Abstract
Since the first description of allergic bronchopulmonary aspergillosis (ABPA) in the 1950s there have been numerous studies that have shed light on the characteristics and immunopathogenesis of this disease. The increased knowledge and awareness have resulted in earlier diagnosis and treatment of patients with this condition. This article aims to provide a summary and updates on ABPA by reviewing the results of recent studies on this disease with a focus on articles published within the last 5 years. A systematic search of PubMed/Medline with keywords of ABPA or allergic bronchopulmonary aspergillosis was performed. All selected articles were reviewed with a focus on findings of articles published from December 2006 to December 2011. The relevant findings are summarized in this paper.
Introduction
Allergic bronchopulmonary aspergillosis (ABPA) is a hypersensitivity lung disease that occurs almost exclusively in patients with asthma and cystic fibrosis (CF) in the setting of bronchial colonization by Aspergillus fumigatus, which is a ubiquitous saprophytic mold species that can cause a variety of pathologies in its host [Geiser et al. 2007]. As depicted in Figure 1, there are five major pulmonary diseases caused by A. fumigatus: ABPA, hypersensitivity pneumonitis, aspergilloma, chronic necrotizing Aspergillus pneumonia, and invasive aspergillosis [Soubani and Chandrasekar, 2002; Zmeili and Soubani, 2007]. ABPA manifests as recurrent pulmonary infiltrates, bronchiectasis and productive cough with mucus plugs in asthmatic and CF patients [Greenberger and Patterson, 1988; Rosenberg et al. 1977]. There are differences in characteristics, prevalence, diagnostic criteria and treatment of ABPA in asthmatics versus CF patients.
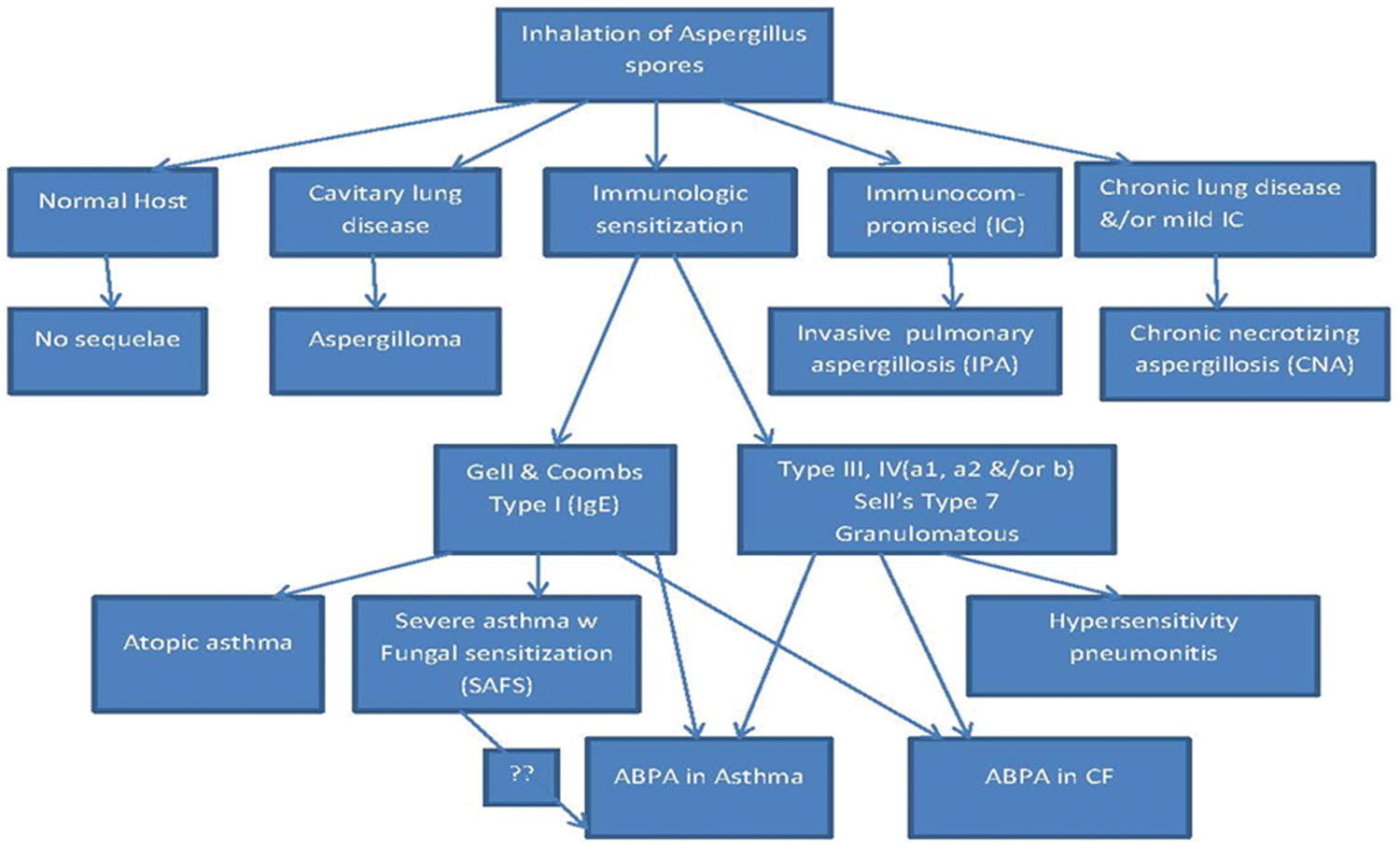
In human hosts with a normal immune system and normal lung architecture, the inhalation of Aspergillus spores results in no disease. If the host is immunocompromised, has abnormal lung architecture or develops hypersensitivity responses to Aspergillus, the following five pulmonary diseases may result: invasive aspergillosis; chronic necrotizing aspergillosis; aspergilloma; hypersensitivity pneumonitis; allergic bronchopulmonary aspergillosis (ABPA) in asthma; or cystic fibrosis (CF). Atopic asthma and severe asthma with fungal sensitization (SAFS) are associated with IgE against Aspergillus.
