Abstract
Acute respiratory distress syndrome (ARDS) is a syndrome of acute lung injury that is characterized by noncardiogenic pulmonary edema and severe hypoxemia second to a pathogenic impairment of gas exchange. Despite significant advances in the area, mortality remains high among ARDS patients. High mortality and a limited spectrum of therapeutic options have left clinicians searching for alternatives, spiking interest in selective pulmonary vasodilators (SPVs). Despite the lack of robust evidence, SPVs are commonly employed for their therapeutic role in improving oxygenation in patients who have developed refractory hypoxemia in ARDS. While inhaled epoprostenol (iEPO) also impacts arterial oxygenation by decreasing ventilation–perfusion (V/Q) mismatching and pulmonary shunt flow, this effect is not different from inhaled nitric oxide (iNO). The most effective and safest dose for yielding a clinically significant increase in PaO2 and reduction in pulmonary artery pressure (PAP) appears to be 20–30 ng/kg/min in adults and 30 ng/kg/min in pediatric patients. iEPO appears to have a ceiling effect above these doses in which no additional benefit may be derived. iNO and iEPO have shown similar efficacy profiles; however, they differ with respect to cost and ease of therapeutic administration. The most beneficial effects of iEPO have been seen in adult patients with secondary ARDS as compared with primary ARDS, most likely due to the difference in etiology of the two disease states, and in patients suffering from baseline right ventricular heart failure. Although iEPO has demonstrated improvements in hemodynamic parameters and oxygenation in ARDS patients, due to the limited number of randomized clinical trials and the lack of studies investigating mortality, the use of iEPO cannot be recommended as standard of care in ARDS. iEPO should be reserved for those refractory to traditional therapies.
Background
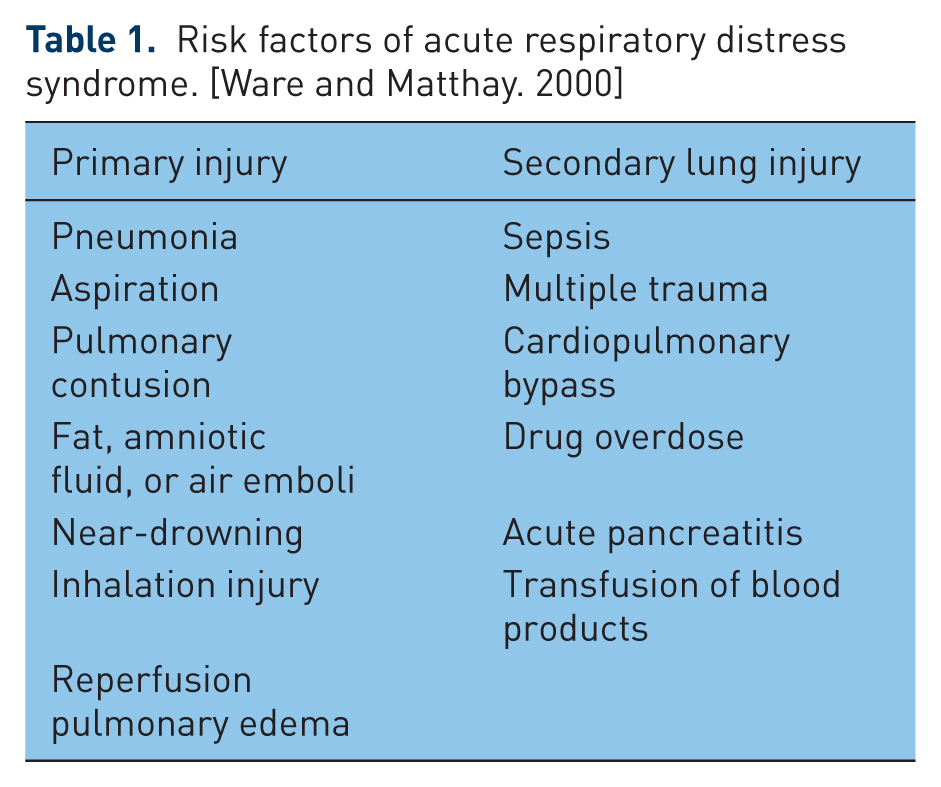
Acute respiratory distress syndrome (ARDS) is a syndrome of acute lung injury that is characterized by noncardiogenic pulmonary edema and severe hypoxemia second to a pathogenic impairment of gas exchange [ARDS Definition Task Force et al. 2012]. ARDS can develop from a multitude of lung insults, both primary and secondary (Table 1) [Ware and Matthay, 2000]. This process results in a right-to-left pulmonary shunt that increases perfusion in areas incapable of ventilation, resulting in severe refractory hypoxemia unresponsive to supplemental oxygen [ARDS Definition Task Force et al. 2012; Ware and Matthay, 2000].
