Abstract
Pulmonary arterial hypertension (PAH) is characterized by pulmonary vascular remodeling that limits the ability of the pulmonary vascular bed to withstand the physiological changes of pregnancy. Historically, pregnancy in PAH carries a high risk to the parturient. Normal pulmonary vasculature can withstand the hemodynamic and physiological changes associated with pregnancy without the development of respiratory symptomatology. However, in the presence of pulmonary vascular remodeling the capacity to handle these changes is compromised. During pregnancy, increase in cardiac output from the increased intravascular volume can lead to right heart failure. Therefore, all patients with PAH of childbearing potential should receive preconception counseling and be advised to use two methods of contraception. Patients with PAH should be advised against continuing pregnancy if they do become pregnant. According to the literature, deterioration in pregnancy mainly occurs in the second trimester and early in the third trimester; immediately postpartum is the most critical time for patients with PAH. In this review, we will discuss the recent advances in the management of parturient patients with PAH.
Keywords
Introduction
Pulmonary arterial hypertension (PAH) is characterized by pulmonary vascular remodeling and carries a high mortality resulting from right ventricular failure [Benza et al. 2012; D’Alonzo, Barst et al. 1991]. PAH is defined hemodynamically by the presence of a mean pulmonary artery pressure (PAP) of at least 25 mmHg with a pulmonary capillary wedge pressure (PCWP) or left ventricular end diastolic pressure of up to 15 mmHg [Badesch et al. 2009]. Pathophysiology of PAH is complex and appears in part from an imbalance between mediators of vasodilatation [e.g. prostacyclin, nitric oxide (NO)] and vasoconstriction (e.g. endothelin-1) in favor of the vasoconstrictive forces, and promotion of vascular smooth muscle cell proliferation in the small pulmonary arteries [Safdar, 2010, 2011; Badesch et al. 2004]. Prior to the availability of specific therapies, PAH (diagnostic group 1) (Table 1) was considered a rapidly fatal illness with a median survival of 2.8 years [D’Alonzo et al. 1991]. Even with the recent advances made in the treatment, prognosis for patients with PAH remains poor [Benza et al. 2010].
Updated pulmonary hypertension classification [Dana Point 2008].

COPD, chronic obstructive pulmonary disease; HIV, human immunodeficiency virus; modified from Simonneau et al. 2009.
Pregnancy presents a serious risk for patients with PAH [Smedstad et al. 1994; Weiss et al. 1998; Yentis et al. 1998]. Normal pulmonary vasculature can accommodate the increased blood volume associated with pregnancy without significant cardiopulmonary compromise. However, in the presence of pulmonary vascular remodeling the capacity to handle the increased blood flow is compromised. Hence, increase in cardiac output from the increased intravascular volume associated with pregnancy can lead to right heart failure. In this regard, deterioration in pregnancy occurs in the second and early in the third trimester, and immediately postpartum is the most critical period for patients with PAH.
Normal physiological changes in pregnancy
Normal physiological changes of pregnancy include a progressive increase in blood volume starting from 6–8 weeks’ gestation [Madden, 2009]. This results in a cumulative increase in blood volume of 40–100% from baseline by the end of gestation. In contrast, red blood cell mass increases by only 20–30%, resulting in a ‘physiologic anemia’ state. The increase in cardiac output parallels the increase in blood volume. Cardiac output increase is accomplished by an increase in stroke volume with some increase in heart rate. The cardiac output increases from 6–8 liters/min by 8–11 weeks to a maximum of 9 liters/min at term. Heart rate increases by 10–20 beats per minute over the course of the pregnancy, peaking in the late second and early third trimester. In addition, heart rate increases during labor in response to uterine contraction, anxiety and pain [Robson et al. 1987; Easterling et al. 1990; van Oppen et al. 1996]. Diastolic blood pressure decreases in mid-pregnancy; however, systolic blood pressure and PAP remain unchanged. It is also important to note that the venous pooling of blood may reduce absorption of drugs administered intramuscularly or subcutaneously. Progesterone mediates the systemic vascular relaxation and the hyperdynamic state results in a progressive decrease in the systemic vascular resistance during pregnancy.