The pathogenesis of ABPA is complex and involves cascades of immunologic reactions including Aspergillus-specific IgE-mediated (type I) hypersensitivity, IgG-mediated immune complex (type III) hypersensitivity, and abnormal cell-mediated immune responses [Knutsen, 2003, 2011]. In genetically susceptible individuals, these hypersensitivity responses in the bronchi and bronchioles result in mucus impaction and inflammatory cell infiltration in bronchial wall and peribronchial tissues, that can progress to organizing pneumonia, bronchiectasis and bronchocentric noncaseating granulomatosis [Greenberger, 2002; Knutsen et al. 2012].
ABPA is generally more common among severe asthmatic patients who are steroid dependent. While the prevalence of ABPA was reported to be as low as 1–2% in asthmatic patients in outpatient clinics [Donnelly et al. 1991; Novey, 1975], this rate was reported as 7–14% among steroid-dependent asthma patients who were referred to specialty clinics [Basich et al. 1981], and was even higher (39%) in patients who have been admitted to the ICU with severe asthma attacks [Agarwal et al. 2010b]. The prevalence of ABPA in CF ranges from 6% to 10% [de Almeida et al. 2006; Chotirmall et al. 2008]. Untreated disease for prolonged periods of time can result in irreversible pulmonary damage. Therefore early detection and treatment of ABPA is critical in preventing serious lung damage such as bronchiectasis [Greenberger, 2003].
Since the first description of ABPA in the 1950s [Hinson et al. 1952], there have been numerous studies that have shed light on the characteristics and immunopathogenesis of this disease. The increased knowledge and awareness have resulted in earlier diagnosis and treatment of patients with this condition. This article aims to provide a summary and updates on ABPA by reviewing the results of recent studies on this disease with focus on articles published within the last 5 years. A systematic search of PubMed/Medline with keywords of ABPA or allergic bronchopulmonary aspergillosis was performed. All selected articles were reviewed with focus on findings of articles published from December 2006 to December 2011. The relevant findings are summarized in this paper.
Diagnostic criteria
The diagnosis of ABPA in asthma is based on clinical features and immunologic sensitivity to A. fumigatus. The Rosenberg–Patterson criteria was described in 1977 for diagnosis of this disease [Rosenberg et al. 1977]. According to the authors, the presence of six of the seven major criteria made the diagnosis likely, and the presence of all seven made it certain.
The criteria were further modified to the minimal essential criteria as detailed in Table 1 [Greenberger, 2003; Schwartz and Greenberger, 1991] which is currently widely used. High-resolution CT (HRCT) of the chest is the imaging modality of choice for the diagnosis of ABPA. There are patients with ABPA who fulfill diagnostic criteria but do not have the CT chest finding consistent with proximal or central bronchiectasis; this group of patients is labeled as serological ABPA (ABPA-S). In contrast, ABPA-CB refers to patients with central bronchiectasis [Greenberger et al. 1993].
The minimal essential criteria for allergic bronchopulmonary aspergillosis (ABPA) diagnosis.

More recently another phenotype of severe asthma has been defined. These asthma patients have positive skin prick test to Aspergillus but their antibody levels to A. fumigatus is below the threshold for diagnosis of ABPA and thus do not fulfill the minimal criteria; this group of patients are classified as severe asthma with fungal sensitization (SAFS) [Agarwal et al. 2011; Denning et al. 2006]. It should be noted that patients with ABPA might not fulfill serologic criteria except when they are in acute or exacerbation stages of the disease. Therefore, it is recommended to repeat the serologic tests in this group of patients during recurrent exacerbation.
There is controversy on the threshold for total IgE level used for diagnosis with some centers using 1000 ng/ml (equivalent to 417 IU/ml) [Greenberger, 2003] and others using 1000 IU/ mL [Agarwal et al. 2006; Pa, 2003]. There are no studies comparing these levels with respect to response to treatment or prognosis in cohorts of asthmatics.
