Abstract
Two recent phase III trials in patients with severe eosinophilic asthma have shown that anti-interleukin 5 (IL-5) therapy with mepolizumab reduces the frequency of asthma attacks, improves symptoms and allows patients to reduce oral glucocorticoid use without loss of control of asthma. An earlier large 616 patient Dose Ranging Efficacy And safety with Mepolizumab in severe asthma (DREAM) study had shown that the only variables associated with treatment efficacy were a prior history of asthma attacks and the peripheral blood eosinophil count. The link between blood eosinophil counts and treatment efficacy is biologically obvious given that IL-5 has a pivotal role in eosinophil production, proliferation and chemotaxis. It is also clinically relevant as the blood eosinophil count is routinely measured and thus readily available in patients with asthma. Recognition of the link between airway or blood eosinophilia and treatment response was also important in the clinical testing of the alternative IL-5 blocker, such as reslizumab, which is currently being evaluated in a phase III randomized controlled trial (RCT) after having shown to improve lung function, improve symptom score and reduce sputum eosinophilia in a smaller phase IIb study. In addition, benralizumab, an IL-5α receptor blocker, has shown good effects in a phase IIb RCT with patients with severe asthma that had sputum eosinophilia and more recently in a phase IIa trial with patients with eosinophilic chronic obstructive pulmonary disease. Therefore anti-IL-5 treatment seems generally effective in eosinophilic asthma, either assessed by blood or airway eosinophilia. This factor together with the impressive clinical efficacy and good safety profile make anti-IL-5 (mepolizumab, reslizumab) and benralizumab (anti-IL-5 receptor α) very promising drugs for the treatment of patients with severe eosinophilic asthma, a subgroup that is in desperate need of better treatments.
Keywords
Introduction
Asthma is a prevalent chronic inflammatory disease of the airways that affects 300 million people worldwide (World Health Organization). From 5% to 10% of patients with asthma have severe disease, requiring extensive treatment to control it or it remains uncontrolled despite extensive treatment [Chung et al. 2014] (Global Initiative for Asthma guidelines: http://www.ginasthma.org/2014). This small subgroup accounts for the majority of morbidity and mortality due to asthma and 60% of the total healthcare costs attributable to asthma. Patients with a recent history of an asthma attack have particularly high annual healthcare costs, estimated to be $1740 (£1035; €1257), three times those of a patient with severe asthma and no history of an attack [Loymans et al. 2014].
Severe asthma is heterogeneous with respect to the clinical problem, the nature of the impairment of lung function and the underlying pathology [Pavord, 2013]. Moreover, many patients apparently have severe asthma primarily because of easily correctable issues with inhaler technique or treatment adherence and others have persistent asthma-like symptoms because of the effect of comorbidity. The recent international European Respiratory Society/American Thoracic Society (ATS) guidelines on severe asthma [Chung et al. 2014] have acknowledged these difficulties and produced what is likely to be the most widely accepted definition. They suggest that the diagnosis of asthma is confirmed and comorbidities addressed before making a diagnosis of severe asthma. The guidelines go on to define severe asthma as requiring treatment with high-dose inhaled corticosteroids (ICS) and long-acting β2 agonists or leukotriene modifier/theophylline for the previous year or systemic corticosteroids for 50% of the previous year to prevent it from becoming ‘uncontrolled’ or remaining uncontrolled’ despite this therapy. Uncontrolled asthma is defined as the presence of at least one of criteria in Table 1. Controlled asthma worsens on tapering of these high doses of ICS or systemic corticosteroids (or additional biologics).
ERS/ATS criteria for uncontrolled asthma.
