Abstract
Background:
Exertional dyspnea is a cardinal feature of chronic obstructive pulmonary disease (COPD) and a major cause of activity limitation. Although dual bronchodilation is more effective than bronchodilator monotherapy at improving resting pulmonary function, it is unclear to which extent this translates into superior relief of exertional dyspnea.
Methods:
We conducted a randomized controlled, double-blind, cross-over trial comparing indacaterol 110 µg/glycopyrronium 50 µg once daily (OD) with tiotropium 50 µg OD in patients with moderate to severe COPD and resting hyperinflation (functional residual capacity >120% of predicted value). The primary outcome was Borg dyspnea score at the end of a 3-min constant speed shuttle test after 3 weeks of treatment. Secondary outcomes included changes in Borg dyspnea score after the first dose of study medication, expiratory flows and lung volumes. Statistical analysis was conducted using a cross-over analysis of variance model with repeated measurements.
Results:
A total of 50 patients with COPD and a mean forced expiratory volume in 1 s of 54 ± 11% (mean ± SEM) predicted participated in the cross-over phase of the trial. Compared with baseline, there was a decrease in dyspnea after the first dose of medication with indacaterol/glycopyrronium [mean −1.00, 95% confidence interval (CI) −1.49 to −0.52] but not with tiotropium alone (mean −0.36, 95% CI −0.81 to 0.08). The reduction in dyspnea after the first dose was statistically significant between the two treatments (mean difference of −0.64, 95% CI −1.11 to −0.17). Despite indacaterol/glycopyrronium providing further bronchodilation and lung deflation throughout the trial, the reduction in dyspnea was not sustained at 3 weeks of treatment (mean between-treatment difference at 3 weeks of 0.09, 95% CI −0.44 to 0.61).
Conclusion:
In comparison with bronchodilator monotherapy, indacaterol/glycopyrronium provided greater immediate exertional dyspnea relief, although this difference was not sustained after 3 weeks of therapy despite evidence of further bronchodilation and lung deflation.
Introduction
Dyspnea and exercise intolerance are among the most debilitating symptomatic manifestations of chronic obstructive pulmonary disease (COPD). 1 Besides its effects on exercise tolerance, persistent dyspnea independently predicts poor clinical outcomes including exacerbations, hospitalizations and even mortality. 2 Together, dyspnea and exercise intolerance lead to a vicious circle that promotes the adoption of a sedentary lifestyle, another important predictor of mortality in COPD.3,4
Alleviating dyspnea and improving exercise tolerance are important therapeutic goals when caring for patients with COPD. 5 Several clinical trials have confirmed the ability of long-acting bronchodilators to enhance exercise tolerance and to reduce the perception of dyspnea in patients with moderate to severe COPD.6–11 Central to these benefits is the efficacy of long-acting bronchodilators to improve expiratory flows and to reduce operating lung volumes during exercise, which translates to less perceived dyspnea.6–14
In order to specifically assess the efficacy of COPD treatments to reduce exertional dyspnea, we developed the 3-minute constant speed shuttle test (3-min CSST).15,16 The main feature of this test is that it allows measuring dyspnea scores at fixed time points, including at the end of the test, enabling pre- and post-intervention comparisons with concrete data. Another important characteristic of the 3-min CSST is that the walking speed is externally imposed, and is designed to produce a standardized exercise stimulus to induce dyspnea. This is not the case for the self-paced 6-min walking test which, by design, does not allow for pre- and post-intervention comparisons of dyspnea perception to be made at a standardized exercise stimulus. 17 Lastly, this test allows for dyspnea measurement during walking, which is the most prevalent and problematic activity of daily life for patients with COPD. 18 The feasibility and reproducibility of the 3-min CSST have been documented in our previous work. 15 We also demonstrated the sensitivity of this test to detect reduced dyspnea perception after bronchodilation with ipratropium bromide compared with placebo in patients with COPD. 16 More recently, this exercise methodology was employed to document the added benefit of dual bronchodilation with tiotropium/olodaterol fixed dose combination to reduce exertional dyspnea compared with tiotropium alone in patients with COPD. 19
