Abstract
Chronic obstructive pulmonary disease (COPD) has a 3-year mortality rate up to 37%, 2–6 times higher than the general population. We present evidence supporting pharmacological therapies to improve patient life expectancy, focusing on inhaled corticosteroids (ICSs) combined with long-acting bronchodilators (LABDs). A reduction in 3-year all-cause mortality (ACM) has been shown in patients with severe COPD treated with fluticasone propionate (an ICS) and salmeterol [long-acting beta-agonist (LABA)], compared with placebo. An observational study of elderly patients with severe COPD and multiple comorbidities suggested ICS+LABD reduce ACM compared with LABD monotherapy. Patients with symptomatic COPD at risk of exacerbations saw a mortality benefit with the ICS/long-acting muscarinic antagonist (LAMA)/LABA combinations fluticasone furoate/umeclidinium/vilanterol (FF/UMEC/VI) or budesonide/glycopyrrolate/formoterol (BUD/GLY/FOR) versus UMEC/VI or GLY/FOR (LAMA/LABA combinations) in the IMPACT and ETHOS trials, respectively. Reduced risk of mortality may be due to modulation of airway inflammation, thereby reducing activation of proinflammatory mediators in the peripheral circulation. Importantly, estimated annual risk reduction for ACM with ICS/LAMA/LABA combinations in patients with COPD is of the same order of magnitude as for statins (patients with coronary disease) and angiotensin-converting enzyme inhibitors (patients with vascular disease). Based on the current data, the pharmacological treatment of COPD appears not only able to improve symptoms and reduce the frequency of exacerbations but is also very promising in improving patient prognosis in the long term.
Introduction
Life expectancy is a key indicator of wellness globally. Since 1900, the global average life expectancy has more than doubled and is now over 72 years. 1
Chronic diseases are probably the most important factor in reducing life expectancy in the developed world. In 2016, according to the Global Burden of Diseases, Injuries and Risk Factors Study group, non-communicable diseases were responsible for more than 800 million years of life lost (YLL), with chronic obstructive pulmonary disease (COPD) reported as one of the major contributors, being responsible for more than 47 million YLL. 2 Data from the Danish registry of COPD have shown the 3-year mortality rate for patients with COPD to be 10%, 23.8%, 17.4%, and 36.9% in those with group A, B, C, and D (Global Initiative on Obstructive Lung Disease 2017 classification) disease, respectively. 3 Comparing these rates with the 3-year mortality rate in an age-matched healthy population, 7.4% in men and 5.0% in women, 4 it is clear that patients with COPD have a mortality rate up to six times higher than the general population, depending on the severity of their disease. Notably, in recent decades, the rate of deaths attributable to COPD has not decreased as significantly as death rates for other non-communicable diseases for which highly successful interventions are available in high-income countries.5,6
To date, only two therapeutic interventions have been shown to increase the life expectancy of patients with COPD: long-term oxygen therapy (LTOT) and smoking cessation. The efficacy of LTOT was demonstrated many years ago in two small randomized trials in patients with severe respiratory failure due to COPD.7,8 More recently, attempts to demonstrate the efficacy of LTOT in patients with less severe COPD have been unsuccessful.9,10 Smoking cessation was shown to increase the life expectancy of patients with mild or moderate COPD in the Lung Health Study. 11 This finding identified smoking cessation as a key intervention in patients with COPD, as had already been demonstrated for many other chronic diseases and also for the general population. 12
Here, we discuss the evidence supporting the efficacy of pharmacological therapies in reducing the risk of death and in turn potentially increasing the life expectancy of patients with COPD, with a particular focus on landmark trials using inhaled corticosteroids (ICSs) in combination with long-acting bronchodilators (LABDs).