Risk factors of acute respiratory distress syndrome. [Ware and Matthay. 2000]
Despite the lack of robust evidence, selective pulmonary vasodilators (SPVs) are commonly employed for their therapeutic role in improving oxygenation in patients who have developed refractory hypoxemia in ARDS [Walmrath et al. 1993]. These agents localize drug to lung parenchyma capable of ventilation, improving ventilation–perfusion (V/Q) mismatch. SPVs commonly used include inhaled nitric oxide (iNO) and the inhaled prostacyclin, epoprostenol (iEPO). Epoprostenol initiates a cascade resulting in vasodilation of pulmonary vascular beds promoting blood flow to areas of active ventilation [Zwissler et al. 1996]. While certain strategies such as protective lung ventilation, prone positioning and neuromuscular blockade have literature suggesting benefit in ARDS patients, SPVs have failed to do so in clinical research and are commonly reserved for patients failing to respond to these therapies [Papazian et al. 2010; Slutsky, 2010; Beitler et al. 2014; Goffi and Ferguson, 2014; The ARDS Network, 2000; Walmrath et al. 1996; van Heerden et al. 2000; Dunkley et al. 2013; Domenighetti et al. 2001; Dahlem et al. 2004; Torbic et al. 2013].
Although iNO improves oxygenation, its cost, need for sophisticated equipment and potential systemic side effects limits its judicious use and promotes the demand for alternative therapies [Zwissler et al. 1996; Walmrath et al. 1996; van Heerden et al. 2000; Dunkley et al. 2013; Dahlem et al. 2004; Domenighetti et al. 2001]. iEPO has been suggested as an alternative to iNO due to its similar efficacy, lower potential for systemic side effects, ease of administration and substantially lower cost [Zwissler et al. 1996]. But although the use of iEPO has gained popularity since it was first publicized for the treatment of ARDS in 1993, substantial efficacy data are lacking [Walmrath et al. 1993]. Literature evaluating its use have been few and thus no studies have evaluated mortality as a primary endpoint [Zwissler et al. 1996; Walmrath et al. 1996; van Heerden et al. 2000; Dunkley et al. 2013; Domenighetti et al. 2001; Dahlem et al. 2004]. While commonly used as a therapy of final resort, a gap in the literature has led to controversy surrounding iEPO in regards to optimal dosing and safety, as well as the target patient population.
The efficacy of SPVs has been debated and their true impact is yet to be determined. Compared with iNO, prostacyclins have demonstrated significantly different pharmacodynamic properties, including some applicable to the treatment of ARDS [Adhikari et al. 2014]. Prostacyclins preferentially enhance the cyclic adenosine monophosphate (cAMP) axis resulting in significantly increased surfactant production, an effect not found with iNO [Rose et al. 1999]. Eisenhut and colleagues first reported the impact of prostacyclins on inflammatory cytokines when they identified a prostacyclin-mediated suppression of tumor necrosis factor (TNF) α synthesis in activated monocytes [Eisenhut et al. 1993]. Release of these inflammatory cytokines produces a profound suppression of prostacyclin release in pulmonary vasculature, potentially shifting the balance towards proinflammatory [Wen et al. 1996]. Given the proinflammatory state of ARDS and the significant expression of interleukin (IL) 6, IL-1, IL-10 and TNF-α, supplementation of prostacyclins may be beneficial. Finally, prostacyclins provide known inhibition of platelet function that may be beneficial given the thrombo-oclusive state of ARDS [Ware and Matthay, 2000; GlaxoSmithKline, 2011]. However, this antiplatelet effect has only been identified with intravenous prostacyclins and has not been substantiated in data utilizing iEPO [GlaxoSmithKline, 2011]. These pleotropic effects differentiate iEPO from iNO and may have a role in ARDS given its known pathology.
Dosing epoprostenol
Adult dosing
While their role in ARDS therapy remains controversial, SPVs are still commonly utilized in clinical practice [Walmrath et al. 1993, 1996; Zwissler et al. 1996; van Heerden et al. 2000; Dunkley et al. 2013; Domenighetti et al. 2001; Dahlem et al. 2004; Torbic et al. 2013]. Controversial use and varying indications have led to significant variance in dosing and thus some assumptions may be made when evaluating currently available literature.
