Abstract
Background:
Non-invasive ventilation (NIV) was one of the first-line ventilation supports for hematopoietic stem-cell transplantation (HSCT) patients with acute respiratory distress syndrome (ARDS). Successful NIV may avoid need for intubation. However, the influence NIV failure had on patients’ outcome and its risk factors were hardly known.
Methods:
In this retrospective observational study, we reported risk factors and incidence of NIV failure in HSCT patients who were admitted to the Intensive Care Unit (ICU) with a diagnosis of ARDS and supported with mechanical ventilation, in a 5-year period. Patient outcomes, such as ventilator-free days, ICU-free days, and ICU mortality were also reported.
Results:
Of all the 94 patients included, 70 patients were initially supported with NIV. NIV failure occurred in 44 (63%) patients. Male sex, elevated serum galactomannan (GM) test, (1-3)-β-D-glucan (BG) assay, or elevated serum creatinine level were risk factors for NIV failure. When compared with the NIV success group, failure of NIV was associated with much fewer ICU-free days (22 versus 0, p < 0.001, Cohen’s d = 0.62) and higher ICU mortality (9.5% versus 75.5%, p < 0.001, Pearson’s r = 0.75). There was no difference in ICU-free days, ventilator-free days and ICU mortality between NIV failure and initial invasive mechanical ventilation (IMV) groups. Patients who failed in NIV support had a higher ICU mortality (75.5%) than those who succeeded (9.5%).
Conclusion:
In a small cohort of HSCT patients with mainly moderate severity of ARDS, male patients with elevated serum GM/BG test or serum creatinine level had a higher risk of NIV failure. Both NIV failure and initial IMV groups were characterized by high mortality rate and extremely low ICU-free days and ventilator-free days; failure of NIV support may further aggravate patient prognosis.
The reviews of this paper are available via the supplemental material section.
Keywords
Introduction
Hematopoietic stem-cell transplantation (HSCT) provides best cure for many hematological diseases. Pulmonary complications of infectious or non-infectious origin are common and often lethal among HSCT patients. Lucena and colleagues reported that in a prospective cohort of 169 HSCT patients, 32% of 1-year mortality is due to pulmonary complications. 1 Characterized by acute onset and bilateral lung infiltrates, acute respiratory distress syndrome (ARDS) is one type of pulmonary complication with various causes. Cortegiani and colleagues 2 reported that the leading cause of ARDS in immunocompromised patients are infections. While in allo-HSCT patients, ARDS has an incidence of about 15%, with an intensive care unit (ICU) mortality of nearly 50%. 3
HSCT patients with ARDS are in a hypoxemic state which requires mechanical ventilation support. However, the best ventilation strategy for this specified group of patients is yet to be decided. In the 2017 European Respiratory Society/American Thoracic Society guideline of non-invasive ventilation (NIV) for acute respiratory failure, 4 NIV was recommended as an early support method in immunocompromised patients. Different studies5–7 have also evaluated the effect of NIV in hematological patients with respiratory failure, but all studies involved both HSCT and non-HSCT patients. Previous studies8,9 of immunocompromised ARDS patients have shown that NIV can reduce the rate of intubation and mortality when compared with standard oxygen therapy in some studies; however, recent research10,11 failed to confirm these findings. Also, there is more NIV failure in immunocompromised patients than nonimmunocompromised ones, 2 and for hematological patients, NIV failure is associated with worse outcome when compared with invasive mechanical ventilation (IMV). 12 For HSCT patients, no specific studies are available yet.
The aim of this study is to determine risk factors for NIV failure in HSCT patients with ARDS and describe the prognosis for these patients.
Methods
The Research and Ethics Committee of Peking University People’s Hospital approved this non-interventional retrospective study on 6 July 2019 (approval number: H19REA-017).
Patients
All data of consecutive patients admitted to Peking University People’s Hospital between January 2014 and December 2018 from our research database were reviewed. JS did the record review and data collection, and YH checked the accuracy of data collection. The inclusion criteria were as follows:
(1) adult patient (age ⩾ 18 years) who had received HSCT;
(2) diagnosed with ARDS.
The exclusion criteria were:
(1) age < 18 years;
(2) using NIV as palliative support;
(3) patients having a decision that precluded intubation.
