Abstract
Non-cystic fibrosis bronchiectasis (NCFBE), a historically under-recognized chronic respiratory condition, is a significant diagnosis currently experiencing a resurgence of interest in its clinical management. Ciprofloxacin is part of the current armamentarium used in the treatment of the recurrent respiratory tract infections seen in NCFBE. Inhaled ciprofloxacin, a novel method of drug delivery for the fluoroquinolone class, is being actively investigated. The inhaled formulation is designed to enhance drug delivery to the site of infection in the lung while minimizing the risk of systemic toxicity. This review summarizes the pharmacology and pharmacokinetics of ciprofloxacin and the rationale for the development of an inhaled formulation for NCFBE. Preclinical and clinical data regarding current development of inhaled ciprofloxacin formulations is also evaluated. Lastly, the anticipated role of inhaled ciprofloxacin in the management of NCFBE is discussed, including future considerations and potential limitations of therapy.
Introduction
Bronchiectasis (BE) is a condition characterized by the abnormal and permanent dilatation of the airways resulting in clinical symptoms of chronic productive cough, airway obstruction, and recurrent infections [Baydarian and Walter, 2008]. It is traditionally classified as either cystic fibrosis bronchiectasis (CFBE) or non-cystic fibrosis bronchiectasis (NCFBE). Historically, NCFBE has received less attention than its counterpart in cystic fibrosis (CF). There has been renewed interest in NCFBE in recent years, as indicated by the emergence of new guidelines designed specifically with the NCFBE population in mind [Chang et al. 2010; Pasteur et al. 2010].
The current management of BE is based largely on limited clinical data and/or expert opinion. Furthermore, the management of NCFBE in particular is often extrapolated from studies of the CFBE population. Guidelines acknowledge the urgent need for evaluation of treatment modalities specifically in the NCFBE population [Pasteur et al. 2010]. The development of new drug products is often spurred by such a significant, unmet clinical need. This particular review will focus on the development of inhaled ciprofloxacin formulations and discuss their anticipated role in the management of NCFBE.
The projected market for these emerging inhaled ciprofloxacin formulations is substantial. While NCFBE is considered an ‘orphan disease’ in most developed countries, the prevalence is likely higher in developing countries where limited healthcare access and malnutrition may contribute to higher rates of NCFBE [Metersky, 2010; King, 2009]. In addition, there is evidence of a growing burden of BE on healthcare systems, as shown in one retrospective study which demonstrated increasing annual rates of BE-associated hospitalizations in the US [Seitz et al. 2010]. The prevalence of NCFBE in the US is approximately 110,000 patients [Weycker et al. 2005] which remains comparatively larger than the estimated US prevalence of CF with 30,000 patients [Cystic Fibrosis Foundation, 2011]. Rates of NCFBE may also be underestimated due to lack of recognition by physicians or misdiagnosis, often as another chronic respiratory condition such as ‘difficult asthma’ or chronic obstructive pulmonary disease (COPD) [Chang et al. 2012]. However, the increasing use of high-resolution computed tomography scans continues to improve diagnosis and awareness of NCFBE. Therefore, the need for safe and effective therapeutic options for the management of NCFBE is likely to only increase with time. Given this current climate, the development of inhaled ciprofloxacin formulations for NCFBE is viewed with significant interest.
NCFBE: clinical features, pathogenesis, and management
The hallmark symptom of NCFBE is chronic productive cough which is often accompanied by fatigue and lethargy. These symptoms may progress to include more serious features such as hemoptysis, chest pain, exertional dyspnea, recurrent infections or, in severe BE, pulmonary hypertension, and systolic and diastolic left ventricu-lar dysfunction [Baydarian and Walter, 2008; Rademacher and Welte, 2011]. The overall condition of BE, composed of both CFBE and NCFBE, is most commonly described as a process or syndrome as opposed to a singular disease entity. BE is a heterogeneous condition associated with a myriad of etiologic factors. The most common risk factors are infections, e.g. severe pneumonia, mycobacterial infection, pertussis, or measles. Other risk factors include impaired mucociliary clearance (CF, Young’s syndrome, Kartagener’s syndrome); immune dysfunction (HIV infection, transplant rejection, hypogammaglobulinemia, allergic bronchopulmonary aspergillosis); exaggerated immune response (rheumatoid arthritis, inflammatory bowel disease); inflammatory pneumonitis (smoke inhalation, aspiration); COPD; mechanical obstruction (foreign body, obstructing cancer); extremes of age; and malnutrition/socioeconomic disadvantage [Feldman, 2011; King, 2009].
The pathogenesis of BE is described as an initial insult to the lung tissue (often infection) which leads to impaired mucociliary clearance resulting in chronic microbial colonization and airway inflammation/suppuration. Prolonged inflammation results in the release of inflammatory mediators, such as protease/elastase and free radicals [Fuschillo et al. 2008; King, 2009], which cause progressive damage to the bronchial epithelium leading to further impairment in mucociliary clearance, microbial colonization, and inflammation. This process is often referred to as the ‘vicious circle’ of BE [Cole, 1986]. Within this model, the two requisite factors for development of BE appear to be persistent infection and defect in host defenses [King, 2009]. Thus, there is sound theoretical support for attempting to decrease the bacterial load in the respiratory tract. As such, antibiotic therapy has become an important component in both the acute and chronic management of NCFBE [Chang et al. 2010; Pasteur et al. 2010].
