Abstract
Introduction
Patients with severe asthma require high levels of medication to maintain good disease control or experience persisting symptoms despite multiple maintenance treatments [American Thoracic Society, 2000]. They account for only 5–10% of patients with asthma, but have a disproportionate impact on healthcare costs [Antonicelli et al. 2004; Godard et al. 2002]. Severe asthma is a heterogeneous disease and, based on the dominant cell type involved in the airway inflammation, the eosinophilic and noneosinophilic asthma (NEA) phenotypes have been described [Simpson et al. 2006, 2005; Wenzel, 2006; Miranda et al. 2004; Green et al. 2002]. Patients with NEA have increased sputum neutrophils and interleukin-8 (IL-8) levels and are less responsive to corticosteroid therapy than those with eosinophilic inflammation [Simpson et al. 2006, 2005]. Add-on therapies such as oral corticosteroids and anti-IgE monoclonal antibody therapy act specifically on eosinophilic inflammation and allergic IgE pathways and, consequently, offer less relief to patients with NEA [Berry et al. 2007]. Effective anti-inflammatory therapies for these patients are needed [Simpson et al. 2008].
Bronchiectasis is characterized by permanent dilatation of the bronchi that arises from chronic inflammation and bacterial colonization. Even though asthma and bronchiectasis are two different entities, their coexistence has been shown in many patients, especially in those with severe asthma [Oguzulgen et al. 2007]. As in patients with NEA, sputum analysis has shown raised concentrations of neutrophils and IL-8 in patients with non-cystic fibrosis (non-CF) bronchiectasis [Tsang et al. 2000; Richmaneisenstat et al. 1993]. Simpson and colleagues demonstrated that a persistent activation of the innate immune system may lead to this similar pattern of airway inflammation in both diseases [Simpson et al. 2007]. However, it remains uncertain whether bronchiectasis is a comorbidity in patients with asthma that makes the disease more difficult to control, or the result of increased levels of matrix metalloproteinases causing airway destruction and remodeling [Oguzulgen et al. 2007; Demedts et al. 2005; Takemura et al. 2004]. Non-CF bronchiectasis is being recognized with increasing frequency around the world and remains a major cause of excess respiratory morbidity [O’Donnell, 2008]. Since treatment is generally only partly successful in patients with bronchiectasis, improved anti-inflammatory medications are needed.
Beneficial effects of macrolides in asthma have been known for almost 50 years and in the past the reported therapeutic benefits were attributed to the inhibition of metabolism of exogenous steroids [Hrvacic et al. 2009; Fost et al. 1999]. Macrolide antibiotics possess antimicrobial activity against bacteria such as Chlamydia pneumoniae and Mycoplasma pneumoniae, which are associated with asthma exacerbations [Cosentini et al. 2008]. Currently, it is hypothesized that the efficacy of macrolides in patients with severe asthma results not only from their antimicrobial effects, but also from their immunomodulatory properties [Hrvacic et al. 2009]. Doses used in asthma patients are lower than those for standard antibiotic therapy and studies showed an improvement in bronchial hyperresponsiveness independent of the action on exogeneous steroid metabolism [Kostadima et al. 2004; Kamada et al. 1993; Ball et al. 1990].
Neomacrolides, such as clarithromycin and azithromycin, have been shown to have anti-inflammatory and/or immunomodulatory effects in addition to their antimicrobial properties [Giamarellos-Bourboulis, 2008; Lopez-Boado and Rubin, 2008; Shinkai et al. 2008; Culic et al. 2001]. Possible mechanisms of these anti-inflammatory effects include inhibition of inflammatory cytokine production, neutrophil migration and mucus hypersecretion [Crosbie and Woodhead, 2009; Lopez-Boado and Rubin, 2008; Tsai and Standiford, 2004]. Clinical improvement after treatment with macrolides in patients with diffuse panbronchiolitis and CF has been associated with these immunomodulatory effects [Kudoh et al. 1998; Koyama and Geddes, 1997; Sakito et al. 1996]. Reductions in IL-8 and neutrophil numbers in induced sputum after macrolide therapy have been demonstrated in several chronic airway diseases characterized by airway neutrophilia including diffuse panbronchiolitis, CF, bronchiolitis obliterans syndrome following lung transplantation and chronic obstructive pulmonary disease (COPD) [Vanaudenaerde et al. 2007; Verleden et al. 2006; Basyigit et al. 2004; Sakito et al. 1996; Oishi et al. 1994].
