Abstract
Background:
Allergic bronchopulmonary aspergillosis (ABPA) is a severe complication in patients with cystic fibrosis (CF), resulting in deterioration of lung function and impairment of overall prognosis. Standard therapy consists of high dosage, long-term corticosteroid treatment. This carries the risk of serious side effects such as immune suppression, diabetes and osteoporosis. Antifungal drugs such as itraconazole may cause interactions with other drugs and drug levels need to be monitored. Omalizumab treatment has been tried in several case studies.
Methods:
This was a retrospective study of six patients (four female, two male, age 4–33 years old) with CF and ABPA treated with omalizumab within an observation period of 7.5 years.
Results:
All patients showed clinical and laboratory stability or even an improvement within the treatment and post-treatment observation period, although omalizumab therapy was less effective in patients with progressed lung disease and long-term ABPA. Side effects of systemic steroids were reduced.
Conclusion:
Omalizumab has the potential to be an additional and solitary treatment option in patients with CF and ABPA. Early onset treatment may be beneficial and patients with early stage of lung disease seem to benefit more.
Introduction
An estimated 1–25% of patients with cystic fibrosis (CF) develop allergic bronchopulmonary aspergillosis (ABPA) during their lifetime. The current prevalence in the German registry is about 6% and increases with age [Wong et al. 2013; Moss, 2010; Fillaux et al. 2014; Stevens et al. 2003]. A higher incidence may occur during pulmonary colonization with Aspergillus fumigatus, sensitization and genetic susceptibility for atopy [Stevens et al. 2003]. Progressive deterioration of lung function is the major complication of the disease and relapses occur frequently [Baxter et al. 2013]. Recommended treatment with systemic steroids very often results in severe side effects, e.g. immune suppression, disturbance of glucose homeostasis, cataracts and osteoporosis [Wong et al. 2013]. Antifungal drugs, e.g. itraconazole, used to reduce the antigen burden, require monitoring of drug levels, may interact with other medications or cause side effects themselves [Ranhawa et al. 2009; Elphick and Southern, 2012].
The pathogenesis of ABPA has still not completely been clarified, but the presence of pathogenic immunoglobulin E (IgE) is one diagnostic hallmark of the disease [Kanu and Patel, 2008]. Omalizumab is a recombinant humanized monoclonal anti-IgE antibody and is an established therapy modality in allergic asthma. First case studies exist reporting the use of omalizumab for the treatment of ABPA in CF patients, with differing results [Randhawa et al. 2009; Brinkmann et al. 2010; Zirbes and Milla, 2008; Van der Ent et al. 2007; Lebecque et al. 2009a, 2009b]. The aim of this study was to describe our experience with omalizumab in patients with CF and ABPA.
Methods
This was a retrospective analysis of patients with CF and ABPA receiving treatment with omalizumab from 2006 to spring 2014. All patients were treated at the Department of Paediatric Pulmonology of the RWTH Aachen University Hospital, the Practice for Paediatric Pulmonology Aachen and the Department of Cystic Fibrosis for Adults, Luisenhospital Aachen. All patients gave their written informed consent for retrospective analysis of their ABPA history. Diagnosis of CF in all patients was approved by cystic fibrosis transmembrane conductance regulator (CFTR) mutations.
Diagnosis of ABPA was based on the criteria of the ABPA Consensus Conference [Stevens et al. 2003] including at least five criteria: (a) asthma/bronchial obstruction; (b) immediate cutaneous reactivity to Aspergillus species; (c) elevated total serum IgE levels (>417 IU/ml or 1000 ng/ml); (d) elevated serum IgE to Aspergillus fumigatus; (e) immunoglobulin G (IgG) to A. fumigatus (precipitines); and (f) central bronchiectasis, new pulmonary infiltrates or mucus plugging. Omalizumab was administered subcutaneously at fortnightly intervals. Dosage was individually adapted to body weight and IgE level at the beginning of treatment – referring to the prescribing recommendation of omalizumab for allergic asthma. If IgE levels were above the limits of the licence, the maximum possible dosage of omalizumab with reference to the body weight was administered (600 mg fortnightly). Treatment was started after approval of off-label therapy by the health insurance companies.