ARDS was diagnosed according to the Berlin definition. 13 Choice of therapy with NIV or intubation and IMV was at the discretion of the clinicians. In our ICU, the NIV is performed with a Philips V60 ventilator (Philips Respironics, Pittsburgh, PA, USA) with a full-face mask, and IMV with Dräger Infiniti v500 ventilator (Dräger Medical, Lübeck, Germany). The inspiratory pressure or target tidal volume, positive-end expiratory pressure (at least 5 cm H2O) and inhaled oxygen concentration were modified to maintain oxygen saturation ⩾ 90%, a tidal volume 6–10 ml/kg of ideal body weight and respiratory rate ⩽ 30 breath/min. Ventilator settings were adjusted based on continuous monitoring of oxygen saturation, respiratory rate, and arterial blood gas.
NIV failure was defined as loss of consciousness or inability to maintain airway patency, or failure to maintain oxygen saturation ⩾ 90% at fraction of inspired oxygen (FiO2) of 100%, or respiratory rate ⩾ 40 breath/min for 1 h. Intubation and IMV were initiated when NIV failure occurred. The criteria were made based on previous studies14–16 and our department’s protocol during the study period. If oxygen saturation cannot be sustained, clinicians will increase the FiO2 stepwise and closely monitor the status of the patient to ensure timely intubation. Similarly, sustained elevation of respiratory rate was used instead of arterial partial pressure of carbon dioxide (PCO2) for prompt intervention without need of waiting for the arterial blood gas (ABG) test.
Data collection
In our database, the following data were recorded or evaluated at ICU admission: (a) demographical characteristics of patients, including sex, age, body mass index; (b) characteristics of HSCT: diagnosis of primary disease, post-transplantation days (days between HSCT and ARDS), donor relation, source of stem cells, allogenic type, acute graft versus host disease (GVHD) at the time of admission; (c) patient status: Acute Physiology and Chronic Health Evaluation II (APACHE II) score, Sequential Organ Failure Assessment (SOFA) score, ABG (evaluated both on admission and after 4 hours’ NIV support) and partial pressure of oxygen (PO2)/FiO2 (P/F Ratio) derived from it, total blood count, blood biochemical examinations; (d) blood biomarkers of infection: C-reactive protein (CRP), procalcitonin (PCT), galactomannan (GM) test, (1-3)-β-D-glucan (BG) assay.
Other data were recorded on the day of discharge or death: (a) dosage of corticosteroid per day; (b) NIV failure rate and associated risk factors; (c) ICU mortality and associated risk factors; and (d) ICU-free and ventilator-free days. Definitions of ICU-free and ventilator-free days are in Appendix 1. In addition, survival time was recorded separately 1 year after admission.
Statistical analysis
The primary objective of our study was to examine the risk factors of NIV failure in this HSCT cohort and describe patient outcome, including ICU mortality, overall survival time, ICU-free and ventilator-free days.
Collected data were recorded into Excel (Microsoft, Redmond, WA, USA) files, while in data analyzing, data files were transcribed into SPSS (version 25, SPSS Inc., Chicago, IL, USA) formatted records and processed. Normal, distributed, continuous variables were expressed as mean ± standard deviation, non-normal, distributed, continuous variables were expressed as median (25–75%). Categorical variables were described as absolute value and percentage. Test of normal distribution was achieved using the Kolmogorov–Smirnov test, and homogeneity of variance using Levene’s test. Comparison of continuous variables among three groups was achieved using the analysis of variance test or the Kruskal–Wallis test, as appropriate. Comparison of two groups of continuous variables was achieved using the Mann–Whitney U test. Comparison of categorical variables was performed using the χ2 test or Fisher’s exact test, as appropriate. Effect sizes of the associations between NIV failure and patient outcomes were reported with Cohen’s d or Pearson’s r, as appropriate. The univariate and multivariate logistic models were used to investigate the effect of demographical and HSCT characteristics, laboratory tests and ICU treatment on NIV failure and ICU mortality. Based on results of previous studies, variables that may be associated with NIV failure and ICU mortality were tested using univariate analysis; only variables with a p value < 0.1 entered the multivariate logistic regression model. A Cox regression curve was constructed to depict the survival rate of patients with a different course of ventilation support, with risk factors of mortality analyzed simultaneously. All tests were two sided and p < 0.05 was considered statistically significant.
Results
Patients
During the study period, 407 hematology patients were admitted to our ICU, including 109 patients that met our inclusion criteria. However, data of survival time were missing in 15 patients, then 94 patients were included for the final analysis.