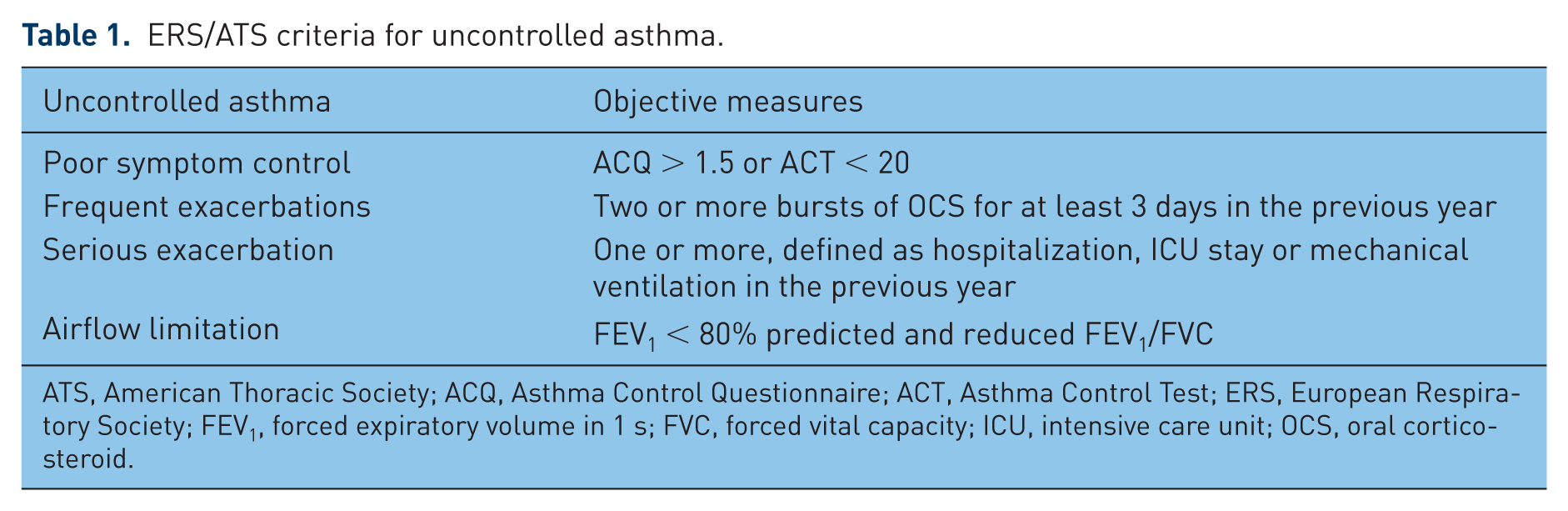
ATS, American Thoracic Society; ACQ, Asthma Control Questionnaire; ACT, Asthma Control Test; ERS, European Respiratory Society; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; ICU, intensive care unit; OCS, oral corticosteroid.
The high level of morbidity, healthcare costs and treatment-related side effects in severe asthma mean that there is a large unmet need for alternative therapies. New treatments should ideally target the pathophysiological mechanism responsible for the morbidity and might therefore have to be adapted for each individual patient in order to match the disease heterogeneity. One particularly fruitful area of development has been the use of monoclonal antibodies targeting T helper 2 (Th2) mediated airway inflammation. It is now known that anti-immunoglobulin E (omalizumab) is only effective in patients with evidence of Th2 airway inflammation [Hanania et al. 2013] and a number of new approaches including anti-interleukin (IL)-4α (dupilumab, which blocks IL-4 and IL-13), anti-IL-13 (lebrikizumab) and anti-IL-5 (mepolizumab, reslizumab and benralizumab) are showing promise in patients with this feature. Among these, anti-IL-5 and specifically mepolizumab has been most extensively investigated in clinical studies.
The clinical effects of mepolizumab and other monoclonal antibodies targeting type 2 inflammation are strikingly consistent, showing a clinically important effect on the frequency of asthma attacks and a small beneficial effect on symptoms and impaired lung function [Bel et al. 2014; Ortega et al. 2014; Pavord et al. 2012]. These findings emphasize that symptoms and impaired lung function are to some extent dissociated from eosinophilic airway inflammation and the risk of asthma attacks. This important conceptual advance means that new ways of assessing airways disease will need to be adopted to realize the full potential of these treatments. In particular, biomarkers of Th2 inflammation will need to be assessed. Several measures are available and suitable for use in clinical practice (Table 1). Whether different combinations of biomarkers are particularly related to the efficacy of different treatments is unclear. This is an important area for further study.
In this review, we will focus on the effectiveness and safety profile of anti-IL-5 drugs for the treatment of patients with severe asthma and briefly discuss upcoming additional treatments.
