Abstract
Indacaterol is a novel, inhaled, ultra-long-acting β2-agonist providing 24-h bronchodilation with once-daily (od) dosing for maintenance use in patients with chronic obstructive pulmonary disease (COPD). This article reviews the bronchodilator properties of indacaterol compared with other treatments used in COPD. Data from five published placebo-controlled studies were reviewed. Two 14-day crossover studies, the first comparing indacaterol 300 µg od with salmeterol 50 µg twice daily (bid), the second comparing indacaterol 150 µg and 300 µg od with tiotropium 18 µg od, assessed forced expiratory volume in 1 s (FEV1) at 24 h postdose (trough). Third, a 14-day crossover study evaluated trough FEV1 following indacaterol 300 µg dosed morning or evening compared with salmeterol 50 µg bid. Fourth, a single-dose study of indacaterol 150 and 300 µg measured FEV1 at 5 min postdose compared with salmeterol/fluticasone 50/500 µg and salbutamol 200 µg. Finally, data from a 1-year study with indacaterol 300 µg and formoterol 12 µg bid were examined to determine whether bronchodilation was maintained long term. In the first two studies, indacaterol increased trough FEV1 after 14 days by a statistically significant and clinically relevant margin over placebo; indacaterol had a greater effect than salmeterol and a similar effect to tiotropium. In the third study, indacaterol had the same effect on trough FEV1 whether dosed in the morning or evening. In the fourth study, the onset of the bronchodilator effect of indacaterol was similar to that of salbutamol. In the fifth study, the bronchodilator effect of indacaterol on trough FEV1 was maintained at a significant and clinically relevant level over 52 weeks, whereas the bronchodilator effect of formoterol diminished over time. To conclude, indacaterol is a highly effective bronchodilator that is superior to or at least as effective as other available long-acting bronchodilators for COPD.
Keywords
Introduction
Treatment guidelines for chronic obstructive pulmonary disease (COPD) describe bronchodilator medications as central to the symptomatic management of COPD [GOLD, 2010]. For patients with moderate to very severe COPD, regular treatment with an inhaled long-acting bronchodilator is recommended; this is more effective and convenient than treatment with short-acting bronchodilators [GOLD, 2010]. The first inhaled long-acting β2-agonists (LABAs), formoterol and salmeterol, induce bronchodilation that lasts for approximately 12 h and are therefore administered twice daily (bid) [Dahl et al. 2001; Boyd et al. 1997]. Tiotropium, the only currently available long-acting anticholinergic bronchodilator, has a duration of action of approximately 24 h and is administered once daily (od) [Donohue et al. 2002; Casaburi et al. 2000].
Indacaterol is a novel, inhaled LABA that is approved in Europe for the maintenance treatment of COPD at doses of 150 µg and 300 µg od. Clinical studies of up to 1 year’s duration have shown that indacaterol provides 24-h bronchodilation with once-daily dosing, and has good overall safety and tolerability [Kornmann et al. 2011; Dahl et al. 2010; Donohue et al. 2010; Feldman et al. 2010]. In addition to these pivotal clinical studies, profiling studies have specifically examined the bronchodilator effect of indacaterol and have included comparisons with other available long-acting bronchodilators, namely tiotropium, salmeterol and formoterol. The objective of the present article is to provide an overview of these profiling studies and to assess how the different aspects of the bronchodilator effect of this novel agent compare with existing drugs, including trough (or predose) forced expiratory volume in 1 s (FEV1), detailed 24-h profiling of the effects on FEV1 and inspiratory capacity (IC), the effects of morning versus evening dosing, and the speed of onset of the bronchodilator effect. The article also evaluates whether tolerance to the bronchodilator effect of indacaterol develops during long-term use. The individual studies are the subject of separate publications [LaForce et al. 2011; Balint et al. 2010; Dahl et al. 2010; Magnussen et al. 2010; Vogelmeier et al. 2010].
Studies reviewed
Summary of study designs for the studies included in this overview.

AM, morning; bid, twice daily; od, once daily; PM, evening.
Summary of demographics and baseline characteristics for the studies included in this overview.

COPD, chronic obstructive pulmonary disease; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity.
Data are for the safety populations of the studies. Data are mean (standard deviation) unless otherwise stated.
Measured at 15 or within 30 min after inhalation of salbutamol.