Figure 1 describes the course of therapy and survival conditions for all included patients. Among these ARDS patients, 70 patients have received NIV as IMV support, and another 24 patients started with IMV (referred to as initial IMV hereafter). NIV failure occurred in 44 (63%) patients with initial NIV, another 26 (37%) patients succeeded with an NIV course, avoiding intubation (these two groups will be referred as NIV success and NIV failure, hereafter). The baseline characteristics of included patients are reported in Table 1. The characteristics were similar between the groups, except for sex ratio (between NIV success and NIV failure groups) and SOFA score (higher in the initial IMV group than in the other two groups).

Course of therapy and survival condition for enrolled patients.
Baseline characteristics of enrolled patients.
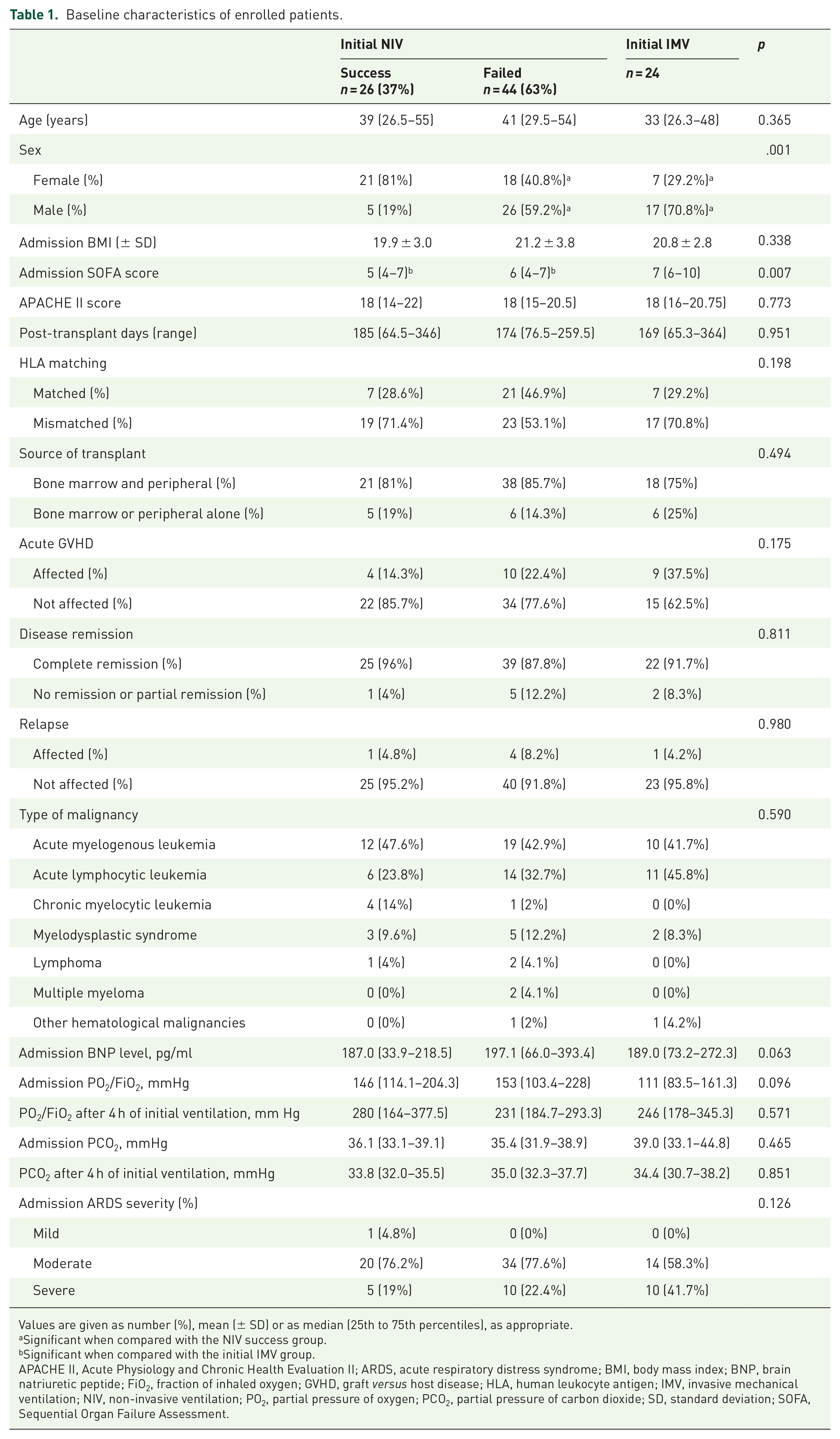
Values are given as number (%), mean (± SD) or as median (25th to 75th percentiles), as appropriate.