IL-5 and the IL-5 receptor
IL-5 is a 115-amino-acid protein that consists of four α helices. In its natural form it consists of a pair of helical bundles by the interdigitation of two identical monomers that contribute a D helix to the other’s A–C helices (Figure 1). This dimer form is important because when the IL-5 dimer is reduced to a pure monomer it loses its biological activity [Milburn et al. 1993].
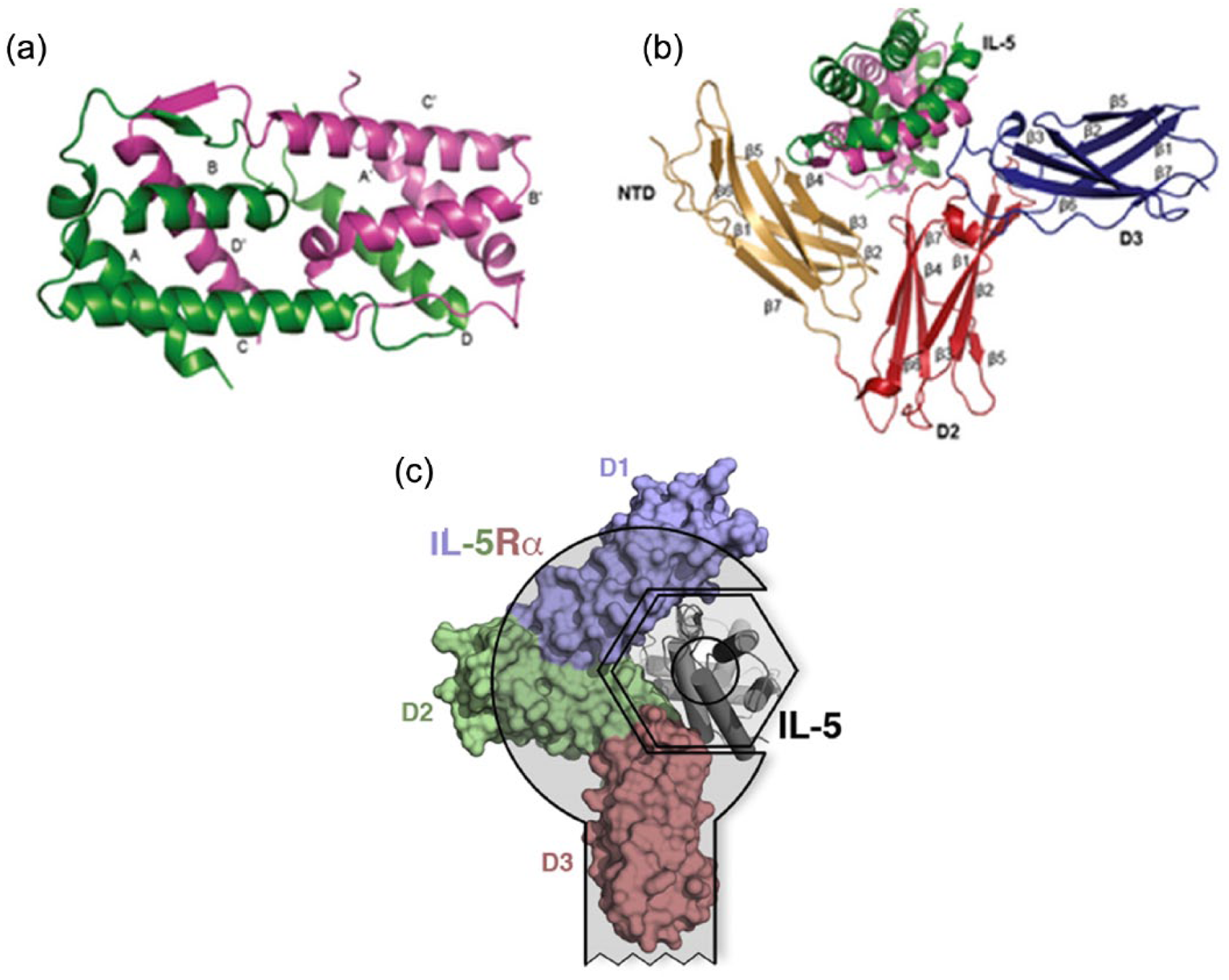
Interleukin 5 (IL-5) cytokine and binary receptor complex structures. (a) Secondary structural elements that make up the IL-5 dimer are shown and individual monomers are coloured green (monomer 1) and pink (monomer 2). The four α helices in each monomer are labelled A–D for monomer 1 and A′–D′ for monomer 2. (b) Binary complex of IL-5:IL-5 receptor α (IL-5Rα) showing the three domains of IL-5Rα (NTD: brown; domain 2: D2, red; domain 3: D3, blue). The β sheets from each domain are labelled β1–β7. (c) IL-5Rα extracellular domain (D1, blue; D2, green; D3, red) covers more than 170° of the ligand four-helix bundle of IL-5 (grey). Figure 1(a, b) is reprinted with permission of Wiley (http://onlinelibrary.wiley.com/doi/10.1111/j.1600-065X.2012.01164.x/full#imr1164-fig-0003); Figure 1(c) is reprinted with permission of Elsevier (http://dx.doi.org/10.1016/j.str.2011.08.015).