The current goals in the management of NCFBE are to identify and treat the underlying cause, provide symptom control, maintain or improve lung function, reduce exacerbations, and improve quality of life [Chang et al. 2010; Pasteur et al. 2010]. Acute pulmonary exacerbations are often treated with short courses of oral antibiotics in the ambulatory setting, yet intravenous antibiotics may be used for more severe illness or resistant pathogens. The choice of antibiotic agent should be based on culture results from pulmonary secretions, e.g. sputum, bronchoscopy washings, as well as clinical severity, patient tolerance, and local susceptibility patterns [Chang et al. 2010; Pasteur et al. 2010].
Chronic bacterial colonization is a clinical feature common to both NCFBE and CFBE. Both the presence and type of pathogen appear to be associated with disease severity. Patients with no pathogens identified from sputum culture generally have the mildest disease [King et al. 2007]. In comparison, chronic colonization with Pseudomonas aeruginosa is of particular concern as it is associated with more frequent pulmonary exacerbations [King et al. 2007], more accelerated decline in lung function [Martinez-Garcia et al. 2007], and impaired quality of life [Wilson et al. 1997]. There is a suggestion that treatment of chronic airway colonization may decrease the progressive inflammatory damage caused by this pathogen. In a small study of 13 severe COPD patients with P. aeruginosa airway colonization, treatment with inhaled tobramycin for 2 weeks was associated with a significant reduction in multiple inflammatory markers, i.e. interleukin-1 beta, interleukin-8, eosinophilic cationic protein, eosinophil count, and a 42% reduction in the 6-month incidence of acute exacerbations [Dal Negro et al. 2008].
The routine treatment of chronic colonization with long-term antibiotics is not currently recommended, but may be considered for patients with frequent exacerbations (≥3–6 per year) or exacerbations of significant severity or prolonged duration [Chang et al. 2010; Pasteur et al. 2010]. As with acute exacerbations, the choice of antibiotic agent should be driven by culture results of pulmonary secretions. The most frequent bacterial organisms isolated from sputum cultures of NCFBE patients are Haemophilus influenzae (30–47%), P. aeruginosa (12–31%), Streptococcus pneumoniae (7–20%), Staphylococcus aureus (3–23%), and Moraxella catarrhalis (2–15%) [Goeminne et al. 2012; King et al. 2007; Li et al. 2005; Nicotra et al. 1995; Pasteur et al. 2000].
The rationale for inhaled antibiotics
The use of inhaled antimicrobials for the management of NCFBE is currently recommended as a potential option for patients with severe infections or frequent exacerbations secondary to chronic bacterial colonization [Chang et al. 2010; Pasteur et al. 2010]. The rationale for inhaled antibiotic therapy is based on enhanced drug delivery at the site of infection which should improve antibacterial efficacy, coupled with potentially lower systemic exposure which should minimize the risk of toxicity [Flume and Klepser, 2002]. Local antibiotic delivery to the lung may be particularly advantageous in patients with damaged or impaired pulmonary-defense systems, as in NCFBE [Danziger, 1986]. Optimal bacterial killing is driven by adequate antibiotic exposure, which is best described by the attainment of target pharmacokinetic–pharmacodynamic (PK-PD) parameters: peak:MIC (minimal inhibitory concentration), AUC (area under the concentration-time curve):MIC, or T (duration of time)>MIC [Craig, 1998; Ambrose et al. 2007]. Interestingly, many antibiotics used to treat respiratory infection, e.g. aminoglycosides, β-lactams, glycopeptides, have poor distribution into the epithelial lining fluid (ELF) of the lung [Rodvold et al. 2011] and potentially poor PK-PD target attainment rates at the site of infection (the lungs). While the specific intrapulmonary PK-PD target for colonization or infection associated with NCFBE is currently unknown, there is sound reasoning to support strategies aimed at significantly increasing intrapulmonary antibiotic delivery for the management of NCFBE.
Current FDA-approved indications for inhaled antibiotics for CF in the United States include tobramycin inhalation solution (TOBI®, Novartis Pharmaceuticals, East Hanover, NJ, USA) and aztreonam inhalation solution (Cayston®, Gilead Sciences Inc., Foster City, CA, USA) [Novartis Pharmaceuticals, 2009; Gilead Sciences, 2012]. Additional agents available in the European Union include tobramycin inhalation powder (TOBI® Podhaler®, Novartis Pharmaceuticals, East Hanover, NJ, USA) and inhaled colistimethate sodium products, i.e. Promixin® (Profile Pharma Ltd, Tangmere, West Sussex, UK), Colomycin® and Colobreathe® (Forest Laboratories, New York, NY, USA) [Forest Laboratories, 2004; European Medicines Agency, 2011, 2012; Medicines and Healthcare Products Regulatory Agency, 2011]. These agents are only indicated for the management of CF patients with P. aeruginosa. There are currently no inhaled antibiotics specifically approved for use in the NCFBE population.
Inhaled ciprofloxacin formulations are currently in development alongside multiple other inhaled antibiotics, including a levofloxacin solution, a liposomal amikacin solution, another tobramycin solution, and a combination formulation of fosfomycin and tobramycin. Of these agents, inhaled liposomal amikacin is the only formulation, aside from inhaled ciprofloxacin, with promising preliminary data specifically in NCFBE patients [O’Donnell et al. 2009]. Levofloxacin is the only other fluoroquinolone in development as an inhalation product. Phase III clinical trials evaluating inhaled levofloxacin in CF patients are currently ongoing [ClinicalTrials.gov identifiers: NCT01180634, NCT01270347].