Patients
Patient No. 1 (female, CF diagnosed at the age of 2 years, Δf508/G542X, severe noncompliance)
At the age of 4 years she had the first diagnosis of ABPA, with a decline in the forced expiratory volume in one second (FEV1) from 73 to 41%. Therapy was performed with oral prednisone and itraconazole. As clinical and laboratory results improved, therapy was discontinued after 16 weeks of treatment.
Two months later (see Figure 1, Patient No. 1) the patient had the first relapse of ABPA (FEV1 55%). Another treatment course of oral prednisone and itraconazole was started; however, 4 weeks later she developed a steroid-induced diabetes. Omalizumab was started and oral prednisone was discontinued within 16 weeks after starting omalizumab. After 32 weeks of treatment, omalizumab therapy was withdrawn (FEV1 51%). However, the first evidence of Pseudomonas aeruginosa colonization was detected during steroid-induced immune suppression within the first 16 weeks.

Course of FEV1 (%), HbA1C (%) and total IgE (kU/l) within each patient during treatment with omalizumab.
Another 10 months later she presented with a second relapse of ABPA (FEV1 29%). Treatment was again started with oral prednisone and itraconazole. Due to progressive deterioration (FEV1 20%), therapy was intensified with omalizumab 5 weeks later. Again, steroids were reduced and FEV1 increased to 29% at week 4 and 42% at week 8. Fulminant infective exacerbations were treated with intravenous (iv) antibiotics. The patient recovered, although she unexpectedly died at the age of 7 years due to septic shock caused by gastrointestinal infection with Clostridium difficile 3 weeks after iv antibiotic treatment.
Patient No. 2 (female, CF diagnosed in infancy, ΔF 508/G542X)
The first diagnosis of ABPA was at the age of 14 years. She presented with pulmonary exacerbation and a deterioration of general condition. FEV1 decreased from 73% to 40% (see Figure 1, Patient No. 2). After treatment with oral prednisone and itraconazole, she developed a steroid-induced diabetes within 7 weeks. Insulin therapy and omalizumab treatment was started. Already 4 weeks later prednisone was reduced to 2 mg/day, FEV1 increased to 58% and diabetes was stable. Omalizumab and steroid treatment was discontinued after 6 months and the patient’s condition had stabilized (FEV1 55%).
However, 25 months later she presented with a relapse of ABPA (FEV1 25%). Treatment was immediately started with prednisone, itraconazole and omalizumab. Prednisone was reduced to 2.5 mg/day within 4 weeks. After 12 months omalizumab was discontinued (FEV1 43%). However, during treatment with omalizumab and after discontinuation, she was still dependent on prednisone 2.5 mg/day since all efforts to discontinue failed due to severe pulmonary obstruction. Insulin is still necessary and osteoporosis was diagnosed at interim.
Patient No. 3 (female, CF diagnosed in infancy, 1717-1G->A/E60X)
The first ABPA was diagnosed at the age of 11 years. Levels of FEV1 declined from 106% to 72%. Treatment was performed with oral prednisone and voriconazole, as itraconazole was not tolerated. The patient developed a weight gain of 10 kg, Cushing syndrome and a first colonization with Pseudomonas aeruginosa within the long-term treatment of 22 months was detected.
Some 56 months later (see Figure 1, Patient No. 3) the patient presented with a relapse of ABPA (FEV 1-decline from 81 to 52%). Due to pathological blood glucose levels, treatment with oral prednisone was reduced quickly. Omalizumab was started 10 weeks after the diagnosis of the relapse and steroids were discontinued within 6 weeks. She had stable glucose levels without insulin treatment and lung function improved. At this moment, she has had 9 months on omalizumab and therapy is planned for at least 12 months.
Patient No. 4 ( male, CF diagnosed at the age of nine years, ΔF 508 homozygous)
ABPA was first diagnosed at the age of 18 years. The first relapse was 36 months and the second 4 months later. Each time treatment with oral prednisone and itraconazole was successful, although during immunosuppressive therapy, the patient was first colonized with Pseudomonas aeruginosa and Stenotrophomonas maltophilia.
