Abstract
Chronic obstructive pulmonary disease (COPD) is a progressive disease in which patients become increasingly disabled by their symptoms and limited in their activities. Health-related quality of life may be profoundly impaired even in the early stages of the disease. Treatment with long-acting inhaled bronchodilators can improve lung function, symptoms and health status and reduce exacerbations of COPD. This review profiles the efficacy, safety and tolerability of indacaterol, an inhaled β2-agonist bronchodilator for once-daily maintenance treatment of patients with COPD. After 12 weeks of treatment with a once-daily dose of 75 µg (the dose approved in the USA and Canada) in patients with moderate to severe COPD, compared with placebo, indacaterol provided significant and clinically relevant levels of bronchodilation [difference in trough forced expiratory volume in 1 s: 131 ml; 95% confidence interval (CI) 104–159; p < 0.001], together with significant reductions in symptom scores (difference in transition dyspnea index total score: 0.84 points; 95% CI 0.37–1.31; p < 0.001) and improvements in health status (difference in St George’s Respiratory Questionnaire total score: −3.8 units; 95% CI −5.6 to −2.0; p < 0.001). The overall safety and tolerability of once-daily treatment with indacaterol 75 µg for 12 weeks did not differ in any substantial aspect from placebo treatment. Indirect comparisons analyzing pooled clinical data and meta-analyses suggest that treatment with indacaterol 75 µg once daily may be effective in reducing exacerbations of COPD, and that its effects on lung function and health status will be comparable with other currently available inhaled long-acting bronchodilators used for COPD. Treatment with indacaterol 75 µg once daily provides effective bronchodilation, improves dyspnea and health status, and has a well characterized profile of safety and tolerability.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is a common problem in the adult population and a leading cause of death. The estimated population prevalence of COPD [Global Initiative for Chronic Obstructive Lung Disease (GOLD) spirometry stage II or higher] in the USA is 12.7% for men and 15.6% for women aged 40 years and over [Buist et al. 2007]. In most age groups, the prevalence of COPD in Canada is also higher in women than men [Public Health Agency of Canada, 2007]. COPD is the third leading cause of death in the USA, accounting for 124,477 deaths in 2007 [CDC, 2010]. In 2004, COPD accounted for about 4% of all deaths in Canada and was the fourth leading cause of death in both men and women in Canada [O’Donnell et al. 2007; Public Health Agency of Canada, 2007]. COPD also presents a considerable burden in subjects of working age [Fletcher et al. 2011], and health status can be significantly impaired even in patients with mild or moderate disease [Jones et al. 2011a, 2011b].
GOLD sets out a recommended strategy for treating patients with COPD. The aims of treatment are to reduce symptoms and their impact on exercise tolerance and health status, and to reduce the risk of future adverse health events [GOLD, 2011], such as exacerbations. While no drug treatment has been shown conclusively to prevent disease progression and reduce mortality in COPD, long-acting bronchodilators have been shown to be effective in reducing symptoms, improving exercise tolerance and health status, and preventing exacerbations [Calverley et al. 2007; Jones et al. 2011a, 2011b; Vogelmeier et al. 2011; Bédard et al. 2012]. Long-acting bronchodilators are therefore at the forefront of pharmacological management strategy for patients with COPD [GOLD, 2011]. Two classes of long-acting bronchodilators are available: long-acting β2 agonists (LABAs) and long-acting muscarinic antagonists (LAMAs). Two agents are available for once-daily administration: the LAMA, tiotropium, and the LABA, indacaterol.
This review focuses on the development and clinical efficacy and safety of the once-daily LABA, indacaterol. Indacaterol was developed as a long-term maintenance treatment for COPD and is approved for use in this indication. Indacaterol is currently approved in all major markets, including the USA and Canada, where it is approved for use at a once-daily dose of 75 µg. In other countries, including those in the European Union, indacaterol is approved at doses of 150 and 300 µg once daily, while in others, such as Japan and China, the approved dose is 150 µg once daily. The phase III clinical trial program includes safety experience gained with treatment for up to 1 year and at doses up to 600 µg. This review will focus on the clinical experience with the 75 µg dose, drawing on results obtained with other doses to illustrate specific points if necessary.
Physical chemistry and pharmacology
Indacaterol is a chirally pure R enantiomer [Battram et al. 2006]. Indacaterol stimulates β2 adrenoceptors on airway smooth muscle in the lungs, resulting in increased levels of the intracellular messenger cyclic adenosine monophosphate, which leads to relaxation of airway smooth muscle and, thus, bronchodilation [Sayers et al. 2009].