Ciprofloxacin: pharmacology and pharmacokinetics
Ciprofloxacin is a second-generation fluoroquinolone currently available in both oral and intravenous (IV) formulations for the treatment of a wide variety of bacterial infections, including lower respiratory tract infections caused by H. influenzae, P. aeruginosa, penicillin-susceptible S. pneumoniae, and M. catarrhalis [Bayer Healthcare Pharmaceuticals, 2009, 2011]. The microbiologic spectrum of ciprofloxacin is characterized by excellent activity against most Gram-negative pathogens, with lesser activity against staphylococci, borderline activity against streptococci, and poor activity against anaerobes. Regarding P. aeruginosa specifically, ciprofloxacin exhibits good activity with an MIC90 historically around 0.5–1.0 µg/ml [McCormack and Grayson, 2010].
Oral formulations of ciprofloxacin are rapidly and well absorbed from the gastrointestinal tract (bioavailability of 70%). Both oral and IV formulations achieve adequate systemic exposure with a steady-state maximum serum concentration (Cmax) of 3–5 µg/ml and a serum AUC of 13–33 µg*h/ml over 8 or 12 hours at commonly used doses [Bayer Healthcare Pharmaceuticals, 2009, 2011]. The half-life (t1/2) of ciprofloxacin is about 4 hours in patients with normal renal function. It is mainly renally eliminated (50–75% drug excreted unchanged in urine); yet, 33–50% of serum clearance is via nonrenal mechanisms [McCormack and Grayson, 2010].
Ciprofloxacin is distributed extensively into bodily tissues, e.g. lung, skin, fat, muscle, cartilage, bone, prostate, and peritoneal fluid. Pharma-cokinetic parameter estimates in sputum are variable in the literature, but overall appear similar to those of serum [Smith et al. 1986; Reed et al. 1988]. Serum ciprofloxacin concentrations achieve a penetration of 46–79% into sputum [Davies et al. 1986; Hoogkamp-Korstanje and Klein, 1986; Smith et al. 1986]. Ciprofloxacin also exhibits measurable penetration into the ELF of the lung and excellent penetration into alveolar macrophages (AMs). Following oral administration of ciprofloxacin 500 mg every 12 h for nine doses, ELF concentrations [mean ± standard deviation (SD)] were 1.9 ± 0.9 and 0.4 ± 0.1 μg/ml, at 4 and 12 h, respectively, and AM concentrations were 34.9 ± 23.2 and 6.8 ± 5.9 μg/ml, respectively [Gotfried et al. 2001].
The favorable spectrum of activity and pharmacokinetics of ciprofloxacin make it an attractive option for the treatment of severe or frequent exacerbations in NCFBE, particularly those due to P. aeruginosa. However, pulmonary concentrations after oral and IV administration of ciprofloxacin are near the MIC90 for many of the respiratory pathogens identified as colonizing NCFBE patients. This leads to a concern for adequate drug exposure of ciprofloxacin at the site of infection. Inhaled ciprofloxacin is being developed as a potential formulation which can address this problem and improve intrapulmonary drug delivery. The route of inhalation also has the potential benefit of minimal systemic exposure which may decrease the risk of toxicity.
Some of the earliest published data regarding the use of inhaled ciprofloxacin was in a rat model performed by Chono and colleagues [Chono et al. 2007]. An intrapulmonary ciprofloxacin solution was delivered at a dose of 200 µg/kg (1/50th of the oral dose) and achieved AUCs (mean ± SD, µg*h/ml) of 103 ± 26, 244 ± 99, and 0.81 ± 0.22 in ELF, AMs, and plasma, respectively. In contrast, oral ciprofloxacin administration at 10 mg/kg resulted in significantly lower intrapulmonary exposure (AUCs of 0.74 ± 0.2 and 14.1 ± 6.5 in ELF and AMs, respectively). An AUC:MIC of >125 is a common exposure target associated with favorable outcomes of ciprofloxacin therapy against Gram-negative aerobes, such as P. aeruginosa, and the Gram-positive organism, S. aureus [Forrest et al. 1993]. These early data from Chono and colleagues suggest inhaled ciprofloxacin is much more likely to achieve this PK-PD target at the site of infection in the lung (AUC:MICs in ELF of 206 or 103 for MICs of 0.5 or 1.0 µg/ml, respectively) compared with oral ciprofloxacin administration. These MIC values are particularly important as 0.5 and 1.0 µg/ml are the respective European Committee on Antimicrobial Susceptibility Testing (EUCAST) and Clinical and Laboratory Standards Institute (CLSI) susceptibility breakpoints for P. aeruginosa [Clinical and Laboratory Standards Institute, 2012; European Committee on Antimicrobial Susceptibility Testing, 2012].
Inhaled ciprofloxacin formulations: preclinical data
There are currently two inhaled ciprofloxacin formulations in development for treatment of chronic bacterial infections in NCFBE: ciprofloxacin dry powder for inhalation (DPI) and dual-release ciprofloxacin for inhalation (DRCFI). There is additional data for a third formulation, ciprofloxacin for inhalation (CFI); however, clinical development of this product was recently halted by the developer [Aradigm Corporation, 2012]. Data concerning CFI is included in this review given its similarity to DRCFI. A summary of all inhaled ciprofloxacin formulations is provided in Table 1.
Inhaled ciprofloxacin formulations.

