Abstract
There has been a recent global resurgence of tuberculosis (TB) fuelled by HIV infection and migration. Childhood TB represents a sentinel event in the community, suggesting recent transmission from an infectious adult. The diagnosis of TB in children is based on chest X-ray, tuberculin skin testing and mycobacterial staining/culture, although the diagnostic yield from these investigations is often lower than in adults. Newer diagnostic tests are being developed and may improve the diagnostic yield in childhood TB. Treatment of TB in children is similar to adults in that short-course multidrug treatment has been adopted as standard therapy in many national TB programmes. Compliance is a major determinant of the success of drug treatment and directly observed therapy has been adopted as a key component of TB treatment programmes. Although uncommon in children, multidrug-resistant TB is also increasing and treatment often involves longer courses of therapy with second-line drugs.
Epidemiology
There is clear evidence that the global incidence of tuberculosis (TB) is increasing. It has been estimated that in 2007 there were 9.27 million incident cases of TB, an increase from 9.24 million in 2006 [WHO, 2009]. An estimated 1.82 million people died from TB, including those coinfected with HIV. Most of the estimated numbers of cases in 2007 were in Asia (55%) and Africa (31%), with small proportions of cases in the eastern Mediterranean region (6%), the European region (5%) and the Americas (3%). Of the 9.27 million incident TB cases in 2007, an estimated 1.37 million (15%) were HIV-positive; 79% of the HIV-positive cases were in the African region and 11% were in the south-east Asian region.
The most profound influence on the incidence of TB is HIV infection, particularly in sub-Saharan Africa, where HIV and TB form a lethal combination, each speeding the other’s progress. HIV infection has been estimated to account for an excess of 34% of new cases [Cantwell and Binkin, 1997]. Other important factors contributing to the global resurgence of TB include poverty, overcrowding, increased travel/immigration, breakdown of TB control programmes, multidrug resistant TB (MDR TB) and incomplete treatment [Raviglione et al. 1995]. In addition, other markers of poverty, such as exposure to indoor air pollution and smoking, have been associated with increasing TB incidence [Lonnroth et al. 2010].
In 1989, the World Health Organization (WHO) estimated that there were 1.3 million annual cases of TB in children under 15 years of age [WHO, 1989]. However, there are many difficulties in estimating the burden of TB in children, including the difficulty in establishing a definitive diagnosis, the increased presence of extrapulmonary disease in young children, the lack of standard case definition and the lower public health priority given to childhood TB compared with that of adult TB. In many resource-limited countries surveillance data are often unreliable due to poor diagnostic facilities and reporting systems [Hershfield, 1991]. The proportion of TB cases in individual countries that occur in children is highly variable. In low-prevalence countries this may be <5% whereas in some high-prevalence countries it is estimated to be four times this figure.
Childhood TB represents a sentinel event within a community, usually indicating recent transmission from an infectious adult with pulmonary disease. Childhood TB is therefore a reflection of TB control in a population.
Natural history and clinical features
It is important to distinguish TB infection [also referred to as latent TB infection (LTBI)] from TB disease. In both there is evidence of TB infection [tuberculin skin test (TST) or blood-based immunological assay], but in latent infection the clinical, radiological or microbiological evidence of disease is lacking. The majority of children (>50%) with TB disease will be asymptomatic. Of those that develop symptoms most will have pulmonary manifestations, while 25–35% of children will have extrapulmonary symptoms [Ussery et al. 1996]. Systemic complaints such as fever, night sweats, anorexia and decreased activity occur less often. The commonest symptoms at presentation in children with TB disease are cough in the preceding 3 months, persistent fatigue and weight loss [Marais et al. 2005].