Another 23 months later (see Figure 1, Patient No. 4) the patient suffered from a third relapse of ABPA (FEV1 29–23%). Treatment was again started with oral prednisone and itraconazole. However, he presented with a deterioration of clinical condition and a deterioration of lung function (FEV1 21%) and omalizumab was added 4 months later. Lung function slowly improved. FEV1 increased (23% after 8 months, 31% after 13 months). Prednisone was reduced to 15 mg/day, although a discontinuation failed several times. No disturbance of glucose homeostasis appeared, but the patient is still in a reduced general condition with high rates of infective exacerbations, steroid- and omalizumab-dependency. Currently, he refuses evaluation for lung transplantation. At this moment, he has had 13 months on omalizumab.
Patient No. 5 (female, CF diagnosed after birth, Δf508/E60X)
The first ABPA was diagnosed at the age of 26 years. The first relapse occurred 6 years later. Both times treatment with prednisone and itraconazole was successful. However, pre-existing insulin-dependent diabetes was aggravated and insulin dosage needed to be adapted each time.
A second relapse occurred (FEV1-decline from 90 to 72%) 12 months after the first relapse (see Figure 1, Patient No. 5). Treatment was immediately started solely with omalizumab. Already 4 weeks later the patient demonstrated a clinical and laboratory improvement (FEV1 82%). She has now had 7 months on omalizumab treatment. Lung function is stable (FEV1 88%), no pulmonary infectious complications are documented and diabetes is stable. Omalizumab therapy is planned for at least 12 months.
Patient No. 6 (male, CF diagnosed in infancy, ΔF 508 homozygous)
The first ABPA was diagnosed at the age of 19 years and successfully treated with oral prednisone and itraconazole. During immunosuppressive therapy, however, multiresistant Staphylococcus aureus in the sputum was detected and 20 months later the patient had a first relapse of ABPA. Another successful treatment with oral prednisone and itraconazole followed; nevertheless he remained dependent on 20 mg/day of oral prednisone.
The second relapse of ABPA (see Figure 1, Patient No. 6) was 35 months later (FEV1 decline from 50 to 38%). Under prednisone and itraconazole, he developed an insulin-dependent diabetes and had frequent pulmonary exacerbations. Omalizumab was added after 2 months; the patient benefited clinically and lung function improved within 4 months (FEV1 63%). Within 2 months of additional treatment with omalizumab, oral prednisone was reduced to 10 mg/day – the first time after 3 years. Glucose homeostasis and lung function is stable. The patient has now been on omalizumab for 9 months and follow-up visits will show if a complete discontinuation of oral prednisone is possible. Omalizumab therapy is planned for at least 12 months.
Results
From 2006 to spring 2014, 51 children and adolescents and 83 adult patients with CF were treated in our centre. Within this time, three (5.8%) children and six (7.2%) adult patients received a diagnosis of ABPA. While three patients underwent established and successful treatment with prednisone and itraconazole, six patients were treated off-label with omalizumab. All of these six patients presented with relapses of ABPA during the observation period. One patient had the first episode of ABPA; the others had the first to third relapse when treated with omalizumab. Indications for omalizumab off-label use were side effects of corticosteroid therapy or treatment failure.
Details of available patient data are displayed in Table 1. The gender distribution was 4:2 (female:male). The average age of the patients at first diagnosis of ABPA was 15.61 years (±7.19, range 4.8–26 years). The average age of starting treatment with omalizumab was 19.47 years old (±9.64, range 5.3–33.2 years). The average duration of observation during and after omalizumab treatment was 19.67 months (±19.19, range 7–56 months).
Data of patients with CF and ABPA treated with omalizumab.

Patient No. 1 and No. 2 with each twice ABPA diagnoses treated with omalizumab (first in the left and second in the right field).
ABPA, allergic bronchopulmonary aspergillosis; CF, cystic fibrosis; IgE, immunoglobulin E; n/a, not applicable; sIgE, specific Immunoglobulin E antibodies; sIgG, specific Immunoglobulin G antibodies; rAsp f4, rAsp f6, specific IgE antibodies to Aspergillus fumigatus recombinant allergens 4 and 6.
