Abstract
Background:
Pediatric bronchiectasis is a common respiratory disease in children. The use of video-assisted thoracoscopic surgery (VATS) for its treatment remains controversial.
Objectives:
The objective of our study was to compare and analyze the clinical efficacy of thoracoscopic surgery and thoracotomy in the treatment of pediatric bronchiectasis and summarize the surgical treatment experience of VATS in children with bronchiectasis.
Design:
Retrospective single-center cohort study.
Methods:
A retrospective analysis was conducted on the clinical data of 46 pediatric patients who underwent surgery with bronchiectasis at the Children’s Hospital of Chongqing Medical University from May 2015 to May 2023. The patients were divided into two groups: the VATS group (25 cases) and the thoracotomy group (21 cases). Comparative analysis was performed on various parameters including basic clinical data, surgical methods, operation time, intraoperative blood loss, transfusion status, postoperative pain, postoperative mechanical ventilation time, chest tube drainage time, length of hospital stay, incidence of complications, and follow-up information.
Results:
There were no statistically significant differences between the two groups of patients in terms of age, weight, gender, etiology, duration of symptoms, site of onset, and comorbidities (p > 0.05). The operation time in the VATS group was longer than that in the thoracotomy group (p < 0.001). However, the VATS group had better outcomes in terms of intraoperative blood loss, transfusion status, postoperative pain, postoperative mechanical ventilation time, chest tube drainage time, and length of hospital stay (p < 0.05). The incidence of postoperative complications in the VATS group was lower than that in the thoracotomy group, although the difference was not statistically significant (p = 0.152). Follow-up data showed no statistically significant difference in the surgical treatment outcomes between the two groups (p = 0.493).
Conclusion:
The incidence of complications and mortality in surgical treatment of bronchiectasis is acceptable. Compared with thoracotomy surgery, VATS has advantages such as smaller trauma, less pain, faster recovery, and fewer complications. For suitable pediatric patients with bronchiectasis, VATS is a safe and effective surgical method.
Introduction
Pediatric bronchiectasis is a chronic respiratory disease that is typically caused by incomplete bronchial development due to congenital factors or damage to the elastic and muscular tissues of the bronchial walls caused by acquired factors. 1 It is characterized by a decline in mucociliary clearance function, persistent or recurrent respiratory tract infections, severe bronchial obstruction, and gradual destruction of the smooth muscles, elastic fibers, and even cartilage of the bronchial walls, ultimately leading to bronchial dilation. The main clinical symptoms include cough, sputum production, wheezing, and chest tightness.2,3 Due to the atypical nature of early symptoms, they are easily overlooked, and the symptoms tend to recur, resulting in a prolonged course of the disease and significant impacts on the growth, development, and quality of life of affected children.4,5
In Western countries, cystic fibrosis is the leading cause of bronchiectasis and one of the most prevalent autosomal recessive genetic diseases. However, in developing countries, the most common causes of bronchiectasis are conditions such as pneumonia, tuberculosis, and foreign body aspiration, and the management of these cases still presents challenges. 6 With the advancements in treatments such as postural drainage, antimicrobial drug use, and anti-inflammatory therapy, there has been an improvement in symptoms for some patients with bronchiectasis. However, surgery remains the only viable option for curing irreversible bronchiectasis.7,8 Video-assisted thoracoscopic surgery (VATS), which has shown promising treatment outcomes in adult pulmonary lobe surgeries, has emerged as a common technique.9,10 Consequently, VATS holds potential as a treatment option for pediatric bronchiectasis. Nonetheless, the safety and efficacy of this approach are uncertain due to factors such as the smaller thoracic cavity in children, limited operative space, frequent concurrent infections in bronchiectasis, delicate lung parenchyma, and the possibility of incomplete fissures. 11 In this study, we conducted a retrospective analysis of clinical data from pediatric non-cystic fibrosis bronchiectasis patients who underwent surgery at the Department of Thoracic Surgery, Children’s Hospital of Chongqing Medical University, between May 2015 and May 2023. The aim of the study was to summarize the treatment experience of VATS in pediatric bronchiectasis cases.