The natural history of TB following infection is summarized in Figure 1. Following initial exposure and infection, the primary complex is characterized by a lung parenchymal infiltrate, commonly subpleural, and regional lymph node enlargement. Most children are asymptomatic at this stage and in the majority of cases the infiltrate and lymphadenopathy will resolve spontaneously. In some children, particularly infants, the lymph nodes continue to enlarge causing pressure effects on surrounding structures such as the bronchi, which may result in partial or complete bronchial obstruction. The primary parenchymal infiltrate may also progress to a caseating lesion, known as progressive primary TB. This results in the rupture into pleural and pericardial spaces leading to pleural or pericardial effusions. Erosion of caseating lesions into pulmonary vessels can result in haematogenous dissemination to the lung (miliary TB) and distant extrapulmonary sites. Older children and adolescents are more likely to develop adult-type reactivation disease or post-primary disease [Harris et al. 1977; Nemir and Krasinski, 1988]. This follows infection acquired after 7 years of age, especially at the time of puberty [Lincoln et al. 1960] and will present with the classic symptoms of fever, malaise, weight loss, night sweats, productive cough, chest pain and haemoptysis. Extensive infiltration and cavitation, especially of the upper lobe of the lung, are usually found on a chest X-ray.

Natural history of tuberculosis.
Extrapulmonary TB is more common in children than adults, occurring in approximately 25% of infants and young children under 4 years of age [Jacobs and Starke, 1993]. Superficial lymphadenitis is the most common form of extrapulmonary TB in children, typically involving the supraclavicular, anterior cervical, tonsillar and submandibular nodes. Without treatment, cold abscess and chronic sinus formation may occur. Central nervous system (CNS) disease, especially TB meningitis, is the most serious complication of TB in children and occurs in about 4% of cases [Kumar et al. 1997]. The overall mortality has been reported to be 13% with approximately half of the survivors developing permanent neurological sequelae [Farinha et al. 2000]. Tuberculomas are less common manifestations of CNS infection usually characterized by solitary brain lesions. Bone and joint TB may involve weight-bearing bones and joints, particularly the vertebrae (Pott’s disease) [Janssens and deHaller, 1990]. Other extrapulmonary manifestations of TB, such as gastrointestinal or renal, are rare in children because of the long incubation periods required following haematogenous dissemination to manifest as disease.
Diagnosis
Microscopy and culture
Microscopic examination of clinical samples for acid-fast bacilli using the Ziehl-Neelsen (ZN) stain has been a standard and rapid diagnostic tool for TB diagnosis. Newer fluorochrome stains, such as the auramine and rhodamine, appear to have better detection compared with the ZN stain [Ba and Rieder, 1999; Steingart et al. 2006]. These tests have been widely used as they are easy to perform, cheap and give rapid results. Recent advances in light-emitting diode technology have widened the applicability of fluorescent microscopy [Marais et al. 2008]. However, because younger children rarely produce sputum, early morning gastric aspirate samples are often collected by aspiration of gastric contents via a nasogastric tube. The yield from microscopy of gastric aspirate samples in children with proven pulmonary TB is less than 20% compared with 75% in adults [Strumpf et al. 1979]. The detection rates on microscopy from other extrapulmonary samples are even lower due to the paucibacillary nature of the disease.