Indacaterol is a partial agonist at the β2 adrenoreceptor. Its mean maximal effect (Emax) at the β2 receptor is 73% that of the reference (full agonist) compound isoprenaline (for comparison, the Emax of the partial agonist salmeterol is 38%) [Battram et al. 2006; Naline et al. 2007]. The lack of any antagonistic effect to the short-acting β2 agonist salbutamol [Naline et al. 2007] suggests that cross tolerance to the two agents in clinical use is unlikely. The relative binding affinities at the three types of β adrenoceptor show selectivity of indacaterol for β2 over β1 and β3 adrenoceptors [Naline et al. 2007].
Studies in human bronchi showed that indacaterol was characterized by a rapid onset of action (similar to salbutamol and formoterol; faster than salmeterol) and a long duration of action (longer than salbutamol, formoterol, and salmeterol) [Naline et al. 2007; Sturton et al. 2008]. The differences between β2 agonists in onset and duration of action may be due to differences in their interaction with cell membranes in the region of the β2 receptors. In vitro work modeling these membrane regions showed several minor differences between indacaterol and salmeterol, including higher partitioning of indacaterol into the microenvironment of the receptor and faster membrane permeation of indacaterol [Lombardi et al. 2009]. Providing β2 agonists by inhalation delivers effective drug levels directly to the bronchiolar epithelium and smooth muscle, and allows reduced doses to provide similar bronchodilation compared with oral and intravenous systemic delivery. Taken together, these effects are likely to contribute to the faster onset and longer duration of therapeutic action of indacaterol [Lombardi et al. 2009].
Pharmacokinetics
Indacaterol is rapidly absorbed after inhalation in humans, reaching peak serum levels (C max) at approximately 15 min. Steady-state serum concentration was achieved after 12–15 days of once-daily dosing [Novartis Pharmaceuticals Corporation, 2011].
Distribution is extensive and serum and plasma protein binding is high. After oral administration, unchanged indacaterol was the main component in serum, and a hydroxylated derivative was the most prominent metabolite. Hydroxylation of indacaterol is predominantly via the isoenzyme cytochrome P450 3A4 [Novartis Pharmaceuticals Corporation, 2011]. Renal clearance makes a very small contribution to elimination (<2% of dose was recovered in urine). After oral administration, at least 90% of the dose was recovered in the excreta, mostly as unchanged drug (54% of dose) and hydroxylated metabolites (23% of dose) [Novartis Pharmaceuticals Corporation, 2011; Kagan et al. 2012]. Pharmacokinetic analyses have shown that no dose adjustments are required for the effect of age, gender, and weight on systemic drug exposure in patients with COPD [Novartis Pharmaceuticals Corporation, 2011]. Patients with mild and moderate hepatic impairment showed no relevant changes in pharmacokinetic variables [Goldsmith et al. 2011].
Drug delivery and administration
Indacaterol gelatine capsules contain indacaterol maleate in a dry powder formulation with lactose as an inactive carrier. Individual capsules are loaded into a single-dose, dry-powder inhaler (Arcapta Neohaler in the USA, and Onbrez Breezhaler in most other countries; Novartis, East Hanover, NJ, USA) fitted with internal pins which pierce the capsule so the patient can inhale the contents. The inhaler has a low internal resistance to the patient’s intake of breath so a low degree of inspiratory effort is required, making it suitable for use by patients with a range of severity of airflow limitation [Pavkov et al. 2010]. Patients’ handling of the indacaterol inhaler has been compared with that of the single-dose dry-powder inhaler used with the LAMA tiotropium (Spiriva HandiHaler, Boehringer Ingelheim, Germany), which works on a similar principle. Although both inhalers scored highly with few errors, patients reported an overall preference for the indacaterol inhaler on the basis of comfort, simplicity of use and confidence that they had taken their medicine correctly [Chapman et al. 2011a].
Indacaterol is taken once daily, and has routinely been administered in the morning in the clinical development studies, given the need for consistent timing of post-dose measurements (in view of diurnal variation of lung function). A study with the indacaterol 300 µg dose taken in the morning or evening reported no difference in bronchodilator effect or in effect on symptoms [Magnussen et al. 2010], providing supporting evidence that the time of once daily dosing does not impact indacaterol efficacy.
Pharmacodynamics
Most of the phase IIb and III clinical studies with indacaterol have evaluated its bronchodilator effect by measuring forced expiratory volume in 1 s (FEV1) at 24 h following the previous dose, also called ‘trough’ FEV1. Another key variable is the area under the curve (AUC) of FEV1 during 4 h post dose, which characterizes the bronchodilator response in the first hours after dosing and therefore includes the peak effect.