Agarwal and colleagues proposed an algorithm for the diagnosis of ABPA starting with screening of all asthmatic patients for Aspergillus sensitization using skin testing [Agarwal et al. 2006; Agarwal, 2009]. Patients who demonstrate immediate hyperreactivity to Aspergillus antigen would be further tested for total IgE level. Depending upon the result, additional tests such as eosinophil count, Aspergillus precipitins and HRCT would be obtained. The diagnosis of ABPA in CF is more controversial due to overlap between clinical and radiological features of ABPA and CF. Traditionally the Nelson criteria was the most commonly used; the most recent diagnostic criteria proposed by the Cystic Fibrosis Foundation Consensus Conference was published in 2003 (Table 2) [Greenberger, 2002, 2003; Stevens et al. 2003].
Criteria for the diagnosis of allergic bronchopulmonary aspergillosis (ABPA) in patients with cystic fibrosis.

(From the ABPA Consensus Conference of the Cystic Fibrosis Foundation [Stevens et al. 2003].)
As many CF patients demonstrate a spontaneous decline in immunologic parameters, such as IgE levels, it has been proposed that the diagnosis of ABPA in CF patients should not be based only on serology and skin test results. Instead, prolonged longitudinal testing with newer markers might be required to make a definite diagnosis [Antunes et al. 2010]. New serological tests such as specific IgE to recombinant A. fumigatus allergens, or thymus and activation regulated chemokine (TARC/CCL17), have been identified as potential diagnostic markers for ABPA in CF [Delhaes et al. 2010].
Clinical features
ABPA usually presents with increased cough, sputum production or wheezing [Safirstein et al. 1973]. Sputum may consist of tan to brownish–black mucus plugs [de Almeida et al. 2006]. The dark mucus plugs are due to increased production of tenacious mucus in the respiratory tract and consist of inflammatory cells including eosinophils, desquamated epithelial cells, and mucin [Kradin and Mark, 2008]. These mucus plugs have been reported in up to 69% of patients in different ABPA series [Agarwal et al. 2006; Agarwal, 2009].
Hemoptysis may occur in late-stage disease (stage V) and is primarily due to severe airway inflammation and bronchiectasis [Kumar, 2003].
Systemic symptoms including low-grade fever, malaise, and weight loss are seen in up to 26% of ABPA patients [Prasad et al. 2009]. The presence of these symptoms in a patient with asthma or CF should prompt an evaluation for ABPA; prior to the diagnosis of ABPA, the combination of fever and productive coughing can be misinterpreted as pneumonia or tuberculosis, especially in context of pulmonary infiltrates on chest X-rays [Agarwal et al. 2006; Ragosta et al. 2004].
In a recently reported cohort of ABPA patients, it was shown that the disease can be present in asymptomatic asthmatics, these patients were diagnosed on routine screening of asthmatics showing that screening can lead to earlier diagnosis of ABPA and possibly better prognosis [Agarwal et al. 2007].
Aspergillus can also cause hypersensitivity in paranasal sinuses manifesting as allergic Aspergillus sinusitis (AAS). It has been proposed that ABPA must be excluded in all patients with AAS and vice versa [Panjabi and Shah, 2011].
Staging
Patterson and colleagues have classified the clinical course of ABPA into five stages that helps to guide the management of the disease [Patterson et al. 1986]. It is important to note that ABPA serology is most likely to be positive in stage I and III (Table 3). The ideal time to send ABPA serology is prior to initiation of prednisone for acute or exacerbation stages (stage I or III).
Allergic bronchopulmonary aspergillosis (ABPA) stages and serologic diagnosis.

Stage I (acute stage) is defined as the initial symptomatic stage of ABPA with radiographic infiltrates, raised IgE levels, and elevated A. fumigatus-specific IgG/IgE. Stage II (remission stage) is the clinical and serologic remission; and is defined as 35–75% decline in IgE levels by 6 weeks, clearing of the chest infiltrates and symptomatic improvement. In stage II, serology may be negative. Stage III (exacerbation stage) is the recurrence of the initial symptoms and up to a twofold increase in serum IgE levels. Stage IV (corticosteroid-dependent asthma stage) is the uncontrolled symptomatic phase and patients in this stage need continuous corticosteroids to control their asthma and prevent recurrence of ABPA symptoms. Serology is often negative in stage IV. Stage V (fibrotic stage) is the fibrocavity disease due to prolonged inflammation; it is diagnosed with presence of upper lobe fibrosis on the chest imaging. If the patient reaches stage V, the fibrotic lesions will not be improved by treatment with corticosteroids, although steroids are often necessary to maintain a bronchodilator response and prevent severe asthma symptoms. ABPA serology may be negative in Stage V. Pulmonary fibrosis is an advanced complication that can result in pulmonary hypertension, right heart failure, and death [Patterson et al. 1986].