Progesterone mediates the increase in minute ventilation, tidal volume and oxygen consumption though respiratory rate remains unchanged. The arterial carbon dioxide level falls to 28–32 torr by term with a plasma bicarbonate level of 18–21 mEq/liter. This gives rise to a sensation of dyspnea in parturient women. Functional residual capacity may decrease by 10–25%, but the total lung capacity remains unchanged due to the compensatory thoracic cage widening. An enlarging uterus may compress the inferior vena cava and the aorta while in the supine position, resulting in reduced venous return. Compression of the aorta and its branches may result in decreased perfusion to the kidneys, lower extremities and utero-placental unit.
Pregnancy is considered a relative hypercoagulable state with development of progressive resistance to protein C and decrease in protein S levels, and increase in clotting factor activity [Marik and Plante, 2008]. This may predispose pregnant patients to form microthrombi.
During labor, fluid shifts may further increase the cardiac output by 10–15%, resulting in an increased preload that stresses the right ventricle and pulmonary vascular bed. Immediately after delivery, uterine contraction causes an auto transfusion of 300–500 ml of blood from the uterus into the maternal circulation [Lane and Trow, 2011]. In addition, venous return may increase from the relief of inferior venacaval obstruction by the gravid uterus. The resultant increase in cardiac output may remain elevated for up to 48 h and the hormonal levels may take up to 6 weeks to normalize. On average, blood loss during vaginal delivery is 10% of total blood volume and 30% during a Cesarean section. These changes impose an extra strain on the already compromised cardiopulmonary system of patients with PAH.
Risk of pregnancy in pulmonary arterial hypertension
Diagnosis of PAH may be made during pregnancy or soon after delivery, suggesting that the hemodynamic changes associated with pregnancy result in unmasking of the disease state (Table 4]. In addition, substantial hormonal changes during pregnancy may lead to the development of PAH in predisposed pregnant women. Historically, maternal mortality in patients with PAH is noted to be 30–50% [McCaffrey and Dunn, 1964; Smedstad et al. 1994; Weiss et al. 1998; Yentis et al. 1998]. It is well established that patients with PAH have a high perioperative risk of about 36–50%. Weiss and colleagues evaluated published data from 1978 through 1996 using the keywords ‘pregnancy’, ‘pulmonary hypertension’, ‘primary pulmonary hypertension’, ‘pulmonary vasculitis’, ‘pulmonary vascular disease’, ‘congenital heart disease’, ‘Eisenmenger’s complex’ and ‘Eisenmenger’s syndrome’ [Weiss et al. 1998]. The overall maternal mortality was noted to be 38%, 36% in Eisenmenger’s syndrome, 30% in primary pulmonary hypertension and 56% in associated PAH (congenital disease, thromboembolism, hepatitis, systemic connective tissue disease and vascular inflammatory disease) [Weiss et al. 1998]. All except three fatalities occurred within 35 days of delivery. In this analysis of published data, late diagnosis and late hospital admission were reported as independent risk factors associated with maternal mortality.
Bedard and colleagues analyzed Medline articles published between January 1997 and September 2007 using the keywords very similar to the Weiss study ‘pregnancy’, ‘pulmonary hypertension’, ‘Eisenmenger’, ‘Eisenmenger’s syndrome’, ‘primary or idiopathic pulmonary hypertension’, ‘pulmonary vascular disease’ and ‘congenital heart disease’ (Table 4) [Bedard et al. 2009]. Next, the authors conducted a comparative analysis with data from the study by Weiss and colleagues (reported data between 1978 and 1996) [Weiss et al. 1998]. Seventy-three parturients were identified between 1997 and 2007, of whom 72% were on PAH-specific therapy. Overall mortality in this group was 25% (38% in the previous era), 17% in idiopathic PAH (30% in the previous era), 28% in congenital heart disease associated PAH (36% in the previous era) and 33% in patients with associated PAH (56% in the previous era). Parturient women who received general anesthesia were four times more likely to die than those receiving regional anesthesia. Primigravidae were at higher risk of death than parturients with previous pregnancies; 78% of deaths occurred within 1 month postpartum (15 deaths postpartum versus three deaths during pregnancy). While 85% of the parturients were on calcium channel blockers, only five were on sildenafil [US Food and Drug Administration (FDA) approved in 2005]. In the older era there were no specific PAH therapies available, however with the advent of PAH-specific therapies, improvement in mortality was noted in all patients with PAH.