Considering the well-documented bronchodilation efficacy of indacaterol/glycopyrronium fixed dosed combination
20
and its capacity to alleviate patient-reported dyspnea during activity of daily life compared with placebo and tiotropium,
21
we designed this randomized controlled, double-blind cross-over study to compare the effects of dual bronchodilation with indacaterol/glycopyrronium
Methods
Study design
This was a randomized, cross-over and double-blind multicenter trial. The primary objective was to compare the effects of indacaterol 110 µg/glycopyrronium 50 µg
Study participants
Participants were recruited from the pulmonary clinics of three university-affiliated hospitals in Canada (Institut universitaire de cardiologie et de pneumologie de Québec, Université Laval in Quebec City, McGill University Health Center in Montreal and Kingston General Hospital, Queens University in Kingston). Participants were selected on the basis of (a) moderate to severe COPD with a post-bronchodilator forced expiratory volume in 1 s (FEV1) between 30% and 79% of the predicted value, an FEV1/forced vital capacity (FVC) ratio of <70%, and evidence of hyperinflation at rest [functional residual capacity (FRC) >120% of predicted value]; (b) aged over 50 years; (c) a cigarette-smoking history >10 pack-years; and (d) a Borg dyspnea score of at least 4 points during the baseline 3-min CSST. The main exclusion criteria were a COPD exacerbation less than 2 months prior to the study, a current diagnosis of asthma, pulsed arterial O2 saturation (SpO2) <85% at rest or during exercise, the presence of another pathology that could influence exercise tolerance and use of home oxygen. Participants experiencing a COPD exacerbation during the study were not allowed to continue. Each institutional research ethics board approved the study and all participants signed an informed consent form.
Visits and follow up
The study required seven visits (Figure 1): three during the run-in and familiarization phase (V1–V3) and two during each of the two treatment phases (V4–V7). At V1, the inclusion/exclusion criteria were reviewed and written informed consent obtained. Spirometry, lung volumes and diffusion capacity (DLCO) were then measured to confirm study eligibility. V2 included a spirometry, a maximal incremental shuttle walking test (ISWT) and completion of the CAT. Participants were then familiarized with the 3-min CSST by doing two practice tests. At V3, patients performed two or three 3-min CSST to determine the shuttle speed that would produce a Borg dyspnea score of at least 4 points (moderate) while being sustainable for the entire 3-min test duration. Participants then entered the first treatment phase, during which they received one of the two study treatments: indacaterol 110 μg/glycopyrronium 50 μg (Ultibro®) once daily (OD) administered in a fixed dose combination with the Breezhaler® device (Novartis AG) + placebo OD administered with the Handihaler® device (Boehringer Ingelheim) or placebo OD administered with the Breezhaler® device + tiotropium 18 μg (Spiriva®) OD administered with the Handihaler® device. The duration of each treatment phase was 3 weeks. There was a 2-week washout period between the two treatment phases. V4 and V6 were the baseline visits for each treatment phase (except for Borg dyspnea score during the 3-min CSST, which was determined at V3). Participants performed spirometry and lung volumes measurements before and 1 h 20 min after receiving the study medication. One 3-min CSST was then performed 2 h 25 min after dosing, at the shuttle speed that was determined during V3. The same procedures were repeated at the end of each study phase (V5 and V7). Total study duration was 11 weeks.

Study design.
Randomization and study blinding
At V4, treatment sequence was randomized in a 1:1 ratio and stratified by site using a centralized computerized system. A numbered envelope associated with a specific treatment order was then delivered to the study team at each site. Only the study pharmacist could associate the number on the envelope with the corresponding treatment sequence. Study blinding was only broken after completion of data analysis. Study medications were pre-packaged; placebo and active treatments had the same external appearance.