It worth mentioning that ABPA does not always evolve sequentially through all of these stages. Early diagnosis and treatment is thought to be associated with a lower risk of advanced disease in the future [Patterson et al. 1986; Tillie-Leblond and Tonnel, 2005].
Imaging studies
ABPA can have a wide spectrum of radiographic appearances.
HRCT of chest with thin (1–2 mm) sections is considered the imaging modality of choice for the diagnosis of ABPA [Lynch, 1998].
The major findings on HRCT chest are central bronchiectasis (CB), air trapping (mosaic attenuation), and high-attenuation mucus (HAM) caused by mucus plugs or mucoid impactions. Some of the other findings include bronchial wall thickening, atelectasis, lobar or whole lung collapse, pulmonary fibrosis, centrilobular nodules, and cavities with or without air-fluid levels [Logan and Muller, 1996].
Central bronchiectasis manifests as dilated bronchi that have a larger caliber (1.5 to >3 times) than the adjacent bronchial artery in the central two thirds of the lung field. Bronchiectasis is further classified to cylindrical, varicose or cystic. Patients with ABPA can have any type of bronchiectasis involving multiple bronchi; upper lobes are more likely to be involved. Of note asthma patients can also have bronchial wall thickening or localized areas of cylindrical bronchiectasis. However, the extent of lung involvement is much less in asthma patients mostly limited to one or two lobes [Mitchell et al. 2000; Ward et al. 1999].
The transient areas of consolidation seen frequently in ABPA are predominately in the upper lobes, caused by bronchial obstruction with mucus plugs. The bronchus filled with mucus results in a band shadow or glove-finger shadow on the plain radiographs that may be relieved by coughing up a mucus plug [McCarthy et al. 1970].
The impacted mucus in the airways can manifest as the ‘tree-in-bud pattern’ on HRCT scan and can have normal or high attenuation. In HAM, the higher concentration of ions such as calcium, iron, and manganese results in higher Hounsfield density when compared with surrounding normal skeletal muscle [Ward et al. 1999].
Classification
Classically ABPA is categorized based on presence of the radiographic finding of central bronchiectasis. Imaging modalities in this way are major tools to predict the outcome and guide treatment. Greenberger and colleagues classified patients as ABPA-S, who otherwise fulfill all diagnostic criteria, but lack demonstrable abnormalities on CT chest, and ABPA-CB, who have central bronchiectasis in the imaging [Greenberger et al. 1993]. Another CT classification categorizes ABPA into mild (ABPA-S), moderate (ABPA-CB) and severe (ABPA-CB with other radiologic findings; ABPA-CB-ORF) [Kumar, 2003].
Multiple studies have shown that patients with ABPA-S developed fewer exacerbations and do not progress to irreversible lung disease [Greenberger et al. 1993; Kumar and Chopra, 2002].
A recent study correlated the chest HRCT findings with the immunological severity in ABPA. Results of this study showed that presence of HAM was the most consistent marker of serological severity in ABPA. Agarwal and colleagues proposed this finding as an alternate method of classifying ABPA [Agarwal et al. 2010a]. The presence of HAM in HRCT of thorax has been shown to be associated with recurrent relapses of ABPA [Agarwal et al. 2007].
Laboratory studies
ABPA is characterized by a Th2 immune response with increased serum IgE, and eosinophilia [Chauhan et al. 1996; Kauffman, 2003]. Most of the laboratory findings in ABPA are based on this hypersensitivity response.
Aspergillus skin test
Cutaneous reactivity on skin-prick testing is the first step for diagnosis of ABPA and is a marker of immediate IgE-mediated response. A negative skin prick test should be followed by an intradermal test, which is more sensitive. The test is positive if a wheal and erythema is observed within 20 min [Ricketti et al. 1984]. Up to 28% of patients with asthma have positive skin test to Aspergillus but as mentioned before this is the initial screening and patients need to fulfill the minimal criteria for diagnosis of ABPA [Agarwal, 2011].
Total serum IgE levels
The IgE level is a useful marker for both diagnosis and follow up of disease activity. The values may reach to 30,000 IU/ml but the cut off needed for diagnosis of ABPA is greater than 417 IU/ml [Ricketti et al. 1984]. Much of the IgE is the result of polyclonal B-cell activation and not specific to Aspergillus [Knutsen, 2011].
Peripheral eosinophilia
An elevated blood eosinophil count of more than 1000 cells/ml is another potentially useful test for the diagnosis of ABPA. However, a recent cohort study has shown that a considerable percentage of ABPA patients fail to show eosinophilia to this extent [Agarwal et al. 2006].
A. fumigates-specific serum IgE and IgG antibodies
An elevated level of A. fumigatus-specific antibodies measured by fluorescent enzyme immuno-assay is another important diagnostic test for ABPA. Patient’s serum IgG and IgE antibodies to A. fumigatus are compared to the pooled serum samples from patients with asthma and a positive Aspergillus skin test. An index higher than two is considered a positive test [Wang et al. 1978].