Deterioration in pregnancy is reported to occur between 20 and 24 weeks of gestation and early in the third trimester [Bonnin et al. 2005]. Bonnin and colleagues reviewed the charts of 14 pregnant women with 15 pregnancies over a period of 10 years (1992–2002) (Table 4) [Bonnin et al. 2005]. The overall mortality in this study was 36%. Two women died before delivery at 12 and 23 weeks of gestation. There were three early deteriorations and five deteriorations that occurred in the third trimester. In cases of early deterioration only one patient survived, suggesting that early deterioration should lead to therapeutic abortion. The authors concluded that sudden hemodynamic instability is associated with a high maternal mortality and right ventricular failure was the most common cause of death.
In a recent study, Katsuragi and colleagues reported data from 42 pregnant Japanese patients with PAH (1982–2007). Eighteen terminated their pregnancies and 24 continued till term (Table 2) [Katsuragi et al. 2012]. In this study the authors divided patient data according to their PAP and showed that patients with mild PAH tolerate pregnancy better then patients with severe PAH [severe PAH defined as pulmonary artery systolic pressure (PASP) > 50 mmHg by echocardiography or mean PAP > 40 mmHg by cardiac catheterization]. They reported one maternal and one fetal death both in patients with severe PAH (overall mortality 4%).
Drugs used to treat pulmonary hypertension in pregnancy.

cGMP, cyclic guanosine monophosphate; INH, inhalation treatment; PDE-5, phosphodiesterase-5.
In a prospective study from 13 participating centers in France, Jais and colleagues reported data from 26 pregnancies with their 3-year outcome (Table 4) [Jais et al. 2012]. A total of 16 pregnancies were successful, six had induced abortion and two had spontaneous abortion. Three women died in the early postpartum period due to right heart failure, whereas one required urgent heart–lung transplantation. The authors reported that the outcome of pregnancies was better in patients with lower pulmonary vascular resistance (PVR 500 ± 352 dyn/s/cm5) whereas patients with a very high PVR (1667 ± 209 dyn/s/cm5) died or required transplantation. Although the numbers are small, 62% of all pregnancies were successful in this study.
Contraception counseling
Preconception counseling is very important for patients with PAH. They are advised to use two methods of contraception and to terminate pregnancy immediately if they do become pregnant. Since endothelin receptor antagonists (ETRAs) are potent teratogens, proof of contraception is required for women of childbearing potential before starting treatment and during treatment. Due to the drug interaction of bosentan with hormonal contraceptives, the possibility of contraceptive failure exists, necessitating the use of a second form of birth control. Bosentan and ambrisentan are tetatogenic and should be stopped immediately when pregnancy is suspected (FDA pregnancy category X). Early PAH deterioration during pregnancy should lead to prompt therapeutic abortion [Smedstad et al. 1994]. We strongly advise against pregnancy in this patient population given the high mortality and termination of pregnancy is recommended.
Risks associated with mode of delivery
Mode of delivery should be carefully selected in parturient patients with PAH as it will influence the outcome. In this regard, Cesarean section is the preferred mode of delivery but requires a multidisciplinary approach as outlined below.
Vaginal delivery
This mode of delivery should be avoided in patients with PAH as it carries a multitude of disadvantages as outlined below.
Valsalva maneuver: this maneuver used during vaginal delivery can decrease venous return. As the cardiac output in a patient with PAH is preload dependent, the ensuing reduction in cardiac output can precipitate cardiopulmonary collapse.
Vasovagal response can decrease the venous return, reduce the preload and lead to cardiopulmonary collapse.