Doses for phase III clinical studies in global development were selected based on investigation of doses of indacaterol from 75 to 600 µg once daily with active and placebo comparisons in a population of patients with COPD. This dose-ranging program included a 2-week adaptive, seamless study [Barnes et al. 2010], using predefined criteria to select two doses (150 µg and 300 µg) for longer-term investigation of safety and efficacy for regulatory submissions. As illustrated in Figure 1, the 150 µg dose was chosen because of its numerical superiority in effects on lung function; the 300 µg dose was also selected for further evaluation of additional efficacy benefits over the longer term, and to further support safety assessment of indacaterol. The safety data from this ongoing study, as well as another ongoing 1-year study of higher doses (300 and 600 µg) [Dahl et al. 2010], showed no safety signal with any dose of indacaterol [Barnes et al. 2010].

Treatment differences versus placebo in (a) trough FEV1 at day 15 and (b) FEV1–AUC1–4h at day 15. Broken lines represent the preset efficacy criteria for dose selection: (a) for trough FEV1, the highest value among the formoterol–placebo difference, the tiotropium–placebo difference, or 120 ml; (b) for FEV1–AUC1–4h, the highest among the formoterol–placebo and tiotropium–placebo differences. Data are least squares means with 95% confidence intervals (analysis of covariance). Data are from the first stage of an adaptive, seamless design study. AUC, area under the curve; FEV1, forced expiratory volume in 1 s; FOR, formoterol 12 µg twice daily; IND, indacaterol (75, 150, and 300 µg once daily); TIO, open-label tiotropium 18 µg once daily [Barnes et al. 2010].
Further investigation of the efficacy of indacaterol at doses less than 150 μg in bronchoreactive patients (as a more sensitive population to differentiate between doses) was conducted at the request of the US Food and Drug Administration (FDA) for US registration to identify the minimum effective dose. In a dose-ranging study [ClinicalTrials.gov identifier: NCT01079130] in patients with persistent asthma (indacaterol 18.75, 37.5, 75, and 150 μg once daily), indacaterol 75 and 150 μg were each more effective than each of the lower doses [Novartis Pharmaceuticals Corporation, 2011] on the primary outcome measure, trough FEV1 at day 15.
In their review of the US new drug application submission of indacaterol 75 µg and 150 µg once daily for COPD, the FDA concluded that both doses showed substantial evidence of efficacy, including improved health status, and had an adequate safety profile, and that the 75 µg dose should be approved for the treatment of patients with COPD [Novartis Pharmaceuticals Corporation, 2011]. Similarly, based on their review of data on quality, safety, and efficacy (including spirometry and symptom-related variables such as dyspnea and health status), Health Canada concluded that the benefit/risk profile of indacaterol 75 µg was favorable for the indication of COPD [Novartis Canada, 2011].
Efficacy
Bronchodilator efficacy
In many of the indacaterol clinical studies, the primary efficacy variable was trough FEV1 after 12 weeks of treatment. Trough FEV1 following morning dosing of a 24 h bronchodilator assesses the bronchodilator effect of treatment at a time when the majority of patients report their symptoms to be worst, that is, on waking and rising [Kessler et al. 2011]. Trough FEV1 gives a conservative estimate of the full clinical benefits of bronchodilators in COPD. Serial spirometry studies indicate that once-daily bronchodilators provide peak airflow improvements, especially during the first 12 h after dosing, which are further sustained throughout the 24 h period prior to trough [Beeh and Beier, 2010]. In the clinical studies with indacaterol, a difference in trough FEV1 relative to placebo of 120 ml was prespecified as clinically relevant. This lies at the midpoint of the 100–140 ml range reported as clinically relevant by an American Thoracic Society–European Respiratory Society task force [Cazzola et al. 2008].
The bronchodilator efficacy of indacaterol 75 µg was confirmed in two 12-week, placebo-controlled studies in a total of 641 patients with moderate to severe COPD. Indacaterol 75 µg significantly increased trough FEV1 by 120 and 140 ml over placebo in the two studies, respectively, after 12 weeks of treatment [Kerwin et al. 2011a]. This represented a difference of 131 ml [95% confidence interval (CI) 104–159; p < 0.001] when results of the two studies were pooled [Donohue et al. 2012].
Onset of action
A fast onset of bronchodilator effect that may be perceptible to patients is one factor that may help improve patient adherence to treatment in COPD [Bourbeau and Bartlett, 2008]. In the two placebo-controlled studies with indacaterol 75 µg, FEV1 at 5 min after the first dose was significantly increased, by 90 and 100 ml compared with placebo [Kerwin et al. 2011a].