Precipitating antibodies against A. fumigatus
Using the double gel immunodiffusion technique, precipitating antibodies can be detected in unconcentrated sera of 70–100% of ABPA patients.
Specific Aspergillus antigens
Few studies have evaluated the role of recombinant Aspergillus allergens in diagnosing ABPA based on their specific binding to IgE in sera of ABPA patients. In reports of testing Asp f1, Asp f2, Asp f3, Asp f4, and Asp f6, it has been shown that Asp f 2, Asp f 4, and Asp f 6 have the highest specificity for measurement of specific IgE and diagnosis of ABPA [Crameri, 1998; Crameri et al. 1998; Kurup et al. 2000]. A recent study showed that measuring the rAspf4 IgE with both anti-Aspergillus enzyme-linked immunosorbent assay (ELISA) and precipitin has sensitivity of 100% for detection of ABPA in CF patients [Fricker-Hidalgo et al. 2010].
TARC
Chemokine (C-C motif) ligand 17 (CCL17) is a small cytokine belonging to the CC chemokine family that is also known as TARC. It was found that CF patients with ABPA have significantly higher serum levels of TARC compared with the rest of CF patients [Hartl et al. 2006]. Another study by Latzin and colleagues longitudinally followed 48 CF patients, of whom 12 had a diagnosis of ABPA according to Nelson’s criteria. They performed repeated measurements of serum total immunoglobulin IgE, specific A. fumigatus IgE and IgG, and specific IgE against recombinant A. fumigatus for 1–8 years. They reported that TARC was a more accurate marker for the diagnosis of ABPA with an accuracy of (93%) for TARC compared with total IgE (74%), or specific IgE against recombinant A. fumigatus-4 (75%) and specific IgE against recombinant A. fumigatus-6 (79%) [Latzin et al. 2008].
Histopathology
Histopathologic findings in patients with allergic bronchopulmonary aspergillosis can be varied. The common findings include mucin filled bronchioles, bronchial lumen containing allergic mucin, necrotic eosinophils, Charcot–Leyden crystals, and at times septate fungal hyphae. The alveolar spaces might be filled by eosinophils admixed with variable number of macrophages. The other finding is bronchocentric granulomatosis, which is manifested by partial replacement of bronchial epithelium by palisading histiocytes [Kradin and Mark, 2008; Chan-Yeung et al. 1971; Slavin et al. 1988].
Pathogenesis
The pathogenesis of ABPA starts with inhalation of A. fumigatus and deep colonization of lungs. The germination and production of hyphae will result in high concentrations of Aspergillus allergens on the respiratory epithelium [Slavin et al. 1988]. Aspergillus produces multiple proteases that can disrupt the epithelial integrity and induce an inflammatory response that will result in enhancing the allergen exposure to the bronchoalveolar lymphoid tissue [Knutsen, 2011].
A. fumigatus detection in sputum was found to be associated not only with A. fumigatus-IgE sensitization but also with neutrophilic airway inflammation and reduced lung function in asthmatics [Fairs et al. 2010].
Aspergillus allergens are processed by antigen-presenting cells (APCs) and presented to CD4+ T cells. The T-cell response is skewed toward Th2 and therefore production of IL- 4, IL-5, and IL-13 [Kumar, 2003]. In a murine model, sensitized mice when challenged by A. fumigatus showed a strong allergic response with accumulation of pulmonary Th2 cells [Chu et al. 1996].
It is proposed that ABPA develops in susceptible patients because of increased frequency and/ or activity of A. fumigatus-specific Th2 CD4+. Furthermore peripheral blood mononuclear cells captured from ABPA patients were found to have an increased sensitivity to IL-4 [Khan et al. 2000]. IL-4 produced by Th2 lymphocytes binds to the IL- 4 receptor (IL-4R) on B cells and results in B-cell proliferation and isotype switching to IgE [Bacharier and Geha, 2000]. This receptor binding can also increase the expression of HLA-DR class II, and CD23 on B-cells. CD23 is the low affinity IgE receptor (FcϵRII), which is an activation marker present on multiple immune cells including B cells, monocytes, eosinophils, and activated T cells. Of note CD23 interacts with CD21 and plays a major role in inducing IgE synthesis by B cells [Bonnefoy et al. 1995, 1996].
IL-4 can also increase the expression of VCAM-1 on endothelial cells and result in recruitment of other immune cells, such as eosinophils and basophils, and enhance the proliferation of fibroblasts, which have a major role in airway remodeling [Moser et al. 1992; Schleimer et al. 1992].
It has also been reported that ABPA patients have an increased expression of CD86 on monocyte-derived dendritic cells [Knutsen, 2011]. CD86 is found on dendritic cells that have the histamine receptor 2, which skews antigen-specific T cells to a Th2 response. It was proposed that the APCs such as monocytes and dendritic cells bearing HLA-DR2 and/or HLA-DR5 and increased sensitivity to IL-4 stimulation may play an important role in skewing A. fumigatus-specific Th2 responses in ABPA [Slavin et al. 1987].