All-cause mortality as an outcome for COPD pharmacological trials
COPD is a complex disease. It has been known for more than 50 years that both the clinical and physiological characteristics vary among patients with COPD, suggesting the presence of more than one type of disease under the same broad definition. 13 In fact, COPD is much more complex than that. Major contributors to this complexity include the variability of symptoms and the rate at which health-related quality of life (HRQoL) deteriorates, in addition to the presence of other diseases frequently associated with COPD, mainly in elderly patients. It is clear that forced expiratory volume in 1 s, a valuable objective measure of lung function deterioration, cannot fully explain the clinical severity of the disease and the clinical impact of any pharmacological intervention. To overcome this problem, many patient-centered outcomes, for example, dyspnoea and HRQoL, or the frequency of exacerbations, an important determinant of disease prognosis in patients with COPD, have been used in pharmacological trials. 14
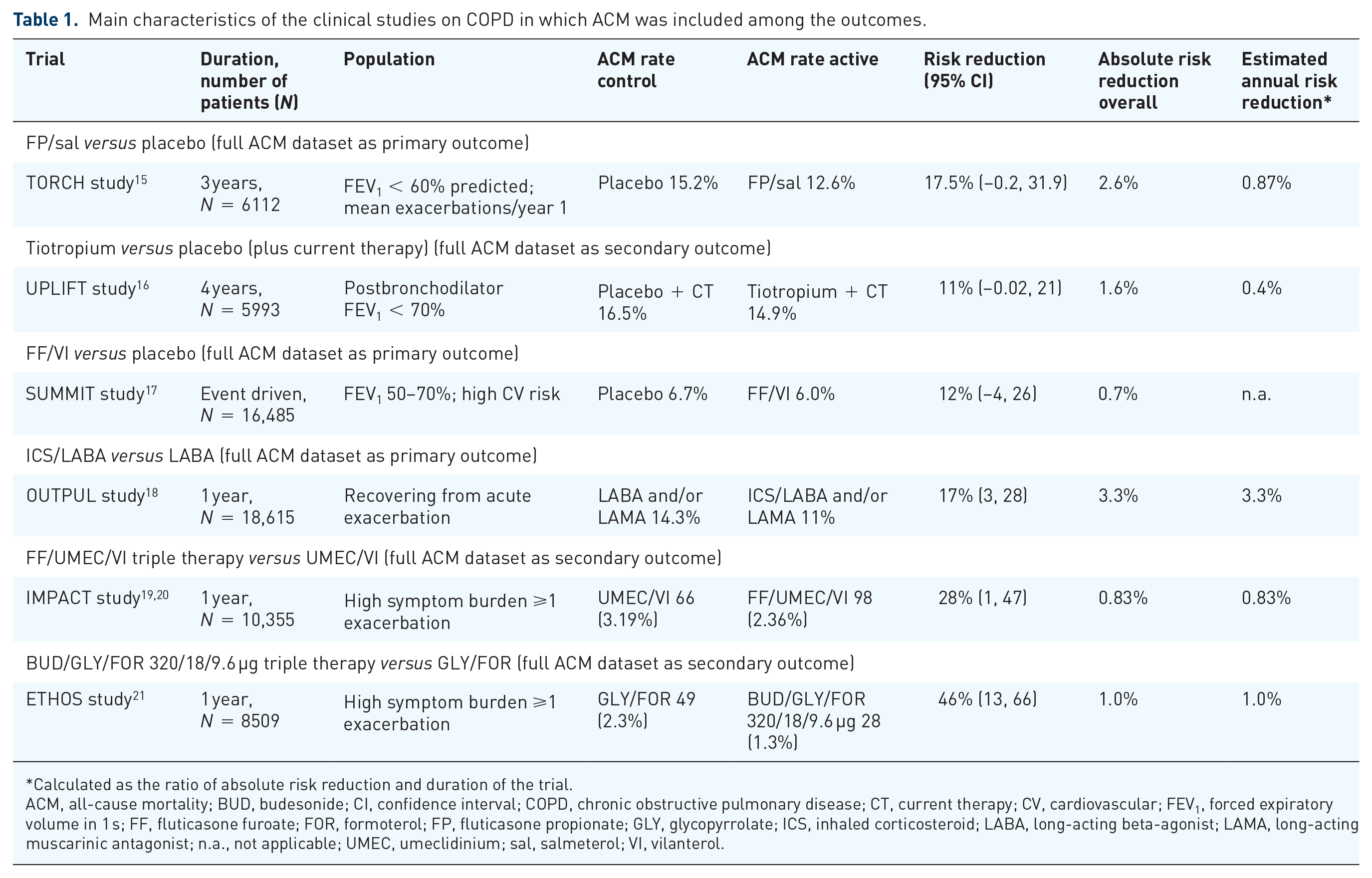
The rate of all-cause mortality (ACM) is a comprehensive prognostic indicator for any disease. Reducing the risk of death may be considered as the best measure of the balance between the advantages and disadvantages of a specific therapeutic intervention. As an outcome, ACM is relatively easy to collect; it is reliable and generally considered as the gold standard in defining the efficacy of a given therapy. Studies in which ACM was the primary or secondary prespecified outcome along with ACM being a prespecified ‘other’ endpoint in the IMPACT trial in patients with COPD are summarised in Table 1.