Long-term maintenance
In the two 12-week studies evaluating indacaterol 75 µg, similar, statistically significant differences between active and placebo treatments were observed after 4 and 12 weeks of treatment (120 ml in one study, and 140 ml in the second study, all p < 0.001) [Kerwin et al. 2011a] demonstrating maintenance of efficacy over the 12-week treatment period. There are no 1-year studies with the 75 µg dose, but the bronchodilator efficacy of indacaterol doses of 150 and 300 µg given for up to 1 year was maintained during long-term treatment, with no evidence of tolerance [Dahl et al. 2010; Chapman et al. 2011].
Symptoms
Breathlessness, or dyspnea, is the most common symptom of COPD and the most troubling for patients [Kessler et al. 2011; Celli et al. 2004]. Most of the clinical studies with indacaterol measured the effect on symptoms using the transition dyspnea index (TDI) [Mahler et al. 1984]. A difference in TDI total score of at least one point between active and placebo treatments or from baseline is considered to be the minimum clinically important difference (MCID) [Mahler and Witek, 2005].
Analysis of pooled data from two 12-week studies showed a difference between indacaterol 75 µg and placebo for TDI total score at week 12 of 0.84 points (95% CI 0.37–1.31; p < 0.001) (Figure 2(a)). Compared with placebo, patients receiving indacaterol had a significantly greater likelihood of achieving the MCID, with an odds ratio of 1.79 (p < 0.001 versus placebo) (Figure 2(b)) [Kerwin et al. 2011a; Donohue et al. 2012].
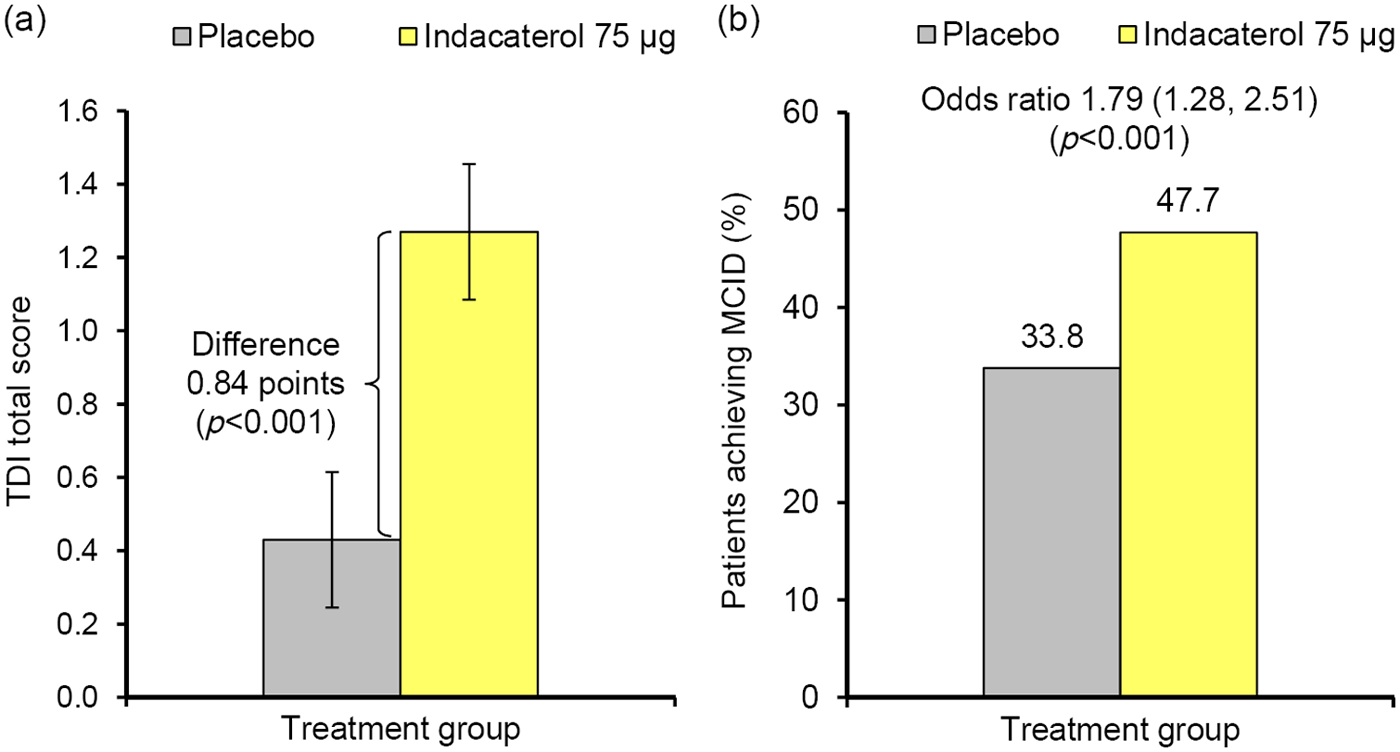
(a) TDI total score and (b) percentage of patients achieving the MCID in TDI total score (≥1 point) after 12 weeks of treatment with indacaterol 75 µg. Data in (a) are least squares means ± standard error. Pooled data from two 12-week, placebo-controlled studies. [Kerwin et al. 2011a; Donohue et al. 2012]. MCID, minimum clinically important difference; TDI, transition dyspnea index.