Methods
Study design
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement 12 (Supplemental File). We reviewed the clinical data of pediatric patients who underwent bronchiectasis surgery at the Department of Thoracic Surgery, Children’s Hospital of Chongqing Medical University, between May 2015 and May 2023. The data collected included demographic characteristics such as age and gender, etiology, symptoms, auxiliary examinations, surgical-related information, postoperative complications, and follow-up results. Preoperative preparations included detailed medical history inquiries, physical examinations, laboratory tests, pulmonary function tests, electrocardiograms, and high-resolution computed tomography (HRCT). HRCT findings included: (1) bronchial luminal dilation exceeding 1.5 times the normal luminal diameter with thickened walls; (2) bronchial diameter-to-accompanying pulmonary artery diameter ratio >0.8 (in the absence of pulmonary hypertension), presenting a ‘signet ring’ appearance in the transverse section 13 ; and (3) loss of the normal pattern of gradual decrease in airway caliber from the center to the periphery, with a ‘tramline’ appearance in the longitudinal section of the bronchus, and bronchi visible within 1 cm below the chest wall 14 (Figure 1).
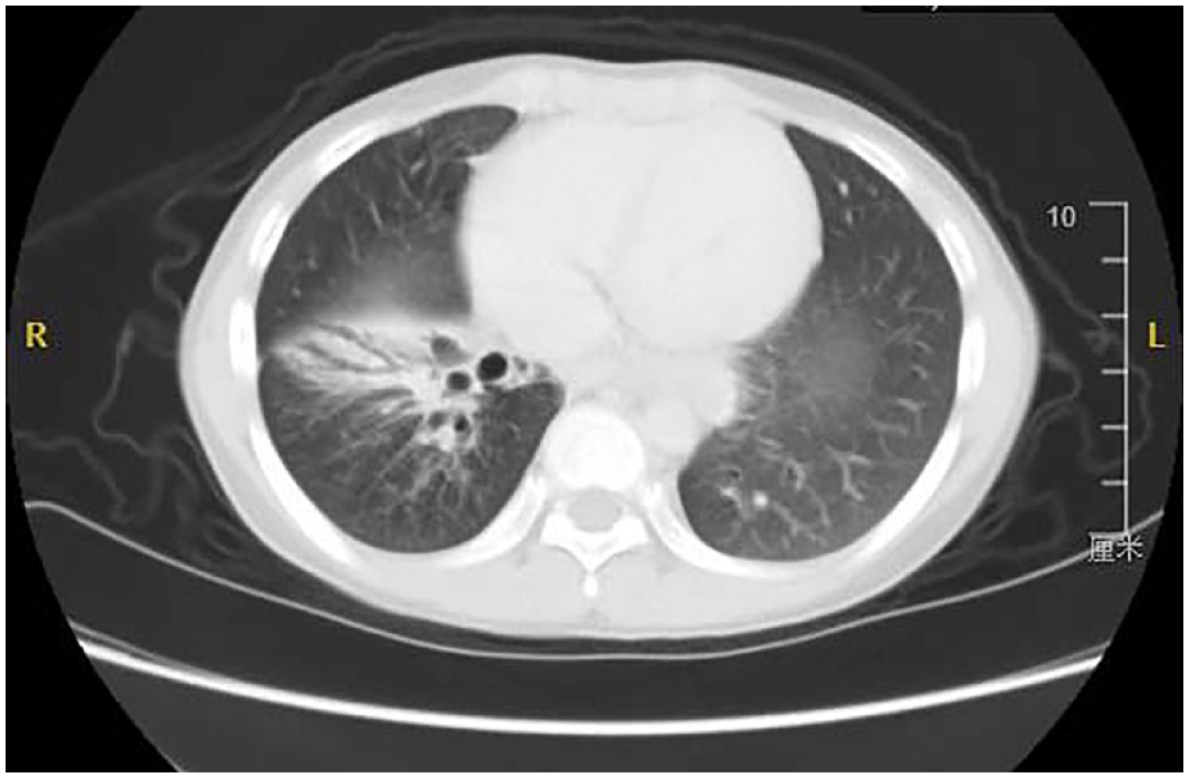
CT scan shows bronchiectasis in the right lower lobe.