Main characteristics of the clinical studies on COPD in which ACM was included among the outcomes.
Calculated as the ratio of absolute risk reduction and duration of the trial.
ACM, all-cause mortality; BUD, budesonide; CI, confidence interval; COPD, chronic obstructive pulmonary disease; CT, current therapy; CV, cardiovascular; FEV1, forced expiratory volume in 1 s; FF, fluticasone furoate; FOR, formoterol; FP, fluticasone propionate; GLY, glycopyrrolate; ICS, inhaled corticosteroid; LABA, long-acting beta-agonist; LAMA, long-acting muscarinic antagonist; n.a., not applicable; UMEC, umeclidinium; sal, salmeterol; VI, vilanterol.
It is well established that LABDs improve symptoms and HRQoL and reduce the frequency of exacerbations in patients with COPD. However, their effect on mortality is less clear. The efficacy of the long-acting muscarinic antagonist (LAMA) tiotropium in reducing ACM was investigated as a secondary outcome in the randomized, double-blind UPLIFT trial. 16 The authors compared 4 years of therapy with either tiotropium or placebo in patients with COPD who were permitted to use all respiratory medications except inhaled anticholinergic drugs. The death rate reduction in patients randomized to tiotropium was statistically significant at the end of the protocol-defined treatment period [hazard ratio (HR) 0.84, 95% confidence interval (CI) 0.73, 0.97], but not 30 days thereafter (HR 0.89, 95% CI 0.79, 1.02). Overall, the results were interpreted as suggestive, but not conclusive, evidence of a mortality benefit with tiotropium in patients with COPD.
In a pooled analysis of seven randomized clinical trials (RCTs), in which ACM was assessed as a secondary outcome, 22 and in two observational studies,23,24 the risk of death was improved in patients with COPD treated with ICSs. Despite a number of important limitations, these studies formed the basis for the design of the first large clinical trial in COPD that had ACM as a primary outcome. The TORCH trial demonstrated a numerical reduction in the 3-year ACM rate in patients with severe COPD treated with the ICS fluticasone propionate (FP) and the long-acting beta-agonist (LABA) salmeterol versus placebo (HR 0.825, 95% CI 0.681, 1.002). 15 Unfortunately, the sample size obtained at the end of the study, after a safety call from an ‘interim analysis’, was insufficient to obtain a statistically significant result.
The SUMMIT trial, another study in which ACM was the primary endpoint, included patients with moderate COPD and history, or increased risk, of cardiovascular disease. 17 In our opinion, the low severity of COPD in the SUMMIT population, along with the higher risk of death from concomitant diseases, were key contributors to the lack of significance of the reduction in ACM with ICS/LABA treatment, in this case, fluticasone furoate (FF) and vilanterol (VI), versus placebo (HR 0.88, 95% CI 0.74, 1.04).