Genetics of ABPA
Studies have identified multiple genetic alterations in the patients with ABPA and proposed the idea that ABPA patients are genetically at risk to develop hypersensitivity to A. fumigatus due to the exaggerated Th2 responses to its antigens. These genetic risks include IL-4 receptor alpha chain (IL-4Ra) polymorphisms, HLA-DR and HLA-DQ polymorphisms, IL-10 promoter polymorphisms, surfactant protein A2 (SP-A2) polymorphisms, cystic fibrosis transmembrane conductance regulator gene (CFTR) mutations, and TLR-9 polymorphisms. The genetic alterations in these genes attributed to ABPA are briefly discussed here.
HLA-DR
The expression of HLA-DR2 and/or DR5 particularly DRB1-1501 and DRB1-1503 genotypes in asthmatic and CF patients is associated with increased risk for ABPA after exposure to A. fumigatus. On the other hand, the presence of HLA-DQ2, especially DQB-0201, provided protection from the development of ABPA [Aron et al. 1999; Chauhan et al. 1996, 1997].
IL-4Ra
There is a high prevalence of gain of function IL-4 receptor polymorphisms in ABPA patients. The single nucleotide polymorphisms (SNPs) of IL-4Ra (ile75val) were seen with significantly higher prevalence in ABPA cases. It was shown that copresence of ile75val with another SNP, gln576arg, resulted in elevated IL-4-dependent CD23 expression in ABPA [Knutsen et al. 2004, 2006; Hershey et al. 1997].
IL-10 promoter polymorphisms
The -1082GG genotype is associated with higher risk for Aspergillus colonization and ABPA in CF patients. This polymorphism is associated with increased IL-10 synthesis [Brouard et al. 2005].
Surfactant protein A gene polymorphisms
Multiple SNPs in SP-A2 collagen region is associated with ABPA; for instance a significantly higher percentage of AGA allele (A1660G) was found in patients with ABPA. Some of these polymorphisms were associated with increased levels of total IgE and eosinophilia in ABPA patients. It is also proposed that changes in conformation or affinity of SP-A2 based on these polymorphisms in the collagen region may affect the function of alveolar macrophages and compromise host defense [Knutsen, 2011; Saxena et al. 2003; Vaid et al. 2007].
CFTR gene mutation
An increased frequency of CFTR mutations is seen in patients with ABPA compared with other asthma patients [Lebecque et al. 2011; Marchand et al. 2001; Miller et al. 1996].
Toll-like receptor gene polymorphisms
ABPA patients are shown to have increased frequency of allele C for the TLR9 (T-1237C) polymorphism compared with control patients [Carvalho et al. 2008]. TLR-9 is involved in recognition of CpG motifs in bacterial and viral DNA. Of note on murine neutrophils, Aspergillus hyphae and conidia signal through TLR-9 [Bellocchio et al. 2004].
Differential diagnosis
There are multiple pulmonary conditions that have similar characteristic and can mimic ABPA These diseases include infectious pneumonia, severe asthma, nondiagnosed CF, pulmonary tuberculosis, sarcoidosis, eosinophilic pneumonia, Churg–Strauss syndrome, and bronchocentric granulomatosis.
It is very important to differentiate ABPA from these conditions and start treatment before development of bronchiectasis and potential subsequent pulmonary hypertension and respiratory failure.
Natural history
As described above ABPA is categorized to five different stages. However, the disease does not necessarily progress from stage one to five and it can have a different natural history in different individuals. Upon presentation and diagnosis, it is not always clear whether the patient will enter remission or have recurrent disease. Following adequate therapy most patients in stage I and III can go to complete remission (stage II), but a complete remission is not necessarily permanent as exacerbations can occur several years later. About 25–50% of patients may relapse, defined by doubling of the baseline IgE levels. However, early diagnosis and treatment is thought to be associated with a lower risk of advancement to stage IV and V [Agarwal et al. 2006; Agarwal, 2009].
Patients diagnosed in stage IV and V require oral glucocorticoids for longer period of time and patients in stage V with widespread bronchiectasis, pulmonary dysfunction and/or corpulmonale may never improve clinically and immunologically despite long-term glucocorticoid therapy [Lee et al. 1987].
Treatment
The goals of therapy in ABPA are to treat acute asthmatic exacerbations, control the inflammation, and limit progressive end-stage fibrotic disease. Patients should also be advised to avoid and clear the sources of fungi such as moldy basements. It is of utmost importance to optimally control the asthma or CF, and the other comorbid conditions patients might have like sinusitis or rhinitis.
Steroids
Oral corticosteroid is the mainstay treatment of ABPA [Stevens et al. 2000a]. Corticosteroids target the inflammatory response triggered by A. fumigatus, which, as detailed above is the cornerstone in pathophysiology of ABPA.