Focusing on the first treatment course with omalizumab (Figure 2), average FEV1 of all patients before diagnosis of ABPA was 67.5% (range 29–91%, ±24.07%). At diagnosis of ABPA, average FEV1 decreased to 46.33% (range 21–72%, ±17.42%). At the beginning of treatment with omalizumab, patients had an average FEV1 of 57.5% (range 23–86, ±22.29%). In all patients 3–4 months after beginning of omalizumab, average FEV1 increased to 62.33% (range 31–85 ±19.77%). Average FEV1 6–9 months after omalizumab was 59.83% (range 31–89%, ±19.47%). Only two patients had a second treatment course with omalizumab 10 and 25 months after discontinuation of the first omalizumab treatment. Under omalizumab these two patients showed an increase of FEV1 and a reduction of steroids.
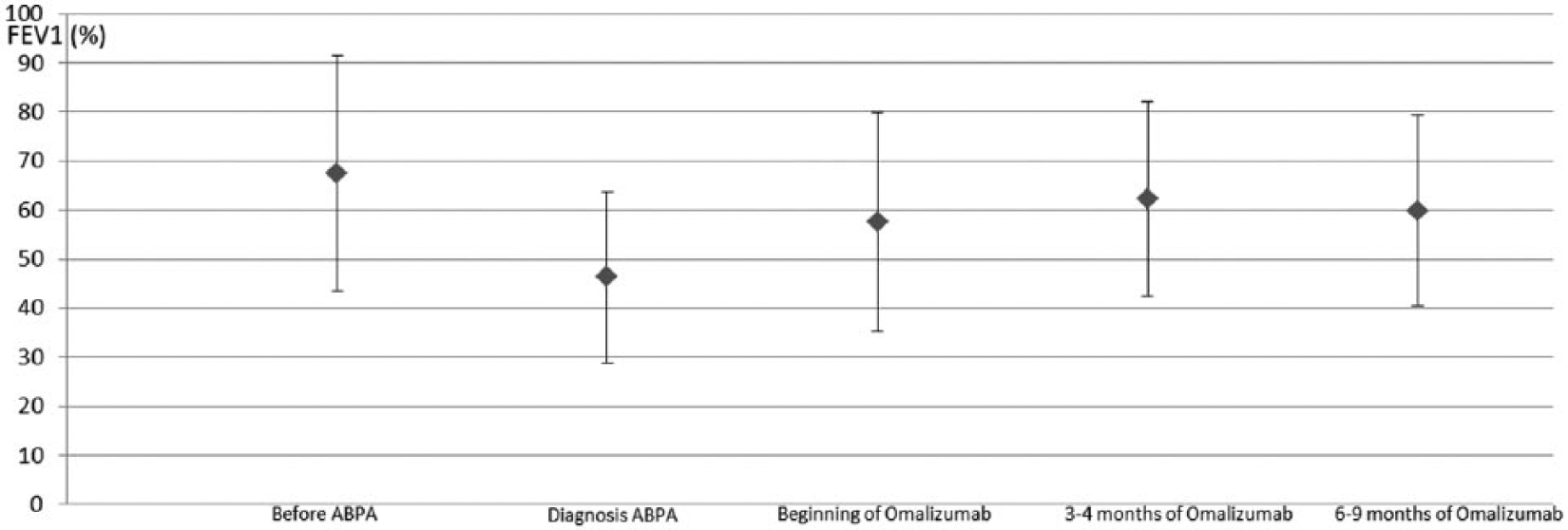
Trendline of average FEV1 (%) (including standard deviation) of all patients during the first treatment with omalizumab.
Within all patients a reduction (4/6 patients) or even cessation (1/6) or absence of systemic steroids (1/6) was observed during treatment with omalizumab. As a result side effects of systemic steroids were reduced. No patient showed adverse reactions to omalizumab. ABPA relapses during treatment with omalizumab did not appear, although relapses occurred after discontinuation of treatment.
In detail, Patient No. 1 showed a good response to omalizumab. During the first course steroids were discontinued and reduced during the second course. Glucose homeostasis was stable. However, under immunosuppression, recurrent infective exacerbations occurred.