Surgical indications included non-diffuse bronchiectasis, failure of medical treatment, and sufficient cardiopulmonary reserve. Failure of medical treatment was defined as the need for multiple hospitalizations due to the impact on learning, growth, or repeated deterioration of the condition. Preoperative selective bronchoscopy was performed to determine the presence of airway abnormalities or foreign bodies, and bronchoalveolar lavage was performed. Antibiotic treatment was administered based on the results of bronchoalveolar lavage fluid or sputum culture combined with drug sensitivity testing. In the thoracotomy group, patients were placed in the lateral decubitus position, and a posterior lateral incision at the fourth or fifth rib was made. The skin and subcutaneous muscles were dissected layer by layer to enter the thoracic cavity. Rib retractors were used to separate the ribs and explore the intrathoracic condition. Arteries were typically addressed before veins to avoid lung congestion. The choice of VATS was primarily based on the comprehensive judgment of the surgeon: absence of a history of recurrent purulent sputum, absence of significant scarring at the pulmonary hilum, mild pleural adhesions, and absence of lymph node calcification. VATS was performed using a three-port technique. Artificial pneumothorax was established to facilitate lung collapse, with a CO2 pressure of 4–6 mmHg and a flow rate of 1–2 L/min. The appropriate surgical approach was chosen based on intraoperative findings, and intraoperative blood gas monitoring was performed. Routine closed chest tube drainage was left in place postoperatively. The potential causes of bronchiectasis in the patients were diagnosed based on intraoperative exploration and postoperative pathological results. Complete resection was defined as the complete removal of anatomically involved pulmonary segments. Follow-up data were obtained through regular outpatient visits or telephone follow-ups.
The surgical outcomes were evaluated according to the following criteria 15 :
Excellent: Complete resolution of symptoms that led to surgery.
Good: Significant improvement in preoperative symptoms, with occasional need for antibiotic treatment.
No change: No improvement in preoperative symptoms, with no reduction in hospitalizations or the need for medication.
Worse: Frequent deterioration of symptoms or worsening of the condition.
Statistical analysis
Data analysis was conducted using SPSS 22.0 statistical software (IBM, Armonk, NY, USA).Continuous variables were expressed as mean ± standard deviation and analyzed using t-tests. Categorical variables were analyzed using the chi-square test. In cases where the theoretical frequency ranged from 1 to 5, the corrected chi-square value was used. When the theoretical frequency was less than 1, Fisher’s exact probability test was applied. A p value less than 0.05 was considered statistically significant.
Results
Between May 2015 and May 2023, a total of 46 pediatric patients with bronchiectasis underwent surgical treatment in our department. Among them, 25 patients underwent thoracotomy, whereas 21 patients underwent VATS. The clinical characteristics of the patients are summarized in Table 1. There were 25 male patients and 21 female patients, with a mean age of 9.50 years (range: 2.50–16.75 years) and an average weight of 29.16 kg (range: 14–55 kg). The main symptoms included cough (65.2%), sputum production (50.0%), and recurrent infections (34.8%). The average duration of symptoms was 29.07 months. The most common cause of bronchiectasis was lung infection (50.0%). The involvement of different lung lobes was similar in both groups, and there were no cases of massive hemoptysis requiring emergency treatment in either group. In the VATS group, two patients had coexisting asthma, whereas in the thoracotomy group, two patients had coexisting asthma and two had coexisting rhinitis. None of the patients in the VATS group had a history of tuberculosis, whereas four patients in the thoracotomy group had a history of tuberculosis. Two patients in the thoracotomy group had forced expiratory volume in the first second of expiration <60% predicted. In the VATS group, four patients had positive sputum/BALF (Bronchoalveolar lavage fluid) cultures, compared to nine patients in the thoracotomy group.
Characteristics of patients with bronchiectasis undergoing VATS and thoracotomy.

p Value < 0.05 was considered to be statistically significant.
CPAM, congenital pulmonary airway malformation; FEV1, forced expiratory volume in the first second of expiration; SD, standard deviation; VATS, video-assisted thoracoscopic surgery.