The crossover study by Zwissler and colleagues compared iEPO with iNO to determine the most clinically effective dose of each agent for improving the partial pressure of oxygen in arterial blood (PaO2) while reducing pulmonary artery pressure (PAP). Mechanically ventilated ARDS patients were first treated with iEPO in incremental doses of 1, 10 and 25 ng/kg/min; after a washout period of 20–30 minutes, the patients were then switched to iNO therapy with incremental doses of 1, 4 and 8 parts per million (ppm). The doses of each medication were aerosolized for 15 minutes and markers of hemodynamics and gas exchange were measured immediately after each dose. Both iEPO and iNO showed a dose-dependent decrease in mean PAP. iEPO resulted in at least a 10% decrease in PAP at all doses in all eight patients with ARDS and pulmonary hypertension, compared with only five of the eight patients receiving iNO [Zwissler et al. 1996]. This could be due in part to the lower dose of iNO used in this study, as doses as high as 80 ppm have been used in ARDS, with most of its effect demonstrated at approximately 20 ppm [Zwissler et al. 1996; Walmrath et al. 1993]. At an iEPO dose of 10 ng/kg/min, there was a 20% decrease in pulmonary vascular resistance (PVR) with no further change at 25 ng/kg/min. iEPO produced a significant increase in PaO2 at doses 10 ng/kg/min (+18%) and 25 ng/kg/min (+24%), with no effect at 1 ng/kg/min. iEPO produced no change in intrapulmonary shunt flow (QsQt), while iNO decreased QsQt at all concentrations tested [Zwissler et al. 1996].
Walmrath and colleagues produced similar hemodynamic findings when they performed a crossover study to compare iEPO and iNO in a similar population. Study subjects were randomized to receive iNO, followed by iEPO, or vice versa, with a 60 minute washout period between the two treatments. Both treatments were titrated to patient response, with a mean iNO dose of 17.8 ± 2.7 ppm and a mean iEPO dose of 7.5 ± 2.5 ng/kg/min. In addition, iEPO produced a significant decrease in PAP from 35.0 to 31.9 mmHg (p < 0.05), while iNO produced no significant change. This decrease in PAP noted with iEPO subsequently resulted in a decrease in PVR, with no effects on systemic vascular resistance (SVR) or mean arterial pressure (MAP). Both iNO and iEPO treatments resulted in significant improvements in the ratio of PaO2 to fraction of inspired oxygen (FiO2) and PaO2 [Walmrath et al. 1996].
The prospective study by van Heerden and colleagues evaluated the dose response to iEPO in nine patients with hypoxemia secondary to ARDS. iEPO was administered over the dose range of 0–50 ng/kg/min, titrated every 30 minutes in increments of 10 ng/kg/min. Although oxygenation, measured using PaO2/FiO2, improved with each titration, a statistically significant difference was only demonstrated between doses of 0 and 50 ng/kg/min [van Heerden et al. 2000]. Therefore, similar to Zwissler and colleagues, the authors concluded that there was no significant difference between various doses of iEPO on indices of oxygenation when effects at different doses are compared [Zwissler et al. 1996; van Heerden et al. 2000]. Both studies also agreed that the greatest improvement in oxygenation was demonstrated between 0 and 10 ng/kg/min with no significant effect on QsQt [Zwissler et al. 1996; van Heerden et al. 2000].
The findings of Dunkley and colleagues mirrored those of Zwissler and colleagues with regard to oxygenation findings in 16 mechanically ventilated adults with ARDS [Zwissler et al. 1996; Dunkley et al. 2013]. The mean starting dose of iEPO was 30 ng/kg/min and was titrated as needed to a maximum dose of 50 ng/kg/min. A total of 10 patients (62.5%) in this study responded to iEPO, all of whom responded at the starting dose (30ng/kg/min). Since 10 of the 16 patients studied responded at the initial dose of 30 ng/kg/min, it may be hypothesized that at least a fraction of these patients may have responded at a lower iEPO dose. The median initial response of these 10 patients was an increase in PaO2/FiO2 by 44.5%. The median maximal increase in PaO2/FiO2 seen was 112.6%. It stands to reason that the much larger response in PaO2/FiO2 in this study was due to the significantly higher dose utilized compared with other trials. Six patients did not respond to iEPO therapy, three of whom were on high frequency oscillatory ventilation (HFOV) [Dunkley et al. 2013].