Patients with at least a 12% and 200 ml improvement in forced expiratory volume in 1s (FEV1) from baseline to 5 min after single doses of active treatment or placebo [Balint et al. 2010].

CI, confidence interval; n, number of patients; NA, not applicable.
p < 0.05; **p ≤ 0.01; ***p ≤ 0.001.
Odds ratios for likelihood of achieving this response versus the comparators listed below.
Study treatments were indacaterol 150 and 300 µg od, tiotropium 18 µg od, salmeterol 50 µg bid, salbutamol 200 µg (single dose), salmeterol/fluticasone 50/500 µg (single dose), and formoterol 12 µg bid. Indacaterol was administered via a single-dose dry powder inhaler (DPI; known in Europe as Onbrez® Breezhaler® [Novartis]); the other study drugs were delivered via their respective proprietary DPIs. Patients who were receiving inhaled corticosteroids before recruitment to the studies continued this treatment at a stable dose and regimen for the duration of the study. Fixed-dose inhaled corticosteroid/LABA combinations were replaced with inhaled corticosteroid monotherapy at equivalent dose and regimen. Salbutamol use as rescue medication was permitted in each study.
In four of the studies [LaForce et al. 2011; Dahl et al. 2010; Magnussen et al. 2010; Vogelmeier et al. 2010], the primary objective was to assess the superiority of indacaterol over placebo in terms of trough FEV1 (defined as the mean of FEV1 measurements at 23 h 10 min and 23 h 45 min postdose) after 14 days [LaForce et al. 2011; Magnussen et al. 2010; Vogelmeier et al. 2010] or 12 weeks [Dahl et al. 2010] of treatment. A difference in FEV1 between indacaterol and placebo of 120 ml was prespecified as clinically important. In the single-dose study, the main objective was to assess the superiority of indacaterol over placebo in terms of FEV1 at 5 min postdose [Balint et al. 2010].
The efficacy outcomes relevant to this overview were as follows.
In the first two studies, comparisons between indacaterol and tiotropium and between indacaterol and salmeterol in their effect on trough FEV1 after 14 days of treatment (steady state). These studies also compared the effect of these treatments on FEV1 measured serially at intervals postdose over 24 h, after 14 days of treatment. The second study also compared indacaterol and salmeterol in their effect on resting IC measured serially at intervals postdose over 24 h, after 14 days of treatment. In the third study, comparisons between the effect on trough FEV1 of indacaterol dosed morning (AM) or evening (PM), together with the comparison between indacaterol and salmeterol, after 14 days of treatment. In the fourth study, comparison between indacaterol and the fixed-dose combination of salmeterol and fluticasone in their effect on FEV1 at 5 min postdose, with the standard rescue medication salbutamol, a short-acting inhaled β2-agonist, as reference. In the fifth study, comparison between the long-term bronchodilator effects of indacaterol and formoterol.
The safety of indacaterol was evaluated in all five studies in terms of adverse events, laboratory tests, vital signs and electrocardiograms.
Efficacy variables were analysed using mixed-model analysis of covariance (ANCOVA), including terms for treatment, period, patient and period baseline FEV1 for the crossover studies (daytime and treatment-by-daytime interaction were included in the model used for the AM versus PM dosing study). Trough FEV1 in the 52-week study was analysed by mixed-model ANCOVA with treatment as a fixed effect and baseline FEV1 and FEV1 reversibility (assessed at screening) as covariates.
Results of studies
Patient demographic and clinical characteristics are summarized in Table 2. Baseline FEV1 and FEV1 reversibility to salbutamol were generally similar across the different studies.
Effects at steady state
Trough FEV1
Figure 1 presents the differences in trough FEV1 between active treatments and placebo after 14 days of treatment in the first two studies. In both studies, there were statistically significant and clinically relevant differences (150–200 ml) between indacaterol and placebo, thus fulfilling the primary objective of the studies [LaForce et al. 2011; Vogelmeier et al. 2010].
Differences in trough forced expiratory volume in 1 s (FEV1) between active treatments and placebo after 14 days of treatment with (a) indacaterol 150 µg, 300 µg, tiotropium 18 µg and placebo once daily (od), and (b) indacaterol 300 µg od, salmeterol 50 µg twice daily and placebo (data are least squares means and (a) 97.5% confidence intervals [CI] or (b) 95% CI) [LaForce et al. 2011; Vogelmeier et al. 2010]. ***p ≤ 0.001 for active treatment versus placebo;†p = 0.011 for indacaterol versus salmeterol. Broken line shows prespecified 120 ml level of clinically relevant difference versus placebo.