Ciprofloxacin dry powder for inhalation
Delivery of inhaled antibiotics in the form of a dry powder is often more convenient for patients compared with the traditional delivery of inhaled antibiotics via ultrasonic or jet nebulizers. Ciprofloxacin DPI (formerly BAY q3939) is being developed by Bayer Healthcare Pharmaceuticals, Inc. (Leverkusen, Germany) for the treatment of chronic P. aeruginosa infections in a variety of respiratory disorders, i.e. CF, COPD, and NCFBE. Ciprofloxacin DPI is a dry powder formulation which utilizes PulmoSphere™ technology, an emulsion-based spray-drying process, to produce highly dispersible, low-density particles which are delivered using the small, handheld T-326 inhaler (Novartis Pharmaceuticals, East Hanover, NJ, USA) [Stass et al. 2008a]. The product is dosed in milligrams of ciprofloxacin DPI, where 50 mg of ciprofloxacin DPI is equivalent to 32.5 mg of ciprofloxacin-betaine, the free base form of the drug. This formulation is currently entering phase III clinical development with a clinical program entitled RESPIRE [Bayer Healthcare Pharmaceuticals, 2012].
Preclinical studies involving the free base component of ciprofloxacin DPI, ciprofloxacin-betaine, provide preliminary support of antibacterial efficacy with this formulation (see Table 2). An in vitro PK-PD hollow fiber model simulated unbound sputum concentrations following administration of ciprofloxacin-betaine 32.5 mg once or twice daily [Knezevic et al. 2008]. The simulated exposure (Cmax 120 µg/ml) was highly and rapidly bactericidal against a wild-type strain of P. aeruginosa (MIC 0.125 µg/ml) with at least a 6 log10 colony forming units (CFU) decrease in bacterial density from baseline within 1 h with both regimens. In another model of rats with P. aeruginosa pneumonia, ciprofloxacin-betaine was administered intratracheally at 2.5 mg/kg once daily and showed a significant reduction in bacterial density in the lung compared to an untreated, infected control group [Endermann and Pickbrenner, 2008]. The study included a second cycle of infection/treatment where ciprofloxacin-betaine exhibited slightly greater mean reduction in bacterial density compared with the first cycle (mean reduction of 1.9 log10 CFU versus 1.7 log10 CFU, respectively).
Preclinical data for inhaled ciprofloxacin formulations.

Published in abstract form only.
AM, alveolar macrophage; AUC, area under the concentration-time curve; BID, twice daily; CFU, colony forming units; CIP, ciprofloxacin; CIP-B, ciprofloxacin-betaine; ELF, epithelial lining fluid; MIC, minimal inhibitory concentration; PK/PD, pharmacokinetic–pharmacodynamic.
Liposomal ciprofloxacin formulations: ciprofloxacin for inhalation and dual-release ciprofloxacin for inhalation
Packaging of the drug in liposomes has many potential advantages for drug delivery. It often confers controlled and prolonged release of drug at the site of action, may protect against drug degradation, and may augment cellular uptake (including uptake by AMs in the lung). Aradigm Corporation (Hayward, CA, USA) has developed ciprofloxacin for inhalation (CFI or Lipoquin™, formerly ARD-3100), a liposomal formulation, and dual-release ciprofloxacin for inhalation (DRCFI or Pulmaquin™, formerly ARD-3150), a mixed formulation comprising both liposomal and unencapsulated drug. Only the latter formulation, DRCFI, is currently being pursued for further clinical development in phase III clinical trials as chronic maintenance therapy for NCFBE [Aradigm Corporation, 2012]. These liposomal formulations are delivered via jet nebulization using a PARI LC Sprint nebulizer and PARI TurboBoy-S compressor [Bilton et al. 2010].
The differing pharmacokinetics of inhaled liposomal versus free ciprofloxacin were illustrated in a study by Chono and colleagues using a rat pneumonia model (see Table 2) [Chono et al. 2008]. Liposomal and free drug were compared at the same dose (200 µg/kg) as two separate formulations in both uninfected rats and those with lipopolysaccharide-induced pneumonia. Liposomal ciprofloxacin showed significantly higher exposure in both ELF and AMs (AUCs of 352 and 4289 μg*h/ml) compared with free ciprofloxacin (AUC 194 and 341 μg*h/ml). Systemic exposure was lower with liposomal ciprofloxacin versus free ciprofloxacin (AUCplasma of 0.6 versus 1.3, respectively). The PK-PD analysis also predicted good antibacterial activity with relatively high AUC:MIC ratios in ELF for both liposomal and free ciprofloxacin (352 and 194, respectively, assuming a MIC of 1.0 μg/ml in both cases).
At this time, the authors are unaware of any published preclinical data for the DRCFI formulation. Further detail regarding the pharmacokinetics of this formulation can be found in the following clinical data section.
Inhaled ciprofloxacin formulations: clinical data
Of note, most of the clinical data presented herein is currently available in abstract form only. Data which are not yet available in fully published form should be interpreted in a preliminary context.