Significant when compared with the NIV success group.
Significant when compared with the initial IMV group.
APACHE II, Acute Physiology and Chronic Health Evaluation II; ARDS, acute respiratory distress syndrome; BMI, body mass index; BNP, brain natriuretic peptide; FiO2, fraction of inhaled oxygen; GVHD, graft versus host disease; HLA, human leukocyte antigen; IMV, invasive mechanical ventilation; NIV, non-invasive ventilation; PO2, partial pressure of oxygen; PCO2, partial pressure of carbon dioxide; SD, standard deviation; SOFA, Sequential Organ Failure Assessment.
Onset time and characteristics of NIV failure
The onset time of NIV failure is depicted in Figure 2, most (65.9%) of the NIV failure occurred within 5–10 days of NIV support initiation, even though the onset time of NIV failure ranged from 0 to 42 days. Characteristics of the NIV failure were divided into three subtypes: (a) acute loss of consciousness (Glasgow Coma Scale score < 8); (b) conscious patients with oxygenation failure (PO2 in ABG < 60 mmHg); (c) conscious patients with worsening hypercapnia (PCO2 in ABG > 50 mmHg). As depicted in Figure 3, at the time of NIV failure onset, 87% of NIV patients had oxygenation failure, 11% had worsening hypercapnia, and only 2% of NIV patients had acute loss of consciousness. In the NIV failure group, 41% (18/44) of patients had an improvement of P/F ratio of at least 100 mmHg after 4 hours’ NIV support.
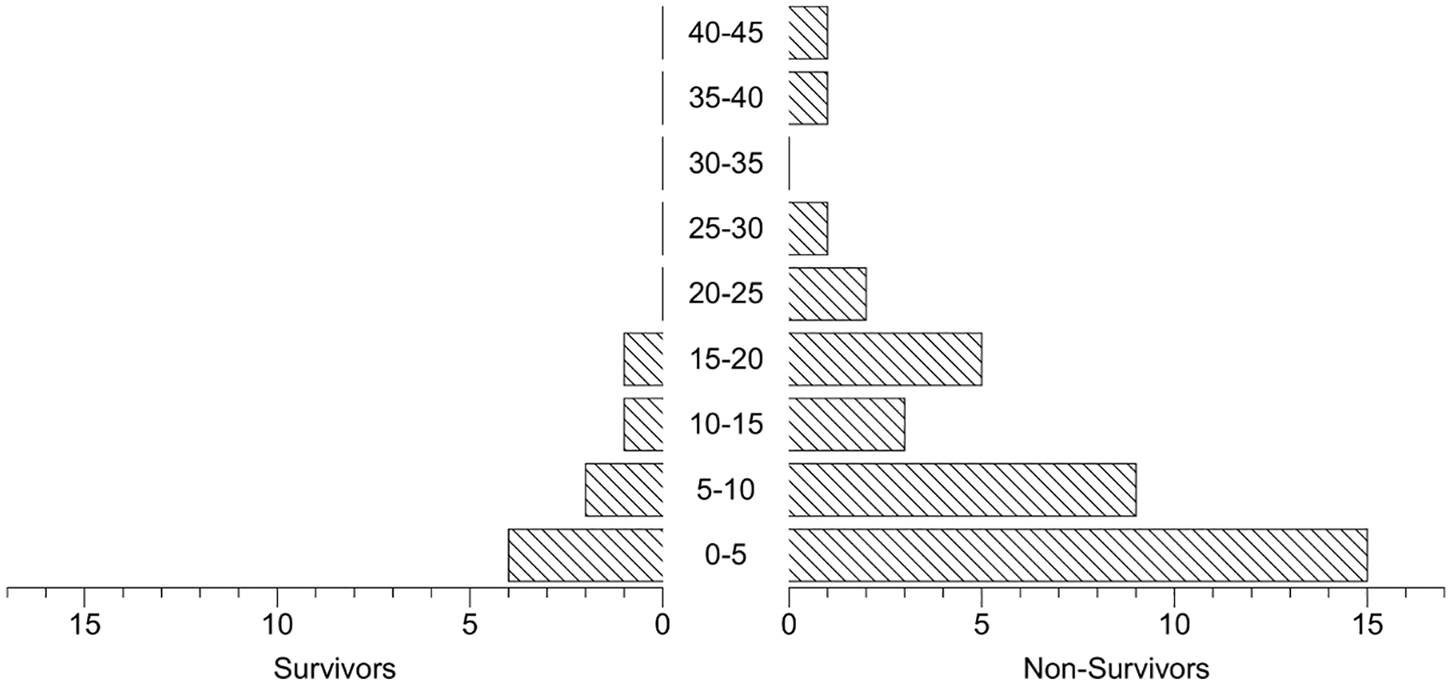
Time (days) to intubation since NIV initiated.