In terms of differences between active treatments, trough FEV1 on Day 14 was numerically greater with indacaterol 150 and 300 µg od than with tiotropium 18 µg od (by 50 and 30 ml, respectively) in the first study (Figure 1) [Vogelmeier et al. 2010]. The p-value of 0.043 for the comparison between indacaterol 150 µg and tiotropium did not reach the level of significance required to demonstrate superiority (which was p = 0.025, in order to allow for multiplicity [Vogelmeier et al. 2010]. In the second study, indacaterol 300 µg od had a greater effect than salmeterol bid on trough FEV1 on Day 14 by a significant margin of 90 ml (Figure 1) [LaForce et al. 2011].
In terms of changes from baseline, the mean change from baseline in trough FEV1 on Day 14 was 160 ml (11.9%) with indacaterol 150 µg, 140 ml (10.2%) with indacaterol 300 µg and 120 ml (8.6%) with tiotropium (placebo: −10 ml, −0.7%) in the first study, and 180 ml (14.1%) with indacaterol 300 µg and 80 ml (6.3%) with salmeterol (placebo: −30 ml, −2.3%) in the second study.
FEV1 at intervals over 24 h postdose
Figure 2 shows serial measurements of FEV1 over 24 h postdose after 14 days of treatment in the first two studies [LaForce et al. 2011; Vogelmeier et al. 2010]. In both studies, FEV1 was statistically significantly greater with indacaterol than with placebo at all time points postdose. Compared with tiotropium (Figure 2a), FEV1 was similar or higher with both indacaterol doses at all time points. Compared with salmeterol (Figure 2b), FEV1 with indacaterol was numerically superior at all time points and statistically significantly greater during the first 4 h following dosing and towards the end of salmeterol’s 12-h duration.
Serial forced expiratory volume in 1 s (FEV1) measured at intervals postdose after 14 days of treatment with (a) indacaterol 150 µg once daily (od), indacaterol 300 µg od, tiotropium 18 µg od and placebo and (b) indacaterol 300 µg od, salmeterol 50 µg twice daily and placebo (data are least squares means ± standard error) [LaForce et al. 2011; Vogelmeier et al. 2010]. Significant differences were observed at each time point between active and placebo treatments (p < 0.001 for indacaterol and tiotropium versus placebo; p < 0.01 for salmeterol versus placebo). *p < 0.05 for indacaterol versus tiotropium and for indacaterol versus salmeterol at time points indicated.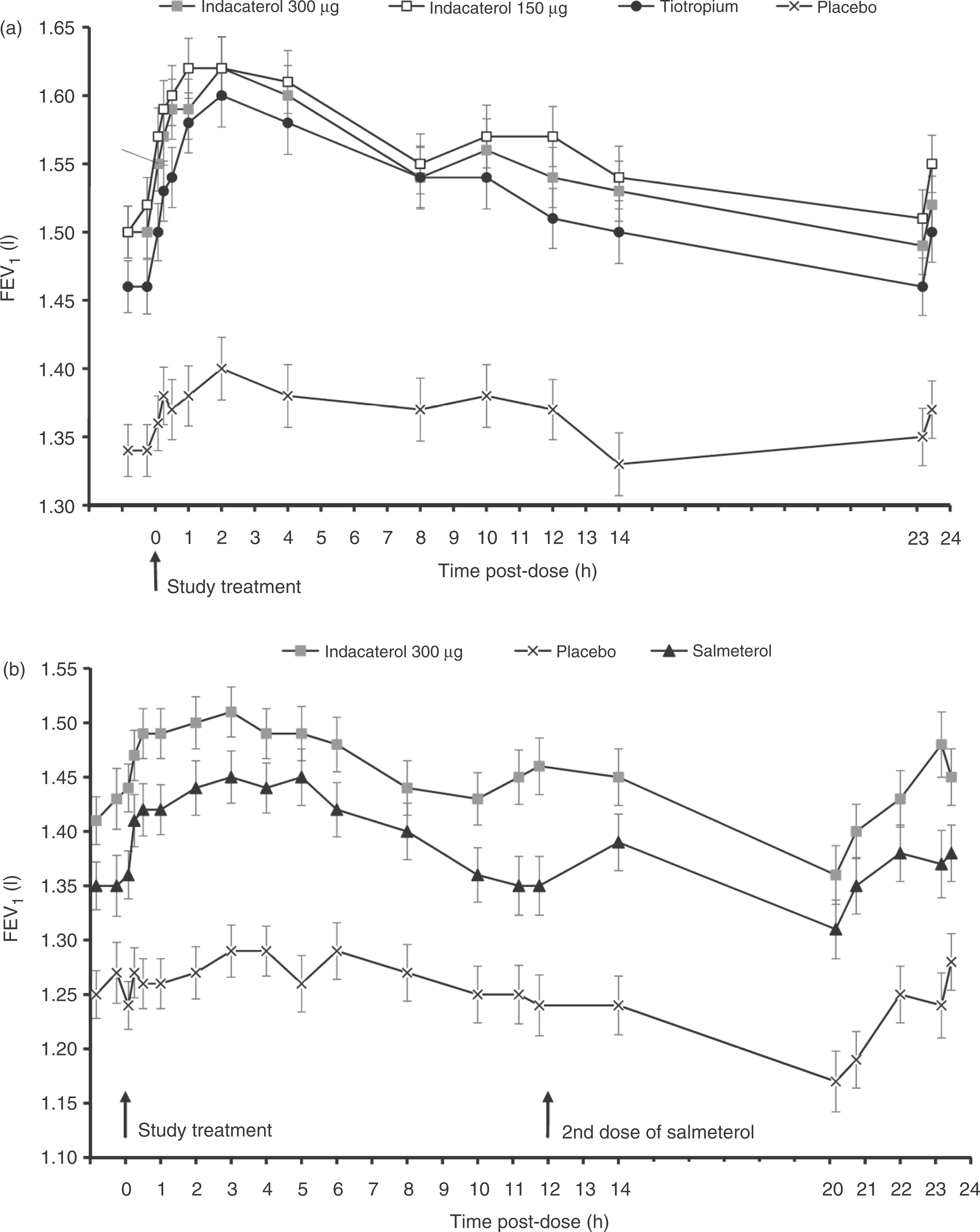
