Abstract
Background:
The benefits of high-flow nasal cannula (HFNC) as primary intervention in patients with acute hypoxemic respiratory failure (AHRF) are still a matter in debate. Our objective was to compare HFNC therapy versus conventional oxygen therapy (COT) in the prevention of endotracheal intubation in this group of patients.
Methods:
An open-label, controlled and single-centre clinical trial was conducted in patients with severe AHRF, defined by a PaO2/FIO2 ratio ⩽200, to compare HFNC with a control group (CG) treated by COT delivered through a face mask, with the need to perform intubation as the primary outcome. The secondary outcomes included tolerance of the HFNC device and to look for the predictive factors for intubation in these patients.
Results:
A total of 46 patients were included (22 in the COT group and 24 in the HFNC group) 48% of whom needed intubation: 63% in the COT group and 33% in the HFNC group, with significant differences both in intention to treat [χ2 = 4.2; p = 0.04, relative risk (RR) = 0.5; confidence interval (CI) 95%: 0.3–1.0] and also in treatment analysis (χ2 = 4.7; p = 0.03; RR = 0.5; IC 95%: 0.3–0.9) We obtained a number needed to treat (NNT) = 3 patients treated to avoid an intubation. Intubation occurred significantly later in the HFNC group. Estimated PaO2/FIO2, respiratory rate and dyspnea were significantly better in the HFNC group. Patients treated with HFNC who required intubation presented significant worsening after the first 8 h, as compared with non-intubated HFNC group patients. Mortality was 22% with no differences. The HFNC group patients were hospitalized for almost half of the time in the intensive care unit (ICU) and in the ward, with significantly less hospital length of stay. A total of 14 patients in the HFNC group (58%) complained of excessive heat and 17% of noise; 3 patients did not tolerate HFNC.
Conclusion:
Patients with severe acute hypoxemic respiratory failure who tolerate HFNC present a significantly lower need for endotracheal intubation compared with conventional oxygen therapy.
Clinical Trial Register
EUDRA CT number: 2012-001671-36
The reviews of this paper are available via the supplemental material section.
Key messages
- What is the key question?
The high-flow therapy is an increasing alternative for the conventional oxygen therapy in patients with acute respiratory failure. But is it really effective? Can it reduce the need of endotracheal intubation in patients with acute and severe respiratory failure?
- What is the bottom line?
This Clinical Trial tries to answer the key question by randomly applying high-flow or conventional oxygen therapy to patients with acute and severe respiratory failure. We also measured the tolerance to the device because in our daily experience, this was not as good as previously published.
- Why read on?
There are only a few Clinical Trials published about this topic,1–3 and only one of them was conducted exclusively in an ICU and in patients who were not immunocompromised. 4 This article, like previous published reports, confirms that endotracheal intubation may be avoided by its application, especially in the most severe cases. This is essential information that must be broadcast to our colleagues that attend this type of patient.
Introduction
Invasive mechanical ventilation (IMV) through an endotracheal tube is the usual supportive treatment for severe acute hypoxemic respiratory failure (AHRF; which we define as a PaO2/FIO2 ratio ⩽ 200), when other techniques have failed, but is associated to major adverse effects leading to greater morbidity, mortality and longer hospital stay.5–7 The evidence supports the use of non-invasive mechanical ventilation (NIV) in certain clinical settings,8–11 but results in poor tolerance during prolonged treatments. High-flow nasal cannula (HFNC), initially introduced in children, 12 and later in adult,s13,14 is now an alternative with promising results in many clinical scenarios, but primarily in AHRF.6,15–17 Its benefits are attributed to the fact that it provides high flow, matching the patient’s inspiratory flow demands, at an ideal temperature and humidity,18,19 which improves oxygenation, produces a certain continuous positive pressure effect on airways, reduces proximal airway dead space, 20 and increases respiratory volumes.21,22 HFNC has also been used in immunocompromised patients,1,23,24 patients with chronic obstructive pulmonary disease (COPD),25,26 and in other different clinical scenarios.27,28
There are some prospective and randomised controlled studies on the use of HFNC as a first-line intervention in patients with AHRF.2–4,29–31A recent meta-analysis of nine randomised trials of patients deemed at risk of respiratory failure and intubation showed that HFNC may decrease the need of intubation, but no differences in mortality rate were observed.1–3,32 However, it is important to highlight that these trials were conducted in different settings, such as intensive care unit (ICU) or emergency department, so this is an important limitation to the final conclusion.