Pain of childbirth may cause sympathetic nervous system stimulation and increase the heart rate and the altered systemic and pulmonary vascular resistance may produce hemodynamic instability. Hence, during this period, vasopressor or inotropic support may be required. If vaginal delivery is chosen, effective analgesia is most important and low-dose epidural analgesia should be considered.
With uterine contraction, approximately 500 ml of blood is diverted from the uterine circulation into the maternal circulation (autotransfusion), leading to increased circulating blood volume. In addition, venous return may increase from the relief of inferior vena cava obstruction by the gravid uterus. This increase in preload can precipitate right heart failure.
Labor induction: agents used to induce labor may precipitate clinical deterioration. Prostaglandin E produces pulmonary vasodilation whereas prostaglandin F2a induces pulmonary vasoconstriction. Low-dose oxytocin can be used to induce labor, but with caution, as it can increase pulmonary vascular resistance.
Cesarean section
Cesarean section is the preferred mode of delivery and should be used unless not available or in cases of emergencies [Kiely et al. 2010; Bonnin et al. 2005]. Cesarean section bypasses the complications associated with labor and the autotransfusion associated with vaginal contractions. However, Cesarean section may result in increased fluid shifts from abdominal surgery, as well as increased pulmonary vascular resistance and increased right ventricular after load imparted by positive pressure ventilation in the setting of mechanical ventilation. The choice of anesthesia is important when Cesarean section is being considered and should be preplanned with the anesthesiologist who should be aware of the hemodynamic changes associated with PAH (see below).
Anesthesia in pregnant patients with pulmonary arterial hypertension
General anesthesia
General anesthesia is known to depress cardiac contractibility (volatile agents), increase pulmonary vascular resistance (positive pressure ventilation), and increase PAP during larngoscopy and intubation [Sorensen and Jacobsen, 1977]. General and epidural anesthesia should be performed early in the labor by an experienced anesthesiologist to minimize the increase in cardiac output that occurs with contraction and pain. In addition, positive pressure ventilation may reduce venous return predisposing the patient to right heart failure.
Epidural or combined spinal–epidural anesthesia
Epidural anesthesia with incremental doses is considered the best approach to regional anesthesia in patients with PAH. However, combined spinal–epidural anesthesia provides a better sensory block than epidural anesthesia alone with no additional risk of hypotension when a very low dose is used [Duggan and Katz, 2003]. Although a newer technique, it is better tolerated and causes fewer systemic side effects, such as hypotension and nausea.
Spinal anesthesia
Spinal anesthesia consists of a single shot of an analgesic agent that produces a brief duration of anesthesia (2 h) and may produce rebound worsening of PAH. Therefore, it should be avoided in this patient population [Bonnin et al. 2005]. In addition, spinal anesthesia may lead to systemic hypotension.
Antepartum monitoring
Close monitoring of pregnant patients with PAH is essential (Figure 1). These high-risk parturients should be followed every 4 weeks until the third trimester and in the third trimester weekly follow up is suggested. Echocardiograms are recommended every 4 weeks and in the third trimester weekly echocardiograms are advised to monitor the cardiac status and adjust PAH medications. Calcium channel blockers should be used if the pulmonary hemodynamics showed a positive vasodilator response (FDA pregnancy category C). Warfarin should be stopped immediately (pregnancy category X), and if indicated, parturients can be started on low molecular weight heparin (LMWH). LMWH should be used if patients are on bed rest or when they get admitted to hospital. We recommend that parturients are advised to avoid constipation as the resulting valsalva may decrease venous return. Elective Cesarean section is advisable when the fetus becomes viable (usually 34 weeks’ gestation). In the reported literature, a multidisciplinary approach has led to a favorable maternal–fetal outcome in parturients with PAH [Kiely et al. 2010]. Therefore, close communication between the obstetrician, anesthesiologist and pulmonary vascular physician that helps in the proper and timely management of these high-risk obstetric patients is strongly recommended [Kiely et al. 2010].