Characteristics of NIV failure.
Risk factors of NIV failure
Based on previous studies, factors that may be associated with NIV failure were analyzed with univariate analysis, including creatinine, serum BG/GM assay, sex, mean steroid dosage per day, blood neutrophil/lymphocyte count, SOFA score, mean arterial pressure, pH value in ABG, PO2/FiO2 before NIV support. Only four factors with a p value < 0.1 entered the multivariate logistic regression model, as described in Table 2. The factors that remained in the model after processing included high serum BG assay [>50 pg/ml, odds ratio (OR) 4.06, 95% confidence interval (CI) 1.13–17.05, p = 0.043], high serum GM assay (>0.2 pg/ml, OR 4.15, 95% CI 1.18–15.85, p = 0.038), high blood creatinine level (>95 umol/l, OR 8.09, 95% CI 1.90–34.39, p = 0.005), and male sex (OR 5.95, 95% CI 1.58–22.37, p = 0.008).
Risk factors of failed NIV.

BG, (1-3)-β-D-glucan; CI, confidence interval; FiO2, fraction of inhaled oxygen; GM, galactomannan; MAP, mean arterial pressure; NIV, non-invasive ventilation; OR, odds ratio; PO2, partial pressure of oxygen; SOFA, Sequential Organ Failure Assessment.
Patient outcomes
Patient outcomes are reported in Table 3. When compared with NIV success group, failure of NIV was associated with extremely low ICU-free days (22 versus 0, p < 0.001) and higher ICU mortality (9.5% versus 75.5%, p < 0.001). The effect size of the association between NIV failure and ICU-free days was calculated with Cohen’s d, which was 0.62. The effect size of the association between NIV failure and ICU mortality was calculated with Pearson’s r, which was 0.75, while there was no difference of ICU-free days, ventilator-free days, and ICU mortality between NIV failure and initial IMV groups. In Table 4, aforementioned outcomes in different subgroups of risk factors of NIV failure are provided; patients with elevated serum GM assay had higher ICU mortality (79.5% versus 50.9%, p = 0.005) and lower ICU-free days (0 versus 0, p = 0.006). Although mean and 25th–75th values were the same between two groups, there were significant (asymptotic significance for two sides: 0.006) differences between two groups by Mann–Whitney U test. The finding was that more patients with a normal GM test had more ICU-free days than patients in the other group. In addition, patient with male sex had higher ICU mortality than females (80% versus 43.2%, p = 0.008).
Outcomes of patients with initial NIV or IMV.

Values are given as median (25th to 75th percentiles) or number (%).
Significant when compared with NIV success group.
Cohen’s d = 0.62.
Pearson’s r = 0.75.
ICU, intensive care unit; IMV, invasive mechanical ventilation; N/A, not applicable; NIV, non-invasive ventilation.
Outcomes of patients influenced by risk factors of NIV failure.

Values are given as median (25th to 75th percentiles) or number (%).
Only applicable for patients with NIV failure and initial IMV.
BG, (1-3)-β-D-glucan; GM, galactomannan; ICU, intensive care unit; IMV, invasive mechanical ventilation; NIV, non-invasive ventilation.
Risk factors for ICU mortality are reported in Table 5. After multivariate analysis, there were three factors remaining in the model: longer time from ARDS onset to intubation (>7 days, OR 3.70, 95% CI 1.182–11.582, p = 0.025), high serum GM assay (>0.2 pg/ml, OR 3.62, 95% CI 1.171–11.198, p = 0.025), while female sex was associated with lower ICU mortality (OR 0.26, 95% CI 0.087–0.777, p = 0.16).
Risk factors of ICU mortality.