Ciprofloxacin dry powder for inhalation
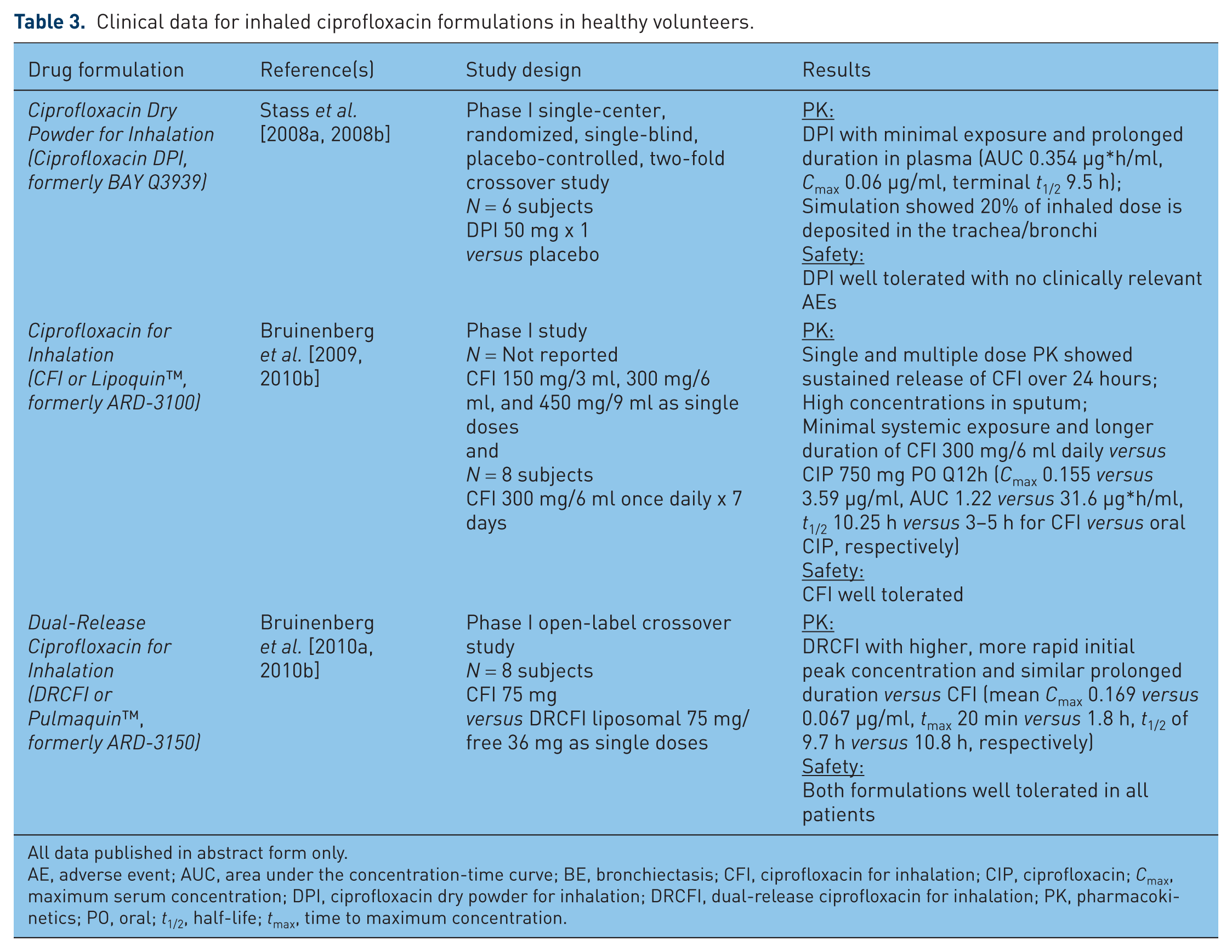
Early clinical development of ciprofloxacin DPI includes phase I studies in healthy volunteers, CF, and COPD patients (data for healthy volunteers summarized in Table 3). These studies provide the first human data describing intrapulmonary pharmacokinetics with ciprofloxacin DPI. The data are fairly consistent across the different patient populations and support the proposed advantage to inhaled delivery, showing high intrapulmonary concentrations of ciprofloxacin with minimal systemic exposure.
Clinical data for inhaled ciprofloxacin formulations in healthy volunteers.
All data published in abstract form only.
AE, adverse event; AUC, area under the concentration-time curve; BE, bronchiectasis; CFI, ciprofloxacin for inhalation; CIP, ciprofloxacin; Cmax, maximum serum concentration; DPI, ciprofloxacin dry powder for inhalation; DRCFI, dual-release ciprofloxacin for inhalation; PK, pharmacokinetics; PO, oral; t1/2, half-life; tmax, time to maximum concentration.
A phase I study of ciprofloxacin DPI 50 mg given as a single dose in healthy volunteers showed minimal systemic exposure with prolonged duration in the plasma with the following mean pharmacokinetics parameters: AUC 0.354 µg*h/ml, Cmax 0.06 µg/ml, terminal t1/2 9.5 h, volume of distribution (Vd) 1262 l, clearance (CL) 91.7 l/h [Stass et al. 2008a, 2008b]. The formulation was well tolerated with no clinically relevant adverse events. Phase I studies in CF and COPD patients showed high intrapulmonary concentrations and confirmed minimal systemic exposure. In CF patients, ciprofloxacin DPI 50 and 100 mg once daily displayed a mean [coefficient of variation (CV%)] Cmax in the lung of 344 (179.4%) and 540 (91.4%) µg/ml, respectively, on day 1 [Stass et al. 2008c]. No accumulation in plasma was noted at steady state on day 7. DPI administration was well tolerated with no reported drug- or inhalation-related adverse events. In a separate phase I study in adolescent CF patients, mean (CV%) Cmax in the lung was relatively lower at 91.1 µg/ml (133.9%) and mean serum t1/2 was relatively shorter at 4.5 h (20.4%) following a single dose of ciprofloxacin DPI 50 mg [Stass et al. 2009]. A phase I study in 16 Japanese patients with mild to moderate COPD demonstrated detectable sputum concentrations of ciprofloxacin 12 h following a single dose of ciprofloxacin DPI 50 mg with a terminal t1/2 of 7.2 h and a penetration ratio (AUCsputum:AUCplasma) of 63 [Tokimatsu et al. 2011]. A second phase I study in 25 patients with mild to moderate COPD evaluated ciprofloxacin DPI 50 mg BID, 50 mg TID, and 75 mg BID versus placebo following both 1- and 9-day dosing [Stass et al. 2011]. Mean Cmax at steady state was at least 300-fold plasma concentrations in all DPI arms. While intrapulmonary concentrations of ciprofloxacin were high, significant inter- and intra-subject variability in sputum concentrations was noted. Phase I studies in moderate to severe COPD patients have shown similar trends of high intrapulmonary exposure and minimal systemic exposure [Kadota et al. 2012; Stass et al. 2012]. The studies in COPD patients had relatively higher rates of adverse events compared with other patient populations. Most adverse events were mild, e.g. bitter taste, nasopharyngitis, and dyspnea; yet a transient decrease in lung function, indicated by a decrease in mean forced expiratory volume in 1 second (FEV1), was noted in some studies [Kadota et al. 2012; Stass et al. 2011]. No severe or serious adverse events were reported.