IL-5 is mainly produced by Th2 lymphocytes, Tc2 cells (type 2 cytotoxic T cells), eosinophils, mast cells and γδ-T cells [Takatsu, 2013]. More recently type 2 innate lymphoid cells (ILC2s) have been shown to be a potentially important additional source. Evidence from inhalation challenge models in mice indicates ILC2s (from bronchoalveolar lavage fluid) become the main source of IL-5 and IL-13 production after challenge with IL-25 and IL-33 [Klein Wolterink et al. 2012]. IL-25 and IL-33 are essential epithelial signalling cytokines that are released after exposure to allergens or viruses. Smaller amounts of IL-5 can be produced by epithelial cells, natural killer (NK) cells and NK T cells.
Cellular production of IL-5 has been described both in the context of innate and adaptive immune responses. It is produced together with IL-4 and IL-13 upon helminth infection; however, in asthma, viral triggers and allergens can lead to IL-5 production as well. In Figure 2 two important asthma-associated triggers are the initiators of two separate cascades that both lead to IL-5 production and eosinophil proliferation and activation. The two triggers are allergens and pathogen/glycolipids/pollutants; the allergic cascade leads to Th2 cell activation and the nonallergic pathway to ILC2 activation. Whether these processes coexist, constantly alternate or are generally fixed in patients with asthma is not yet known.

The roles of T helper 2 (Th2) cells and type 2 innate lymphoid cells (ILC2s) in eosinophilic asthma. In the cascade that is driven by an allergic stimulus (1), eosinophilic airway inflammation and bronchial hyperresponsiveness (BHR) are driven by adaptive Th2 cells that produce interleukin (IL)-5, IL-13 and IL-4, the latter driving immunoglobulin E (IgE) synthesis. In the other cascade (2) that is driven by smoking, pollutants and pathogens, ILC2s produce IL-5 and IL-13 and thus cause eosinophilia and BHR. This latter pathway does not depend on adaptive immunity but interacts with the adaptive pathway; there is no specific allergen involved and ILC2s produce little IL-4, so there is no associated IgE response from B cells. CRTH2, chemoattractant receptor-homologous molecule ;PGD2, prostaglandin D2; LTE4, Leukotrien E4; GM-CSF, Granulocyte-macrophage colony-stimulating factor; TSLP, Thymic stromal lymphopoietin.
IL-5 induces B-cell activation and eosinophil proliferation and differentiation in mice. In humans it has been proven to prolong survival and terminal maturation, to influence chemotaxis, increase adhesion to endothelial cells and enhance eosinophil effector functions. The influence of IL-5 on other cell types is less clear. There is evidence that IL-5 can positively influence survival in sepsis independent of its effect on eosinophils by stimulating neutrophils and monocytes via the IL-5 receptor which is known to be expressed during sepsis [Linch et al. 2012].