In Patient No. 2 we saw a very good response to omalizumab at the first use, steroids were reduced and diabetes improved. Before the second omalizumab course CF-related lung disease had worsened and treatment with steroids had to be continued despite side effects (diabetes and osteoporosis). Lung function remained at much lower levels than before and after the first ABPA.
Patient No. 3 improved significantly during omalizumab treatment and steroids were completely discontinued within 6 weeks. Lung function – before ABPA and during omalizumab treatment – is almost normal.
Patient No. 4 had an already severely reduced lung function when, during the third relapse of ABPA, omalizumab was added due to hesitant response to steroids and itraconazole. Even though the patient is stable on additional omalizumab he is still steroid-dependent on a lower dosage of prednisone and has high rates of infective exacerbations.
In Patient No. 5 we saw successful ABPA treatment with exclusive omalizumab therapy at the second ABPA relapse, and recovery was quick and good. FEV1 before and during ABPA treatment with omalizumab is almost normal.
Patient No. 6 needed continuously 20 mg/day prednisone for 3 years after the first ABPA relapse. He developed diabetes. After the second relapse omalizumab was introduced and prednisone was halved due to clinical improvement.
Discussion
The risk of developing ABPA is higher in CF patients than in the asthmatic population (1–2%). Within the past two decades, an increase of ABPA in CF patients has been observed. Reasons for this might be awareness of this complicating disease, and availability of diagnostic algorithms and procedures [Agarwal, 2009]. It is known that sensitization to Aspergillus depends on the mode and frequency of exposure. Patients with CF have frequent treatment with antibiotics – systemic and inhaled. Observations suggest that this might be a risk factor for colonization of the bronchial tree with Aspergillus or other fungi. Moreover, CF patients have abnormal mucus and spores of Aspergillus might be trapped within the bronchial airways, increasing the frequency and/or activity of Aspergillus fumigatus-specific Th2 CD4+ cells [Stevens et al. 2003]. Furthermore, in CF patients and persons with heterozygous carriage of CFTR mutations, ABPA seems to be more frequent as a result of genetic susceptibility [Stevens et al. 2003; Muro et al. 2013].
It is not clear whether treatment of colonization of the bronchial tissue with Aspergillus fumigatus might reduce the incidence of ABPA development, especially as oral antifungals may cause drug interactions and side effects [Moss, 2010]. Study groups have tried to identify markers, e.g. Galaktomannan, Aspergillus polymerase chain reaction (PCR), to differentiate between colonization and infection of Aspergillus in CF patients, but clear results are absent. Observations show that colonization with Aspergillus in CF patients is associated with a higher risk of sensitization to Aspergillus and development of ABPA [Moss, 2010; Fillaux et al. 2014; Baxter et al. 2013]. Additionally, colonization with Aspergillus is associated with deterioration of lung function, higher rates of hospital admissions, higher frequency of colonization with Pseudomonas aeruginosa and pathologic radiologic findings of the lung in CF patients [Fillaux et al. 2014; Speirs et al. 2012].
Systemic steroids are the mainstay of ABPA therapy. The anti-inflammatory effects result in inhibition of phospholipase A2-activity, arachidonic acid metabolism, chemotaxis, cell adhesion, tissue infiltration of inflammatory cells and production of interleukin-1 (IL-1) and tumour necrosis factor (TNF) [Stevens et al. 2003]. Dosage recommendations exist, either for oral prednisone or iv pulse methylprednisolone [Cohen-Cymberknoh et al. 2009]. It is well-known that long-term use of systemic steroids may cause infections due to immune suppression, disturbance of glucose homeostasis, hypertension, adrenal insufficiency, weight gain, cataracts and osteoporosis. Second-line treatment with antifungal drugs, e.g. itraconazole and voriconazole, reduces the antigen burden. Treatment of ABPA in CF patients with antifungals can be of value, but also has several drawbacks, e.g. interactions with other drugs, side effects and problems finding the right drug level. Moreover, a direct therapeutic effect remains to be shown [Elphick and Southern, 2012; Speirs et al. 2012; Jat et al. 2013].