Phase II studies have evaluated the safety and efficacy of ciprofloxacin DPI in both NCFBE and CF patients, with varying results, although the primary outcome differed between the two patient populations (data for NCFBE patients summarized in Table 4). A phase II study was conducted in 124 stable NCFBE patients whose sputum cultures were positive for predefined potential respiratory pathogens, i.e. P. aeruginosa (mucoid or non-mucoid), S. aureus, S. pneumoniae, H. influenzae, M. catarrhalis, Enterobacteriaceae, Stenotrophomonas maltophilia or Achromobacter xylosoxidans [Wilson et al. 2012]. Subjects were randomized to receive ciprofloxacin DPI 50 mg BID or matched placebo for 28 days (with 56 days of follow up). Chronic macrolide use was allowed as anti-inflammatory therapy if treatment had been in place for at least 30 days prior to randomization. The primary objective of reduction in sputum bacterial load at end of treatment (EOT) showed significantly greater reduction in the ciprofloxacin DPI group versus placebo (decrease of 3.62 versus 0.27 log10 CFU/g, respectively, p < 0.001) [Wilson et al. 2012]. Secondary outcomes also showed significantly greater eradication rates of baseline respiratory pathogens (35% versus 8% at EOT in the ciprofloxacin DPI versus placebo groups, respectively, p = 0.001) as well as trends in favor of ciprofloxacin DPI with reduction in C-reactive protein, fewer pulmonary exacerbations, and improvement in health status impact as measured by St. George’s Respiratory Questionnaire (SGRQ) scores. The formulation was well tolerated overall with few treatment-related adverse events. These data are promising as ciprofloxacin DPI enters phase III clinical trials for the chronic management of NCFBE.
Clinical data for inhaled ciprofloxacin formulations in non-cystic fibrosis bronchiectasis.

All data published in abstract form only, except for Wilson et al. [2012].
Data from Serisier [2012].
AE, adverse event; AUC, area under the concentration-time curve; BE, bronchiectasis; BID, twice daily; CFI, ciprofloxacin for inhalation; CFU, colony forming units; CI, confidence interval; CIP, ciprofloxacin; Cmax, maximum serum concentration; COPD, chronic obstructive pulmonary disease; CRP, C-reactive protein; DPI, ciprofloxacin dry powder for inhalation; DRCFI, dual-release ciprofloxacin for inhalation; EOT, end of treatment; FEV1, forced expiratory volume in 1 second; HR, hazard ratio; NCFBE, non-cystic fibrosis bronchiectasis; ORBIT, Once-Daily Respiratory Bronchiectasis Inhalation Treatment Trial; PK, pharmacokinetics; SGRQ, St. George’s Respiratory Questionnaire; t1/2, half-life; tmax, time to maximum concentration.
Another phase IIB study was performed in 286 stable CF patients colonized with P. aeruginosa [Dorkin et al. 2011]. Subjects were randomized to treatment with either ciprofloxacin DPI 50 mg BID, 75 mg BID, or matching placebo for 28 days (with 28 days of follow up). The primary objective of change in FEV1 from baseline at day 28 showed no significant difference between groups. Reanalysis by pooling the two treatment groups versus the two placebo groups demonstrated a statistically significant, yet small (2%) improvement in FEV1 with treatment versus placebo [Serisier, 2012]. Given these borderline favorable outcomes, it is unclear whether Bayer will continue clinical development of ciprofloxacin DPI in CF patients at this time.
Liposomal ciprofloxacin formulations: ciprofloxacin for inhalation and dual-release ciprofloxacin for inhalation
Early clinical development of the liposomal ciprofloxacin formulations, CFI and DRCFI, include phase I studies in healthy volunteers and NCFBE patients (see Tables 3 and 4). Similar to the phase I ciprofloxacin DPI data, these studies support the pharmacokinetic advantage of inhaled delivery, again reporting high sputum concentrations of ciprofloxacin with minimal systemic exposure. A phase I, dose escalation study in healthy volunteers evaluated single doses of CFI 150 mg/3 ml, 300 mg/6 ml, and 450 mg/9 ml as well as a regimen of 300 mg/6 ml nebulized once daily for 7 days [Bruinenberg et al. 2009]. This study demonstrated low systemic exposure of CFI 300 mg versus oral ciprofloxacin 750 mg Q12h with mean Cmax of 0.155 versus 3.59 μg/ml and AUC of 1.22 versus 31.6 μg*h/ml for CFI versus oral ciprofloxacin, respectively. Significantly longer duration was also noted with the CFI formulation (t1/2 10.25 h versus 3–5 h).