In the study by Domenighetti and colleagues, the authors examined the relationship between morphology of ARDS and the dose–response with iEPO. After obtaining baseline hemodynamic function tests on 15 patients, they titrated iEPO from 2 ng/kg/min to a maximum dose of 40 ng/kg/min. The stages of titration were 2, 4, 10, 20 and 40 ng/kg/min, with each step lasting 15 minutes. At the end of each stage the authors measured PAP, systemic arterial pressure, blood gases and pH to determine the most efficacious dose and to determine if the patient responded to therapy. A patient was considered a responder if they showed an increase in PaO2 ⩾ 1 kPa or a PaO2/FiO2 increase of ⩾10%. There were eight patients who responded to iEPO therapy, all of whom were in the secondary ARDS group. The average iEPO dose used in this study was 34 ± 9 ng/kg/min. However, in those patients who responded to iEPO therapy (secondary ARDS patients), an average dose of 32 ± 1 ng/kg/min was used [Domenighetti et al. 2001].
Pediatric dosing
Possibly the best quality data come from the only prospective randomized controlled trial (RCT), by Dahlem and colleagues, in which the authors investigated the effects of iEPO on oxygenation in 14 mechanically ventilated children with acute lung injury (ALI) as defined by the American–European consensus conference on ARDS from 1994 [Bernard et al. 1994; Dahlem et al. 2004]. It should be noted that since the publication of the study by Dahlem and colleagues, this definition of ALI/ARDS has been replaced by the newer Berlin definition [Dahlem et al. 2004; ARDS Definition Task Force et al. 2012]. Changes in oxygenation were measured by calculation of the oxygenation index (mean airway pressure × 100 × PaO2/FiO2) after iEPO doses of 10, 20, 30, 40 and 50 ng/kg/min. After treatment with iEPO, when compared with placebo, there was a significant improvement in oxygenation index at 30 ng/kg/min by 26% (range 3–35%). Doses at 20, 40 and 50 ng/kg/min produced results trending toward significance when compared with placebo [Dahlem et al. 2004]. The authors noted that, in pediatric populations, a smaller nebulizer tube is used compared with that used in adult patients and this may explain why higher doses of iEPO (30 ng/kg/min compared with 20 ng/kg/min) are needed for pediatric patients to respond [Dahlem et al. 2004; Coleman et al. 1996; Thomas et al. 1991]. However, this study provides interesting data suggesting that iEPO demonstrates a bell-shaped response curve with regards to improved oxygenation and possibly a dosing ceiling in this indication [Dahlem et al. 2004].
Safety profile
The use of vasodilators in the treatment of ARDS has long been debated as its benefit has yet to be identified and it has been associated with serious adverse events. iNO has been associated with platelet aggregation, worsening hypoxemia, renal failure and methemoglobinemia [Zwissler et al. 1996; Siobal, 2004]. iNO has also been associated with cytotoxic properties through the formation of peroxynitrite in the presence of oxygen free radicals. The formation of this cytotoxic metabolite may result in damage to DNA, lipid peroxidation and eventual cell death [Adhikari et al. 2007]. iEPO has not been associated with these detrimental cytotoxic effects [Tabrizi et al. 2012; Vane and Botting, 1995]. It has been suggested that iNO be abandoned in ARDS patients regardless of hypoxemia severity due to lack of mortality benefit and an increased risk of renal dysfunction [Adhikari et al. 2007]. Although intravenous (IV) epoprostenol may be associated with worsening V/Q matching, hypotension, inhibition of platelet aggregation, and tachycardia, it has been postulated that iEPO’s selectivity for the pulmonary vasculature and this method of administration may minimize or even eliminate these adverse events in most patients [Tabrizi et al. 2012; Bone et al. 1989; Walmrath et al. 1996; van Heerden et al. 2000; Dunkley et al. 2013].
Van Heerden and colleagues examined the safety profile of iEPO during their study of nine patients [van Heerden et al. 2000]. At acidic or neutral pH, iEPO is rapidly hydrolyzed to its inactive major metabolite, 6-keto PGF1α, which is more stable in circulation than the parent compound and easily measurable in plasma as a surrogate for iEPO systemic absorption [Rosenkranz et al. 1981; van Heerden et al. 2000]. Though a significant increase in plasma levels of 6-keto PGF1α with increasing doses of iEPO was observed, this was not associated with any systemic hypotensive effect. At doses of 0–50 ng/kg/min, there was no significant dose-related change in PAP, although there was a trend toward an increase in MAP and cardiac index (CI) [van Heerden et al. 2000]. The authors noted that the lack of effect was most likely due to the fact that the study population had ‘a relatively normal baseline value’ for these parameters. The authors failed to show a dose-dependent effect of iEPO on platelet inhibition, likely due to impaired platelet function resulting from the severity of their illnesses [van Heerden et al. 2000].