Inspiratory capacity
The effects of indacaterol and salmeterol on resting IC over 24 h following dosing on Day 14 in the second study [LaForce et al. 2011] are shown in Figure 3. At all time points postdose, indacaterol 300 µg od was statistically significantly superior to placebo, and numerically or statistically significantly superior to salmeterol 50 µg bid.
Inspiratory capacity (IC) measured after 14 days of treatment with indacaterol 300 µg once daily (od), salmeterol 50 µg twice daily (bid) and placebo (data are least squares means ± standard error) [LaForce et al. 2011]. Indacaterol was statistically significantly superior to placebo at each time point; salmeterol was statistically significantly superior to placebo except at 50 min predose and 3 h, 8 h, 10 h, 11 h 10 min and 23 h 45 min postdose; *indacaterol was statistically significantly superior to salmeterol at time points indicated. Data from the 3-h postdose time point are excluded from the figure as one patient on indacaterol reported an out-of-range value [12.37 l].
Effect of morning and evening dosing
In the third study [Magnussen et al. 2010], indacaterol increased trough FEV1 by 200 ml relative to placebo whether dosed in the morning or evening (Figure 4). The evening measurement of trough FEV1 (24 h after indacaterol dosed PM and 12 h after salmeterol dosed AM) was higher with indacaterol than with salmeterol (treatment difference of 110 ml, p < 0.001). The morning measurement of trough FEV1 (24 h after indacaterol dosed AM and 12 h after salmeterol dosed PM) was numerically higher with indacaterol versus salmeterol (treatment difference of 50 ml, p = NS).
Differences in trough forced expiratory volume in 1 s (FEV1) between active treatments and placebo after 14 days of treatment with indacaterol 300 µg once daily (dosed morning or evening) and salmeterol 50 µg twice daily (data are least squares means with 95% confidence intervals) [Magnussen et al. 2010]. **p < 0.01, ***p < 0.001 for active treatment versus placebo. Trough values were measured 24 h after the previous dose of indacaterol and 12 h after the previous dose of salmeterol.