The observational OUTPUL study, performed in a cohort of elderly patients with severe COPD and multiple comorbidities, suggested that the addition of an ICS to a LABD reduces the risk of ACM versus LABD monotherapy (HR 0.83, 95% CI 0.72, 0.97). In this study, the effect of ICSs was even more efficient in a subanalysis of the frequent exacerbators included in the cohort (HR 0.63, 95% CI 0.44, 0.90). 18
ACM was included as a prespecified outcome in the recent IMPACT trial, in which patients with symptomatic COPD and at risk of exacerbations were randomly assigned in a 2:2:1 ratio to one of the following treatments: ICS/LABA/LAMA with FF/umeclidinium/VI (FF/UMEC/VI), ICS/LABA with FF/VI, or LABA/LAMA with UMEC/VI. 19 Including off-treatment data, the reduction in the risk of ACM was significantly reduced in patients treated with FF/UMEC/VI compared with UMEC/VI (HR 0.71, 95% CI 0.51, 0.99). On the contrary, the reduction in the risk of ACM with FF/UMEC/VI versus FF/VI (HR 0.90, 95% CI 0.67, 1.20) was not statistically significant. 20 However, caution was warranted in the interpretation of these results due to the amount of missing data. This has since been addressed in the collection of additional vital status follow up with 99.6% of patients having vital status recorded at week 52 of the study. These data also demonstrated a statistically significant reduction in the risk of ACM in patients treated with FF/UMEC/VI versus UMEC/VI (HR 0.72, 95% CI 0.53, 0.99). 20 A sensitivity analysis, using the tipping point method, 25 was used to assess the impact the 0.04% missing data at week 52 had on the results and confirmed the robustness of the results. 20
ACM was also included as a prespecified outcome in the ETHOS trial, which randomized patients with symptomatic COPD and a history of exacerbations in the previous year in a 1:1:1:1 ratio to ICS/LAMA/LABA with budesonide/glycopyrrolate/formoterol fumarate (BUD/GLY/FOR) at either a 320 µg or 160 µg dose of BUD, LAMA/LABA with GLY/FOR or ICS/LABA with BUD 320 µg/FOR. 21 Including off-treatment data, triple therapy with BUD 320 µg/GLY/FOR demonstrated a lower risk of death compared with both GLY/FOR (HR 0.54, 95% CI 0.34, 0.87) and BUD/FOR (HR 0.78, 95% CI 0.47, 1.30). 21 In contrast, the triple therapy with the lower ICS dose (BUD 160 µg/GLY/FOR) only demonstrated a lower risk of death compared with GLY/FOR (HR 0.79, 95% CI 0.52, 1.20), with a higher risk of death compared with BUD/FOR reported (HR 1.13, 95% CI 0.72, 1.80). 21 It should be noted that owing to the testing hierarchy employed in the ETHOS trial no inferences regarding the statistical significance of the ACM results could be made. 21
ACM reduction with ICSs in patients with COPD in comparison to ACM reduction with treatments for other chronic diseases
Although the two RCTs where ACM was the main outcome gave nonsignificant results for reasons we have outlined above, we believe that on the basis of the available evidence, the probability of reducing the risk of death of patients with symptomatic COPD at risk of exacerbations using ICS therapy added to LABDs is quite high. It is important to evaluate the public health relevance of the results obtained in the IMPACT trial in which, for the first time, a benefit on ACM has been prospectively demonstrated by pharmacological treatment.