Use of rescue albuterol
In the clinical studies with indacaterol, patients were provided with the short-acting β2 agonist albuterol, which they could use as required for symptom relief or ‘rescue’. Efficacy of treatment was assessed in terms of changes in puffs per day of albuterol used, and the proportion of days when no albuterol was required, both recorded in patient diaries. A patient’s need to use rescue albuterol may indirectly reflect their level of symptoms [Witek and Mahler, 2003; Partridge et al. 2010].
In the two 12-week placebo-controlled studies, patients treated with indacaterol 75 µg recorded a greater decrease from baseline in the use of rescue albuterol (by 1.2 puffs per day and 0.7 puffs per day; p < 0.001), and used albuterol on fewer days (differences of 13.7% and 8.4% of days; p < 0.01) compared with those receiving placebo [Kerwin et al. 2011a]. Pooled results from the two studies are depicted in Figure 3.

Effect of indacaterol 75 µg on use of albuterol as rescue medication over 12 weeks of treatment: (a) change from baseline in mean daily number of puffs of rescue albuterol used and (b) percentage of days without rescue albuterol use. Data are least squares means ± standard error. Pooled data from two 12-week, placebo-controlled studies [Kerwin et al. 2011a; Donohue et al. 2012].
Health status
COPD has a profound impact on patients’ everyday lives and activities that is not always fully reflected in measurements of airflow limitation and may be more accurately measured by patient-reported outcomes such as health status [Jones and Agusti, 2006; Jones et al. 2011a, 2011b]. In the indacaterol clinical studies, health status was assessed using the St George’s Respiratory Questionnaire (SGRQ) [Jones et al. 1992]. A difference from baseline or from placebo of −4 units is considered the MCID [Jones, 2005].
In the two studies with indacaterol 75 µg, mean SGRQ total scores decreased from baseline with indacaterol treatment by more than the MCID by week 12 (changes from baseline of −5.8 and −4.9 in the two studies, respectively) [Kerwin et al. 2011b]. Analyzing pooled data from the two studies, compared with placebo, indacaterol improved SGRQ total score by −3.8 units (95% CI −5.6 to −2.0; p < 0.001) (Figure 4(a)). The odds ratio for achieving the MCID at week 12 was significantly greater with indacaterol than with placebo (Figure 4(b)).

(a) SGRQ total score and (b) percentage of patients achieving the MCID in SGRQ total score (≥4 units) after 12 weeks of treatment with indacaterol 75 µg. Data in (a) are least squares means ± standard error. Pooled data from two 12-week, placebo-controlled studies [Kerwin et al. 2011a; Donohue et al. 2012]. MCID, minimum clinically important difference; St George’s Respiratory Questionnaire.
Comparative studies
Apart from dose-ranging studies, no phase III clinical trials to date have included head-to-head comparisons of indacaterol 75 µg once daily with other long-acting maintenance monotherapies, although other doses have been evaluated. Cope and colleagues therefore conducted a meta-analysis to assess the comparative efficacy of indacaterol 75 μg versus tiotropium, salmeterol, and formoterol. Their analysis was based on a synthesis of currently available randomized clinical trial evidence identified from a systematic literature review as well as an individual patient data meta-analysis of randomized clinical trials with indacaterol, and focused on the outcomes of trough FEV1 and health status as measured by the SGRQ. For FEV1, indacaterol 75 μg was expected to be more effective than formoterol (difference of 70 ml in trough FEV1) and comparable with tiotropium and salmeterol; and was expected to be comparable with all active treatments for health status [Cope et al. 2012b].
At doses of 150 or 300 µg (higher than the North American approved dose of 75 µg), three studies have compared treatment with indacaterol with the once-daily LAMA tiotropium [Vogelmeier et al. 2010; Donohue et al. 2010; Buhl et al. 2011]. A 12-week, head-to-head comparison of indacaterol 150 µg and tiotropium 18 µg under blinded conditions (n = 1598) reported that the two treatments had similar bronchodilator effects on trough FEV1.The effects of indacaterol were significantly greater than tiotropium 18 µg at week 12 for dyspnea (TDI total score) and health status (SGRQ total score), with significantly higher odds ratios with indacaterol than with tiotropium for achieving the MCID in both measures at week 12 [Buhl et al. 2011].