In the VATS group, intraoperative pleural adhesions were observed in six cases. Three patients were converted to thoracotomy due to bleeding (n = 2) and fused fissure (n = 1). The distribution of surgical procedures is shown in Table 2. The majority of patients underwent lung lobe resection, followed by bilateral lobe resection, lobe and segment resection, and segment resection. In the VATS group, two cases had incomplete resection, compared to five cases in the thoracotomy group. Compared to the thoracotomy group, the VATS group had longer operative time (201.71 ± 54.12 versus 131.60 ± 43.22, p < 0.001). However, the VATS group had lower intraoperative blood loss (40.95 ± 19.46 versus 69.00 ± 31.29, p = 0.001), less postoperative pain (Wong–Baker Pain Rating Scale, 3.81 ± 1.88 versus 6.00 ± 2.16, p = 0.001), shorter mechanical ventilation time postoperatively (4.05 ± 1.80 versus 5.48 ± 2.34, p = 0.025), shorter chest tube drainage time (6.48 ± 1.99 versus 8.04 ± 2.93, p = 0.044), and shorter hospital stay (10.90 ± 2.75 versus 14.04 ± 3.43, p = 0.002) than the thoracotomy group. Six patients in the thoracotomy group required intraoperative or postoperative blood transfusion (p = 0.025), and there were no perioperative deaths in either group. Although not statistically significant, the VATS group had fewer postoperative complications than the thoracotomy group (23.8% versus 44.0%, p = 0.152) (Table 3).
Perioperative data for patients with bronchiectasis undergoing VATS and thoracotomy.

p-Value < 0.05 was considered to be statistically significant.
VATS, video-assisted thoracoscopic surgery.
Postoperative complications for patients with bronchiectasis undergoing VATS and thoracotomy.

p Value < 0.05 was considered to be statistically significant.
VATS, video-assisted thoracoscopic surgery.
All patients completed the follow-up period, with an average duration of 12.21 months (range: 1–38 months). In the thoracotomy group, 20 patients (80.0%) achieved an ‘excellent’ postoperative outcome, 3 patients (12.0%) were categorized as ‘good’, and 2 patients (8.0%) showed ‘no change or worsening’ in their condition. In the VATS group, 19 patients (90.5%) had an ‘excellent’ postoperative outcome, 2 patients (9.5%) were classified as ‘good’, and none of the patients experienced ‘no change or worsening’. Although the outcomes of VATS appeared to be superior to those of thoracotomy surgery, the difference between the two groups was not statistically significant (p = 0.493) (Table 4).
Outcomes of patients with bronchiectasis undergoing VATS and thoracotomy.

p Value < 0.05 was considered to be statistically significant.
VATS, video-assisted thoracoscopic surgery.
Discussion
Bronchiectasis is a chronic suppurative disease in which the bronchial walls are damaged, deformed, and dilated due to factors such as recurrent infections, mucus obstruction, or congenital developmental defects. The incidence of bronchiectasis varies in the research, and there are significant differences observed among different ethnic groups.2,14,16,17 The overall prevalence of bronchiectasis has decreased in recent years due to factors such as the widespread use of antibiotics and the implementation of preventive vaccinations. 18 However, with advancements in our understanding of the disease, improvements in diagnostic criteria, and the widespread use of HRCT imaging, the detection rate of bronchiectasis has significantly increased compared to previous estimates.
HRCT is a non-invasive and highly accurate method for diagnosing bronchiectasis as it directly displays the morphology, distribution, and location of bronchial dilation. 19 Bronchoscopy can detect lesions such as foreign body inhalation, tracheobronchomalacia, and airway deformities. Bronchial mucosal biopsy can be used to diagnose primary ciliary dyskinesia, and bronchoalveolar lavage can be performed for microbial and cytological examination, serving as an important means to identify potential causes in pediatric patients. 18 Preoperative pulmonary function tests help assess the patient’s condition and evaluate treatment outcomes.20,21 Children with bronchiectasis may exhibit obstructive ventilatory dysfunction, restrictive ventilatory dysfunction, or relatively normal lung function. In our study, the majority of patients had relatively normal lung function, which is one of the reasons for the overall favorable prognosis postoperatively. Other auxiliary investigations are also employed to identify the underlying causes of bronchiectasis. For example, blood leukocyte count, neutrophil, and lymphocyte classification counts can aid in diagnosing immunodeficiency disorders, esophageal pH monitoring is used for diagnosing gastroesophageal reflux disease, and sweat tests can assist in diagnosing cystic fibrosis. 22
Drug therapy plays a significant role in controlling the progression of bronchiectasis. It includes physical therapy, antimicrobial agents, 23 anti-inflammatory treatment, and bronchodilators. However, for pediatric patients with bronchiectasis whose symptoms and quality of life are adversely affected and do not respond to drug therapy, surgical treatment can significantly improve outcomes. In cases of bronchiectasis involving multiple lung lobes, where small lesions are difficult to identify or remove, incomplete resection can also be considered. 15 The goal of surgery is to remove the primarily affected areas while preserving as much normal lung tissue as possible. This helps reduce hospitalization time and antibiotic usage, thereby improving the quality of life. Importantly, surgery can also delay the progression of the disease by preventing infection in adjacent lung regions.