The IL-5 receptor is a type I cytokine receptor, which is a form of transmembrane receptor that recognizes and responds to cytokines that have four α helical strands (Figure 1). The receptor consists of a ligand specific α subunit and a signal transducing β subunit (βc). This common β subunit is expressed on a variety of leukocytes and also forms the IL-3 and granulocyte macrophage colony-stimulating factor (GM-CSF) receptor together with the IL-3 and GM-CSF α subunits. Upon binding of the IL-5 receptor, complex tyrosine kinases such as JAK2 and Lyn are being activated through the common β subunit, resulting in an activated intracellular signalling pathway. The IL-5 receptor is present on eosinophils and basophils and exerts its aforementioned functions after binding.
IL-5 and the IL-5 receptor (IL-5R) have been investigated in most detail in eosinophils. IL-5 strongly influences eosinophil dynamics throughout the human body. Intravenously administered IL-5 induces a rapid decline in peripheral blood eosinophils within 0.5–2 h followed by a marked eosinophilia lasting up to 72 h in patients with asthma [Stirling et al. 2001]. Inhalation of IL-5 causes a drop in metacholine PC20 after 24h, blood eosinophilia and increased ECP in sputum (shi et al 1998 AJRCCM, Jan 157 (1), 204–209). However, not all studies have shown this [van Rensen et al. 2001] so the local effect after inhalation remains a matter of scientific debate.
It is clear that IL-5 greatly influences eosinophil biology and therefore is a logical target in diseases that are associated with eosinophils. Therefore it is currently tested in a specific subgroup of patients with chronic obstructive pulmonary disorder (COPD), with evidence of eosinophilic airway inflammation [Bafadhel et al. 2011, 2012]. Other eosinophilic diseases occurring with or independently of asthma such as allergic rhinitis, nasal polyposis, eosinophilic eosophagitis, hyper-eosinophilic syndrome, atopic dermatitis and the Churg–Strauss syndrome could possibly benefit from IL-5 blockage.
Eosinophilic inflammation in asthma
In a subgroup of patients with asthma, high numbers of eosinophils are present in upper and lower airways, in the lung tissue and in peripheral blood. BAL fluid and sputum of these patients contains eosinophils and often these eosinophils persist despite treatment with ICS. Three studies have aimed to adjust ICS treatment based on sputum eosinophilia and found that this strategy effectively reduced exacerbations [Chlumsky et al. 2006; Green et al. 2002; Jayaram et al. 2006]. This finding strongly suggests that the presence of airway eosinophilia is a clinically relevant finding and suggests that blocking IL-5 or its receptor might be a promising strategy to treat asthma. The fact that IL-5 does not strongly influence other cell types suggests that it is safe to target.
Anti-IL-5 treatments
Mepolizumab
Mepolizumab was the first of the currently available IL-5 antagonists to be tested in a RCT in asthma. Mepolizumab is an IL-5-specific humanized immunoglobulin G1 (Ig G1) monoclonal antibody, which directly binds to IL-5, thereby stopping it from binding to its receptor on the surface of eosinophils and other cells. Leckie and colleagues tested mepolizumab in a group of 24 patients with mild atopic asthma [Leckie et al. 2000]. The aim of the study was to assess the effect of a single dose of mepolizumab on blood and sputum eosinophils, airway hyperresponsiveness (AHR) and the late asthmatic reaction to inhaled allergens. The results clearly showed a lowering of the blood eosinophil count lasting up to 16 weeks and lower sputum eosinophils after 4 weeks; however, it had no effect on AHR or the late asthmatic response after allergen challenge. The strong biological effect but absence of effect on airway dysfunction led many to argue that eosinophilic airway inflammation was not associated with clinically relevant outcomes.