In an effort to optimize pharmacokinetics of ciprofloxacin, free, unencapsulated ciprofloxacin was coformulated with liposomal ciprofloxacin to develop the new product, DRCFI. This formulation was designed to increase Cmax and provide a shorter time to maximum concentration (tmax) while maintaining a similarly prolonged duration, or half-life (t1/2). A second phase I study compared the pharmacokinetics of CFI 75 mg versus DRCFI (liposomal 75 mg/free 36 mg) in eight healthy volunteers and evaluated DRCFI alone in six NCFBE patients [Bruinenberg et al. 2010a, 2010b]. The DRCFI formulation was found to have higher, more rapid initial peak concentration in healthy volunteers with a mean Cmax 0.169 versus 0.067 μg/ml and mean tmax 20 min versus 1.8 h with DRCFI versus CFI, respectively. The DRCFI formulation maintained a similar duration to the CFI formulation with mean t1/2 of 9.7 h in healthy volunteers and 9.4 h in NCFBE patients (versus 10.8 h for CFI in healthy volunteers). Both formulations were reportedly well tolerated in all patients.
The safety and efficacy of CFI has been evaluated in multiple phase II clinical trials in both NCFBE and CF patients (data for NCFBE patients summarized in Table 4). The first phase II study in NCFBE included 36 patients given either CFI 150 or 300 mg nebulized once daily for 28 days; however, this study was not placebo-controlled [Bilton et al. 2009a, 2009b, 2010]. The results demonstrated a significant reduction in sputum P. aeruginosa density from baseline with a mean decrease of 3.5 and 4 log10 CFU for the 150 and 300 mg groups, respectively. Minor improvements in lung function were seen (measured 14 days after the last dose). FEV1 improved 1.3% (p < 0.015) and 0.7% (p<0.496) from baseline in the 150 and 300 mg groups, respectively. CFI was reportedly well tolerated with no serious adverse events; yet, fewer adverse events and pulmonary exacerbations were noted in the 150 mg group versus the 300 mg group.
The second phase II study in NCFBE was entitled the Once-Daily Respiratory Bronchiectasis Inhalation Treatment (ORBIT-1) Trial. It was a randomized, double-blind, placebo-controlled trial in 96 NCFBE patients designed to evaluate lower doses of CFI 100 or 150 mg nebulized once daily for 28 days [Bilton et al. 2011]. The results again showed a significant decrease in sputum bacterial density in both treatment arms compared with placebo (mean decrease of 3.8 log10 CFU in the 100 mg group) [Serisier, 2012].
In the CF population, phase II data regarding the efficacy of CFI appears more favorable than data for the ciprofloxacin DPI formulation. A study in 22 CF patients showed a significant reduction in sputum P. aeruginosa density accompanied by a significant improvement in FEV1 of 6.9% from baseline (p = 0.04) [Bruinenberg et al. 2008]. This trial did not include a placebo control.
DRCFI has been evaluated in one 24-week phase II randomized, double-blind, placebo-controlled clinical trial (ORBIT-2) (see Table 4). It included 42 NCFBE patients with chronic P. aeruginosa infection randomized to either DRCFI (liposomal 150 mg/free 60 mg) or placebo nebulized once daily for three treatment cycles (28 days on, followed by 28 days off). The results indicated a significant reduction in mean sputum P. aeruginosa density at 28 days of 4.2 log10 CFU in the DRCFI group versus 0.1 log10 CFU in the placebo group compared to baseline (p = 0.004) [Serisier et al. 2011a, 2011b, 2012]. Fewer subjects in the DRCFI group required supplemental antibiotics for respiratory-related infections (eight subjects versus 17 subjects in the DRCFI versus placebo groups, respectively; p = 0.05). Therapy with DRCFI also demonstrated a significant delay in median time to first exacerbation (134 days versus 58 days; p = 0.046). There were fewer respiratory adverse events in the DRCFI group compared with placebo and all other adverse events were of similar severity and incidence.
Lastly, there is clinical evidence suggesting the relative antibacterial efficacy of the two liposomal formulations may differ. Serisier and colleagues presented data from placebo-controlled studies in NCFBE patients indicating greater and more sustained bacterial killing with DRCFI versus CFI [Serisier et al. 2012]. At 14 days, the mean reduction in sputum P. aeruginosa density in log10 CFU compared to baseline (versus placebo) was 3.90 (versus 1.69) for DRCFI and 3.27 (versus 0.57) for CFI. At 28 days, the mean reduction was 4.44 (versus an increase of 0.15) for DRCFI and 2.94 (versus 0.87) for CFI. This data, in concert with phase II clinical trial results, suggests DRCFI is a promising future option for chronic maintenance therapy of NCFBE. DRCFI is currently entering phase III clinical trials for this indication [Aradigm Corporation, 2012].
Future considerations
The overall tolerability of these inhaled ciprofloxacin formulations is a significant concern which must be adequately explored prior to the incorporation of these new treatments into clinical practice. Current safety data for inhaled ciprofloxacin formulations is limited, yet appears favorable. No drug-related serious adverse events have been reported in the clinical trials to date. Studies in NCFBE patients report the inhaled ciprofloxacin formulations were well tolerated. The most detailed safety data currently available in NCFBE patients is from the published phase II study of ciprofloxacin DPI [Wilson et al. 2012]. Exacerbations were the most frequently reported adverse event in this trial and other, less common treatment-emergent adverse events included dysgeusia and headache. A particular safety concern in the development of inhaled ciprofloxacin is the risk of respiratory adverse events, e.g. bronchospasm, dyspnea, chest pain, wheezing, as these have been noted to occur with use of other inhaled antimicrobials in NCFBE patients [Barker et al. 2000]. In the study by Wilson and colleagues, cough, hemoptysis, and bronchospasm were reported, but the incidence was low overall and similar between the ciprofloxacin DPI group versus the placebo group [Wilson et al. 2012].