Since patients with NEA and/or bronchiectasis have increased neutrophils and IL-8 in their airways, the addition of macrolides to the treatment could be effective in patients with severe asthma and/or bronchiectasis. In a small monocentric randomized placebo-controlled trial, Simpson and colleagues showed a significant reduction in IL-8 and neutrophils in induced sputum of patients with severe asthma after treatment with clarithromycin for 8 weeks [Simpson et al. 2008]. Although the total asthma quality of life score improved with clarithromycin treatment, no significant changes in symptom scores or lung function were reported.
We therefore performed a retrospective observational cohort study and evaluated the safety and efficacy of add-on therapy with neomacrolides on pulmonary function tests and symptom scores in patients with severe asthma and/or bronchiectasis.
Methods
Design
This was a retrospective, single-center, observational cohort study, conducted at the Ghent University Hospital in Ghent, Belgium. A cohort of patients with severe asthma and/or bronchiectasis, receiving low-dose neomacrolides (clarithromycin or azithromycin) as add-on treatment for 3–8 weeks, was established. The ethics committee of Ghent University Hospital approved the study (IRB 2007/206). Patients were treated with clarithromycine (n = 97) 500 or 250 mg once daily or with azithromycine (n = 11) 500 or 250 mg three times a week. After 3–8 weeks of add-on treatment with low-dose macrolides, pulmonary function tests (in patients with asthma) and symptom scores (in both patient groups) were evaluated.
Patients
Male and female non-CF, adult patients with severe asthma and/or bronchiectasis, treated with neomacrolides as add-on treatment between June 2004 and September 2008, were selected. All patients were followed by a respiratory physician at the polyclinic for respiratory medicine at Ghent University Hospital. They were included in the care path for patients with severe asthma and/or bronchiectasis and were unresponsive to current maximal maintenance therapy. Therefore, a trial with low-dose neomacrolides was started as add-on therapy in this subgroup of patients, selected based upon persistent respiratory symptoms and/or frequent exacerbations. Immunocompromised patients and patients diagnosed with allergic bronchopulmonary aspergillosis were excluded. Diagnosis of asthma was made according to American Throracic Society (ATS)/European Respiratory Society (ERS) criteria [Bateman et al. 2008; Global Initiative for Asthma, 2006; Laszlo, 2006]. Based on clinical manifestations, pulmonary function tests, smoking history and high-resolution CT (HRCT) scan, patients were diagnosed with severe asthma (with or without bronchiectasis) or with bronchiectasis without asthma. After 3–8 weeks of treatment, patients were divided into responders and nonresponders based on the median improvement in forced expiratory volume in one second predicted (FEV1; %) in asthma patients and symptom score in patients with bronchiectasis.
Assessments
Personal history, physical examination, pulmonary function tests, allergy skin prick tests, measurement of the fraction of nitric oxide in exhaled breath (FeNO), blood laboratory evaluations such as blood eosinophilia and total IgE, and high-resolution CT scan of the thorax were performed prior to the add-on treatment with macrolides in the same standardized way. Remodeling of proximal airways in severe asthma has been shown to be associated with impaired lung function and neutrophilic airway inflammation [Gupta et al. 2010]. We evaluated proximal airway remodeling by assessing the thickening of the right upper lobe apical segmental bronchus (RB1) on HRCT, which has been shown to be a good proxy for overall airway remodeling [Aysola et al. 2008]. After identifying RB1 on the HRCT images, the lumen area of RB1 (LA) and wall area (WA) were measured. Both measurements (LA and WA) were corrected for body surface area (BSA). The percentage wall area was calculated as follows: % WA = WA/TA x 100. In patients with bronchiectasis only, HRCT-scans were scored using an adaptive scoring system as described previously by Helbich and colleagues and Balla and colleagues [Helbich et al.; Balla et al.].