This interpretation was supported by two subsequent clinical trials. Flood-Page and colleagues treated 24 patients with mild asthma with three doses of 750 mg mepolizumab or placebo over 20 weeks [Flood-Page et al. 2003a]. The biological effect of mepolizumab on peripheral blood eosinophilia was confirmed (67–100% decrease) in the second trial and found to be associated with a ~24% reduction in eosinophil numbers in airway mucosal biopsy and a 52% drop in percentage of eosinophils in the bone marrow [Flood-Page et al. 2003b]. However, again mepolizumab had no effect on more clinically relevant outcomes including AHR, forced expiratory volume in 1 s (FEV1) and peak flow measurements. In a larger parallel group RCT 250 mg and 750 mg of mepolizumab or placebo was given for 12 weeks to 362 patients with persistent asthma who were symptomatic despite treatment with inhaled corticosteroids [Flood-Page et al. 2007]. The following clinical parameters were assessed: morning peak expiratory flow, FEV1, daily β2 agonist use, symptom scores, exacerbation rates and quality of life measures. None of these parameters significantly improved, although this study did show a trend to a reduced rate of asthma attacks in patients randomized to the highest dose of mepolizumab. With respect to safety, all three of these early RCTs showed none or minimal adverse events caused by mepolizumab.
The results of these RCTs led to considerable doubt in the paradigm that eosinophils are damaging effector cells in asthma [Flood-Page et al. 2003a]. However, two factors suggested that a different interpretation was possible. First, cross-sectional studies in patients with asthma, particularly those with more severe disease, showed considerable dissociation between eosinophilic airway inflammation on one hand and symptoms and lung function on the other. A significant number of patients with symptomatic asthma and good evidence of airway dysfunction were found to have noneosinophilic asthma and therefore little prospect of responding to an anti-eosinophil treatment. Second, studies investigating the effect of additional doses of corticosteroids in asthma (FACET) and trials evaluating the effects of anti-inflammatory treatment targeted to suppress eosinophilic airway inflammation showed that the main clinical impact of these interventions was a reduced risk of asthma attacks. Therefore it seemed possible that mepolizumab might have a clinically important effect on the frequency of asthma attacks if the treatment was specifically targeted to patients with evidence of eosinophilic airway inflammation.
Two subsequent studies did this. Nair and colleagues selected patients with persistent sputum eosinophilia and symptoms despite oral and inhaled prednisone treatment [Nair et al. 2009]. They treated 11 patients with placebo and nine patients with 750 mg mepolizumab monthly for 4 months and found a large reduction in exacerbation rate and evidence of an oral glucocorticoid-sparing effect in the treatment arm. Only one patient out nine in the treatment arm suffered from an exacerbation (noneosinophilic) versus 10 out of 11 in the placebo arm. The secondary outcome measures showed a modest improvement in FEV1, no effect on the day-to-day symptoms and a significant improvement of the Juniper Asthma Control Questionnaire (ACQ). Haldar and colleagues included 61 patients with eosinophilic asthma defined as a sputum eosinophil count greater than 3% on at least one occasion over the past 2 years, refractory asthma according to ATS criteria and at least two exacerbations in the past year despite high corticosteroid treatment [Haldar et al. 2009]. The most important finding was a reduced exacerbation rate in the treatment group (relative risk 0.57, 95% confidence interval 0.32–0.92). The asthma-related quality of life scores improved in the mepolizumab group; however symptoms, FEV1 or AHR did not significantly improve over time.
The DREAM trial that followed up the two aforementioned studies recruited 621 patients with recurrent severe asthma exacerbations and evidence of eosinophilic inflammation [Pavord et al. 2012]. One of the aims of this study was to identify biomarkers of eosinophilic airway inflammation that were less invasive than induced sputum and were therefore more applicable in clinical practice. Patients were randomized into four groups: a placebo group and three treatment groups receiving 75, 250 and 750 mg of mepolizumab every 4 weeks. The primary outcome of the study was clinically significant asthma exacerbations over a year. These were reduced by 48% in the 75 mg group, 39% in the 250 mg group and 52% in the 750 mg group. Secondary outcomes were rate of exacerbations requiring admission, visits to the emergency department, blood and sputum eosinophil counts, prebronchodilator FEV1, and scores on the Asthma Quality of Life Questionnaire (AQLQ) and ACQ. Out of these, blood and sputum eosinophil counts were reduced; however, small and nonsignificant effects on FEV1, AQLQ and ACQ were observed. Because the DREAM study involved a large population and all three doses of mepolizumab had an equivalent clinical effect, there was considerable power to investigate baseline factors associated with a treatment response. Of the many variables assessed, only the prior exacerbation history and the baseline blood eosinophil count were associated with drug effects became apparent when the blood eosinophil count was above 0.15 × 109/liter. Another notable finding of the study was that the lowest dose of mepolizumab evaluated was as clinically effective as the higher dose on exacerbation rates and blood eosinophil counts but did not reduce sputum eosinophil counts significantly. This suggests that the main effect of treatment is to reduce circulating eosinophils and thus the influx of cells in response to increased signalling from the airway. Finally, the efficacy of the lowest intravenous dose meant that it was possible that subcutaneous administration would be effective.