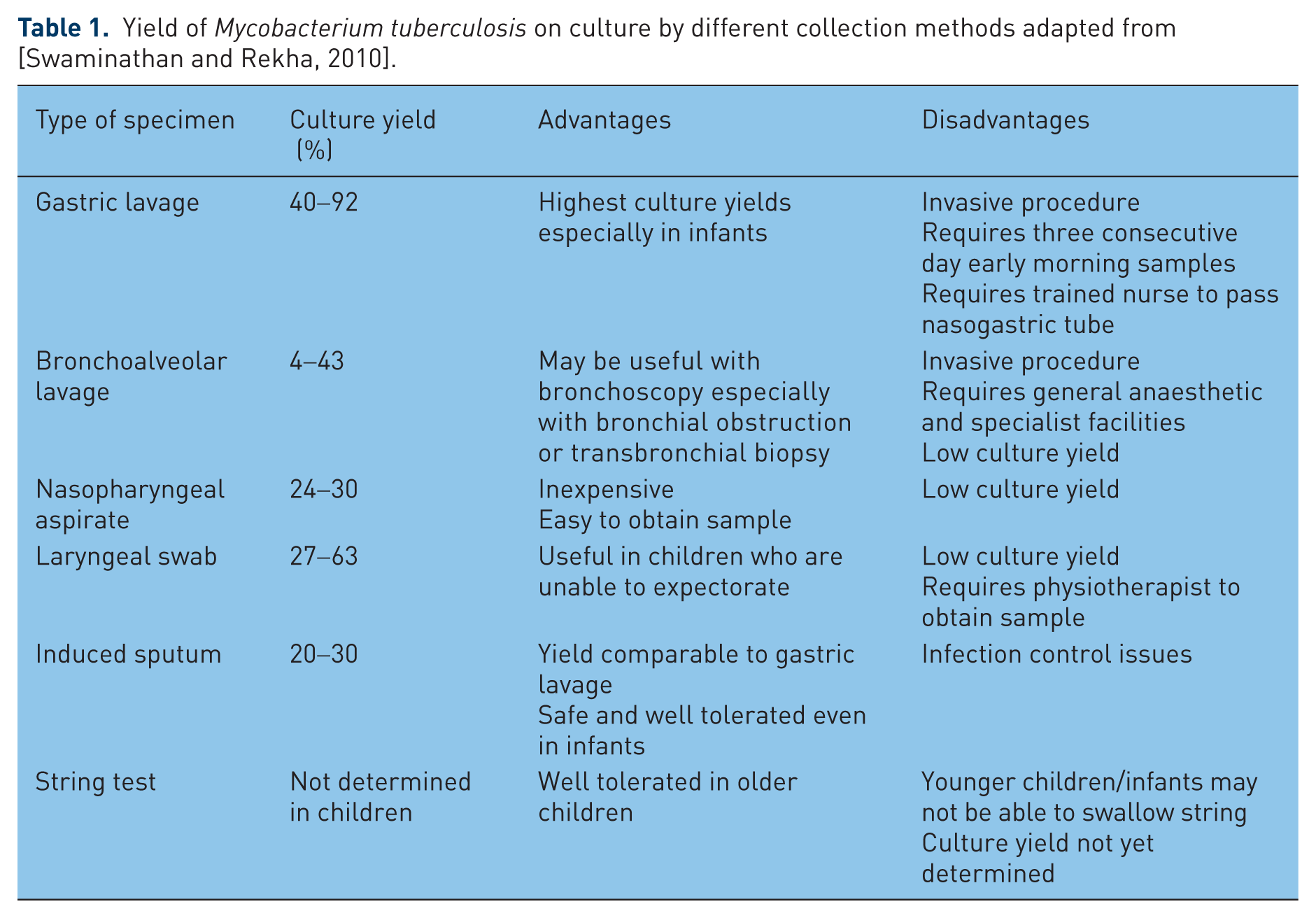
Mycobacterial culture of sputum or gastric aspirates has provided a more useful method of diagnosis in children with suspected pulmonary TB. Table 1 summarizes the culture yield from different collection methods (adapted from [Swaminathan and Rekha, 2010]). Three consecutive morning gastric aspirates yield Mycobacterium tuberculosis in 40–92% of cases and may be higher in infants [Vallejo et al. 1994]. The role of bronchoscopy in evaluating children with pulmonary TB is controversial. The culture yield is usually lower than for three properly obtained gastric aspirates [Abadco and Steiner, 1993; Somu et al. 1995]. The culture yield from other body fluids or tissues from children with extrapulmonary TB is usually less than 50% due again to the paucibacillary nature of the disease [Starke and Taylor-Watts, 1989].
Yield of Mycobacterium tuberculosis on culture by different collection methods adapted from [Swaminathan and Rekha, 2010].