Allowed medication during the study
After confirming study eligibility and throughout the 3-week run-in period, participants received open-label ipratropium (Atrovent® MDI 20 μg/puff, four puffs QID, Boehringer Ingelheim). This medication was stopped 12 h before V3. Ipratropium was also allowed during the washout periods and was stopped 12 h before V4 and V6. As-needed salbutamol (Ventolin® MDI 100 μg/puff, two puffs every 3–4 h PRN, GlaxoSmithKline) was allowed throughout the study, except that it had to be stopped 6 h prior to V3, V4, V5, V6 and V7. Long-acting bronchodilators were prohibited after V1 and throughout the study. Inhaled corticosteroids were continued at the same dosage as before the study. Phosphodiesterase-4 inhibitors and leukotriene antagonists were also permitted.
Measurements
Pulmonary function testing
Spirometry, lung volumes and DLCO were measured according to standard operating procedures. Measured values were expressed as percentages of their normal predicted values. 24
ISWT
The ISWT was performed to characterize the exercise capacity of the study participants and to exclude those with an exercise-induced desaturation (SpO2 <85%). As originally described by Singh
Dyspnea questionnaires and CAT
The baseline dyspnea index was administered at baseline (V4 and V6), while the TDI and CAT were administered at the end of each treatment phase (V5 and V7), prior to dosing.
3-min CSST
Participants were instructed to walk in a corridor at a constant and externally imposed speed for 3 minutes and were asked to rate their dyspnea and leg fatigue with Borg’s modified 0–10 scale 27 at prespecified moments during the test and at the end. Participants were directed to follow the audio signal for the entire 3 min of the test or until they became symptom limited. They were instructed to walk between two cones set up in the hospital corridor, positioned as for the ISWT, pacing their speed in a way not to wait at the cones for the next audio signal. At V3, and following a published algorithm for shuttle speed selection, 19 patients were asked to perform at least two tests at two different speeds in order to determine, among the five different walking speeds (2.5, 3.25, 4, 5, and 6 km/h), the highest shuttle speed that could be sustained for the entire 3 min and that would produce a Borg dyspnea score of at least 4 points at end-exercise. In doing so, our objective was to induce a sufficiently high level of dyspnea to be amenable to therapy. If patients were not able to carry through the 3 min at 2.5 km/h or could not reach a Borg dyspnea score of at least 4 points at the quickest speed, they were excluded.
Cardiac and ventilatory measures
During each exercise test, minute ventilation (VE), breathing frequency (Bf) and tidal volume (VT) were measured breath-by-breath using a portable cardiopulmonary exercise testing system (Jaeger® Oxycon Mobile), calibrated according to the standard operating procedures. These values were obtained by averaging the last 15 s of each minute.
Safety monitoring
Participants were screened at each visit for any adverse event and were invited to contact the team if any symptom or medical event appeared during the study, namely a COPD exacerbation, a hospitalization, intolerance to treatment and/or any other significant event. All adverse events were reviewed to assess whether the study drugs or the testing could be the cause of these events. Vital signs and 12-lead electrocardiogram were also monitored during all exercise tests.
Statistical analysis
Sample size calculation
We calculated that 40 completed patients would be necessary to identify a change of 0.75 in Borg dyspnea score between the two treatments, with an α of 0.05 and a β of 0.85. We purposely decided to power our study to detect a Borg dyspnea score difference lower than what is considered as the clinically meaningful difference for this variable (1 point) 28 in order to maximize statistical power. Expecting that 20% of patients would fail to reach a Borg dyspnea score of 3 or more at the 3-min CSST, we planned on recruiting up to 60 participants.
Data analysis
Results are reported as mean ± SEM. For the primary outcome analysis of Borg dyspnea scores as well as for the secondary variables, comparisons of the values were made using an ANOVA model for a 2 × 2 cross-over design with repeated measurements within periods. The sources of variation included in the model were the sequence, period, treatment and time effect, as well as the following interaction terms: sequence * period, period * time, sequence * time and treatment * time. The normality assumption was verified by the Shapiro–Wilk statistic applied on the model residuals, and the homogeneity of variances by the traditional residual plots. All analyses were performed using SAS (SAS Institute Inc., NC, version 9.4) at the 0.05 significance level.