First-dose effects
Onset of action (bronchodilation at 5 min postdose)
In the fourth study [Balint et al. 2010], FEV1 at 5 min postdose was statistically significantly higher with both doses of indacaterol than with placebo, with treatment differences of 100–120 ml (both p < 0.001). Both doses of indacaterol had a greater effect than salmeterol/fluticasone, with treatment differences of 50–70 ml (both p < 0.01), and a similar effect to salbutamol (differences of 10–30 ml; p = NS). The effects on FEV1 at 5 min postdose expressed as change from baseline are displayed in Figure 5. Patients taking indacaterol were significantly more likely to achieve improvements in FEV1 from baseline of 12% and 200 ml at 5 min postdose compared with placebo and with salmeterol/fluticasone, with no significant differences compared with salbutamol (Table 3).
Change from baseline (%) in forced expiratory volume in 1 s (FEV1) at 5 min after single doses of indacaterol 150 µg, indacaterol 300 µg, salmeterol/fluticasone 50/500 µg, salbutamol 200 µg and placebo (data are least squares means ± standard errors) [Balint et al. 2010]. ***p < 0.001 for active treatment versus placebo;††p < 0.01,†††p < 0.001 for indacaterol or salbutamol versus salmeterol/fluticasone. Incidence (% of patients) of adverse events overall and the most commonly occurring with indacaterol treatment: pooled data from 6 months’ treatment with indacaterol, formoterol, salmeterol, tiotropium or placebo [Dahl et al. 2010; Donohue et al. 2010; Kornmann et al. 2010]. COPD, chronic obstructive pulmonary disease; IND 150, indacaterol 150 µg od; IND 300, indacaterol 300 µg od; IND 600, indacaterol 600 µg od; FOR, formoterol 12 µg bid; PBO, placebo; SLM, salmeterol 50 µg bid; TIO, tiotropium 50 µg od; URTI, upper respiratory tract infection.

The bronchodilator effect of indacaterol was also measured at 5 min postdose in the first two studies. In the blinded comparison with tiotropium, FEV1 at 5 min postdose on Day 1 was 120–130 ml higher following indacaterol (150 and 300 µg) than after placebo (both p < 0.001), and 80 ml higher with both indacaterol doses than with tiotropium (both p < 0.001) [Vogelmeier et al. 2010]. In the second study, FEV1 at 5 min postdose on Day 1 was 110 ml higher with indacaterol 300 µg than with placebo, and 50 ml higher than with salmeterol (both p < 0.001) [LaForce et al. 2011].
Efficacy during long-term use
The fifth study, the 1-year comparison of indacaterol 300 µg od and formoterol 12 µg bid, offered the opportunity to assess whether the bronchodilator effect was maintained over the long term [Dahl et al. 2010]. As can be seen in Figure 6, the effect of indacaterol on trough FEV1 relative to placebo increased slightly from the first dose to Week 12, and remained at a similar level at Week 52. With formoterol 12 µg bid, however, the greatest effect was observed at Day 1, with a decline of some 40% in effect by Week 12, and a further 30% decline from Week 12 to Week 52.
Differences between active [indacaterol 300 µg once daily (od) and formoterol 12 µg twice daily (bid)] and placebo treatments in trough forced expiratory volume in 1 s (ml) after first dose (Day 1) and after 12 and 52 weeks of treatment (data are least squares means ± standard error) [Dahl et al. 2010]. *p < 0.05, ***p < 0.001 versus placebo; †p < 0.05, †††p < 0.001 versus formoterol. Patient numbers for indacaterol 300 µg od, formoterol 12 µg bid and placebo were, respectively, 391, 382 and 384 at Day 1, 389, 379 and 371 at Week 12, and 320, 300 and 279 at Week 52.
Safety and tolerability of indacaterol
Overall, indacaterol was well tolerated in the five studies summarized in this overview, and the results are reported separately [LaForce et al. 2011; Balint et al. 2010; Dahl et al. 2010; Magnussen et al. 2010; Vogelmeier et al. 2010]. The safety profile of a new treatment is better judged from long-term studies. These have shown that the overall rates of adverse events were generally comparable across patient groups treated with indacaterol, placebo or other long-acting bronchodilators, and most events with indacaterol were mild or moderate in severity [Kornmann et al. 2011; Dahl et al. 2010; Donohue et al. 2010]. Table 4 summarizes the overall incidence of adverse events and those most commonly occurring, based on data from these studies [Kornmann et al. 2011; Dahl et al. 2010; Donohue et al. 2010]. The incidence of class-related side effects of inhaled β2-agonists (e.g. hypokalaemia, hyperglycaemia or prolonged QTc interval) was low or comparable with placebo [Kornmann et al. 2011; Dahl et al. 2010; Donohue et al. 2010].