Sputum induction with nebulized 5% saline has more recently been used safely in young infants in resource-limited settings and has culture yields equivalent to those from three consecutive early morning gastric aspirates [Zar et al. 2000]. There are concerns regarding the spread of TB to other patients and staff and it is recommended that sputum induction be performed with appropriate infection control procedures (e.g. negative pressured cubicles), and by appropriately trained staff.
Tuberculin skin test
A positive TST reaction is a hallmark of primary infection with M. tuberculosis. After initial infection, most children show tuberculin reactivity in 3–6 weeks, but occasionally it can take up to 3 months after initial infection. Tuberculin reactivity usually remains positive for the lifetime of the patient, even after treatment for TB [Hsu, 1983].
The Mantoux test is the standard TST and uses 2 tuberculin units of purified protein derivative. The Mantoux test involves the intradermal injection of purified protein derivative solution into the most superficial layer of the skin, raising an immediate wheal. The subsequent induration (rather than erythema) is measured as millimetres after 48–72 h. Percutaneous multipuncture devices, such as the Heaf test and Tine test, are now no longer in common use. In the UK, a TST is regarded as positive with induration of greater than 5 mm in those without prior Bacille Calmette Guérin (BCG) vaccination and greater than 15 mm for those who have received BCG vaccination. WHO guidelines differ slightly in that a positive TST is regarded as positive with induration greater than 10 mm for those without prior BCG vaccination and greater than 15 mm for those with BCG vaccination history [WHO, 1997]. The US guidelines also differ in that TST cutoffs are based on risk categorization. The cutoff of greater than 5 mm has also been suggested for children with HIV infection [Pickering et al. 2009].
TST suffers from false-negative and false-positive results. Up to 10% of otherwise normal children with culture proven TB have a negative TST [Starke and Taylor-Watts, 1989]. False-negative TST may occur in children with debilitating or immunosuppressive illnesses, malnutrition or other severe infections. False-positive TST results may be a problem particularly following BCG vaccination. False-positive TST may also arise with environmental non-TB mycobacteria [Larsson et al. 1993]. Skin reactivity can be boosted, probably through antigenic stimulation, by serial testing with TST in many children and adults who received BCG [Sepulveda et al. 1988].
Radiology
Chest X-ray is widely used for the detection of pulmonary TB, including hilar lymphadenopathy and lung parenchymal changes. The sensitivity and specificity of chest X-ray in children are low (67% and 59% for chest lymphadenoapthy respectively) with large intra- and interobserver variability [Swingler et al. 2005]. Cavitatory disease is uncommon in younger children but is often seen in adolescents (Figure 2), who may develop adult-type postprimary disease [Khan and Starke, 1995]. Miliary TB has the characteristic ‘snowstorm’ appearance on chest X-ray (Figure 3). More recently computed tomography (CT) imaging has been useful in demonstrating early pulmonary disease, such as cavitation, and intrathoracic hilar lymphadenopathy [Andronikou et al. 2004]. CNS disease, such as TB meningitis or tuberculoma, may also be identified on CT imaging, especially as contrast demonstrates meningeal enhancement (Figure 4). Magnetic resonance imaging has been found to be useful for musculoskeletal TB, particularly involving bones and joints [De Backer et al. 2006].

Chest X-ray of cavitatory tuberculosis.

Chest X-ray of miliary tuberculosis.