Data regarding the safety of inhaled ciprofloxacin in other patient populations are limited as well. The adverse event of mild to moderate dysgeusia has also been described in CF patients [Stass et al. 2009] and a wider array of adverse events has been reported in COPD patients, including bitter taste, nasopharyngitis, dyspnea, and transient decreases in lung function (FEV1) [Kadota et al. 2012; Stass et al. 2011, 2012; Tokimatsu et al. 2011].
The potential for long-term administration of inhaled ciprofloxacin also raises additional toxicity concerns. The risk of systemic adverse events, such as central nervous system effects, QTc prolongation, tendinitis, or arthropathy, is likely low given minimal systemic absorption of these formulations [Owens and Ambrose, 2005]. It is also unknown if long-term local administration to the airways can have unexpected effects on pulmonary tissues.
Ultimately, adverse event data from the pending phase III trials for both ciprofloxacin DPI and DRCFI should offer more comprehensive information regarding the safety of inhaled ciprofloxacin in the NCFBE population. Animal studies and post-marketing surveillance will also be necessary in order to examine the risk of toxicity with inhaled ciprofloxacin over time.
A potential limitation of the use of inhaled ciprofloxacin is the risk of emergence of bacterial resistance to the fluoroquinolones or, more specifically, to ciprofloxacin with long-term therapy. Some data indicate development of resistance with long-term oral ciprofloxacin in BE is associated with clinical deterioration [Rayner et al. 1994]. Driving resistance to fluoroquinolones via inhaled administration would potentially eliminate an important oral antipseudomonal option and may increase the need for parenteral antibiotics in the future. The 2010 British Thoracic Society guideline for NCFBE currently recommends against the use of long-term oral fluoroquinolones until further studies are available which better elucidate the risk of resistance and its associated outcomes [Pasteur et al. 2010]. Characterizing this potential risk with inhaled ciprofloxacin will be crucial to determining its place in therapy.
To date, the only available information regarding the emergence of resistance with inhaled ciprofloxacin is from the phase II study of ciprofloxacin DPI in 124 NCFBE patients [Wilson et al. 2012]. The study reported MICs of respiratory pathogens increased to >4 µg/ml in six subjects in the ciprofloxacin DPI group whereas this occurred in no subjects in the placebo group. The resistant pathogens included P. aeruginosa (4/6), S. maltophilia (1/6), and H. influenzae (1/6). In most cases (n = 4 subjects), bacterial resistance was transient and subsequent respiratory cultures grew bacterial isolates with MICs in the susceptible range by the end of the study. However, one subject with mucoid P. aeruginosa had persistent resistance out to day 92 of the study (MICs ranging from 4 to 64 µg/ml over the follow-up period). Follow-up resistance data on the remaining subject were unavailable due to premature study discontinuation. Details regarding the clinical outcomes of these particular subjects were not described.
Given the currently published data, it is also unclear if there is any supportive evidence for the use of inhaled ciprofloxacin in NCFBE patients with acute pulmonary exacerbations. The clinical data for both ciprofloxacin DPI and DRCFI provide preliminary evidence of efficacy in a population of stable NCFBE patients with chronic respiratory pathogen colonization or infection [Serisier et al. 2011a, 2011b, 2012; Wilson et al. 2012]. Further data from fully published clinical trials are needed in order to clearly describe the generalizability and full therapeutic utility of inhaled ciprofloxacin therapy.
If inhaled ciprofloxacin formulations are ultimately approved for clinical use and the aforementioned issues are addressed, inhaled ciprofloxacin will present a valuable new treatment option for the chronic management of NCFBE. The particular strength of inhaled ciprofloxacin in this setting will be that clinical development was geared specifically for the NCFBE population. This will eliminate any issues of generalizability that arise from extrapolating research findings from other patient populations, e.g. CF or COPD. It will also garner important new information regarding the clinical outcomes of chronic antibiotic management in NCFBE patients.
Conclusions
The development of inhaled ciprofloxacin presents a promising new therapy for the chronic management of NCFBE. The DPI formulation is a dosage form which may be more convenient in regards to drug delivery and portability, yet will likely require twice-daily dosing. The liposomal formulation is designed to have a prolonged duration which will likely provide a more desirable once-daily administration schedule. Encapsulation in liposomes may also provide benefits in terms of improved cellular uptake and antibacterial efficacy; however, the clinical impact of these latter properties remains unclear.
Expert groups, such as the British Thoracic Society and the Society of Infectious Diseases Pharmacists, support the conduct of well-designed clinical trials to carefully investigate the efficacy, safety, and optimal use of the inhaled method of antibiotic delivery [Le et al. 2010; Pasteur et al. 2010]. The overall aim of chronic antibiotic therapy in NCFBE is the reduction of exacerbations [Pasteur et al. 2010]. Evaluation of such new therapies should therefore incorporate outcomes associated with exacerbations, e.g. exacerbation frequency, quality of life, and length of hospitalizations [Le et al. 2010; Pasteur et al. 2010]. Phase III randomized, controlled trials for the two different inhaled ciprofloxacin formulations are currently planned and should provide much needed information regarding the use of this therapy in this historically under-recognized patient population.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
M.H. Gotfried receives research support from Gilead Sciences, Inc. J. Justo and L.H. Danziger declare no conflicts of interest.