In patients with AHRF admitted to an ICU and treated with HFNC as the first-line intervention, the only published clinical trial did not demonstrate a decrease in intubation, except in severe patients with PaO2/FiO2 < 200 (post hoc analysis). 4 Also, they described a significant difference in 90-day mortality in favour of high-flow oxygen.
On the other hand, it is as important to consider that the time of intubation could be delayed due to HFNC therapy and that this could carry worse clinical outcomes in this group of patients. 33
The aim of this study was to compare the efficacy of HFNC with standard oxygen therapy in patients with AHRF, with the need for intubation as the primary outcome, and to learn their degree of tolerance.
Methods
Study design
The study was designed as an open-label, controlled and single-centre clinical trial, to compare HFNC (intervention group or HFNC group) versus conventional oxygen therapy (COT; control group or GC) in patients with AHRF.
The study was approved by the Ethics Committee of the University Hospital of La Princesa, Madrid and was conducted in a single Spanish ICU between January 2013 and December 2015.
Patients
We included consecutive cases of patients older than 18 years, of both sexes, with AHRF of any etiology that did not meet exclusion criteria after signing informed consent. AHRF was defined as: a PaO2/FIO2 ratio ⩽ 200 or SpO2/FIO2 ratio ⩽ 160 (SpO2 = peripheral oxygen saturation by pulse oximetry); and respiratory rate > 30 r/min for at least 30 min. Exclusion criteria included: nasal anatomical deformity, Glasgow coma score <12, urgent need for intubation or criteria for NIV (exacerbation of COPD with hypercapnic acidosis, PaCO2 > 45 mmHg, and pHa < 7.3025,34,35 or respiratory insufficiency due to cardiogenic pulmonary edema), 36 acute agitation, limited communication, hemodynamic instability, or any patient who, at the discretion of the investigator, could not comply with the protocol. Even if the investigator did not consider that the patient was able to understand or sign the informed consent, or simply if the patient is conditioned to participate because of the vulnerability of the situation, this was a sufficient reason for exclusion. To guarantee this process, the attending physician (not involved with the study) was witness to it.
Randomisation
Randomisation was simple, blinded, by balanced blocks of six patients to compare the HFNC group versus COT group in patients with AHRF.
The process was started by any physician in charge in the ICU who detected patients who met inclusion criteria. Once consent was offered and signed by the patient, randomization was performed through sealed opaque envelopes. The physician takes the next envelope and this was opened in sight of the patient. The therapy is obviously not blinded.
Procedures
Patients included in the COT group continued with the standard treatment via a venturi mask maintaining the same initial FIO2 (0.6–1) upon admission to ICU. If necessary for desaturation, we raised the FIO2 to the minimum required to maintain a SpO2 ⩾93%. We decided to maintain the COT because this is the classical and not yet improved therapy in terms of mortality, length of stay and need for invasive ventilation of patients with ARF with no exclusion criteria previously defined: basically instability, altered mental state, urgent need for intubation or criteria for NIV. 1
In the HFNC group, high flow oxygen was administered by a HFNC system (Optiflow®, Fisher & Paykel, Maidenhead, UK), using the same FIO2 applied with the venturi mask at admission in ICU (0.6–1). We started with a flow of 20 l/min, and then increasing 5–10 l/min every 1–3 min according to patient tolerance, up to a maximum 50 l/min. The initial temperature was 34°, increasing to 37°C with flows ⩾ 30 l/min.
Once target settings for HFNC (50 l/min and 37°C) were reached, the programmed measurements were initiated: 10, 20 and 30 min and 2, 4, 8, 12, and 24 h. From the second day onwards, they were taken every 12 h. The COT group was treated via a venturi mask with the same initial FIO2, and taking the same programmed measurements. Measurements could be added in both groups according to the judgment of the attending physician.