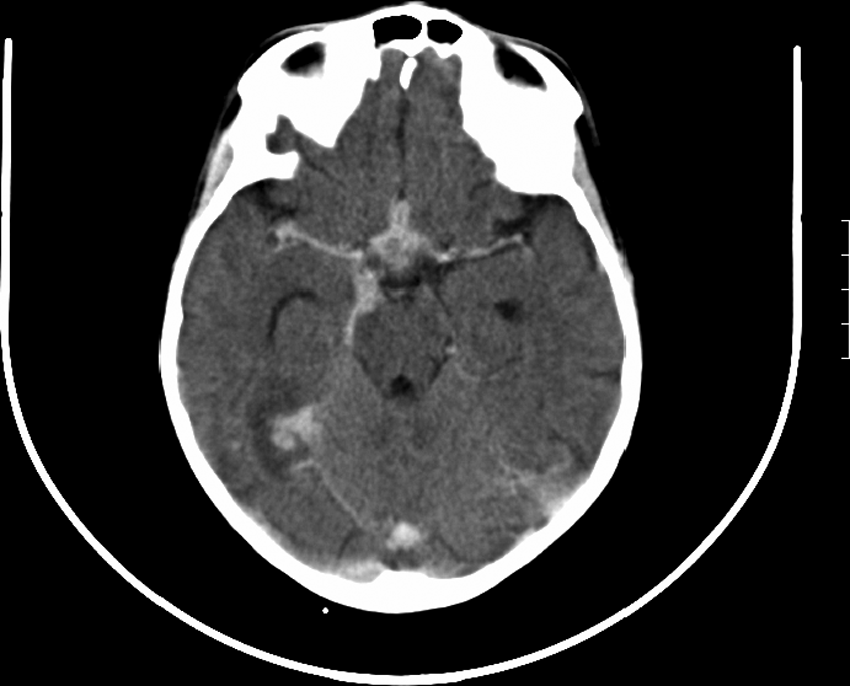
Head computed tomography scan of tuberculosis meningitis.
Molecular diagnostics
While detection of live M. tuberculosis is likely to remain the gold standard for the diagnosis of TB, nucleic acid amplification assays (NAAs) for the detection of mycobacterial DNA or RNA are increasingly being developed for clinical use. Commercial NAAs have been extensively evaluated in adults. These tests offer high specificity (85–98%), high sensitivity for smear-positive TB (pooled estimate 96%) but poorer sensitivity for smear-negative TB (pooled estimate 66%) [Nicol and Zar, 2011]. Their performance in children has not been thoroughly evaluated; however, limited studies to date suggest that their performance in children is likely to be similar to that in smear-negative adults due to the paucibacillary nature of TB in children. Polymerase chain reaction (PCR) has been used successfully in identifying many infectious agents and used for TB diagnosis. The sensitivity of a good quality PCR would be expected to be 90–100% and 60–70% on smear-positive and smear-negative culture-positive respiratory samples respectively [Eisenach et al. 1991; Noordhock et al. 1994; Watterson and Drobniewski, 2000]. However PCR performs less well in children with sensitivity of 40–60% compared with clinical diagnosis [Starke et al. 1993; Smith et al. 1996; Gomez-Pastrana et al. 1999; Fauville-Dufaux et al. 1996] and similar to standard cultures, which have a sensitivity of 30–40%. PCR detection in other body fluids or tissues, such as cerebrospinal fluid, appears to be lower. At present, PCR methods have a limited role in the diagnosis of TB in children but may be useful when the diagnosis is not easily established using standard methods.
NAAs have also been used to identify rifampicin resistance by identifying the rpoB gene of M. tuberculosis. This gene accounts for over 95% of rifampicin resistance and because rifampicin resistance is usually accompanied by isoniazid resistance (monoresistance is rare) this test is used as a marker for MDR TB. Recently, WHO endorsed one such NAA, the Xpert MTB/Rif test (GeneXpert, Cepheid, Sunnyvale, CA, USA), for rapid testing directly from sputum in adults. It is not clear how these tests will perform in children with TB, which typically is paucibacillary in nature.