Other randomized clinical trials with doses higher than 75 µg once daily have compared indacaterol with the twice-daily LABAs formoterol and salmeterol. Indacaterol proved to be the more effective bronchodilator in terms of its effect on trough FEV1 after 12 weeks compared with both salmeterol 50 µg [Kornmann et al. 2011; Korn et al. 2011] and formoterol 12 µg [Dahl et al. 2010], and provided a greater reduction in dyspnea (TDI total score at week 12) compared with salmeterol [Kornmann et al. 2011; Korn et al. 2011].
Indacaterol has not been directly compared with the fixed-dose combinations of inhaled corticosteroids (ICS) and LABAs in multiple-dose studies. In the absence of such comparisons, Cope and colleagues conducted a network meta-analysis of data from available randomized clinical trials of indacaterol 75 µg and the ICS/LABA combinations fluticasone/salmeterol and budesonide/formoterol [Cope et al. 2012a]. They reported that indacaterol 75 µg would be expected to provide comparable efficacy for lung function (trough FEV1), health status (SGRQ) and breathlessness (TDI).
Exercise capacity and lung hyperinflation
Air trapping in the lungs at rest or with activity is a fundamental part of reduced lung function in most patients with COPD, and acute increases in air trapping during exercise or exacerbations (‘dynamic hyperinflation’), as measured by reductions in inspiratory capacity or increases in end-expiratory lung volumes (e.g. increased functional residual capacity), are accompanied by dyspnea and impaired gas exchange. Patients can be severely limited in the physical activities they are able to undertake, and this contributes to their impaired health-related quality of life [Laveneziana et al. 2007]. Bronchodilator treatment can allow improved lung emptying, providing intervention at an early point in this downward spiral of disease [O’Donnell and Laveneziana, 2006; Cooper, 2006].
The effects of indacaterol on exercise capacity and hyperinflation were evaluated in two studies, both using an indacaterol dose of 300 µg. A 3-week crossover study (n = 90) reported improved exercise endurance time with indacaterol 300 µg compared with placebo after the first dose and after 3 weeks. After 3 weeks, indacaterol 300 µg also increased resting and end-exercise inspiratory capacity, an indirect measure of dynamic hyperinflation [O’Donnell et al. 2011]. A smaller, 2-week crossover study (n = 27) showed similar effects, with improvements with indacaterol 300 µg relative to placebo in resting and dynamic inspiratory capacity and exercise endurance time [Beeh et al. 2011].
Exacerbations
A history of frequent exacerbations of COPD is taken as an indicator of future risk, and increased exacerbations are associated with increased mortality in COPD [Hurst et al. 2010; GOLD, 2011]. Therefore, prevention of moderate to severe exacerbations (requiring systemic corticosteroids, antibiotics, or hospitalizations) has become a high priority for COPD pharmacotherapies.
Data on the effect of indacaterol 75 µg on COPD exacerbations are limited. The two 12-week studies with indacaterol 75 µg once daily included exacerbations only as an exploratory efficacy variable, particularly because these studies, which were designed to measure lung function and symptoms, were too short in duration to assess COPD exacerbations. (Exacerbations are subject to seasonal variation and so should ideally be studied over treatment periods of at least 1 year [Jenkins et al. 2012].) In addition, patients were not selected for these studies on the basis of a history of, or risk of, exacerbations. Overall numbers of COPD exacerbations were very low in both studies and, as expected, given the short duration and low overall rate, results were inconclusive.
Donohue and colleagues investigated the effect of indacaterol on COPD exacerbations (as an efficacy variable) using all available pooled data from clinical studies of at least 12 weeks in duration, taking into account the different treatment durations to produce an ‘annualized’ exacerbation rate with all approved indacaterol doses (i.e. 75, 150, and 300 µg). All indacaterol doses were associated with similar, significant reductions in exacerbation rates versus placebo, and the effect of the indacaterol 75 µg dose was not statistically significant from any of the other active treatments included in this analysis (Figure 5) [Donohue et al. 2011].