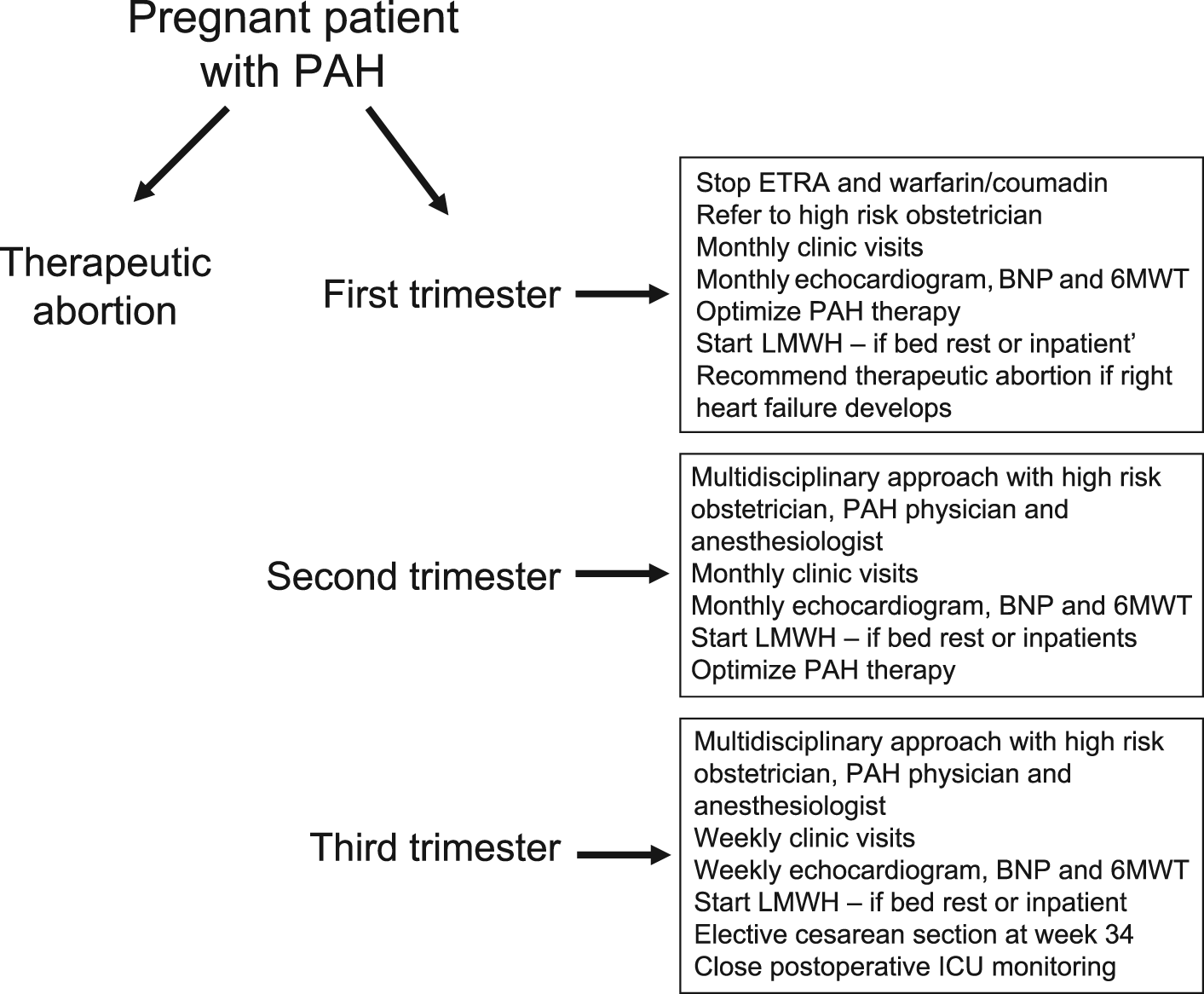
Recommended evaluation and follow up of a pregnant patient with pulmonary arterial hypertension. BNP, brain natriuretic peptide; ETRA, endothelin receptor antagonist; ICU, intensive care unit; LMWH, low molecular weight heparin; PAH, pulmonary arterial hypertension; 6MWT, 6 min walk test.