Results
Between June 2016 and November 2018, we screened 95 patients, of which 50 entered the cross-over phase of the trial (Figure 2). The majority of participants who were excluded (

Flow chart.
Baseline demographics and participants characteristics (

Data are mean (SEM) unless indicated otherwise.
ACE, angiotensin-converting enzyme inhibitors; ARB, angiotensin II receptor blockers; DLCO, carbon monoxide diffusion capacity; FEV1, forced expiratory volume in 1 s; FRC, functional residual volume; FVC, forced vital capacity; ICS, inhaled corticosteroids; LABA, long-acting β2-agonists; LAMA, long-acting muscarinic antagonists; MRC, medical research council; PDE4, phosphodiesterase-4 inhibitor.
Baseline incremental shuttle walk test (ISWT) performance and walking speed selection for the 3-min constant speed shuttle test (3-min CSST).
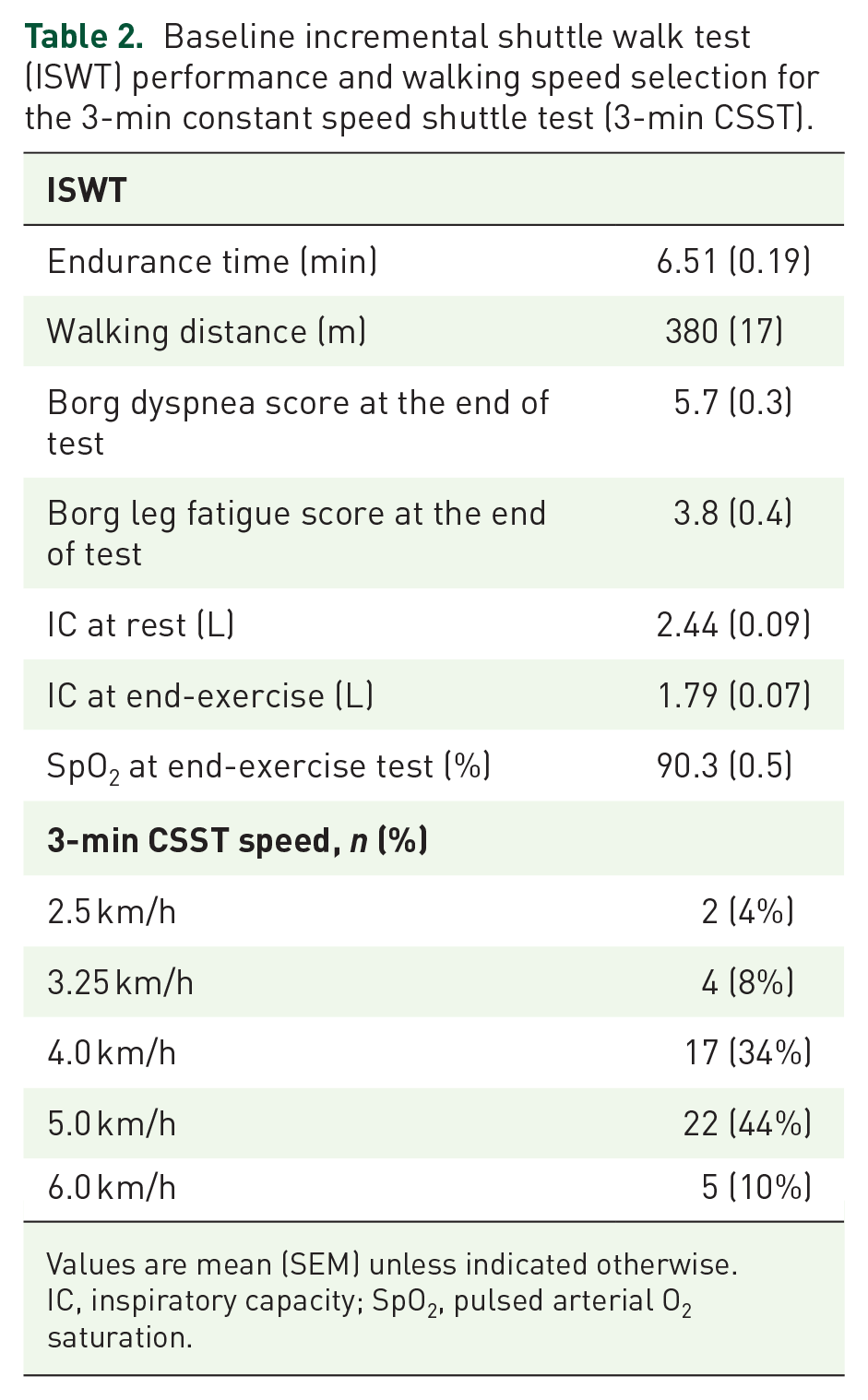
Values are mean (SEM) unless indicated otherwise.