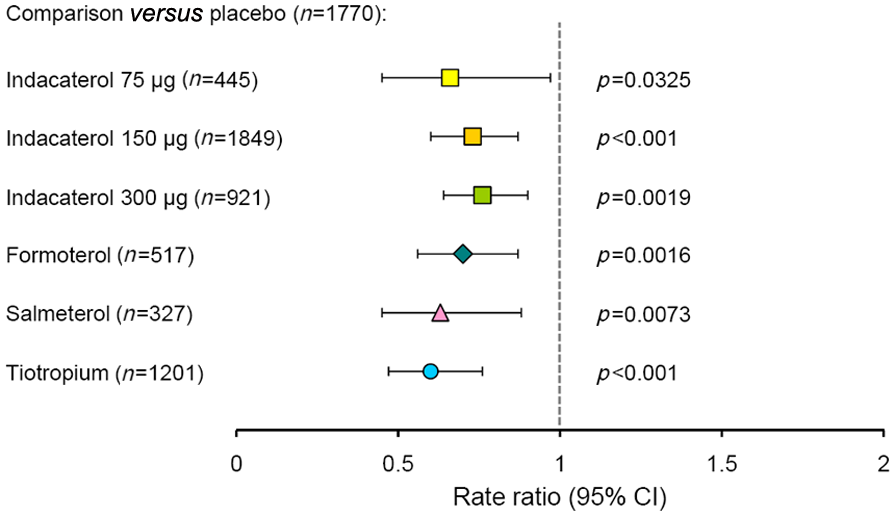
Annualized rate of chronic obstructive pulmonary disease exacerbations compared with placebo. Rate ratios less than 1 (to the left of the vertical broken line) indicate an improvement relative to placebo. Results from pooled analysis of safety data from indacaterol clinical studies. Tiotropium was administered open label in 35% of this treatment group [Donohue et al. 2011]. CI, confidence interval.
Safety
Safety and tolerability have been closely monitored and extensively reported during the indacaterol development program, both in individual studies and in pooled data analyses.
In the two 12-week studies of indacaterol 75 µg and placebo [Kerwin et al. 2011a], the overall frequency of adverse events was similar between treatment groups (Table 1). Of those events that were more common with indacaterol treatment, nearly all were mild or moderate in severity (cough, 21 out of 22 events were mild or moderate; nasopharyngitis, 17 out of 18; headache, 13 out of 14; bronchitis, 10 out of 10; urinary tract infection, 10 out of 10; oropharyngeal pain, 8 out of 8). COPD worsening was less common with indacaterol than with placebo and most cases were mild or moderate (26 out of 28 events).
Adverse events [n (%)] occurring in at least 2% of indacaterol treatment group: pooled data from two 12-week studies [Donohue et al. 2012; Kerwin et al. 2011a].

COPD, chronic obstructive pulmonary disease.
In the two 12-week studies [Kerwin et al. 2011a], serious adverse events occurred in eight patients (2.5%) treated with indacaterol 75 µg and in 13 (4.1%) of the placebo group. The only serious events occurring in more than one indacaterol-treated patient were noncardiac chest pain in two patients and COPD worsening in two. None of the serious adverse events was suspected to be related to study treatment. Two patients died, both in the placebo group.
The safety and tolerability of higher doses of indacaterol (150, 300 and 600 µg) have been evaluated during treatment periods of up to 1 year, with no untoward findings or concerns raised [Chapman et al. 2011b; Dahl et al. 2010].
A safety analysis of pooled clinical data by Donohue and colleagues provides the most comprehensive report of indacaterol safety to date, including nearly 5000 patients treated with indacaterol (75, 150, 300, or 600 µg) in studies of 3–12 months in duration [Donohue et al. 2011]. As in individual studies, the most common adverse events with indacaterol were COPD worsening, nasopharyngitis, and headache; most cases were mild or moderate and incidence was generally similar to placebo and other active treatments. The risk of COPD worsening as an adverse event was significantly reduced with indacaterol compared with placebo, and the incidence of COPD worsening as a serious adverse event, including events leading to hospitalization, was numerically lower in all indacaterol treatment groups compared with placebo. There were very few deaths during indacaterol treatment, and the relative risk of all-cause death was reduced with indacaterol (all doses combined) relative to placebo (Figure 6).

Risk of death relative to placebo, adjusted for length of time on treatment. Results from pooled analysis of safety data. Tiotropium was administered open label in 35% of this treatment group [Donohue et al. 2011]. CI, confidence interval.
The cardiovascular safety of indacaterol has been examined, including the type of analysis of major cardiovascular adverse events (MACE) advocated by the FDA as the preferred method to analyze potential cardiovascular and cerebrovascular risks for new therapies [FDA, 2008]. This involves a broad search for terms relating to the ‘hard’ outcomes of myocardial infarction, cerebrovascular events, and nervous system hemorrhages, and a more focused search for terms best matching myocardial infarction and stroke. In the pooled analysis of Donohue and colleagues, the MACE analysis showed that hazard ratios versus placebo were nonsignificantly reduced (<1) for all indacaterol doses, demonstrating no association with indacaterol treatment for either the broad or more focused definition of cardiovascular events [Donohue et al. 2011]. Worth and colleagues reported specifically on the cardio- and cerebrovascular safety of indacaterol treatment (150, 300 and 600 µg) in a pooled analysis of a subset of these patients, and their results also showed no potential for increased risks in these areas with indacaterol [Worth et al. 2011].