Peripartum monitoring
Close monitoring of the hemodynamic status of these parturients is important. An arterial line and a central venous catheter may be sufficient for monitoring the hemodynamic status obviating, the need for Swan-Ganz catheter placement. A central venous catheter can be used to monitor the right atrial pressure and mixed venous oxygen saturation, and also to facilitate rapid fluid infusion, and if required, blood draws. The use of a Swan-Ganz catheter may predispose PAH patients to increased risk of pulmonary artery rupture or thrombosis. Therefore, routine use of these catheters is not recommended, but a small and select group of patients may benefit from close monitoring.
Postpartum period
The most critical period for acute decompensation is postpartum. Therefore, patients should be observed in the intensive care unit for several days postpartum. Anticoagulation should be used during this period, especially in patients with idiopathic PAH and chronic thromboembolic pulmonary hypertension. We usually use LMWH during this period. Volume expansion of up to 40–50% occurs in pregnancy, peaks in the third trimester and is sustained for up to 24 weeks postpartum. Fluid from the extravascular space also shifts into the intravascular compartment postpartum, and the vulnerable period is childbirth and immediately postpartum. It may take up to 6 months for these patients to return to their previous baseline state. However, it is important to note that not all patients with PAH go back to their baseline cardiopulmonary state after childbirth.
Treatment options
Agents used to treat pregnant patients with PAH are outlined in Table 2. If a patient with PAH becomes pregnant, ETRAs should be stopped immediately due to their teratogenic effects (pregnancy risk category X) (Table 3) [Spence et al. 1999].
US Food and Drug Administration assigned risk category for pulmonary arterial hypertension drugs.

B, Animal reproduction studies have failed to demonstrate a risk to the fetus and there are no adequate and well controlled studies in pregnant women.
C, Animal reproduction studies have shown an adverse effect on the fetus and there are no adequate and well controlled studies in humans, but potential benefits may warrant use of the drug in pregnant women despite potential risks.
X, Studies in animals or humans have demonstrated fetal abnormalities or there is positive evidence of human fetal risk based on adverse reaction data from investigational or marketing experience, and the risks involved in use of the drug in pregnant women clearly outweigh potential benefits.
Prostacyclin analogues
Epoprostenol and treprostinil
Epoprostenol has been successfully used to treat PAH in pregnant women. There are several case reports of the use of intravenous prostanoids in PAH [Garabedian et al. 2010; Goland et al. 2010; Easterling et al. 1999; Badalian et al. 2000; Stewart et al. 2001; Geohas and McLaughlin, 2003]. In one report, Geohas and McLaughlin successfully treated a pregnant woman with Eisenmenger’s syndrome with intravenous epoprostenol who presented during the last trimester (34 weeks and 3 days of gestation) [Geohas and McLaughlin, 2003]. She improved clinically with intravenous epoprostenol infusion that was rapidly titrated over a 24 h period to 9 ng/kg/min and then she underwent a Cesarean section with the delivery of a boy.
In another case report, a pregnant patient with PAH presented at 26 weeks’ gestation with progressive exertional dyspnea [Stewart et al. 2001]. At 32 weeks’ gestation, she was started on intravenous epoprostenol that was titrated up to 10 ng/kg/min and successfully underwent a Cesarean section under epidural anesthesia at week 36. No infant or mother mortality was reported at 2-year follow up. Another report described a patient with severe PAH who was on long-term epoprostenol infusion therapy and became pregnant with twins [Badalian et al. 2000]. She required uptitration of her epoprostenol dose, and in addition, she was treated with NO during delivery and the postpartum period with a good outcome. Goland and colleagues reported two cases of pregnant patients who presented at the last trimester with severe PAH. Both were treated with sildenafil and intravenous epoprostenol during the reminder of their pregnancies and the postpartum period with a good outcome [Goland et al. 2010].