Immunodiagnosis
Due to the limitations of TST, particularly cross reactivity with BCG immunization and environmental mycobacteria, newer diagnostic tests have been the developed based on in vitro T-cell-based interferon-γ release assays (IGRAs) in response to stimulation to TB-specific antigens (ESAT-6 and CFP10). These antigens are present in M. tuberculosis complex but absent from all strains of Mycobacterium bovis BCG, and almost all environmental mycobacteria. Two IGRAs, the QuantiFERON-TB Gold assay (Cellestis Ltd, Carnegie, Victoria, Australia), and the T SPOT-TB assay (Oxford Immunotec, Oxford, UK) are available. Both tests measure interferon-γ release from T cells in response to using methods such as enzyme-linked immunosorbent assay and enzyme-linked immunospot (ELISPOT) assay [Lalvani et al. 2001]. The QuantiFERON-TB Gold cultures whole blood with the TB-specific assays and measures interferon-γ in the culture supernatant, whereas the ELISPOT cultures peripheral blood mononuclear cells and directly counts the number of interferon-γ secreting cells.
IGRAs have been studied in LTBI and active disease in different geographic settings. As there is no gold standard for LTBI, exposure gradients have been used and comparison made with TST. Overall, although evidence is limited, the results show that IGRAs have modest predictive value, perhaps of the same magnitude as TST. For the diagnosis of LTBI, there is high agreement between the IGRAs, but much discordance (mostly TST positive/IGRA negative) between the IGRA test and TST. The high specificity of IGRAs may be useful in reducing the number of low-risk children who receive preventative therapy [Ling et al. 2011]. Furthermore, IGRAs cannot distinguish between LTBI and active disease not dissimilar to the TST. For active disease, IGRAs have suboptimal sensitivity and therefore cannot be used in isolation to rule out TB disease in children. While IGRAs may be used to help support a diagnosis of TB in combination with the TST and other investigations, they should not be a substitute, or obviate the need for, appropriate specimen collection.
Treatment
Latent tuberculosis infection
The rationale for treating LTBI is the significant risk of progression to TB disease in untreated infection, especially in children. The risk is greatest in younger children (<5 years of age) and in the first 18 months following transmission. The highest risk is in infants and young children in whom a risk of disease progression of 40–50% is widely quoted [Haas, 2000; Miller et al. 1963]. The risk decreases gradually through childhood to adult levels of around 10%.
Treatment of LTBI is highly effective in reducing the risk of TB disease, especially in children, in whom efficacy is well over 90%. A study of over 4000 children with tuberculin reactivity showed a 60-fold increased risk of developing TB disease if prophylactic therapy was not given, equivalent to a number needed to treat (NNT) of 50 LTBI cases to prevent one TB disease [Curry, 1967; Blyth et al. 2004]. A meta-analysis of over 73,000 adults and children exposed to MTB in various settings indicated that prophylactic therapy gave a relative risk of TB disease of 0.4 compared with no prophylaxis, a NNT of approximately 90 [Smieja et al. 2000].
Various regimens have been suggested for LTBI treatment, but most have not been subjected to randomized, controlled trials in children. Six months of isoniazid therapy is the cornerstone of prophylactic therapy and the current recommendation for LTBI in many countries, including the USA, the UK and Australia [WHO, 1982]. Alternative regimens for LTBI are widely used in children, but often with less of an evidence base. Isoniazid and rifampicin for 3 months is well tolerated and of high efficacy in children [Bright-Thomas et al. 2010]. A meta-analysis in adults showed that this combination was equally efficacious as 6 months of isoniazid monotherapy [Ena and Valls, 2005]. Rifampicin monotherapy for 4 months is also used, but infrequently in children [Reichman et al. 2004]. In adults with silicosis, who have a high risk of disease progression from LTBI, 3 months of rifampicin was as efficacious as 6 months of isoniazid [Hong Kong Chest Service/Tuberculosis Research Centre, Madras/British Medical Research Council, 1992], but there are no studies in children. Data, again only from adults, indicate that the combination of rifampicin and pyrazinamide potentially causes fatal hepatotoxicity and is best avoided [Ijaz et al. 2006; McElroy et al. 2005].