Permissive hypercapnia and resultant acidosis is commonly accepted during the treatment of ARDS. This decreased pH likely facilitates faster degradation of the parent compound to its metabolite, thus minimizing the possibility of the parent compound acting on platelets, which possibly explains the absence of impaired platelet function [Rosenkranz et al. 1981]. It is hypothesized that platelet inhibition within the pulmonary circulation may be beneficial in ARDS patients as platelet and immune cell aggregation may contribute to the disease process. Even though this study failed to show a risk of systemic effects with the use of iEPO due to its small sample size and variability in baseline function of the study population, a relationship cannot be dismissed [van Heerden et al. 2000].
In the studies by both Zwissler and colleagues and Walmrath and colleagues, the researchers showed that, unlike IV epoprostenol, iEPO will decrease PAP without effecting SVR or MAP. Their findings also suggest that iEPO improves QsQt which is not seen, or potentially worsened in ARDS patients treated with IV epoprostenol [Zwissler et al. 1996; Walmrath et al. 1996].
Dunkley and colleagues found the most common adverse event observed with iEPO was hypotension (n = 3, 19%) [Dunkley et al. 2013]. However, it should be noted that one of these patients mistakenly received IV epoprostenol, which is more commonly associated with hypotension [Dunkley et al. 2013; GlaxoSmithKline, 2011]. Other adverse events noted could easily be attributed to the acuity of the studied patients. The retrospective nature of the study prevents correlation between these adverse effects and iEPO [Dunkley et al. 2013].
Torbic and colleagues compared the safety of iNO and iEPO in critically ill patients with either ARDS (n = 63), acute right ventricular failure (n = 36), or cardiac decompensation following a heart or lung transplant (n = 6). Patients were started on an SPV after having failed traditional therapies; the vasodilator was chosen at the discretion of the attending physician (iNO or iEPO). Patients were initiated on doses of iNO 1–80 ppm or iEPO 10–50 ng/kg/min, both titrated to patient response. This study showed no difference between the two therapies with respect to efficacy or safety [Torbic et al. 2013].
In the pediatric population (<18 years old) the only prospective trial, while small (n = 14), failed to identify any adverse effects in children receiving iEPO [Dahlem et al. 2004]. Brown and colleagues evaluated the use of iEPO in the pediatric population with acute pulmonary hypertension via retrospective chart review; of the 20 patients in the study, 13 were neonates. A total of six of the 20 patients experienced at least one side effect related to iEPO therapy; five of these six patients were neonates. The most common side effect reported was a drop in systolic blood pressure requiring fluid boluses or vasoactive agents. After subgroup analysis, it was determined that while some patients displayed a significant improvement in oxygenation, patients less than 30 days of age had a higher incidence of adverse events [Brown et al. 2012]. The significant difference in volumes of distribution, surface area to body mass ratios, and absorption kinetics in pediatric and neonatal populations may explain the higher incidence of adverse events and thus identify them as a population requiring more aggressive monitoring.
Patient populations
In the study by Domenighetti and colleagues, iEPO was administered to 15 patients with ARDS to determine a relationship between the etiology of ARDS and the response to iEPO [Domenighetti et al. 2001]. This study was unique in that it demonstrated a significant difference in oxygenation outcomes between patients presenting with primary ARDS and those with secondary ARDS. In primary ARDS, iEPO induced a reduction in PaO2/FiO2 (from 146 ± 16 to 135 mmHg ± 17; p < 0.05) and PaO2 (from 87 ± 2 to 79 mmHg ± 2; p < 0.02), whereas in patients presenting with secondary ARDS, there was an increase in PaO2/FiO2 (from 161 ± 23 to 171 mmHg ± 22: p < 0.01) and PaO2 (from 76 ± 4 to 84 mmHg ± 4; p < 0.01). This shows improved oxygenation in patients with secondary ARDS and a potential to worsen oxygenation in patients with primary ARDS. There were eight responders in this study, all of whom were in the secondary ARDS group, and seven nonresponders, all but one of whom were in the primary ARDS group [Domenighetti et al. 2001]. This may be explained by the fact that ARDS caused by primary insult is characterized more by lung tissue consolidation, whereas ARDS caused by secondary insult is characterized by interstitial edema and alveolar collapse. This in conjunction with potentially fibrotic lung parenchyma, and vasculature may prevent response and increase the potential for dead space perfusion in these populations [Gattinoni et al. 1998]. The study demonstrated that a more favorable response to iEPO may be seen in patients with secondary ARDS compared with primary ARDS [Domenighetti et al. 2001]. The authors hypothesized that the worsening oxygenation in primary ARDS is due to iEPO accumulating in alveolar areas that are not active, whereas in secondary ARDS, iEPO predominately accumulates in functional alveoli [Gattinoni et al. 1998]. This hypothesis is supported by the results of computed tomography imaging utilized by Domenighetti and colleagues [Domenighetti et al. 2001].