Time from ARDS onset to intubation, for initial IMV patients, the value was 0.
ARDS, acute respiratory distress syndrome; BG, (1-3)-β-D-glucan; CI, confidence interval; GM, galactomannan; ICU, intensive care unit; IMV, invasive mechanical ventilation; NIV, non-invasive ventilation; OR, odds ratio.
Figure 4 depicts the survival curve of patients with different ventilation supports using Cox regression analysis, adjusted for underlying differences between groups (sex and admission SOFA score) and possible risk factors of ICU mortality (time to intubation, serum GM assay, creatinine). Log-rank test showed that when compared with the NIV success group, survival rate was significantly lower in the NIV failure (p < 0.001) and initial IMV groups (p < 0.001), while between the latter two groups, there was no difference (p = 0.395).
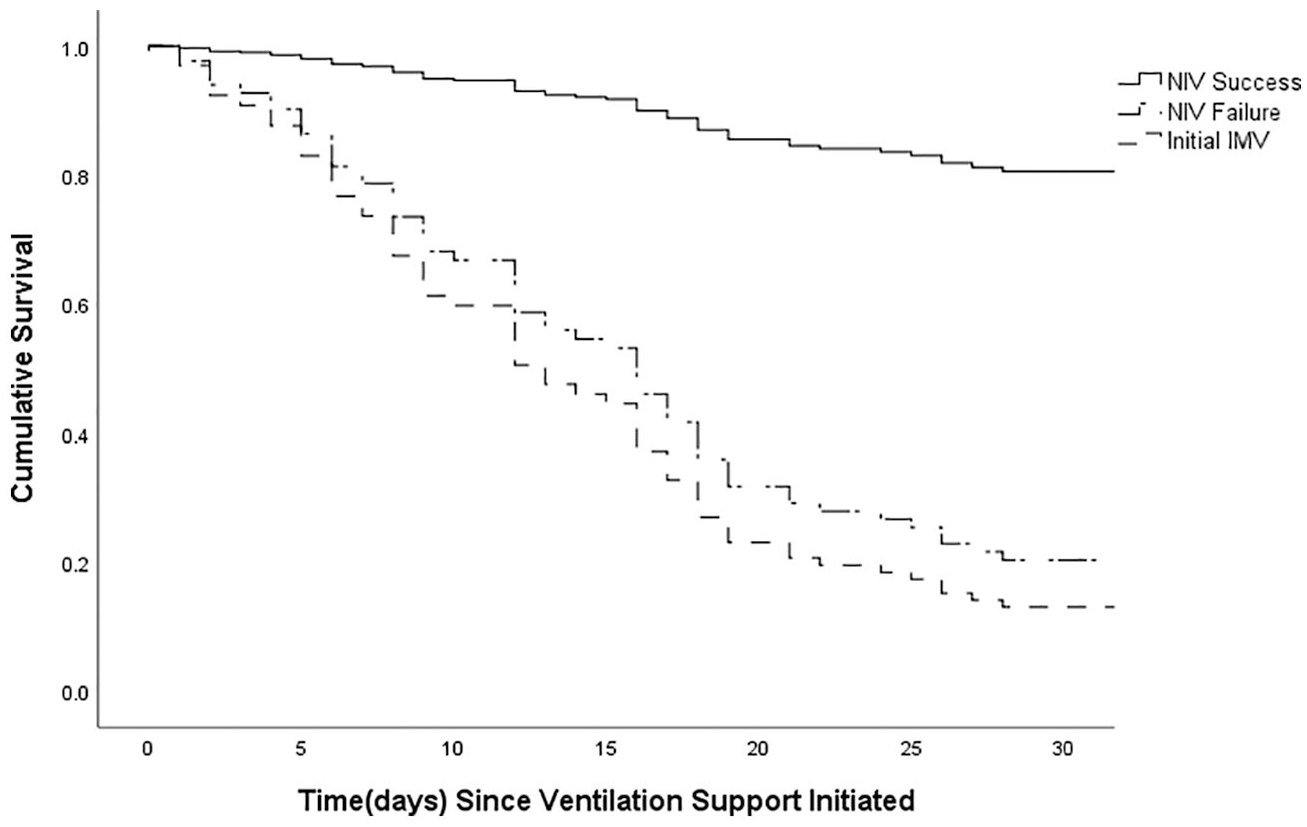
Cox regression curves for the probability of survival among patients having NIV success, NIV failure and initial NIV groups after adjusting for baseline differences and risk factors of ICU mortality.