Spirometry was performed using a Viasys Sensormedics VMAX Spectra 22 according to the ATS/ERS task force guidelines for lung function testing [Laszlo, 2006]. Prior to therapy and after 3–8 weeks of treatment, four respiratory symptoms (including dyspnea, cough, nocturnal cough and sputum purulence/volume) were scored with a semiquantitative symptom score ranging from 0 to 2 and combined with a total symptom score ranging from 0 to 8. After 3–8 weeks, pulmonary function tests were performed only in the patients diagnosed with severe asthma. Severe exacerbations of asthma or bronchiectasis were defined as episodes needing use of systemic corticosteroids, disease-related emergency room visits or hospitalizations [Reddel et al. 2009].
Statistical methods
Results for continuous variables were expressed as mean ± standard deviation (SD), categorical variables were expressed as a proportion. Descriptive analyses evaluated sociodemographic and clinical variables of patients with asthma and patients with bronchiectasis only. A paired-sample Wilcoxon signed rank test was used to compare pulmonary function and symptom score between baseline and after 3–8 weeks of therapy. The Mann–Whitney U-test and chi-square test were used to compare continuous and categorical variables between asthma patients and patients with bronchiectasis and between responders and nonresponders. Multivariate logistic regression analysis was used to identify possible predictors for therapeutic response. All statistical analyses were performed using IBM SPSS Statistics, version 18 (SPSS Inc., Chicago, IL). A p-value of less than 0.05 was considered significant.
Results
Patients
A total of 137 adult, non-CF patients with severe asthma and/or bronchiectasis were treated with low-dose neomacrolides as add-on treatment. Four patients stopped macrolide treatment due to side effects, including rash (n = 1), diarrhea (n = 2) and dysgeusia (n = 1). After 3–8 weeks of treatment, 108 patients were available for evaluation (Figure 1): clarithromycin was used by 97 patients and azithromycin by 11 patients. The clinical characteristics and medication use of the patients with asthma and patients with bronchiectasis are shown in Table 1. Patients with asthma were younger, had a lower FEV1 (%) and FEV1/forced vital capacity (FVC) ratio, and were more frequently allergic compared with patients with bronchiectasis only. In both groups there was a female predominance. Forty percent of the patients with asthma had a severe asthma exacerbation in the last 12 months and 62% of the patients with bronchiectasis had at least one lower respiratory tract infection (LRTI) in this period.

Flowchart of the patient population. CF, cystic fibrosis.
Patient characteristics.

Lung function and symptom score in patients with severe asthma
Nearly all patients with asthma (45 of 47; 96%) were treated with (high-dose) inhaled corticosteroids and long-acting β2-agonists. In all 47 asthma patients, symptom scores were available after 3–8 weeks. Total symptom score decreased significantly (p < 0.001) with a mean procentual improvement of 58.0%. In 37 of the 47 patients with severe asthma, pulmonary function tests were available after 3–8 weeks of add-on treatment. The mean prebronchodilatation FEV1 (%) raised significantly (p = 0.001) from 72.5% to 82.2%. The median difference in FEV1 (%) was 7.0%. Responders were defined as patients having more than 7.0% improvement in FEV1 (%).
Symptom score in patients with bronchiectasis only
Symptom scores for dyspnea, cough, nocturnal cough and sputum purulence and volume all decreased significantly and as a result the mean total symptom score decreased significantly from 4.61 to 1.93 (p < 0.001). The median procentual improvement in total symptom score was 60%. Responders were defined as patients having more than 60% improvement in total symptom score after 3–8 weeks of therapy.