A similar finding was noted in the study by Pacheo and colleagues, which evaluated ARDS patients treated with iEPO, comparing hospital survivors with nonsurvivors to identify predictors of mortality [Pacheco et al. 2014]. Through logistic regression, Pacheo and colleagues noted that trauma, as the etiology of ARDS, was independently associated with a lower risk of hospital mortality [Pacheco et al. 2014]. This study, however, is retrospective in nature and is only able to provide an association. Ventilation techniques utilized may also predict response in patients receiving iEPO.
In the study by Dunkley and colleagues, three of the four patients on HFOV did not respond. The single responding patient experienced the lowest improvement in oxygenation of the studied population. One patient on HFOV who did not respond initially to iEPO was transitioned to continuous positive airway pressure. Immediately following the transition, the patient began to respond to iEPO therapy with an increase in PaO2/FiO2 by 27% (from 83 to 105 mmHg) [Dunkley et al. 2013]. The lack of effect of iEPO when combined with HFOV is most likely explained by ineffective drug deposition within the pulmonary vasculature caused by the high frequencies used [Vane and Botting, 1995].
While use of iEPO provides the benefit of improving oxygenation through preferential dilation of well-ventilated areas of the lungs, it also provides hemodynamic benefits by relieving strain on the right ventricle by reducing PVR. This hemodynamic alteration appears to have the most profound effect on patients with reduced right ventricular function at baseline. This is likely due these patients lacking sufficient intrinsic cardiac output to overcome the increased pulmonary pressure induced by profound hypoxemia, mechanical ventilation and a number of other potential causes of pulmonary hypertension seen in critically ill patients [Radermacher et al. 1990; De Wet et al. 2004]. Though iNO has been shown to improve PAP and oxygenation, several studies have failed to show the increase in cardiac output seen with iEPO [Radermacher et al. 1990; De Wet et al. 2004; Bocchi et al. 1994; Haraldsson et al. 1998; Rich et al. 1999].
However, after the successful use of iEPO in adult patients with ARDS and other hypoxic respiratory states, iEPO began to be utilized in pediatric patients refractory to iNO or when iNO was unavailable. Kelly and colleagues reported the successful use of iEPO in four term infants with persistent pulmonary hypertension of the newborn (PPHN) who did not respond adequately to iNO. Of the studied infants, three had improved oxygenation and reduced PAP, and were extubated within three weeks of the initiation of iEPO. The authors noted no changes in systemic blood pressure, heart rate or temperature during inhalation of iEPO in any of the four infants studied [Kelly et al. 2002].
The use of iEPO in pediatric patients (<18 years old) with acute pulmonary hypertension was studied via retrospective chart review by Brown and colleagues. After subgroup analysis, the authors concluded that only pediatric patients less than 30 days of age (neonates) with PPHN or meconium aspiration syndrome (MAS) had significant improvement in their oxygenation index. However, since this age group also had a higher incidence of adverse events, caution should be exercised when initiating this therapy in neonates. Although iEPO is used with success in pediatric patients including neonates, large prospective controlled studies are needed to elucidate its role and to determine its safety profile in this patient population [Brown et al. 2012].
Cost benefits
The study conducted by Torbic and colleagues compared the therapeutic costs of the SPVs and showed that iNO exceeded the cost of iEPO per patient anywhere from 4.5 times (US$3930 ± 4210 versus US$838 ± 996, p < 0.0001), when using low iNO contract pricing, to 17 times (US$14240 ± 15255 versus US$838 ± 996, p < 0.0001) when using high iNO contract pricing. In the authors’ subgroup analysis, iEPO was found to be significantly less costly than iNO across all three subgroups (ARDS, heart/lung transplant, acute right ventricular failure). However, it is worth noting that this pricing information is specific to the research institution. While the doses of iEPO and iNO that were used were within the range specified by the institution’s protocol, specific mean doses were not mentioned. Nevertheless, the cost difference between iNO and iEPO, as reported in Torbic and colleagues, can be considered clinically significant even for today’s therapies and it is generally accepted that iEPO is a less costly alternative to iNO [Torbic et al. 2013]. A study conducted by De Wet and colleagues also demonstrated the cost-effectiveness of using iEPO in place of iNO for reducing pulmonary hypertension in patients undergoing cardiothoracic surgery. They found that the daily cost of iEPO was US$150 compared with US$3000 for iNO. Though in a different patient population, this study further exhibits the opportunity for significant cost savings with the utilization of iEPO rather than iNO [De Wet et al. 2004].