Discussion
To our knowledge, this is the first study focusing on failure of NIV in HSCT patients with ARDS. A sum of 37% patients in our study avoided intubation through the support of NIV, while 63% of NIV-supported HSCT patients failed the initial therapy, requiring intubation and IMV. In a previous study by Molina and colleagues, 17 hematological patients with ARDS had a high failure rate (60.3%) of NIV. In their cohort, only 23.7% (71/300) were HSCT patients, while patients included in our study were all after HSCT. In a previous study of various populations,15,16,18 rate of NIV failure varied from 37% to 54%. It is clear that HSCT patients have a higher rate of NIV failure after ARDS.
Onset time and characteristics of NIV failure
Previous studies8,19,20 reported that most incidents of NIV failure happened in the first 48 h following support initiation, while in our study, a large proportion (63%, Figure 2) of NIV failure occurred within 10 days of NIV therapy. In our cohort, most cases (83%) of NIV failure onset were characterized by oxygenation failure (Figure 3). For patients who failed NIV, 49.4% of them had an elevated P/F ratio of at least 100 mmHg after 4 h of NIV therapy. Thus, HSCT patients with ARDS often have only impaired oxygenation function and have a rather wide range of NIV failure onset time than other groups; early improvement of oxygenation cannot preclude late failure incidence, which suggests that identifying risk factors of NIV failure is critical for prognostic estimation.
Risk factors of NIV failure
High level of serum G/GM assay (>50 pg/ml for G assay and >0.2 pg/ml for GM assay) was a leading factor of NIV failure in this study. G assay, and especially GM assay, are sensitive biomarkers for fungal infections, which can effectively predict invasive fungal disease in HSCT patients.21–23 Invasive fungal infections, including fungal pneumonia, occurs frequently and is associated with high mortality in the HSCT population 24 despite preventive antifungal therapy, and often requires mechanical ventilation support.25,26 A multinational cohort study by Azoulay and colleagues reported that immunocompromised patients with pulmonary fungal infections had high risk of intubation and IMV. 27 Also, as depicted in Tables 4 and 5, a serum GM assay higher than 0.2 pg/ml was associated with elevated ICU mortality and very few ICU-free days for both NIV and initial IMV patients. Therefore, HSCT patients with fungal infection were at high risk of NIV failure, and their prognoses were poor.
In addition, the multivariate regression indicated that an increased creatinine level (>95 umol/l) was associated with NIV failure, which is in accordance with previous studies.7,28 Renal failure and multiple organ failure are risk factors for ICU mortality in HSCT patients,29,30 and also a relative contraindication of NIV. 31 However, in our study cohort, there was no significant difference of admission SOFA score, as an indicator of multiple organ failure, between NIV success and NIV failure groups. It is possible that the severity of organ failure may be aggravated over time, as reported in other studies,2,7 but related data were not available in the present research. In order to prevent NIV failure, it is reasonable to evaluate organ functions regularly in this population. In our study, male sex was also identified as a risk factor of NIV failure, which has been reported in other studies.12,32 Some researchers 12 have conjectured that this may be caused by higher cardiovascular risks in males, which could not be tested with the data we had.
The severity of ARDS has been reported as a strong risk factor for NIV failure in several studies,7,15,17,18 which focused on hematological and other types of patients. Nevertheless, this correlation did not exist in the current study. It can be explained by the composition of our cohort; patients with initial NIV were mainly in the moderate-to-severe class of ARDS, and there was only one patient with mild severity, which makes comparison based on severity ineffective.
Outcome of included patients
In this study cohort, patients with NIV failure had a significantly higher rate of ICU mortality when compared with the NIV success group (Table 3). Through multivariate analysis, one risk factor of ICU mortality discovered by logistic regression was the elevated serum GM assay level (>0.2 pg/ml). As reported in a previous study, 24 GM had a high specificity for invasive aspergillosis, which was one of the leading causes of mortality and poor prognosis in HSCT patients. No difference of survival was found between NIV failure and initial IMV groups, as depicted by the Cox regression-derived survival curve (Figure 4) that adjusted for possible risk factors of ICU mortality. Moreover, patients in both groups had extremely low ventilator-free days and ICU-free days (Table 3).