Subsequent phase III RCTs evaluated this possibility in a population of patients with severe asthma, more than two exacerbations in the last year and a blood eosinophil count greater than 0.15 × 109/liter at screening or greater than 0.3 × 109/liter within the last year. The primary outcome was the degree of reduction of oral glucocorticoid use in one study and exacerbation rate in the other [Bel et al. 2014; Ortega et al. 2014]. Mepolizumab reduced the baseline oral glucocorticoid use by 50%, reduced exacerbations and improved ACQ significantly in the treatment arm of the smaller study (n = 135) [Bel et al. 2014]. In the larger study (n = 621) exacerbation rate was reduced by 47% among patients receiving 75 mg intravenous mepolizumab and 53% among those receiving 100 mg subcutaneous mepolizumab [Ortega et al. 2014]. There was also a modest mean increase in baseline FEV1 of 100 ml in the treatment groups compared with placebo and the St George’s Respiratory Questionnaire (SGRQ) scores sig-nificantly improved in those groups. A near to significant improvement was also seen in the ACQ scores. These mepolizumab studies are the first to show both clear biological effects and effects on clinically significant outcome measures, such as the exacerbation rate, glucocorticoid use, FEV1, ACQ and SGRQ. The more clear evidence of efficacy against these outcomes may reflect selection of a more responsive population as a result of use of the peripheral blood eosinophil count.
Reslizumab
Reslizumab is a monoclonal antibody like mepolizumab that is directed at the IL-5 molecule itself. In a study with monkeys it showed a long-term effect that could still be measured after 6 months [Egan et al. 1999]. This makes it a promising compound for clinical use. In an early study with patients with asthma, reslizumab exhibited a terminal half life of approximately 25 days [Kips et al. 2003]. With higher dosages (1.0 mg/kg) it showed an effect on peripheral blood eosinophilia up till 30 days after the administration of the drug. In a more recent RCT 106 patients with poorly controlled, eosinophilic asthma, assessed by induced sputum, were included and randomly assigned to receive placebo or reslizumab treatment. Reslizumab showed to significantly improve lung function, reduce sputum eosinophils and improve ACQ overall, and especially in a subgroup with nasal polyps. The researchers did report a higher percentage of nasopharyngitis in the treatment arm and one patient had to stop reslizumab infusions because of worsening of asthma. The results of two large phase III trials with reslizumab that are currently being performed might give clearance about the occurrence of adverse events. Nasopharyngitis is one of the most reported side effects in mepolizumab studies as well, however occurrence was similar in the placebo arms.
Benralizumab
Benralizumab is a monoclonal antibody directed against the α chain of the IL-5R. It has been proven to lower peripheral blood eosinophil count, reduce tissue eosinophilia and lower sputum eosinophil counts in a similar way to mepolizumab [Laviolette et al. 2013]. In a phase IIb dose-ranging study 324 patients with eosinophilic and noneosinophilic asthma were randomized and allocated to receive either placebo or benralizumab in different dosages [Castro et al. 2014]. Eosinophilic status was determined by using the eosinophil/lymphocyte and eosinophil/neutrophil index. Benralizumab in dosages of 20 mg and 100 mg reduced exacerbations in patients with uncontrolled asthma and high peripheral blood eosinophilia (>0.3 × 109/liter). In this particular group ACQ and FEV1 also improved.
The results of a recent RCT with patients who suffer from eosinophilic COPD are very much in line with the findings in patients with asthma [Brightling et al. 2014]. Patients with eosinophilic COPD are characterized by having sputum eosinophilia and more frequent exacerbations then their noneosinophilic counterparts. Interestingly, again the subgroup with peripheral blood eosinophilia showed an improvement in SGRQ scores, Chronic Respiratory Questionnaire-Self Assessed Standardized, and FEV1 in the post hoc analysis.
