulu, Hawaii. The abstract is available at CHEST, 164(4), pp. A3575-A3576; CHEST, 164(4), pp. A3586–A3587.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