Another risk factor was longer time (>7 days) elapsed from ARDS onset to intubation. This could be explained by insufficient ventilation support provided by NIV and the delay of more effective IMV therapy. As mentioned previously, patients with initial IMV support were in a more severe condition than those with NIV, similarly poor outcomes in NIV failure and initial IMV groups indicate that the failure of NIV support may further aggravate patient prognosis, or, as supported by previous studies,33,34 NIV may not be appropriate for all ARDS patients. Recent studies2,27 on immunocompromised patients also showed no advantage of NIV as first-line therapy for ARDS, in terms of lowering mortality and intubation rate. This phenomenon also prompts making more rational criteria for NIV support and developing proper methods to discriminate patients with risk of NIV failure, to improve patient outcomes in this particular population. However, no recommendations can be made, based on our descriptive study. Research with more rigorous methodologies are needed to further evaluate the role of NIV in HSCT patients.
Limitations
Our study had several limitations. First, as a result of the single center and retrospective nature, we cannot rule out possible confounding factors for patients’ prognosis and NIV failure. Also, our unit is experienced in NIV support for HSCT patients; these experiences may not be applicable to other centers with less experience of mechanical ventilation in hematological patients. Second, the small dataset may impede the analysis of possible risk factors in this population, and, as mentioned previously, this study was unable to evaluate evolution of disease severity overtime (e.g. SOFA score, infection biomarkers, etc.), as aggravation of disease severity may be related to NIV failure and higher mortality.7,35 However, this is the first study focused on NIV failure in HSCT patients with ARDS. Our findings that failed NIV was associated with a worse outcome should prompt more methodologically rigorous studies on this topic.
Conclusion
In a small cohort of HSCT patients with mainly moderate severity of ARDS, NIV failure had a high incidence and was associated with increased mortality. Patients with male sex, elevated serum GM/BG test or serum creatinine level had a higher risk of NIV failure. Both NIV failure and initial IMV groups were characterized by a high mortality rate and very few ICU-free and ventilator-free days; failure of NIV support may further aggravate patient prognosis.
Supplemental Material
Appendix_A._Definition_of_ICU_Free_Days_and_Ventilator_Free_Days – Supplemental material for Risk factors of non-invasive ventilation failure in hematopoietic stem-cell transplantation patients with acute respiratory distress syndrome
Supplemental material, Appendix_A._Definition_of_ICU_Free_Days_and_Ventilator_Free_Days for Risk factors of non-invasive ventilation failure in hematopoietic stem-cell transplantation patients with acute respiratory distress syndrome by Jiawei Shen, Yan Hu, Huiying Zhao, Zengli Xiao, Lianze Zhao, Anqi Du and Youzhong An in Therapeutic Advances in Respiratory Disease
Supplemental Material
Author_Response_1 – Supplemental material for Risk factors of non-invasive ventilation failure in hematopoietic stem-cell transplantation patients with acute respiratory distress syndrome
Supplemental material, Author_Response_1 for Risk factors of non-invasive ventilation failure in hematopoietic stem-cell transplantation patients with acute respiratory distress syndrome by Jiawei Shen, Yan Hu, Huiying Zhao, Zengli Xiao, Lianze Zhao, Anqi Du and Youzhong An in Therapeutic Advances in Respiratory Disease
Supplemental Material
Reviewer_1_v.1 – Supplemental material for Risk factors of non-invasive ventilation failure in hematopoietic stem-cell transplantation patients with acute respiratory distress syndrome
Supplemental material, Reviewer_1_v.1 for Risk factors of non-invasive ventilation failure in hematopoietic stem-cell transplantation patients with acute respiratory distress syndrome by Jiawei Shen, Yan Hu, Huiying Zhao, Zengli Xiao, Lianze Zhao, Anqi Du and Youzhong An in Therapeutic Advances in Respiratory Disease
Supplemental Material
Reviewer_2_v.1 – Supplemental material for Risk factors of non-invasive ventilation failure in hematopoietic stem-cell transplantation patients with acute respiratory distress syndrome
Supplemental material, Reviewer_2_v.1 for Risk factors of non-invasive ventilation failure in hematopoietic stem-cell transplantation patients with acute respiratory distress syndrome by Jiawei Shen, Yan Hu, Huiying Zhao, Zengli Xiao, Lianze Zhao, Anqi Du and Youzhong An in Therapeutic Advances in Respiratory Disease
Footnotes
Author contribution(s)
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Supplemental material
The reviews of this paper are available via the supplemental material section.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