Predictors of therapeutic response
In the subpopulation of patients with severe asthma, responders were older (55.0 vs. 47.0 years, p = 0.042) and tended to have a longer duration of disease (29.0 vs. 9.0 years, p = 0.052) compared with nonresponders. Although not statistically significant, responders had a higher BMI, lower FEV1 and less allergy. There was no significant difference in percentage wall area between responders and nonresponders in patients with severe asthma. Clinical characteristics of responders and nonresponders in the asthma population are compared in Table 2.
Characteristics of responders and non-responders in patients with severe asthma.

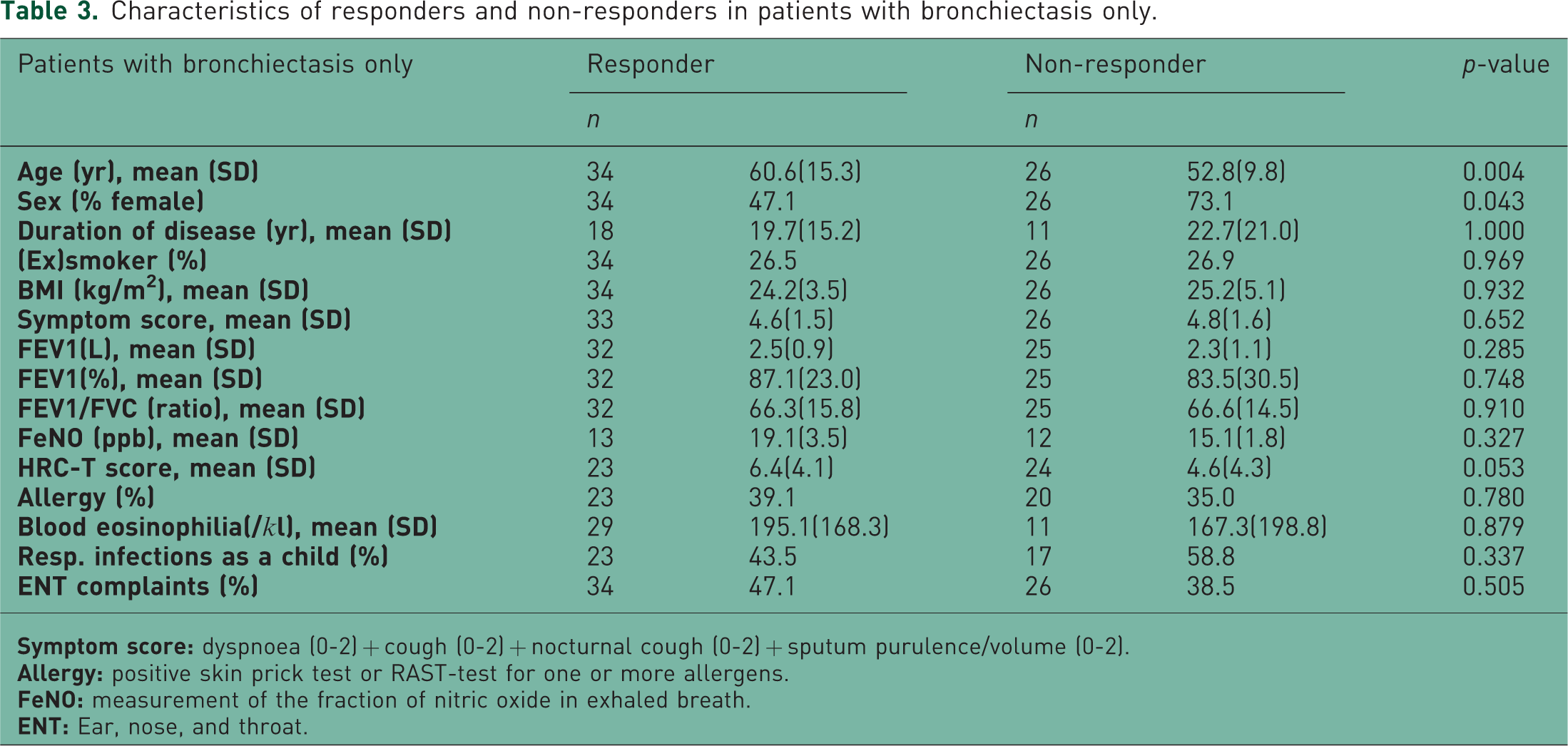
In the patients with bronchiectasis only, responders were older (61.0 vs. 53.0 years, p = 0.004), but there was no difference in duration of disease. Responders were more frequently male (53 vs. 27%, p = 0.043). There was a nonsignificant trend towards higher HRCT score for bronchiectasis in responders (6.4 vs. 4.6; p = 0.053). Characteristics of responders and nonresponders in patients with bronchiectasis are compared in Table 3. The multivariate logistic regression analysis showed that age and male sex were independent predictors for treatment response in patients with bronchiectasis. The odds ratio (OR) for age was 1.06 with 95% confidence interval (CI) 1.01–1.12; OR for male sex was 4.47 with 95% CI 1.14–17.51.
Characteristics of responders and non-responders in patients with bronchiectasis only.
Discussion
In this retrospective observational study, we report a significant improvement in pulmonary function and symptom score in patients with severe asthma after 3–8 weeks of add-on treatment with clarithromycin or azithromycin. Previous studies on the use of macrolides in patients with asthma have shown conflicting results, probably in part due to the heterogeneity of the study populations [Richeldi et al. 2005]. In a study of 55 patients with chronic stable asthma, Kraft and colleagues found improvement in FEV1 only in patients who were PCR+ for Mycoplasma pneumoniae or Chlamydia pneumonia [Kraft et al. 2002]. Two studies have shown significant reductions in bronchial hyperresponsiveness in patients with asthma after 8 weeks of treatment with clarithromycin, but neither showed any significant change in FEV1 [Kostadima et al. 2004; Amayasu et al. 2000]. In a more recent study, Simpson and colleagues showed improvement in quality of life in patients with severe refractory asthma, but no improvement in bronchial hyperresponsiveness or FEV1 was observed [Simpson et al. 2008].