IC, inspiratory capacity; SpO2, pulsed arterial O2 saturation.
At baseline, mean Borg dyspnea score at the end of the 3-min CSST was 5.15 (0.25). After 3 weeks of treatment (Figure 3(a); Table 3), both treatments provided statistically significant relief of exertional dyspnea. There was a 0.66 [95% confidence interval (CI) −1.21 to −0.10)]reduction in Borg dyspnea score from baseline with indacaterol/glycopyrronium while the corresponding value for tiotropium was 0.80 (95% CI −1.34 to −0.26), with a non-significant between-treatment difference of −0.15 (95% CI −0.40 to 0.70). After the first dose of study medication (Figure 3(b); Table 3), the decrease in dyspnea was significant only for indacaterol/glycopyrronium (mean −1.00, 95% CI −1.49 to −0.50) but not with tiotropium (mean −0.36, 95% CI −0.81 to 0.09), with a significant between-treatment difference of −0.63 (95% CI −1.09 to −0.17).

Borg dyspnea scores at the end of the 3-min constant speed shuttle test after 3 weeks of treatment (a), and after the first dose of study medication (b).
Borg dyspnea score at the end of the 3-min constant speed shuttle test (CSST) after 3 weeks of treatment and after the first dose.

Values are mean (SE) and 95% confidence intervals (in square brackets).
Indicates statistical significance (
Ind/Gly, indacaterol/glycopyrronium; Tio, tiotropium.
The impact of both treatment regimens on pulmonary function is provided in Table 4 and Figure 4. Both treatments significantly improved expiratory flows and this was accompanied by evidence of lung deflation, notably increased inspiratory capacity (IC) and reduced FRC. Compared with tiotropium, indacaterol/glycopyrronium proved superior, as indicated by a 90 ml difference in FEV1 and a 140–170 ml difference in slow vital capacity (SVC) (both
Pulmonary function tests before and after first dose and 3 weeks of treatment.

Values are mean (SEM) and 95% confidence intervals (in square brackets). Baseline values for the pulmonary function variables are the average of those obtained before the administration of study medication at visits 4 and 6.
Indicates statistical significance (
FEV1, forced expiratory volume in 1 s; FRC, functional residual capacity; FVC, forced vital capacity; IC, inspiratory capacity; Ind/Gly, indacaterol/glycopyrronium; RV, residual volume; SVC, slow vital capacity; Tio, tiotropium.

Changes from baseline in slow vital capacity (SVC), forced vital capacity (FVC), forced expiratory volume in 1 s (FEV1), inspiratory capacity (IC), functional residual capacity (FRC) and residual volume (RV) after the first dose ((a) and (c)) or after 3 weeks of therapy ((b) and (d)) of either indacaterol/glycopyrronium or tiotropium alone. Results are expressed in L ((a) and (b)) and in percentage change from baseline ((c) and (d)).