Inhaled steroids or antifungals and oral montelukast are not beneficial in ABPA [Moss, 2010; Rundfeld et al. 2013]. Several case reports detail using omalizumab for ABPA treatment in CF [ElMallah et al. 2012]. Omalizumab is a monoclonal anti-IgE antibody, thus blocking docking of specific IgE on mast cells and basophils. During treatment it reduces free IgE, but anti-IgE–IgE complexes may result in elevations of total IgE. Thus, monitoring of total IgE antibody titres alone delivers no sufficient information of disease activity. Moreover, IgE can also be produced against other antigens [Mroueh and Spock, 1994]. Detection of free-IgE levels might be helpful, though only selected laboratories perform this by solid phase immunoenzymetric assay [Zirbes and Milla, 2008].
A downregulation of IgE receptors is an important consequence of omalizumab treatment, thus reducing inflammatory activity [Zirbes and Milla, 2008; ElMallah et al. 2012; Collins et al. 2012]. Moreover, omalizumab reduces airway eosinophilia and decreases IL-4+ cells in allergic asthma. These factors might also be relevant in ABPA [Tanou et al. 2014].
A significant steroid-sparing effect of omalizumab in children with severe asthma was recently documented in a therapeutic trial [Brodlie et al. 2012]. Clinical parameters, e.g. increase of FEV1, decrease of exacerbations, reduction of systemic steroids and subjective improvement [asthma control test (ACT)] were signs of a therapy response to omalizumab within this group. In omalizumab-treated CF patients with ABPA, laboratory findings (e.g. decrease of free IgE, serum eosinophil levels, markers of systemic inflammation) have not been fully evaluated [Collins et al. 2012].
Until now, no completed randomized, double-blind and controlled multicentre trial has been completed regarding omalizumab treatment for ABPA. The only study was finished preterm (ClinicalTrials.gov Identifier: NCT00787917, Novartis 2008, www.clinicaltrials.gov). Only 14 patients were recruited and results have not been published. Existing case studies mainly showed good clinical responses [Zirbes and Milla, 2008; Collins et al. 2012]. However, it is an expensive off-label therapy and publications exist reporting cases in which omalizumab treatment was not successful. In particular, omalizumab failed in a CF patient with ABPA for 4 years when treatment was started. After an initial response ABPA worsened under attempts to reduce systemic steroids [Brinkmann et al. 2010]. This is in concordance with what we found in this long-term observation. All patients in a good clinical condition, with early introduction of omalizumab showed a good response – irrespective whether it was the first diagnosis of ABPA or a relapse. In progressed lung disease, long-term diagnosis of ABPA or higher stages of ABPA, we observed that significant improvement was limited. Nevertheless, treatment with omalizumab kept our two patients (No. 2 and 4) on a stable level of lung function with lower doses of systemic steroids required. However, progression of CF-related lung disease can only be slowed by ABPA treatment with or without omalizumab since infections, inflammation and structural changes of the pulmonary framework have ongoing other pathomechanisms.
As this was a retrospective study within collaborating units but with different therapists, no clear treatment strategy existed for when and how omalizumab was introduced. A randomized, multicentre study should include strategies of treatment and parameters should comprise lung function, quality of life, laboratory parameters (including free IgE, serum and sputum eosinophils, and omalizumab blood levels), clinical and vital signs, and comedication. Therapy regimens should focus on finding the right treatment option for different phenotypes of ABPA patients with CF. Long-term observations should generate recommendations for how to reduce or discontinue omalizumab treatment preventing ABPA relapses.
Conclusion
In our retrospective analysis we have the impression that omalizumab is of value in treating ABPA in patients with CF. In particular, an effect of early treatment was observed and the potential for keeping patients stable with lower doses or absence of systemic steroids was determined. Nevertheless, only randomized clinical trials can answer questions regarding whether treatment with omalizumab is effective in the long term, which dosage regimen is the most effective, how long treatment should last and what kind of patients should be selected for treatment. Until then, we propose standard therapy of ABPA with systemic steroids and systemic antifungal drugs as first-line medication. In cases of nonresponse, an absence of an increase in lung function and a lack of falling IgE levels, as well as drug-induced side effects, omalizumab treatment should be considered.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
None of the authors has any financial or personal relationship with other people or organizations to disclose that could inappropriately influence (bias) their work.