Alternative prostacyclins
Iloprost
Though iNO and iEPO are commonly used for the treatment of ARDS, other SPVs have been studied. Iloprost is a carbacyclin derivative of PGI2 that is traditionally used for the treatment of pulmonary hypertension, but like iEPO, has theoretical benefits in ARDS patients. However, there is minimal evidence to support its use for this indication [Actelion Pharmaceuticals, 2004]. Benefits of iloprost may include lower viscosity and longer halflife (20–30 minutes) compared with iNO or iEPO, attributes which may better facilitate its nebulization [Actelion Pharmaceuticals, 2004; Grant and Goa, 1992]. However, the longer halflife of iloprost has been associated with an increased incidence of systemic hypotension compared with iNO [Siobal, 2004; Hoeper et al. 2000]. It is hypothesized that iloprost would have better clinical results when administered intermittently, as opposed to continuous nebulization as in the setting of iEPO or iNO [Hoeper et al. 2000; Siobal, 2004]. Sawheny and colleagues administered iloprost to 20 patients with pulmonary hypertension and ARDS and demonstrated an improvement in V/Q matching without deleterious effects on hemodynamics [Sawheny et al. 2013]. However, this is the only trial known utilizing iloprost in ARDS patients. Given the low rate of adverse effects with the utilization of SPVs, this trial easily could have been underpowered to show a difference.
In pediatrics, iEPO has been proposed to cause injury to the lung epithelium due to being diluted in an alkaline base (pH ~10). It is hypothesized that, since iloprost is diluted in a neutral base, it may be less harmful to the delicate epithelium of the lungs. An early case series proposed benefit of using inhaled iloprost as rescue therapy in two neonatal patients. Initially, both patients showed dramatic improvement in oxygenation, but despite best efforts to save them, both patients died within 3 weeks of initiating iloprost [De Luca et al. 2007].
Ivy and colleagues studied the short-term and long-term effects of iloprost in ambulatory children with pulmonary arterial hypertension. They showed that iloprost produced similar effects to iNO in regards to efficacy; however, in some patients iloprost caused marked bronchoconstriction and other adverse effects leading to discontinuation of therapy. In addition, some patients experience clinical deterioration on inhaled iloprost therapy requiring rescue IV therapy. While this is a different population, it is unknown which patients may have iloprost-induced bronchoconstriction and reduced lung function [Ivy et al. 2008].
Alternatively, a case series of 10 infants given iloprost as rescue therapy did not show this bronchoconstriction but did have significant oxygenation improvement (PaO2/FiO2 increase) and a decline in respiratory rate after initiation. The authors concluded that nebulized iloprost in addition to noninvasive respiratory support is feasible and safe in infants with impending hypoxemic respiratory failure. However, given the retrospective nature involving two interventions, it is difficult to decipher the true treatment effect and adverse effect profile of iloprost [Piastra et al. 2012]. In addition, iloprost has shown mixed effects in animal models of ARDS with an improvement in PAP, but not in oxygenation [Senturk et al. 2012]. Because of these mixed results and limited data, the use of iloprost in adult or pediatric patients cannot be endorsed.
Conclusion
Regular utilization of SPVs for the treatment of ARDS may be debatable. While they provide improvements in a number of abnormal parameters in ARDS patients, their benefit has not been substantiated. Epoprostenol provides effects that potentially alter the pathophysiological process including inflammation, hypoxia, surfactant production, hypoxic vasoconstriction and thrombo-occlusive effects of ARDS. Despite significant advances in the area, mortality remains high among ARDS patients [ARDS Definition Task Force et al. 2012]. This high mortality and a limited spectrum of therapeutic options have left clinicians searching for alternatives, spiking interest in SPVs. Lack of literature associated with improved outcomes has not dissuaded institutions from relying on SPVs in the care of refractory patients.