The effects of indacaterol on blood glucose, plasma potassium, electrocardiogram changes (QTc interval), and vital signs have been routinely monitored in clinical studies and evaluated in analyses of pooled data. Indacaterol, over a range of doses, showed little or no effect on these safety variables and any reported changes were clinically insignificant [Kerwin et al. 2011a; Worth et al. 2011; Donohue et al. 2012]. Importantly, Donohue and colleagues found no dose relationship with indacaterol (75–600 µg) for any of the categories of adverse events in their report, with only minor exceptions for vital signs (judged clinically insignificant) and for cough following inhalation. In clinical trials, patients experienced a transient cough after inhalation of indacaterol at a mean of 14–18% of attended visits, increasing in line with indacaterol dose. This cough usually occurred within 15 s following inhalation and lasted for no more than 15 s [Donohue et al. 2011]. The cough following inhalation was not associated with bronchospasm, exacerbations, deteriorations of disease or loss of efficacy, and therefore need not be considered to be a safety concern [Donohue et al. 2011]. Postmarketing experience with a cumulative patient exposure of approximately 296,995 patient years across indacaterol doses has been consistent with the overall clinical trial safety database (Novartis, unpublished data).
Precautions and contraindications
Indacaterol is a long-acting β2-adrenergic agonist and may cause stimulation of the adrenergic nervous system (side effects such as tremor, heart racing, nervousness, and headache can be seen with β2-agonist inhalers).
For patients with GOLD stage I or II airflow limitation who have few symptoms and a low risk of COPD exacerbations (group A patients, according to the categories in the current GOLD strategy), the recommended first choice of treatment is a short-acting bronchodilator for use on an as-needed basis [GOLD, 2011]. Treatment with a combination of short-acting bronchodilators (β2 agonist plus antimuscarinic), or a long-acting bronchodilator, would be a second choice in these group A patients [GOLD, 2011].
Summary and conclusions
Indacaterol 75 µg is an effective bronchodilator providing clinically relevant improvements in FEV1 on once daily dosing over a 12-week treatment period. Studies with higher doses have shown that bronchodilator effectiveness is maintained during up to 1 year of treatment. Studies have shown that indacaterol 75 µg provides important benefits in terms of improving symptoms and health status in patients with COPD, with significantly more patients achieving the MCID in these two measures.
Further data are required to evaluate the effect of indacaterol 75 µg on hyperinflation (or ‘air trapping’), one of the hallmarks of COPD, as well as its ability to increase exercise duration and the activity level of patients with COPD. Favorable effects on resting and dynamic hyperinflation and exercise capacity have been reported at higher indacaterol doses. Indirect comparisons with placebo and active treatments obtained from analyses of pooled data suggest that indacaterol 75 µg may be effective in reducing COPD exacerbations; longer-term prospective studies are required to confirm this finding.
Indacaterol 75 µg has a good overall safety and tolerability profile. Although longer-term data (>12 weeks) with this dose are lacking, other studies using up to eightfold higher doses for up to 1 year have also reported a good profile of safety and tolerability. Based on its efficacy and safety, indacaterol has now been licensed in all major markets as a long-term, once-daily maintenance bronchodilator treatment for airflow obstruction in patients with COPD.
The efficacy, safety, and tolerability data reviewed here suggest that in clinical use the therapeutic index of indacaterol 75 µg is likely to be high and appropriate for long-term use treating patients with moderate to severe COPD. Physicians may initiate treatment with indacaterol 75 µg once daily as an inhaled bronchodilator with a well characterized safety profile (doses up to 600 µg studied in clinical trials) that provides a significant improvement in lung function, reduction in rescue medication, and improvements in patient-reported outcomes of dyspnea and health status.
Footnotes
Acknowledgements
The authors would like to thank Sarah Filcek (CircleScience) for editorial assistance, including writing assistance, styling and submission, with this manuscript. With direction from the authors, Sarah Filcek drafted the manuscript that was edited significantly for intellectual content and scientific accuracy by the authors. The final draft submitted to the journal was approved by the authors.
Funding
Funding for editorial assistance was provided by Novartis Pharmaceuticals Corporation. The indacaterol research described in the manuscript was funded by Novartis.
Conflict of interest statement
Dr Kerwin has served on advisory boards or speaker panels for AstraZeneca, Forest, Ironwood, Mylan, Pearl, Pfizer, Sanofi Aventis, Sunovion, and Targacept. He has received travel reimbursement from Merck, Forest, and Novartis. He has conducted pharmaceutical-sponsored multicenter clinical research trials for approximately 70 pharmaceutical companies. Dr Williams is an employee of Novartis.