Simpson and colleagues have demonstrated that neutrophilic airway inflammation is associated with a good response to clarithromycin [Simpson et al. 2008]. Recently, using cluster analysis in the Severe Asthma Research Program (SARP), five distinct clinical phenotypes of severe asthma have been identified [Moore et al. 2010]. Interestingly, cluster 3, a unique group of mostly older obese women with late-onset nonatopic asthma, and cluster 5, a group with severe airflow obstruction and late-onset nonatopic asthma, have the highest percentages of neutrophils in induced sputum [Moore et al. 2010]. Older age, female predominance, overweight and the relatively low prevalence of allergy, as observed in our patients with severe asthma, are in line with the clinical characteristics of clusters 3 and 5. These clinical characteristics (older age and less allergy) are even more pronounced in the responders, suggesting that neutrophilic airway inflammation might predominate in these patients with severe asthma and explain the therapeutic benefit of macrolides. However, in our study, no examination of airway inflammation as a response determinant or outcome measure has been made.
In patients with bronchiectasis only, we observed a significant improvement of dyspnea, cough, nocturnal cough, sputum purulence/volume and total symptom score. Despite the fact that trials of macrolide treatment in non-CF bronchiectasis are limited, there is some consistent evidence of a decrease in exacerbation frequency and sputum volume [Crosbie and Woodhead, 2009; Cymbala et al. 2005]. In a prospective open-label study on 39 adult patients, Davies and Wilson reported a decrease in exacerbation frequency, in the need for i.v. antibiotics and in symptoms after treatment with low-dose azithromycin [Davies and Wilson, 2004]. Other studies have been conducted in children or with macrolides other than azithromycin and clarithromycin [Yalcin et al. 2006; Tsang et al. 1999; Koh et al. 1997]. These studies correspond with our findings that sputum volume or purulence was decreased after neomacrolide therapy.