Conclusion
The clinical value of anti-IL-5 treatment continues to be confirmed by trials using various dosages, different ways of administration (intravenous/subcutaneous) and, most importantly, different patient subgroups. Overall patient selection in combination with a suitable primary endpoint has been the key strategy to prove the benefit of mepolizumab. The results from benralizumab and reslizumab trials strengthen this concept. Primary endpoints have been evolving from AHR and lung functional measurements in early studies to exacerbation frequency and glucocorticoid-sparing effects in recent trials. Patient selection went from patients with mild asthma to patients with uncontrolled asthma and airway eosinophilia and eventually to patients with peripheral blood eosinophilia and frequent exacerbations.
The efficacy of anti-IL-5 has been described in two different ways: on the one hand by assessing biomarkers and on the other by looking at clinical markers (Table 3). A strong biological effect has been shown in all studies. Anti-IL-5 dramatically lowers peripheral blood eosinophilia, lowers the percentage of bone marrow eosinophils and lowers airway eosinophils in all patients with asthma regardless of their clinical or inflammatory phenotype. This effect lasts up to 30 days; benralizumab has a more sustained and perhaps more marked effect. The clinical effects of treatment are very much influenced by patient selection and are therefore more difficult to assess in a systematic way. The most important lesson is that there should be a correlation between the patient selection criterion (e.g. peripheral blood eosinophilia) and the primary outcome of the study, such as exacerbation frequency. Therefore it has only been in recent trials that the clinical value of IL-5 has been established by showing a reduction in exacerbation frequency, improvement of symptoms scores, quality of life and even lung function (FEV1) in patients who have severe eosinophilic asthma.
The safety profile of mepolizumab has been up to par with the placebo arms in the RCTs performed (Table 2). Benralizumab has been characterized by a solid safety profile as well. Reslizumab needs more data to support its safety profile; in the one RCT that was performed there was a higher incidence of nasopharyngitis. However, overall, anti-IL-5 treatment appears to be safe for clinical use.
Safety table for mepolizumab.

AE, adverse event; SAE, severe adverse event.
Efficacy table for mepolizumab.

+, clinically improved; 0, measured and no effect observed; N/A, not attributable/not measured; unclear, measured, not enough data points for conclusion.
ACQ, Asthma Control Questionnaire; AHR, airway hyperresponsiveness; eos, eosinophil; FeNO, fraction of exhaled nitric oxide; FEV1, forced expiratory volume in 1 s; IL, interleukin; QLQ, Quality of Life Questionnaire; RCT, randomized controlled trial.
The clinical efficacy and safety of anti-IL-5 treatment has been very promising and makes it an attractive alternative to oral corticosteroids in patients with severe eosinophilic asthma. Although it is likely to be a costly treatment, this patient group accounts for the majority of the asthma-related healthcare costs, suffers extensively from the side effects of high doses of systemic glucocorticoid treatment, and therefore is very much in need of a drug that is able to control asthma exacerbations with minimal side effects.
Footnotes
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
Funding
BH declares that he has received sponsorship from Novartis and GSK to attend international scientific meetings. LX has received funding from NIHR Biomedical Research Centre Programme, British Medical Association, Oxfordshire Health Services Research Committee Research Grant and NIH. IDP has received speaker’s honoraria for speaking at sponsored meetings from Astra Zeneca, Boehringer Inglehiem, GSK. He has received honoraria for attending advisory panels with Almirall, Astra Zeneca, Boehringer Ingelheim, GSK, MSD, Schering-Plough, Novartis, Dey, Napp. He has received sponsorship to attend international scientific meetings from Boehringer Ingelheim, GSK, Astra Zeneca and Napp. He is Chief Medical Advisor to Asthma UK, a member of the UK Department of Health Asthma Strategy Group, a member of the BTS SIGN Asthma guideline group and joint editor in chief of Thorax. Neither IDP nor any member of his family has any shares in pharmaceutical companies.