Discussion
At steady state (Day 14), indacaterol was consistently associated with clinically and statistically significant improvements in trough FEV1 compared with placebo [LaForce et al. 2011; Magnussen et al. 2010; Vogelmeier et al. 2010]. The differences of 150–200 ml appreciably exceeded the prespecified minimal clinically important difference (MCID) of 120 ml, which is the midpoint of the 100–140 ml MCID range proposed by an American Thoracic Society/European Respiratory Society task force [Cazzola et al. 2008]. The bronchodilator efficacy of indacaterol remained undiminished during long-term use. These results confirm the suitability of indacaterol for maintenance use with a once-daily dosing regimen. Improved bronchodilation in the morning is likely to be of particular benefit to COPD patients, this being a time of day when many report symptoms to be at their worst and find it difficult to commence or undertake their daily activities [Partridge et al. 2009]). In addition, in comparison with treatments that are dosed more frequently, the simplified regimen of once-daily dosing is likely to aid adherence and long-term persistence with prescribed medication [Bourbeau and Bartlett, 2008; Breekveldt-Postma et al. 2007].
Indacaterol is licensed for use in COPD in many countries of the world at once-daily doses of 150 and 300 µg. Initial dose-finding research showed that the 150 µg dose was the lowest indacaterol dose with optimal bronchodilator efficacy [Barnes et al. 2010]. Although the two doses were not significantly different in their bronchodilator effect, as reflected in the studies reviewed here, both the 150 µg dose and the next highest dose of 300 µg were taken forward for further investigation of longer term and clinical effects [Donohue et al. 2010]. The 300 µg dose appears to have advantages in terms of added efficacy in controlling dyspnoea [Jones et al. 2011], and the option of a higher indacaterol dose may be useful in clinical practice.
Trough FEV1 was significantly greater with once-daily indacaterol than with both the active comparators. The additional trough FEV1 improvement of 30–50 ml seen here with indacaterol over tiotropium [Vogelmeier et al. 2010] suggests that indacaterol is at least as effective a bronchodilator as tiotropium. The additional improvement versus tiotropium is consistent with the findings of a previous larger study with tiotropium (given open label), in which a statistically significant difference of 50 ml in trough FEV1 between indacaterol and tiotropium was observed after 12 weeks of treatment [Donohue et al. 2010]. Despite the open-label administration of tiotropium in that study, the tiotropium–placebo differences were consistent with previous blinded comparisons [Casaburi et al. 2002; Donohue et al. 2002; Casaburi et al. 2000]. More recently, a preliminary report of a blinded comparison of indacaterol and tiotropium in approximately 1600 patients showed similar effects on trough FEV1 after 12 weeks but a greater effect of indacaterol on symptoms and health status [Dunn et al. 2010].
The treatment difference of 90 ml in trough FEV1 between indacaterol and salmeterol was statistically significant, and exceeded the 52 ml superiority previously demonstrated for tiotropium compared with salmeterol [Donohue et al. 2002]. Improvements in trough FEV1 with indacaterol compared with salmeterol and formoterol have also been demonstrated after treatment periods of 3–12 months [Korn et al. 2011; Kornmann et al. 2011; Dahl et al. 2010]. Indeed, the statistically significant and clinically important differences that remained evident during 1 year’s treatment with indacaterol contrasted with the declining bronchodilator effect of twice-daily formoterol [Dahl et al. 2010]. There are previous reports of some loss of bronchodilator effect of formoterol over 1 year of treatment [Calverley et al. 2003a; Szafranski et al. 2003], although other 1-year studies reported that efficacy was maintained [Rennard et al. 2009; Rossi et al. 2002].
The studies evaluating serial postdose spirometry showed that once-daily dosing of indacaterol provided significant increases in FEV1 over placebo at all time points during the 24-h postdose period [LaForce et al. 2011; Vogelmeier et al. 2010]. Treatment differences well above the MCID were observed as early as 5 min following the initial dose [Balint et al. 2010], when patients are likely to perceive an immediate benefit. The bronchodilator effect of indacaterol at this time was greater than that observed following tiotropium and salmeterol. Compared with salmeterol, the treatment differences between indacaterol and salmeterol were greatest towards the end of salmeterol’s 12-h duration of effect. This more consistent bronchodilator efficacy could in turn provide improved symptom control over the course of 24 h. Other studies in which serial spirometry was conducted after longer treatment periods (12 weeks), although at generally fewer postdose time points than in the studies reviewed here, show broadly similar results, with indacaterol superior to placebo and formoterol or salmeterol at all time points and at least as effective as tiotropium [Korn et al. 2011; Dahl et al. 2010; Donohue et al. 2010].