The effects of indacaterol/glycopyrronium and tiotropium on TDI and CAT scores are shown in Table 5. As can be seen, at week 3 there was a 0.99-point improvement in TDI with indacaterol/glycopyrronium leading to a 1.27-point between-treatment difference. Although this value is considered to be above the clinically meaningful threshold for this variable, 29 this difference did not reach statistical significance. Changes in CAT scores with both therapies were small and statistically non-significant.
Transitional dyspnea index (TDI) total scores and COPD Assessment Test scores (CAT) after 3 weeks of treatment.

Values are mean (SEM) and 95% confidence intervals (in square brackets). Baseline values for the CAT are the average of those obtained before the administration of study medication at visits 4 and 6.
Indicates statistical significance (
CAT, COPD Assessment Test; COPD, chronic obstructive pulmonary disease; Ind/Gly, indacaterol/glycopyrronium; TDI, transition dyspnea index; Tio, tiotropium.
Cardiopulmonary responses to exercise are depicted in Figure 5. Compared with baseline, indacaterol/glycopyrronium led to small reductions in Bf with increases in VT, whose statistical significance was variable over time during the 3-min CSST and visits. There were no statistically significant differences for these variables between the two treatments.

Breathing frequency (Bf), tidal volume (VT) and minute ventilation (VE) during the 3-min constant speed shuttle test after the first dose of study medication ((a), (c), and (e)) and after 3 weeks of treatment ((b), (d) and (f)).
No safety concerns were seen with either medication. Seven patients experienced a COPD exacerbation during the trial (indacaterol/glycopyrronium,
Adverse events.
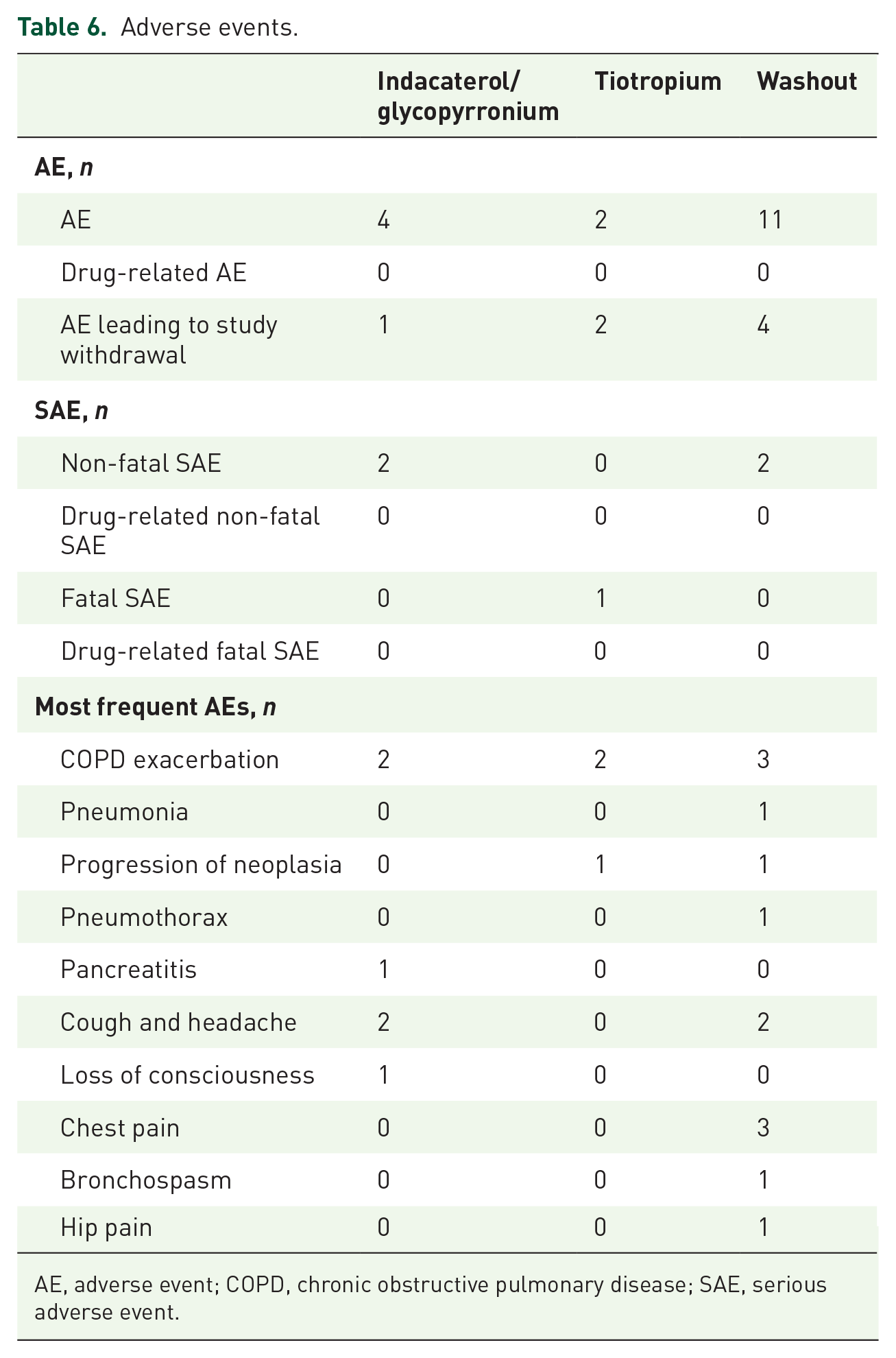
AE, adverse event; COPD, chronic obstructive pulmonary disease; SAE, serious adverse event.
Discussion
In the present study, indacaterol/glycopyrronium provided greater bronchodilation and lung deflation compared with tiotropium alone, and this translated into further exertional dyspnea relief after the first dosage of medication. Despite maintaining its efficacy to improve expiratory flows and lung volumes throughout the study, the added benefits of indacaterol/glycopyrronium on exertional dyspnea were not apparent after 3 weeks of treatment.
Two active bronchodilator treatments were assessed and compared in this trial. As expected from their known efficacy profile, both indacaterol/glycopyrronium and tiotropium provided potent bronchodilation and lung deflation on order of magnitude consistent with previous reports.8,20,30 These beneficial effects on lung function were accompanied by a significant reduction in dyspnea intensity ratings at the end of the 3-min CSST immediately after the first dose of indacaterol/glycopyrronium, and after 3 weeks. Tiotropium alone reduced exertional dyspnea perception after 3 weeks of treatment but not after the first dose, a finding that is consistent with the previously reported progressive efficacy build-up over a 3–6-week course of treatment. 