The minimum FIO2 required to maintain a SpO2 ⩾ 93% was selected. In the HFNC group it increased progressively from 0.6 to 1; and in the COT group from 0.6 via venturi mask with 15 l/min to closed venturi with reservoir and 30 l/min. No direct FIO2 measurements were performed in any group. For the analysis, we estimated the ratio between these measurements and arterial oxygen partial pressure (PaO2/FIO2), taking those of the device in the HFNC group and 0.6 or 1 in the COT group. The PaO2 was measured by arterial gasometry in all cases.
Because of the strict acute respiratory insufficiency (ARI) inclusion criteria, all along the process we were aware of incoming criteria of intubation. The indication for intubation was performed by the attending physician, not the investigator, and was defined by one or more of these criteria: respiratory rate > 35 rpm, PaO2 <60mmHg with maximal FIO2, PaCO2 > 55 mmHg, deterioration of consciousness, paradoxical breathing, and use of accessory muscles.
In all patients, we recorded: the need for intubation (time: before in hours and after in days), death (date and cause), clinical respiratory parameters (respiratory rate, SpO2), arterial blood gases (pH, pO2, pCO2, bicarbonate), side effects or intolerance, days in ICU, length of hospital stay and patient comfort: assessment of dyspnea and comfort using self-perceived analogue scales (modified Borg scale for the measurement of dyspnea and visual analogue scale for comfort).37,38 In both scales the patient, scores with 0–10 points, being 0 the best and 10 the worst situation, at basal time and then 10, 20 and 30 min and 2, 4, 8, 12 and 24 h from the branch inclusion. Confounding variables were: acute pulmonary diseases (etiological diagnosis, number of affected quadrants on chest X-ray: inclusion and worst time), previous comorbidity (chronic pulmonary or neuromuscular diseases, CHARLSON index), need for sedoanalgesia, APACHE II, number of organ failures other than the lung (hemodynamic, renal, liver, coagulopathy, encephalopathy): at inclusion, intubation, and worst evolution, moment at admission and at ICU discharge.
Outcomes
The primary outcome was the proportion of endotracheal intubated patients. Follow up until hospital discharge, death, or scheduled endotracheal intubation for any procedure was made.
Our secondary outcomes were:
-To study tolerance of the HFNC device
-To look for predictive factors for intubation in this group of patients
Statistical analysis
The estimated sample size was 92 patients per study branch, considering that intubation would be independent, 80% in the COT group, and 25% lower in the HFNC group (alpha error = 5%, beta error = 20%, Yates correction). The statistical analysis was performed by Stata software version 13.1 (Statacorp, College Station, TX, USA).
A descriptive analysis was carried out, analyzing the comparability of the groups and comparing by intention to treat: bivariate (parametric or nonparametric) and multivariate (linear regression for repeated measures or Cox proportional risk regression). The independent variables included in the models were those that demonstrated a relationship with p ⩽ 0.20 with the dependent variable. The backward step by step inclusion criterion of variables were used. Statistical significance was set at p < 0.05. Kaplan–Meier curves were plotted to assess the time to endotracheal intubation and discharge from hospital.
The strength of the association was measured with the relative risk or hazard ratio and the population estimate with a 95% confidence interval. The number needed to treat (NNT) was calculated to avoid an intubation.
The data monitoring overseeing was performed by the Foundation of Biomedical Research, La Princesa University Hospital.
Results
Inclusion
During the study period (January 2013 to December 2015), 2352 patients were admitted. Of these, 236 patients presented ARI (in 174 it was the reason for admission), with 63% intubated (95% CI: 57–69%), 78.5% during the first day (95% CI: 71–84%). A total of 58% were men, with a mean age of 63 years (SD: 14.5 years, median: 50 years P25–75: 56–75 years), and a mean APACHE II score of 19 points (SD: 7 points; median: 18 points P25 –75: 14–23 points); 18.6% died (95% CI: 14–24%). Of patients with AHRF, 60% met the inclusion criteria. Consent was requested from 58 patients, and 46 of those signed, 24 were included in the HFNC group and 22 in the COT group (Figure 1). We did not find significant differences between patients with AHRF and the study sample. We found no significant differences between the groups in any of the initial variables (Table 1).