Tuberculosis disease
Several controlled and observational trials of 6-month therapy in children with pulmonary TB caused by organisms known or presumed to be susceptible to the first-line drugs have been published [te Water Naude et al. 2000; Tsakalidis et al. 1992; Kumar et al. 1990; Biddulph, 1990; Reis et al. 1990; Jacobs and Abernathy, 1985; Varudkar, 1985; Al-Dossary et al. 2002]. Although 6 months of therapy with isoniazid and rifampicin has been shown to be effective for treatment of hilar adenopathy and pulmonary TB, a three-drug regimen (isoniazid, rifampicin and pyrazinamide) has been shown to have success rates of greater than 95% and low adverse reaction rates. In general, extrapulmonary TB in children can be treated with the same regimen as pulmonary disease; however there are no data from children and extrapolations have been made from studies in adults. Meningitis and disseminated TB, however, may not be adequately treated with 6 months duration and longer treatment durations of 9–12 months are recommended. The optimal treatment of TB in children and adolescents with HIV infection is unknown. Treatment durations of at least 9 months have been suggested [Pickering et al. 2009]. Treatment schedules, policies and drug doses as advocated by a number of national and international bodies often differ. Tables 2 and 3 compare drug doses and regimens recommended in the UK, the USA and by WHO [WHO, 2010; National Collaborating Centre for Chronic Conditions, 2006; Pickering et al. 2009]. Traditionally, anti-TB regimens have included bactericidal and bacteriostatic drugs that have required treatment for long periods, between 18 and 24 months. More recently, multidrug regimens have been used with more rapid microbiological cure rates that allow shorter durations of therapy (short-course chemotherapy). Isoniazid, rifampicin and pyrazinamide are mainstays of anti-TB therapy. Other agents often used in children include streptomycin and ethambutol.
Recommended doses (mg/kg) of first-line standard antituberculous drugs for children [World Health Organization, 2010; National Collaborating Centre for Chronic Conditions, 2006; Pickering et al. 2009].

Recommended treatment schedules for tuberculosis disease in children [World Health Organization, 2010; National Collaborating Centre for Chronic Conditions, 2006; Pickering et al. 2009].

If resistance suspected.
E, ethambutol; H, isoniazid; NICE, National Institute for Health and Clinical Excellence; R, rifampicin; S, streptomycin; TB, tuberculosis; Z, pyrazinamide.
Adverse reactions to anti-TB therapy occur in children on therapy, but generally the drugs are well tolerated in children. Gastrointestinal reactions such as nausea, vomiting and abdominal pain are common, particularly in the first few weeks of therapy. In most cases these reactions can be managed symptomatically. Isoniazid and rifampicin may be hepatotoxic causing elevation of serum aminotransferase levels (significant if three or more times the upper limit of normal). These hepatotoxic abnormalities are rarely severe in children and modest increases in aminotransferases generally resolve spontaneously. All drugs used in treating TB can cause skin rash, which is usually minor and may be managed symptomatically. Isoniazid has also been associated with symptomatic pyridoxine deficiency, particularly in severely malnourished children. Supplemental pyridoxine is indicated in these malnourished children as well as in breastfeeding infants (dose 5 mg in infants from birth to 1 month, 5–10 mg in infants and children under 12 years and 10 mg in children 12–18 years). Pyrazinamide is generally well tolerated in children and rarely causes hepatic dysfunction. Ethambutol has been associated with retrobulbar neuritis, although it appears to be safe in children older than 5 years of age at a dosage of 15 mg/kg/day and also in younger children without undue fear of side effects [Trebucq, 1997]. It is often appropriate to obtain a baseline ophthalmological assessment in younger children before starting ethambutol therapy. This should be repeated after 1–2 months.
Compliance is a major determinant of the success of drug treatment – compliance of the physician in prescribing the optimum appropriate regimen and monitoring it, and compliance of the patient in taking the medication as prescribed. Compliance in children is further compounded by the fact that children may have mechanical difficulties in taking medications, many of which are not specifically packaged or produced in paediatric formulations. Difficulties with taste, consistency of formulations and gastrointestinal toxicity may be important factors in children that may dramatically affect treatment compliance.