7 As a result of these differential responses to indacaterol/glycopyrronium and tiotropium, there was a clinically meaningful one-point reduction in Borg dyspnea score 28 at the end of the 3-min CSST in favor of indacaterol/glycopyrronium after the first dose but this difference was not sustained after 3 weeks of therapy.
In line with previous trials,20,30 we found that indacaterol/glycopyrronium provided superior bronchodilation, as evidenced by a 90 ml difference in trough FEV1
Our findings are somewhat at variance with the OTIVATO trial, in which dual bronchodilation with tiotropium/olodaterol was associated with a statistically significant 0.36-point reduction in Borg dyspnea score compared with tiotropium after 6 weeks of therapy,
19
as opposed to a non-significant between-treatment difference in the present study. There are several potential explanations for this apparent discrepancy between the two trials. First, the time frame was different; in OTIVATO, each treatment phase lasted 6 weeks as compared with 3 weeks in the present trial. In this regard, it is interesting to note that in the OTIVATO trial, the further reduction in exertional dyspnea between dual
Whether or not dual bronchodilation further improves dyspnea and exercise capacity outcomes over what can be achieved with one long-acting bronchodilator alone has been a long-standing debate because clear treatment differences do not always emerge in individual trials.30,37,38 We consider that the present results are in line with the clinical research experience in comparing dual
The 3-min CSST was designed to allow a specific evaluation of the impact of therapies such as bronchodilators on exertional dyspnea. 16 Because of its fixed duration, the 3-min CSST does not allow us to quantify the impact of therapies on exercise capacity. Together with the OTIVATO trial, we showed that the 3-min CSST can be used in the context of multicenter trials. With this testing modality, in both trials we succeeded in generating clinically important ratings of exertional dyspnea that were amenable to therapy (Borg ⩾ 4). There are, however, some limitations to our trial. The relatively small number of patients, even with a cross-over design, could have underpowered this trial and makes the generalizability of the data uncertain. Despite the fact that this was taken into consideration in the statistical plan, a carry-over effect with this type of design cannot be entirely excluded. Moreover, although we elected for a relatively short 3-week treatment period in order to minimize participant attrition, this decision might have influenced the possibility for the dual bronchodilation to fully express its potential to further alleviate exertional dyspnea compared with monobronchodilation.