Indacaterol has been routinely dosed in the morning in clinical studies in COPD. Dosing in the evening also appears to be effective in providing full 24-h bronchodilation, with the same effect on trough FEV1 as that occurring with morning dosing. Thus, patients will receive the same full 24-h bronchodilation whether they prefer (or need) to take their once-daily dose of indacaterol regularly in the morning or in the evening. The bronchodilator efficacy of the once-daily anticholinergic tiotropium has also been reported to be similar whether dosed AM or PM [Calverley et al. 2003b].
The measurement of FEV1 is important for the diagnosis and severity staging of COPD, and is required by health authorities as a measurement of response for new therapies seeking registration. However, other spirometric measures such as IC may correlate better with measures of clinical response such as exertional dyspnoea and exercise capacity [Cooper 2006; O’Donnell et al. 1999]. Resting IC measures the inspiratory expansion in volume following exhalation during tidal breathing, and therefore indirectly infers functional residual capacity. This is reduced in patients with COPD as a result of air trapping, resulting in the syndrome of hyperinflation. The effect of indacaterol on the resting IC profile shown here [LaForce et al. 2011] was statistically superior to placebo at all time points; by contrast, salmeterol lost significance versus placebo at numerous time points. Resting IC has been shown to correlate with dynamic hyperinflation (i.e. hyperinflation occurring during exercise) and with dyspnoea on exercise in COPD [O’Donnell et al. 2001; Belman et al. 1996]. The hypothesis that indacaterol may have favourable effects on dynamic hyperinflation and exercise capacity was confirmed in a recent report from a placebo-controlled crossover study in which indacaterol 300 µg statistically significantly improved IC, both at rest and following exercise, along with exercise endurance time after 3 weeks of treatment [O’Donnell et al. 2011].
Conclusions
The studies summarized in this overview present a comprehensive profile of the bronchodilator effects of indacaterol, showing a full 24-h bronchodilator effect and demonstrating its suitability for once-daily dosing. Bronchodilation was maintained above the clinically important level of difference compared with placebo during the 24-h postdose period and with repeated dosing. Similar bronchodilator effects were achieved when indacaterol was taken regularly in the morning or in the evening, and the onset of action was fast. In addition, the effect of indacaterol on IC suggests that the effective and sustained bronchodilation may translate into benefits for patient outcomes (such as exertional dyspnoea and exercise capacity). Indacaterol was at least as effective as tiotropium and superior to salmeterol in its bronchodilator effect. By contrast with the twice-daily LABA formoterol, the bronchodilator effect of indacaterol was maintained long term. Indacaterol should be of value for the maintenance treatment of patients with COPD as a highly effective and well-tolerated once-daily bronchodilator.
Footnotes
Conflict of interest statement
Novartis provided financial support for the conduct of this research, and was involved in designing the studies, collecting, analysing and interpreting the study data, writing the study reports, and the decision to submit this paper for publication.
CV has received fees for presentations at symposia sponsored by (in alphabetical order) AstraZeneca, Boehringer, Chiesi, GlaxoSmithKline, Janssen-Cilag, Novartis, Nycomed, Pfizer and Talecris, and for consulting from (in alphabetical order) AstraZeneca, Boehringer, GlaxoSmithKline, Janssen-Cilag, Novartis, Nycomed and Talecris.
HM has received fees for membership of advisory boards of various pharmaceutical companies (Altana, Boehringer Ingelheim, AstraZeneca) and for speaking (Boehringer Ingelheim, AstraZeneca, Chiesi, Altana).
CLF has received fees for speaking from Merck and Novartis, and receives funds for clinical research activities from GlaxoSmithKline, Novartis, Boehringer Ingelheim, Sepracor, Alcon, Medimmune and Forest.
RO and BK are employees of Novartis.
Acknowledgements
The authors thank the patients and staff at the centres participating in the studies. Sarah Filcek of ACUMED (a professional medical writer funded by Novartis) and David Young (Novartis) assisted in the preparation of the manuscript.