Patient eligibility and randomisation scheme.
Comparability of study groups.

min, minutes; (P25–75), 25th–75th Percentile; SD, standard deviation.
We analyzed the subgroup of patients who were not intubated in the first 24 h. They were 30: 19 in the HFNC group and 11 in the COT group. There were no significant differences (p = 0.17) in APACHE II score between therapy groups.
Outcomes
A total of 22 patients (48%) were intubated, 16 during the first day: 14 (63%) in the COT group and 8 (33%) in the HFNC group. The HFNC group patients required significantly less intubation (six less patients requiring intubation as compared with the COT group), both in intention to treat [χ2 = 4.2; p = 0.04, relative risk (RR): 0.5, 95% confidence interval (CI): 0.3–1.0) and in treatment analysis (χ2 = 4.7; p = 0.03; RR = 0.5; CI 95%: 0.3–0.9). In the Cox regression, the intubation occurred later in the HFNC group (p = 0.05), being significant in the patients who were finally treated with HFNC (p = 0.02) (Figure 2). The NNT was 3.3 patients treated to avoid an intubation in intention to treat analysis (95% CI: 3.05–3.58). In the HFNC group, those who needed intubation presented a significant PaO2/FIO2 decrease during 8–24 h of treatment, compared with those who were not intubated (p = 0.02) (Figure 3).
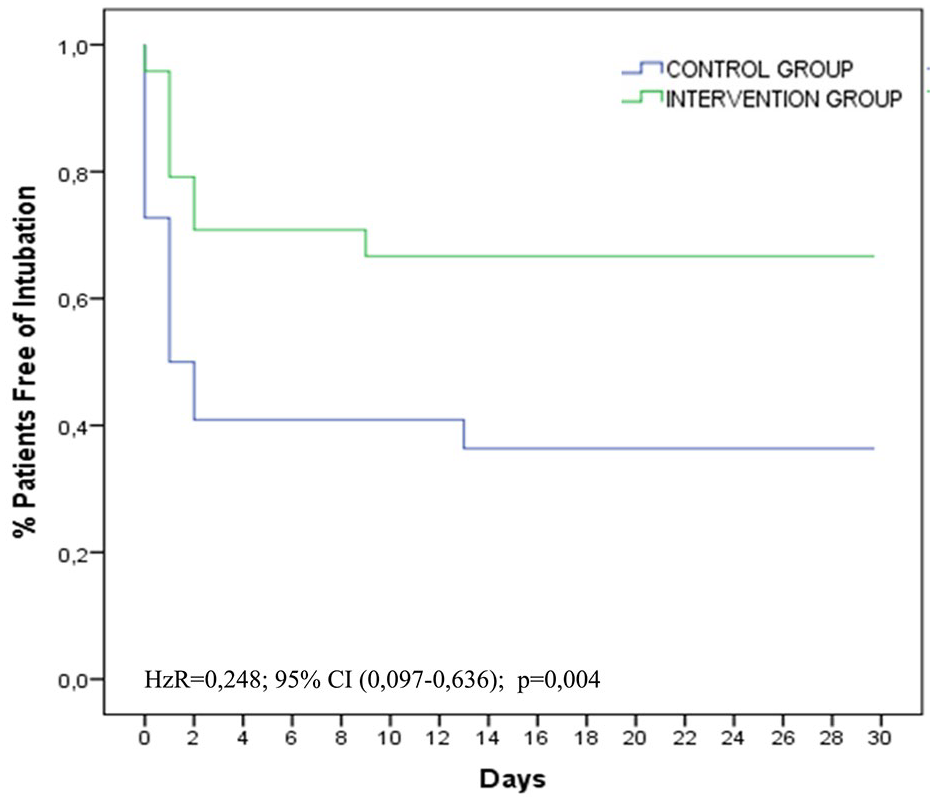
Kaplan–Meier plot of patients free of endotracheal intubation.

Progression between PaO2/FiO2 during the first 24 h in Intervention Group patients according to the need for intubation.