Increased treatment costs and adverse events associated with iNO will likely lead to its abandonment and the need for alternative options [Adhikari et al. 2007]. Use of iEPO has shown a significant decrease in PVR and PAP, and an increase in PaO2 and PaO2/FiO2 without affecting SVR or MAP [Zwissler et al. 1996; Walmrath et al. 1996; van Heerden et al. 2000; Dunkley et al. 2013; Domenighetti et al. 2001; Dahlem et al. 2004; Torbic et al. 2013]. Compared with iNO, iEPO may also provide some pleotropic effects outside of improved oxygenation and thus outcomes with iNO likely cannot be extrapolated to iEPO [Adhikari et al. 2014; Rose et al. 1999; Eisenhut et al. 1993; Wen et al. 1996]. While iEPO also impacts arterial oxygenation by decreasing V/Q mismatching and pulmonary shunt flow, this effect is not different from iNO. Improved oxygenation was seen at iEPO doses as low as 10 ng/kg/min; however, the most effective and safest dose for yielding a clinically significant increase in PaO2 and reduction in PAP appears to be 20–30 ng/kg/min in adults and 30 ng/kg/min in pediatric patients [Zwissler et al. 1996; Walmrath et al. 1996; Dahlem et al. 2004].
Current data appear to support that there may be a ceiling effect for the improvement in oxygenation above these doses in which no additional benefit may be derived [Zwissler et al. 1996; Walmrath et al. 1996; van Heerden et al. 2000; Torbic et al. 2013; Dunkley et al. 2013; Domenighetti et al. 2001; Dahlem et al. 2004]. iNO and iEPO have shown similar efficacy profiles; however, they differ with respect to cost and ease of therapeutic administration. [Zwissler et al. 1996, Walmrath et al. 1996, van Heerden et al. 2000, Dunkley et al. 2013, Domenighetti et al. 2001, Dahlem et al. 2004, Torbic et al. 2013]. The cost expenditure to treat one patient with iEPO has been shown to be 20% of the cost of treating one patient with iNO [Dahlem et al. 2004; De Wet et al. 2004].
Even though iEPO may be a safe, more cost-effective option than iNO, its use has not been associated with changes in clinical outcomes such as morbidity or mortality. This may be explained by the lack of study populations large enough to show any mortality benefit or identification of those populations that would derive the most benefit [Dahlem et al. 2004; Domenighetti et al. 2001; Dunkley et al. 2013; Torbic et al. 2013; van Heerden et al. 2000; Walmrath et al. 1996; Zwissler et al. 1996]. As identified in a number of therapeutic interventions in ARDS, efficacy may be directly correlated to the time of initiation in respect to onset of disease [Papazian et al. 2010; Beitler et al. 2014; Goffi and Ferguson, 2014]. A number of the studies evaluating iEPO failed to provide time to initiation data, and those that did had times ranging from 2 to as long as 13 days [van Heerden et al. 2000; Dahlem et al. 2004; Torbic et al. 2013; Tabrizi et al. 2012]. This may further impact the outcomes and the effect that may be extrapolated from currently available data.
From a review of the current literature (Table 2), the most beneficial effects of iEPO have been seen in adult patients with secondary ARDS as compared with primary ARDS, most likely due to the difference in etiology of the two disease states, and in patients suffering from baseline right ventricular heart failure [Domenighetti et al. 2001; Gattinoni et al. 1998; Bocchi et al. 1994; Haraldsson et al. 1998]. Given poor drug deposition of iEPO during HFOV, which is most commonly used in ARDS, its use in this ventilation mode should be dissuaded [Goffi and Ferguson, 2014; Dunkley et al. 2013]. Though iEPO has demonstrated improvements in hemodynamic parameters and oxygenation in ARDS patients, due to the limited number of randomized clinical trials and the lack of studies investigating mortality, the use of iEPO cannot be recommended as standard of care in ARDS. iEPO should be reserved for those refractory to traditional therapies, a conclusion substantiated by a recent Cochrane review [Afshari et al. 2010; Dahlem et al. 2004; Domenighetti et al. 2001; Dunkley et al. 2013; Torbic et al. 2013; van Heerden et al. 2000; Walmrath et al. 1996; Zwissler et al. 1996].
Studies evaluating the use of inhaled epoprostenol in ARDS patients.

ARDS, acute respiratory distress syndrome; iNO, inhaled nitric oxide; iEPO, inhaled epoprostenol; NS, normal saline.
In the event salvage therapy with iEPO is utilized, current literature supports the methods, dose and patient populations detailed above. The results of this review were based on limited data; for iEPO to be considered as a first-line agent, further prospective research will be required to identify benefit and to improve understanding of ideal populations.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