Directly observed therapy (DOT, short-course chemotherapy) has become a cornerstone for TB control across the globe. DOT is only one of five key elements. These include government commitment to sustained TB control activities, case detection by sputum smear microscopy, standardized treatment regimens of 6–8 months for all confirmed smear-positive cases with DOT for at least 2 months, a regular uninterrupted supply of essential anti-TB drugs and a standardized recording and reporting system. DOT has been adopted by 148 of 210 countries worldwide and almost 55% of the world’s population lives in countries providing DOT. WHO recommends that the DOT strategy is applicable to all patients with TB, including children in whom high success rates (over 95%) can be achieved [WHO, 1994]. Many countries have adopted a universal DOT policy, whereas others such as the UK use a selective policy for those who may be unreliable in taking their therapy [National Collaborating Centre for Chronic Conditions, 2006].
Multidrug-resistant tuberculosis
Single, multiple and multidrug resistance is increasing worldwide. Isoniazid resistance has been found in 6.8–7.2% of isolates in children under 15 years in England and Wales from 1995 to 1999. Multidrug resistance (defined as resistance to isoniazid and rifampicin) over the same period was 0.5–0.7%. Higher levels of resistance occur in ethnic minority groups, especially those from the Indian subcontinent and sub-Saharan Africa. As children have lower rates of TB isolation, MDR TB is often initially only identified in the adult index case or in other contacts.
Treatment of patients with drug-resistant TB should only be carried out by specialists with appropriate experience in the management of such cases. The commonest isolated drug resistance is to isoniazid, which is why it is particularly important to add ethambutol as a fourth agent when isoniazid resistance is suspected, or in those patients at higher risk of resistance. Treatment should be continued for at least 9–12 months, initially with rifampicin, pyrazinamide and ethambutol for 2 months followed by rifampicin and ethambutol for the complete duration of therapy. Isolated drug resistance to other first-line drugs is unusual and appropriate therapy must begin based on recommended guidelines [National Collaborating Centre for Chronic Conditions, 2006]. Rifampicin resistance most commonly occurs in conjunction with isoniazid resistance (called MDR TB). Treatment should be carried out by a specialist with substantial experience in managing complex resistant cases and only in hospitals with appropriate isolation facilities. Such treatment should also be monitored closely for drug toxicity but more importantly to ensure compliance. In most cases treatment will involve five or more drugs and for durations of at least 2 years. Several alternative anti-TB drugs may need to be used, although the efficacy of these drugs has not been evaluated in children. The drugs that have been used previously include injectable aminoglycosides (streptomycin, amikacin, capreomycin, kanamycin), ethionamide/prothionamide, cycloserine, quinolones (ciprofloxacin, ofloxacin, moxifloaxacin), rifabutin, macrolides (azithromycin, clarithromycin), and para-amino salicylic acid. Newer classes of drugs, such as diarlquinolone TMC207, are being studied for the treatment of MDR TB, but with limited data in children [Diacon et al. 2009].
Corticosteroids
Corticosteroids have been found to be beneficial in situations when the host response to M. tuberculosis contributes to significant tissue damage. Corticosteroids have been shown to significantly decrease mortality and long-term neurological sequelae in patients with TB meningitis [Girgis et al. 1991; Schoeman et al. 1997]. Children with bronchial obstruction due to enlarged lymph nodes may also benefit from corticosteroid therapy [Nemir et al. 1967]. Corticosteroids may also be of benefit in extensive pulmonary TB, pericardial effusion and pleural effusion. A dosage of 1–2 mg/kg (maximum of 60 mg) for 4–6 weeks is recommended, followed by a period of weaning doses.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
The authors declare no conflicts of interest in preparing this article.