VATS offers advantages such as fewer postoperative complications, reduced pain, shorter hospitalization, and improved cosmetic outcomes. Additionally, VATS helps prevent long-term complications associated with thoracotomy, such as shoulder muscle weakness, spinal curvature, and chest wall deformities. 24 Therefore, VATS has become the preferred approach for adult lung lobe surgery. However, as one of the most technically demanding minimally invasive procedures in the pediatric population, the complexity of the disease process and surgical anatomy, the risk of bleeding, and unique anesthesia challenges have limited the number of attempted surgeries. 11 Specifically, there is limited literature reporting on the application of VATS in bronchiectasis surgery, especially in children. Therefore, we have summarized the clinical data of pediatric patients undergoing bronchiectasis surgery in our center in recent years. In our study, VATS procedures had a longer operating time compared to thoracotomy, which may be attributed to the difficulty in maintaining single-lung ventilation and the presence of adhesions in the thoracic cavity, requiring additional time for management. Moreover, children have smaller thoracic cavities, which limit the available space for manipulation and increase the complexity of the procedure. However, pediatric patients in the VATS group demonstrated superior intraoperative blood loss, postoperative ventilator support time, chest tube drainage duration, and length of hospital stay compared to the thoracotomy group. The differences between the two groups were statistically significant. These findings may be attributed to the magnification provided by VATS, which allows for more thorough handling of small blood vessels during tissue dissection. Additionally, the avoidance of rib spreaders in VATS helps prevent rib fractures and associated bleeding. Furthermore, VATS, with its smaller incisions, reduced pain, and faster postoperative recovery, contributes to its overall advantages in pediatric bronchiectasis surgery.
For the majority of bronchiectasis patients, resection of the affected lung lobe can alleviate or eliminate clinical symptoms.25–27 In our study, the overall improvement rate was 95.7%. However, no significant difference was observed between the VATS and thoracotomy groups, which is similar to findings in other studies involving adults.28,29 This suggests that recommending VATS for suitable bronchiectasis patients is reasonable. During VATS surgery, there was one case of prolonged air leakage, which resolved spontaneously following continuous drainage, antimicrobial treatment, and nutritional support. Gentle and careful dissection should be performed, and a water seal test should be conducted to check for air leakage. Reinforcing the bronchial stump with prolene suture can effectively prevent bronchopleural fistula. Traditional notions suggest that handling infected or purulent lesions with thoracoscopy may lead to contamination of the pleural cavity or wounds, resulting in empyema and incisional infections. 30 However, in our study, only one patient in the thoracotomy group experienced an incisional infection. This may be attributed to the practice in VATS procedures of placing excised lung tissue in specimen bags and thoroughly irrigating the thoracic cavity with warm water, reducing the risk of infection.
This study has certain limitations. First, it is a retrospective study, and the selection criteria for thoracoscopic surgery are not well-defined. Therefore, there may be potential selection bias in choosing the surgical approach. Second, the sample size included in the study is relatively small, and further randomized controlled trials with larger sample sizes are needed to validate the safety and efficacy of VATS in children with bronchiectasis.
Conclusion
In conclusion, for irreversible bronchiectasis, early surgical intervention can significantly improve the prognosis. Performing appropriate thoracoscopic surgery in eligible patients can achieve rapid postoperative recovery and reduce postoperative complications.
Supplemental Material
sj-docx-1-tar-10.1177_17534666241228159 – Supplemental material for Video-assisted thoracoscopic surgery for non-cystic fibrosis bronchiectasis in children
Supplemental material, sj-docx-1-tar-10.1177_17534666241228159 for Video-assisted thoracoscopic surgery for non-cystic fibrosis bronchiectasis in children by Fengxia Ding, Zhengxia Pan, Chun Wu, Hongbo Li, Yonggang Li, Yong An, Jiangtao Dai, Gang Wang and Bo Liu in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