Likewise, the higher the number of affected quadrants on the radiograph at baseline, the higher the risk of intubation (RR quadrant = 2.1, 95 CI%: 1.2–3.2, p = 0.009), and the higher the number of organs affected, the earlier the need for intubation [intention to treat analysis (IAT): RR 4 versus 1 organ = 6.7 95% CI: 1.2–38.3, p = 0.03].
We found significant differences in PaO2/FIO2, PaCO2, and respiratory rate between both groups, significantly improving in the HFNC group (Figure 4). The sensation of dyspnea, measured by a self-perceived Borg scale, was also statistically better in the HFNC group during the first 24 h (p = 0.02). Hemodynamic variables, pH, and bicarbonate improved over time; but without differences between groups.

Comparison of clinical and blood gases parameters between groups during the first 24 h. Analysis for treatment ATT.
Of the 46 patients, 36 were discharged from the hospital and 10 died intubated (22%, 95% CI: 12–36%), but none died during the intubation procedure. Mortality in the HFNC group was 25% versus 18% in COT group (p = 0.7). Shock with multi-organ failure was the cause of death in all except one of the HFNC group’s patients, who died of ARDS due to a progression of rheumatoid arthritis. We found no differences in time until death. In the multivariate logistic regression analysis, we found that the number of affected quadrants on the radiograph [odds ratio (OR) = 3.6, 95% CI 1.15–11.77, p = 0.028] and the number of affected organs at the time of inclusion (OR = 4.86, 95% CI 1.65–14.35, p = 0.004) were significantly related to death. We also identified a negative relationship between HFNC group and death, meaning that there is a trend for higher mortality risk in the HFNC group, but this is not significant (p = 0.08). We did not find any variables independently and significantly related to the time of death.
The HFNC group patients had shorter length of stay both in ICU and hospital. However, this difference was significant only in terms of hospital stay. These differences were even greater when analysing the patients who survived; (log rank, p = 0.03) (Figure 5).

Kaplan–Meier plot of the length of hospital stay in both groups.
Comfort
Of the 24 HFNC group patients, 14 complained of heat (58%, 95% CI: 39–75.5%), 4 of noise (17%, 95% CI: 7–36%), and 3 did not tolerate the device because of the displeasing heat, without any measurement (12.5%, 95% CI: 4–31%). In the rest, it was only necessary to reduce the flow setting. Nevertheless we found no significant difference between both groups (HFNC group and COT group) in overall comfort measured by analogic self-perceived scales.
Discussion
In this study, we found that the use of HFNC as primary intervention in patients with AHRF may reduce the need for endotracheal intubation.
Differences in intubation rate between both groups were significant and similar to other non-randomised studies,39,40 and to the results obtained from the recently published meta-analysis by subgroups,32,41 where benefits similar to ours (OR = 0.5) were found. Even though intubation criteria were identical in both groups, intubation occurred significantly later in the HFNC group. We attribute this difference to the improvement observed on day 1 of some of the intubation criteria (dyspnea, respiratory rate, and estimated PaO2/FiO2 ratio), in patients treated with HFNC. Findings similar to those described in the literature, which are attributed to the benefits of HFNC.42–44
In the HFNC group, we saw that, in the patients who finally were intubated, the PaO2/FIO2 ratio improvement trend does not continue from the first 8 h to its application. Our explanation to this finding is that these patients may need more positive end-expiratory pressure (PEEP) levels that the device cannot offer. Probably, the initial improvement of dyspnea and gasometric parameters can delay the intubation in some cases and this could worsen the outcome, as described in some studies. 33
The strengths of our study are that the randomisation of the patients and analysis of the variables were blinded. Ours is also a pragmatic study, including any kind of patients with AHRF and with other organ dysfunction, so our results are more generalisable.
Our principal limitation was that participation was offered to one-third of eligible patients, with 10% of these patients refusing. These percentages are very similar to those reported by Frat and colleagues about inclusion in their Clinical Trial. Because of the ARI status, a large group of patients with inclusion criteria were unable to understand or to sign the informed consent, and a family member was often not available in that moment. Furthermore, participation was not offered by the investigator in other cases because the clinical situation seemed to condition them. We were very rigorous about this, and these facts explain the large losses of potentially recruitable patients.