The most accepted treatment dose for oral prednisone is 0.5 mg/kg/day [Greenberger, 2002]. The duration of steroid therapy should be individualized in each patient based on the clinical course. The most commonly used strategy is 2 weeks of daily therapy with 0.5 mg/kg/day followed by alternate days for 6–8 weeks, then tapered by 5–10 mg every 2 weeks [Greenberger, 2002].
Most patients require prolonged low-dose corticosteroid therapy to control their symptoms and decrease the rate of relapse [Capewell et al. 1989; Stevens et al. 2000a]. Close follow up is needed during the first year, patients are considered to be in complete remission if no exacerbations happen in the 3 months after discontinuing steroid therapy. Patients who have evolved to stage IV cannot be taken off prednisone as it will cause exacerbations, and should be managed with continuous daily or alternate-day corticosteroid therapy if possible [Agarwal, 2009; Greenberger, 2002].
Total serum IgE is a useful marker to guide treatment as it is correlated with disease activity, the level should be checked every 6–8 weeks after the initiation of corticosteroid treatment, then every 8 weeks for 1 year [Ricketti et al. 1984]. During this prolonged follow up, one can determine the lowest level as a baseline for each patient. In many patients, the IgE levels do not decrease to normal values.
A repeated chest roentgenogram or CT of the lung after 4–8 weeks of treatment is needed to demonstrate that infiltrates have cleared. Also pulmonary function tests should be monitored to check the lung function during treatment [Greenberger, 2003].
Inhaled corticosteroids
Inhaled corticosteroids (ICSs) are usually needed to control the symptoms of asthma, but studies have failed to demonstrate a role for inhaled corticosteroids in preventing the progression of lung damage in patients with ABPA [Research Committee of the British Thoracic Association, 1979; Heinig et al. 1988]. A recent study followed a group of ABPA-S patients treated with formoterol/budesonide (24–1600 µg/day) for a period of 6 months. Results showed symptomatic deterioration and rise of 99.3% in the median IgE level in this group. Nineteen out of 21 of those same patients were treated with oral corticosteroids afterwards and had resolution of symptoms and significant decline in serum IgE [Agarwal, 2011]. These results confirmed that inhaled steroids alone should not be used for treatment of ABPA.
Intravenous methylprednisolone
A few small studies have used intravenous methylprednisolone in patients with CF and ABPA [Thomson et al. 2006]. Patients who received high-dose intravenous methylprednisolone (10–15 mg/kg/day) plus itraconazole showed clinical and laboratory improvement with increase in forced expiratory volume in 1 second FEV1 and decrease in serum IgE levels and total eosinophil counts [Cohen-Cymberknoh et al. 2009]. Larger randomized controlled studies are needed to evaluate the role of this treatment in ABPA.
Antifungals
Antifungals have an adjunctive role in treatment of ABPA by decreasing the burden of fungal organisms and antigenic stimulation (Table 4).
Antifungals and omalizumab in treament of ABPA
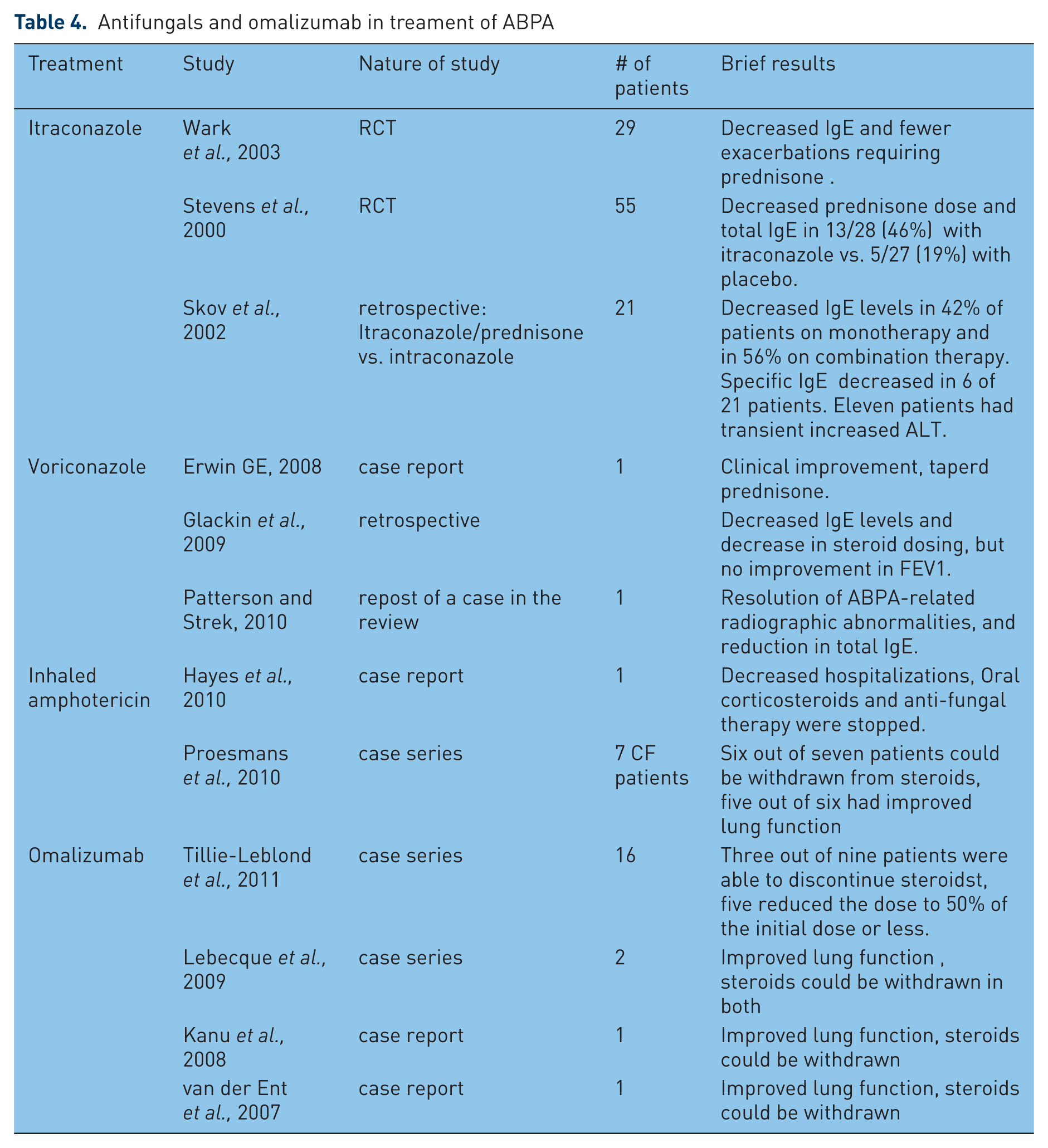