Patients should be started on intravenous epoprostenol as it offers advantages such as a short half life, allowing for rapid dose titration [Barst et al. 1996]. Patients should continue to receive epoprostenol infusion uninterrupted throughout their pregnancy, including during the Cesarean section and postpartum period. Patients can be slowly taken off prostanoids several months postpartum if they remain clinically stable. Thrombocytopenia associated with prostacyclin therapy can increase the propensity to bleed and may require transfusion of platelets and desmopressin [Chin et al. 2009]. It may be difficult for patients to continue on inhaled prostacyclin therapy during delivery and postpartum for multiple reasons. Therefore, it is advisable for such patients to change to intravenous therapy during pregnancy and throughout the postpartum period. There are also reports of the use of treprostinil in parturients with PAH. This may be a treatment option for patients who are not candidates for epoprostenol therapy.
Iloprost
Animal studies have revealed evidence of fetotoxicity and impaired fertility at intravenous doses ranging from 0.01 to 0.5 mg/kg/day [Battenfeld et al. 1995]. Iloprost has been shown to be teratogenic in rats [Battenfeld et al. 1995]. There are no controlled data in human pregnancy. Iloprost is a pregnancy category C drug and only recommended for use during pregnancy when there are no alternatives and the benefits outweigh the risks. To date, no congenital malformations in the fetus or mortality have been reported that are related to iloprost use in parturients.
A case report has been published on a patient with systemic lupus erythematosus associated PAH who became pregnant while taking bosentan and sildenafil [Streit et al. 2009]. Due to the known teratogenic effects, bosentan was stopped immediately (5 weeks’ gestation). Inhaled iloprost was added to sildenafil at 35 weeks’ gestation when an increase in PAP was noted. Cesarean section was performed at 37 weeks’ gestation, and both mother and baby were reported to be alive at the time of last follow up. Elliot and colleagues reported on the outcome of three pregnant patients with PAH who were treated with iloprost early in their pregnancies [Elliot et al. 2005]. These patients were treated with iloprost starting as early as 8 weeks (patient 1), 19 weeks (patient 2) and 18 weeks (patient 3). They were admitted between 24 and 36 weeks’ gestation and delivered between 25 and 36 weeks’ gestation by Cesarean section under epidural anesthesia. Surprisingly, the authors reported no mother or infant mortality, although one baby was delivered at 25 weeks and 5 days of gestation. This patient was initially treated with nebulized iloprost that was changed to intravenous iloprost after she had cardiopulmonary collapse at week 25 and was successfully resuscitated. She delivered a boy 5 days later with an APGAR score of 8 at 1 min and 9 at 5 min.
Curry and colleagues reviewed data from 12 pregnancies in nine patients between 1995 and 2010 and reported two maternal deaths (17% mortality), nine live births and three first-trimester miscarriages (Table 4) [Curry et al. 2012]. Maternal complications included five postpartum hemorrhages and one wound hematoma that required evacuation. All patients were delivered by Cesarean section under general anesthesia, except for one elective and one emergency Cesarean section performed under regional block. PASP ranged from 38 to 106 mmHg in all patients. Two parturients who died had a PASP of 104 mmHg and 55–60 mmHg, respectively. Both had undergone emergency Cesarean sections, had received boluses of oxytocin during the third stage and developed tachyarrythmias. Eight parturients were treated with sildenafil, one with intravenous iloprost, one was on nebulized iloprost and sildenafil, one received diltiazem (pregnancy risk category C) and one patient was on no PAH specific medication. Intravenous iloprost is not FDA approved for use in the USA. We did not find any reported cases of inhaled treprostinil used to treat parturient patients with PAH.
Summary of selected studies.

Bos, bosentan; CCB, calcium channel blocker; CHD-PAH, congenital heart disease associated PAH; CS, Cesarean section; CSA, continuous spinal anesthesia; CSE, combined spinal–epidural anesthesia; CTEPH, chronic thromboembolic pulmonary hypertension; CVD-PAH, collagen vascular disease associated PAH; EA, epidural anesthesia; GA, general anesthesia; HPAH, hereditary PAH; ilop, iloprost; iPAH, idiopathic pulmonary arterial hypertension; IV Epo, intravenous epoprostenol; Left hrt, left heart disease; new dx, new diagnosis; NO, inhaled nitric oxide; NR, not reported; prepreg, prepregnancy; Prost, prostacyclin analogue; Reg, regional anesthesia; SC trep, subcutaneous treprostinil; sild, sildenafil; spinal, spinal anesthesia; VD, vaginal delivery.