Although there may have been a selection bias due to these factors, we did not find any significant differences between eligible patients and the study sample, either in the initial characteristics or in the need for intubation (similar to COT group); neither were there differences in mortality, which ensures the representativeness of the sample. Eligible patients who were not included in the trial were treated with COT and intubated if needed as usual. In the patients included, both therapy groups were comparable in the lack of significant differences in the initial variables. Although APACHE II score was slightly higher in the COT group, we attributed it to the higher percentage of patients intubated in the first 24 h (86% versus 66%) and not for severity differences at the baseline. So we could not remove the intubation parameter from the score, we made a sensitivity analysis excluding the intubation. We analyzed the subgroup of patients who were not intubated in the first 24 h in which there were no significant differences in APACHE II score between therapy groups.
Despite not achieving the expected sample size, we have achieved sufficient statistical power to find significant differences between groups in the main study variable; therefore our results are consistent. Our estimated sample size was probably oversized because we did not expect that the intervention would be so efficient in avoiding intubation.
The intervention was not blind, and this could lead to a bias in the main variable. However, the percentage of intubated patients in the COT group is similar to that found in the total number of eligible patients, and is slightly lower than in other studies (including the French clinical trial); therefore, we believe that the intubation indication was adjusted to the needs and evolution of patients.
HFNC group patients had ICU stays and hospitalisation times that were almost 50% shorter, with a significantly shorter hospital stay, which we attributed to lower morbidity. Mortality in the HFNC group was 25% versus 18% in COT group without statistical difference. We identified a higher, but not statistically significant, mortality trend in the HFNC group. However, the sample size of this study does not provide enough statistical power to demonstrate this difference, if it really exists. It should be noted that there were no deaths either before or during intubation, and the principal cause of death in these patients was multi-organ failure.
The observed mortality (22%) was slightly higher, but not significant, than that described by the French group, with an overlap of confidence intervals. In addition, our patients were more severe. We found that the number of quadrants in the radiograph and the number of affected organs at the time of inclusion were the two variables that were significantly related to death. Although we did not find variables independently and significantly related to the time of death. One HFNC group patient died of AHRF, with ARDS, intubated 36 h after entering the study. That raised the question of whether HFNC might negatively affect prognosis through delay of intubation, as reported by other authors.33,45,46 According to our results, the improvement trend in PaO2/FiO2 with HFNC which does not continue in the 8 h posterior to its application, should alert us to perform intubation.
A progressive adaptation to HFNC was attempted, but its tolerance was worse than described,28,46,47 58% presented discomfort, due mainly to heat and also due to noise. Three patients could not be treated (12.5%). This worse result could be due to our patients being more severe. Nevertheless the overall tolerance, measured by comfort scales, was not different from that of the COT group; probably because the side effects were palliated by the significant improvement of dyspnea sensation in the HFNC group. These conclusions were recently reported for the first time by Mauri et al. 48
During the study, a multicentre clinical trial with a similar objective was conducted at an ICU, 4 but with a third treatment option (NIV) and assigning about 100 patients per group. Unlike our work, the AHRF was less severe (PaO2/FIO2⩽300) and no significant difference in the need for intubation at 28 days was found, but a decrease in mortality at 90 days was seen in the HFNC segment. In a post hoc analysis, the authors verified that the need for intubation was cut by half in more severe patients treated with HFNC. 4 Patients in our sample presented AHRF with estimated PaO2/FiO2 ⩽ 200. The other clinical and blood gas characteristics were similar to those described in the French study. 4
Although some meta-analyses have failed to show consistent and convincing benefit in clinically meaningful outcomes, a meta analysis published recently by Rochwerg and colleagues showed similar conclusions to ours in the primary outcomes. 32 They found a decreased risk of intubation in patients treated with HFNC with no differences in mortality or comfort. However, unlike our trial, they found no differences in the length of stay or dyspnea improvement.
In order to secure and support the clinical benefits of HFNC, such as intubation rates and survival, more randomized clinical trials are needed.