A number of antifungals have been tried to treat the ABPA. The most highly accepted agent is itraconazole that has been proved to have significant therapeutic benefit in randomized, controlled trials [Stevens et al. 2000a; Wark et al. 2003]. The addition of itraconazole for 16 weeks resulted in a significant increase in the likelihood of a clinical response (46% versus 19%). In this trial, response was defined as at least 50% reduction in the steroid dose, at least 25% reduction in the serum IgE, and one of the following: at least 25% improvement in exercise tolerance or pulmonary function tests, or resolution of pulmonary infiltrates. The rate of adverse events was similar in the two groups [Stevens et al. 2000b]. Another randomized controlled trial showed that subjects receiving itraconazole had a decrease in sputum eosinophils of 35% per week, with no decrease seen in the placebo arm. Itraconazole also significantly reduced the serum IgE. There were fewer exacerbations requiring oral corticosteroids in those treated with itraconazole compared with the placebo group [Wark et al. 2003]. The recommended dose for itraconazole is 200 mg twice daily for 4–6 months, and then tapered over 4–6 months. Further pooled analysis of studies confirmed that itraconazole could decrease the IgE levels but could not significantly improve the lung function [Agarwal, 2009]. Of note, the levels of drug might be altered in some patients due to potential interaction with multiple other medications. Owing to the higher cost, side effects, and potential toxicity of this drug, the use of itraconazole is recommended only after trial of steroids is proven to be insufficient [Elphick and Southern, 2000; Greenberger, 2002; Camuset et al. 2007; Glackin et al. 2009; Patterson and Strek, 2010].
A few case reports have shown acceptable benefit from using voriconazole, which is a newer antifungal azole with higher bioavailability [Glackin et al. 2009; Patterson and Strek, 2010]. A recent review has reported that 70–80% of patients who tolerated either voriconazole or posaconazole have responded to this treatment [Hogan and Denning, 2011]. No prospective controlled trials have been conducted to evaluate this drug in ABPA.
Case studies have reported successful use of aerosolized amphotericin B for the treatment of ABPA in CF [Hayes et al. 2010]. Another small study showed improve in lung function and decreased steroid need in six out of seven CF patients with recurrent or difficult to treat ABPA treated with nebulized amphotericin B [Proesmans et al. 2010]. There are no prospective controlled trials of amphotericin for the treatment of ABPA.
Anti-IgE monoclonal antibody
Omalizumab has been shown beneficial in few reports. A recent small trial of omalizumab in 16 patients with ABPA showed a reduction in number of exacerbations and systemic steroid need, but no improvement of the respiratory function [Tillie-Leblond et al. 2011]. A randomized, controlled trial is needed to better investigate the role of omalizumab in treating ABPA.
Footnotes
This work was supported by the Ernest S. Bazley Grant to Northwestern Memorial Hospital and Northwestern University
LCG is the Co-Director of the Northwestern University Allergy-Immunology Laboratory which analyzes sera for allergic bronchopulmonary aspergillosis.