Activity-related dyspnea can be assessed with specific task-based dyspnea questionnaires such as the TDI. One important difference between using a task-based questionnaire
Conclusion
In comparison with bronchodilator monotherapy, dual bronchodilation provided greater immediate exertional dyspnea relief but this difference was not sustained after 3 weeks of therapy despite sustained expiratory flow and lung deflation benefits throughout the trial.
Supplemental Material
Reviewer_1_v.1 – Supplemental material for Relieving exertional dyspnea during the 3-min constant speed shuttle test in patients with COPD with indacaterol/glycopyrronium versus tiotropium: the RED trial
Supplemental material, Reviewer_1_v.1 for Relieving exertional dyspnea during the 3-min constant speed shuttle test in patients with COPD with indacaterol/glycopyrronium versus tiotropium: the RED trial by Jessie Beaulieu, Dennis Jensen, Denis E. O’Donnell, Cynthia Brouillard, Lauren Tracey, Sandra Vincent, Éric Nadreau, Emmanuelle Bernard, Sarah Bernard and François Maltais in Therapeutic Advances in Respiratory Disease
Supplemental Material
Reviewer_2_v.1 – Supplemental material for Relieving exertional dyspnea during the 3-min constant speed shuttle test in patients with COPD with indacaterol/glycopyrronium versus tiotropium: the RED trial
Supplemental material, Reviewer_2_v.1 for Relieving exertional dyspnea during the 3-min constant speed shuttle test in patients with COPD with indacaterol/glycopyrronium versus tiotropium: the RED trial by Jessie Beaulieu, Dennis Jensen, Denis E. O’Donnell, Cynthia Brouillard, Lauren Tracey, Sandra Vincent, Éric Nadreau, Emmanuelle Bernard, Sarah Bernard and François Maltais in Therapeutic Advances in Respiratory Disease
Footnotes
Acknowledgements
The authors thank Gaetan Daigle MSc who performed the statistical analyses and Mickaël Martin for his assistance in preparing the figures.
Author contribution(s)
Conflict of interest statement
DJ reports grants from Boehringer Ingelheim and Novartis during the conduct of this study; grants and personal fees from AstraZeneca, and grants from Tilray outside the submitted work. DEO reports grants from AstraZeneca and Boehringer Ingelheim during the conduct of this study; and personal fees for serving on speaker bureaus, consultation panels and advisory boards from Almirall, AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Novartis and Pfizer outside the submitted work. FM reports grants from AstraZeneca, GlaxoSmithKline, Boehringer Ingelheim, Sanofi, and Novartis during the conduct of this study, and personal fees for serving on speaker bureaus and consultation panels from Boehringer Ingelheim, Grifols and Novartis outside the submitted work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This investigator-initiated research was funded by Novartis AG. LT was funded by a Canada Graduate Scholarship – Master Award (CGS-M) from the Canadian Institutes of Health Research (CIHR). DJ holds a Canada Research Chair in Clinical Exercise and Respiratory Physiology (Tier 2) from the Canadian Institutes of Health Research.
Supplemental material
The reviews of this paper are available via the supplemental material section.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