In conclusion, we believe that patients with severe AHRF, who do not require immediate intubation should be treated using a HFNC. However, if, after an initial improvement, this trend does not continue from the first 8–12 h, endotracheal intubation should be considered.
Supplemental Material
Author_Response_1 – Supplemental material for High-flow nasal oxygen reduces endotracheal intubation: a randomized clinical trial
Supplemental material, Author_Response_1 for High-flow nasal oxygen reduces endotracheal intubation: a randomized clinical trial by Ricardo Andino, Gema Vega, Sandra Karina Pacheco, Nuria Arevalillo, Ana Leal, Laura Fernández and María Jesús Rodriguez in Therapeutic Advances in Respiratory Disease
Supplemental Material
Author_Response_2 – Supplemental material for High-flow nasal oxygen reduces endotracheal intubation: a randomized clinical trial
Supplemental material, Author_Response_2 for High-flow nasal oxygen reduces endotracheal intubation: a randomized clinical trial by Ricardo Andino, Gema Vega, Sandra Karina Pacheco, Nuria Arevalillo, Ana Leal, Laura Fernández and María Jesús Rodriguez in Therapeutic Advances in Respiratory Disease
Supplemental Material
Reviewer_1_v.1 – Supplemental material for High-flow nasal oxygen reduces endotracheal intubation: a randomized clinical trial
Supplemental material, Reviewer_1_v.1 for High-flow nasal oxygen reduces endotracheal intubation: a randomized clinical trial by Ricardo Andino, Gema Vega, Sandra Karina Pacheco, Nuria Arevalillo, Ana Leal, Laura Fernández and María Jesús Rodriguez in Therapeutic Advances in Respiratory Disease
Supplemental Material
Reviewer_1_v.2 – Supplemental material for High-flow nasal oxygen reduces endotracheal intubation: a randomized clinical trial
Supplemental material, Reviewer_1_v.2 for High-flow nasal oxygen reduces endotracheal intubation: a randomized clinical trial by Ricardo Andino, Gema Vega, Sandra Karina Pacheco, Nuria Arevalillo, Ana Leal, Laura Fernández and María Jesús Rodriguez in Therapeutic Advances in Respiratory Disease
Supplemental Material
Reviewer_2_v.1 – Supplemental material for High-flow nasal oxygen reduces endotracheal intubation: a randomized clinical trial
Supplemental material, Reviewer_2_v.1 for High-flow nasal oxygen reduces endotracheal intubation: a randomized clinical trial by Ricardo Andino, Gema Vega, Sandra Karina Pacheco, Nuria Arevalillo, Ana Leal, Laura Fernández and María Jesús Rodriguez in Therapeutic Advances in Respiratory Disease
Supplemental Material
Reviewer_2_v.2 – Supplemental material for High-flow nasal oxygen reduces endotracheal intubation: a randomized clinical trial
Supplemental material, Reviewer_2_v.2 for High-flow nasal oxygen reduces endotracheal intubation: a randomized clinical trial by Ricardo Andino, Gema Vega, Sandra Karina Pacheco, Nuria Arevalillo, Ana Leal, Laura Fernández and María Jesús Rodriguez in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
We thank the Clinical Trials Unit of La Princesa Hospital for their help in monitoring the study. We thank Spanish Ministry of Health, Social Services, and Equality for the funding (N° Exp.: EC11–103). And also we thank Antonio Reyes for his support and advice in the creation of this study and Fernando Suárez for his advice reviewing the manuscript.
Authors Note
The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Author contribution(s)
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received a funding from: Spanish Ministry of Health, Social Services, and Equality. (N° Exp.: EC11-103). The funder of the study had had no role in study design, data collection, data analysis, data interpretation; or writing of the report.
Conflict of interest
The authors declare that there is no conflict of interest.
Ethics approval and consent to participate
The study was approved by the Ethics Committee of the University Hospital of La Princesa, Madrid. The study was reviewed and approved by University Hospital La Princesa Institutional Review Board. All study participants, or their legal guardian, provided informed consent prior to study. Consent for sharing data was not obtained but the presented data were anonymized and the risk of identification is low.
Supplemental material
The reviews of this paper are available via the supplemental material section.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