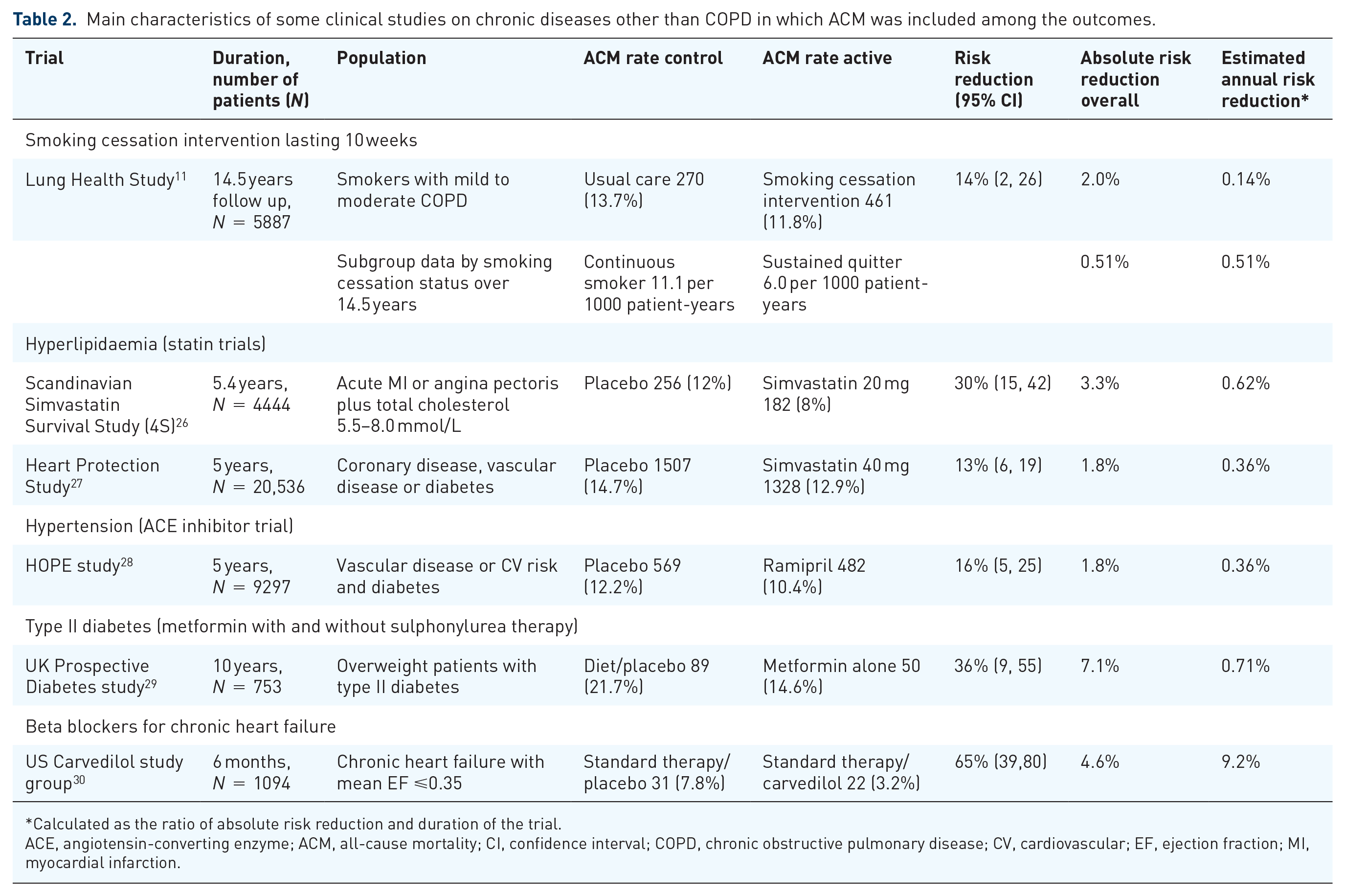
The estimated ACM annual risk reduction obtained by well-established therapies and interventions in other chronic noncommunicable diseases can set up a scenario to compare with the IMPACT results (Table 2). These include smoking cessation, the only evidence-based, life-saving therapeutic intervention suggested for all patients with COPD, as well as established life-saving therapies for hyperlipidemia, hypertension, diabetes, and chronic heart failure, all of which are noncommunicable diseases or disorders that contribute substantially to increasing the YLL burden.
Main characteristics of some clinical studies on chronic diseases other than COPD in which ACM was included among the outcomes.
Calculated as the ratio of absolute risk reduction and duration of the trial.
ACE, angiotensin-converting enzyme; ACM, all-cause mortality; CI, confidence interval; COPD, chronic obstructive pulmonary disease; CV, cardiovascular; EF, ejection fraction; MI, myocardial infarction.
Of course, our choice of therapies and diseases for which there is well-established evidence of ACM reduction is arbitrary. Those included should not be understood as an attempt to draw up a complete list of all such therapies that have similar characteristics. They should be used as a list of possible references against which to measure the importance for public health of the results obtained in the IMPACT study. Nonetheless, Table 2 highlights that the estimated annual risk reduction for ACM with FF/UMEC/VI in COPD is similar to statins in patients with coronary disease, angiotensin-converting enzyme inhibitors in patients with vascular disease and metformin in patients with diabetes who are overweight.
This comparison was based on the annualized absolute risk reduction (aARR), which can be derived from the ratio of the risk reduction measured at the end of each trial to the duration of the observation in years, under the assumption of a constant death rate. Of course, the translation of each aARR to number of deaths avoided each year is dependent on the number of patients who are eligible to be treated with the reference treatment used in each study. COPD has a very high prevalence globally, with no less than one-third of all patients referred to outpatient clinics presenting with the most serious manifestations of the disease, having a high level of symptoms and a high frequency of exacerbations. 3 Based on the results of the IMPACT and ETHOS trials, it is indicated that symptomatic patients at risk of exacerbations can reduce their risk of dying if treated with ICS-containing triple therapy compared with dual bronchodilator therapy. The translation of the estimated aARR for ACM in the IMPACT trial to the large population of treatable patients has the potential to significantly change the public health burden of COPD.