Not specified.
Inhaled nitric oxide
There are several reports of using inhaled NO in pregnant patients with PAH (Table 4) [Robinson et al. 1999; Weiss et al. 2000]. In one case report, a pregnant patient with PAH with atrial septal defect and human immunodeficiency virus who presented at 30 weeks’ gestation was treated with sildenafil and inhaled NO. She went into spontaneous labor and delivered vaginally despite planned Cesarean section [Ng and Yip, 2012]. Inhaled NO was slowly tapered off, and sildenafil was continued. She was started on subcutaneous enoxaparin and digoxin and continued on antiretroviral agents. In another case report, a primigrada with severe PAH was treated with inhaled NO after elective intubation and underwent successful assisted vaginal delivery after having epidural anesthesia [Lam et al. 2001]. A patient with Eisenmenger syndrome was treated with inhaled NO during pregnancy [Goodwin et al. 1999]. Inhaled NO was discontinued 48 h after delivery, and the patient died 2 days later despite direct infusion of prostacyclin in the pulmonary artery. Inhaled NO can be successfully used in selected parturients with PAH.
Phosphodieasterase-5 inhibitors
There are several case reports of sildenafil use in pregnant patients with PAH [Goland et al. 2010; Ng and Yip, 2012; Tacoy et al. 2010; Villanueva-Garcia et al. 2007]. In one case report, a parturient with Eisenmenger syndrome was treated during pregnancy, delivery and postpartum with L-arginine and sildenafil with good infant and patient outcome [Lacassie et al. 2004]. It is important to note that L-arginine is not FDA approved in the USA. A patient was diagnosed with idiopathic PAH after successful full-term pregnancy and Cesarean section with epidural anesthesia. The postpartum course was complicated by progressive dyspnea and lower limb edema and she was started on sildenafil with a favorable outcome [Tacoy et al. 2010].
Sildenafil has other beneficial effects such as uterine artery vasodilation and has been shown to improve uterine muscle wall thickness in patients who have had in vitro fertilization with previous poor endometrial response [Sher and Fisch, 2002]. In addition, sildenafil and NO are being used successfully to treat preterm, term, neonatal and childhood pulmonary hypertension [Travadi and Patole, 2003; Karatza et al. 2005]. Tadalafil, a long-acting phosphodiesterase-5 inhibitor, is FDA approved to treat patients with PAH, but there are no reported studies of the use of tadalafil in parturient patients with PAH.
Conclusion
Pregnancy in patients with PAH is associated with a high maternal mortality rate. Preconception counseling must be undertaken in every patient with PAH of child-bearing potential. Patients are advised to use two methods of contraception and to terminate pregnancy immediately if they become pregnant. A comprehensive and careful discussion between PAH specialists and pregnant women with PAH is strongly recommended to advise them about the risks for themselves and their babies. Close clinical monitoring including monthly follow-up visits in the first and second trimester and weekly clinic visits in the third trimester is recommended. Patients with worsening right heart failure, especially early in the pregnancy, should be given the option to terminate their pregnancy. However, if they plan to continue with the pregnancy, PAH therapy should be augmented to improve the chances of a favorable outcome. In this regard, a multidisciplinary approach with a high-risk obstetrician, a pulmonary vascular physician and an anesthesiologist is strongly recommended. Despite the multidisciplinary approach, mortality remains high for pregnant patients with PAH.
Footnotes
Acknowledgements
The author appreciates the editorial help of Ms Janice Brister in the preparation of this manuscript.
Funding
This study was supported by the National Institute of Health grant K23HL-093214 to ZS.
Conflict of interest statement
Dr. Safdar is on advisory board and provided consultancy for Actelion Pharmaceuticals, Gilead Sciences and United Therapeutics.