In patients with bronchiectasis, we found that responders were older, more frequently male and had a higher HRCT score for bronchiectasis compared with nonresponders. After correcting for covariates using multivariate logistic regression analysis, age (OR age 1.06, 95% CI 1.01–1.12) and male sex (OR male sex 4.47, 95% CI 1.14–17.51) appeared to be independent predictors for good response on neomacrolide therapy. We put forward the hypothesis that a male predominance in responders might explain the connection between non-CF bronchiectasis and diffuse panbronchiolitis (DPB), which is an idiopathic inflammatory airway disease, most common in eastern countries, that evolves to bronchiectasis, respiratory failure and death when untreated [Poletti et al. 2006]. As in patients with bronchiectasis, the airways in patients with DPB are frequently colonized by Pseudomonas aeruginosa and have increased numbers of neutrophils and cytokines as IL-8 [Poletti et al. 2006; Koyama and Geddes, 1997; Sakito et al. 1996]. Thus, DPB is linked to bronchiectasis in its pathophysiology and in its evolution. Since DPB is slightly more frequent in males [Poletti et al. 2006] and since macrolides are very effective in the treatment of DPB [Kadota et al. 2003; Koyama and Geddes, 1997], the link between bronchiectasis and DPB could well explain the male predominance for therapeutic response observed in our study.
As in all observational studies, our study has several limitations. First, the retrospective nature of our study could be associated with poorer data quality (e.g. missing data) in comparison with prospective studies. Since biological parameters as FeNO and blood eosinophilia were not determined repeatedly in this study, we focused on clinically relevant outcomes, i.e. pulmonary function and symptom scores. Second, the statistical analyses are based on the per protocol population, implicating that results are only available from those patients who had a re-evaluation after 3–8 weeks. Third, the uncontrolled design of the study likely overestimates the treatment effects. Finally, this study was conducted in a single center and therefore these findings should be replicated in larger multicenter controlled studies in patients with severe asthma. Despite these limitations, observational studies provide useful and important information about treatment effects in a ‘real-world’ setting in addition to randomized controlled trials.
In summary, we conclude that neomacrolides may be useful as add-on therapy in patients with severe asthma and/or bronchiectasis. Older age in patients with severe asthma and older age, male gender and a higher HRCT score for bronchiectasis in patients with bronchiectasis only may predict therapeutic response. Future large, randomized controlled studies are required to gather more evidence for the implementation of macrolides in these patients. We therefore refer to the ongoing multicenter double-blind placebo-controlled randomized AZIthromycine in Severe ASThma study (AZISAST) (see www.clinicaltrials.gov).
Footnotes
Acknowledgements
All authors have contributed substantially to the submitted work and have read and approved the final manuscript. In particular, Dr Brusselle, Dr Joos and Dr Coeman performed the design of the study, data acquisition, analysis, and drafting of the manuscript. Dr Van Durme, Dr Bauters and Dr Demedts participated in data acquisition and analysis. Dr Smeets performed the analyses of the HRCT of the lungs. Dr Deschepper contributed crucially to the statistical analysis.
We acknowledge the contribution of Dr Tom Holvoet and Dr Lies D’Hooghe to the HRCT analysis and the revision of the manuscript.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Dr M. Coeman declares no conflict of interest.
Dr Y. van Durme declares no conflict of interest.
Dr F. Bauters declares no conflict of interest.
Dr I. Demedts declares no conflict of interest.
Dr E. Deschepper declares no conflict of interest.
Dr P. Smeets declares no conflict of interest.
Professor Dr G. Joos declares no conflict of interest.
Professor Dr G. Brusselle declares no conflict of interest.