Potential mechanisms explaining the effect of ICS/LABA/LAMA therapy on mortality
Several hypotheses may be considered in the attempt to explain the reduction in risk of ACM by triple therapy in patients with COPD. First of all, triple therapy significantly reduced the rate of moderate-severe exacerbations, with a greater effect than either ICS/LABA or LABA/LAMA combinations in patients with symptomatic COPD and at risk of exacerbations.19,21,31 Exacerbations are a well-established predictor of short- and long-term mortality, particularly in patients with frequent exacerbations. 32 Short-term mortality during and after an acute COPD exacerbation may be due to the consequences of respiratory failure and the associated increased risk of severe cardiovascular events,33–35 probably caused by the respiratory failure itself, but also by platelet activation and release of inflammatory mediators from the lung.36–39
Consistent with the anti-inflammatory properties of ICSs, some studies have reported an increased incidence of nonfatal pneumonia; 40 however, these events are more than offset by the beneficial effects of these drugs, as indicated by the reduction in ACM. Furthermore, other long-term side effects, like metabolic, cardiovascular, and musculoskeletal diseases, in patients with COPD treated with moderate/high-dose ICSs are fairly rare and frequently not different from those observed in non-ICS-treated patients.41,42
According to the systemic inflammation theory, the activation of proinflammatory mediators in the peripheral circulation, associated with chronic persistent airway inflammation, may lead to negative consequences, both in the cardiovascular system and in other organs and systems.43–45 In support of this theory, C-reactive protein (CRP), proinflammatory cytokines, and endothelium-derived microparticles have been detected in the blood of patients with COPD, increasing with COPD severity.46–49 Some of these systemic biomarkers increase further during an acute exacerbation, supporting a potential contribution to a poor outcome of exacerbation. Furthermore, persistently high levels of serum CRP after an acute exacerbation have been associated with a higher risk of a second exacerbation37,50 and the persistence of at least two of the six inflammatory biomarkers studied in the ECLIPSE study, that is, white blood cells, high sensitivity CRP, interleukin (IL)-6, IL-8, fibrinogen, and tumor necrosis factor-alpha, was associated with a poor prognosis. 43
Despite the fact that we do not have a clear demonstration that triple therapy including ICSs is able to modify the release of these mediators from the lung, it is reasonable to think that the inflammatory process of the airways in COPD may be modulated by pharmacological treatment. This was demonstrated in a randomized, placebo-controlled study conducted at two centers in The Netherlands, in which FP therapy reduced airway inflammation in patients with moderate-to-severe COPD. 51 Furthermore, reductions in moderate-to-severe exacerbations with several pharmacological treatments were greatest in patients with COPD and an eosinophilic phenotype.52–54 Therefore, more studies are needed to verify that the effect of ICS-containing treatments on ACM is associated with a significant reduction in specific biomarkers of airway and systemic inflammation.
Conclusion
In our opinion, the available evidence about the pharmacological treatments of COPD strongly suggests the possibility of significantly reduced risk of ACM and in turn the potentially improved life expectancy with triple ICS/LABA/LAMA therapy in patients with symptomatic COPD and at risk of exacerbations compared with UMEC/VI. Currently only FF/UMEC/VI and higher dose BUD/GLY/FOR have been shown to significantly reduce ACM in COPD, although we believe that similar effects will be achieved with other ICS/LABA/LAMA combinations.
On the contrary, no demonstration has been obtained as regards the possibility of bronchodilators alone, as both single therapy and LABA+LAMA combinations, to significantly reduce ACM, as assessed by some systematic reviews or observational studies.24,55 However, the greater efficacy of triple therapy compared with ICS/LABA on ACM, although not statistically significant, might be related to the additive effect of two bronchodilators on the risk of exacerbations.
Reducing ACM is the goal of therapies for chronic conditions. Although the general perception is that such an ambitious therapeutic goal is not achievable for COPD, we believe it is now time to change the paradigm of treatment for patients with COPD who are at risk of exacerbations, moving from the relief of symptoms to the improvement of long-term prognosis. Of course, we are confident that therapies for more specific targets in COPD will be available in the future. However, at the present time, ICS/LABA/LAMA association therapy is already able to provide a high probability of prognostic improvement and we hope that future guidelines and recommendations from international initiatives move towards a less uncertain stance on this crucial point.
Footnotes
Acknowledgements
The authors thank Valentina Di Boscio, Neil Martin, Carmen Stabile, Roberta Tosatto, and Gudrun Weiss of GlaxoSmithKline for their contributions to the development of this paper.
Author contributions
The authors meet the criteria for authorship as recommended by the International Committee of Medical Journal Editors, take responsibility for the integrity of the work as a whole, contributed to the writing and reviewing of the manuscript, and have given final approval for the version to be published. AC, PP, and RP were involved in the design of the paper, the acquisition of data, and data analysis/interpretation. ML was involved in the design of the paper and data analysis/interpretation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Editorial support (in the form of grammatical editing and referencing) was provided by Philip Chapman, at Fishawack Indicia Ltd, UK, and was funded by GlaxoSmithKline.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RP was employed in the role of Global Medical Expert and now acts as a consultant for GlaxoSmithKline Italy; PP received institutional and personal grants for education and research from AstraZeneca, Chiesi, GlaxoSmithKline, Menarini, Mundipharma, Novartis, and Sanofi; AC received institutional and personal grants for education and research from GlaxoSmithKline, AstraZeneca, Sanofi, and Chiesi.
