Abstract
Keywords
Introduction
Budesonide/formoterol (Symbicort® Turbuhaler®) 1 maintenance and reliever therapy (Symbicort SMART®) is an established treatment concept for patients with moderate to severe asthma. It has been found to reduce severe exacerbations and maintain similar asthma control or result in better daily asthma control at a lower drug load compared with traditional strategies of inhaled corticosteroid/long-acting β2-agonist (ICS/LABA) plus a short-acting β2-agonist (SABA) [Bateman et al. 2010; Buhl and Vogelmeier, 2007]. The safety of budesonide/formoterol maintenance and reliever therapy has been well documented [Sears and Radner, 2009].
To achieve good asthma control, patients with severe asthma may require high doses of ICS, often in combination with a LABA. Although the budesonide/formoterol maintenance and reliever therapy concept in general has demonstrated good asthma control as measured by the Global Initiative for Asthma guidelines, [Bateman et al. 2010; Global Initiative for Asthma, 2009] a question remains whether this control can be maintained when these high-dose ICS patients are treated according to the budesonide/formoterol maintenance and reliever therapy concept when the daily ICS dose may be reduced.
We have reported the results of a pan-European budesonide/formoterol maintenance and reliever therapy (EuroSMART) study [Aubier et al. 2010]. This was an open-label, randomized 6-month study comparing two maintenance doses of budesonide/formoterol, 160/4.5 µg 1 × 2 and 2 × 2, in 8424 asthmatics ≥18 years of age with symptoms when treated with ICS without or with concomitant use of LABA. Among the total study population (8053 randomized patients), 1339 patients were prescribed a daily dose of 1600 µg of budesonide (or equivalent doses) or more at baseline and of these, a total of 110 patients used doses above 2000 µg/day. These patients were considered to be a group at potential risk when shifting their treatment from daily high-dose ICS (HD stratum) to the budesonide/formoterol maintenance and reliever therapy concept. In this pre-planned secondary analysis, this HD stratum was compared with a medium-dose ICS stratum (MD stratum;
Material and methods
Study design
This was an open-label, randomized, parallel-group, 6-month, multicentre, multinational study performed in 14 European countries in patients with moderate to severe asthma who were symptomatic despite daily use of an ICS with or without LABA. Patients were 18 years or older and had at least a 6-month documented history of asthma according to the American Thoracic Society definition [American Thoracic Society, 1987]. Patients had a history of SABA use for symptom relief during the last month and must have been on maintenance therapy with ICS for at least 1 month at a constant daily dose of at least 500 µg beclomethasone dipropionate, or another ICS in an equivalent dose [Global Initiative for Asthma, 2009].
Patients older than 40 years of age and with a smoking history of 10 pack-years or more were excluded, as were those with a diagnosis of chronic obstructive pulmonary disease. Further inclusion and exclusion criteria have been previously reported [Aubier et al. 2010].
After the run-in period, eligible patients were randomized to one of the following two treatments: budesonide/formoterol maintenance and reliever therapy 160/4.5 µg per dose, one dose twice daily (1 × 2 group) or two doses twice daily (2 × 2 group). The primary variable was the time to the first severe asthma exacerbation.
Assessments
There were four visits in the study: before and after the 2-week run-in period (Visits 1 and 2), after 3 months’ treatment (Visit 3) and after 6 months’ treatment (Visit 4). During the run-in period and during the 14 days prior to Visits 3 and 4, the patients recorded in a notebook the number of as-needed inhalations, the number of inhalations taken as maintenance treatment, asthma symptoms during 24 hours and night-time awakenings due to asthma.
The five-item Asthma Control Questionnaire (ACQ5) [Juniper et al. 1999] excluding forced expiratory volume in 1 second (FEV1), as FEV1 was not measured at all clinics, was filled in and use of SABA (as budesonide/formoterol should be used as reliever medication) was recorded via self-administration at Visits 2, 3 and 4. In large studies, the exclusion of these two questions has been shown not to alter the validity and accuracy of the questionnaire [Juniper et al. 2005].
The primary efficacy variable was time to first severe asthma exacerbation, defined as deterioration in asthma requiring oral or parenteral corticosteroids either for at least 3 days, or leading to hospitalization, or emergency room visit or other patient-initiated unscheduled visits to a healthcare centre. The exacerbation data were retrospectively obtained from patient records and from the patients themselves. Compliance with treatment was not formally monitored as the study aimed to mimic a real-life clinical practice as much as possible. Safety was evaluated as reports of serious adverse events and adverse events leading to discontinuation from the study.
The study was performed according to Good Clinical Practice, the Declaration of Helsinki and the study protocol was approved by all local ethics committees. All patients gave their written informed consent for participation
Statistical analysis
Time to first asthma severe exacerbation was compared using a Cox proportional hazard model, stratified by country and with treatment as a factor. The total number of exacerbations was compared between the treatments using a Poisson regression model controlling dispersion with country and treatment as factors and total time in study as an offset variable. The change in ACQ5 scores, daytime symptoms, awakenings and lung function were analysed using an analysis of variance (ANOVA) model with baseline value as covariate. All analyses were performed separately for each stratum.
Results
Baseline characteristics of patients using a high, medium or low dose of inhaled steroids expressed as budesonide equivalent.

Mean values (standard deviation) are shown unless stated otherwise. The
ACQ5, five-item Asthma Control Questionnaire; ANOVA, analysis of variance; BMI, body mass index; FEV1, forced expiratory volume in 1 second; ICS, inhaled corticosteroid; LABA, long-acting β2-agonist; N/A, not applicable; PEF, peak expiratory flow.
Rows 1, 7, 8, 9, 11, 12 and 13: all means are different. Rows 4, 10 and 14: ≥1600 µg stratum different from 400 µg and 401–1599 µg strata, which are similar. Row 5: ≤400 µg stratum different from 401–500 µg and ≥1600 µg strata.
Patients with the highest use of ICS at baseline had also experienced more exacerbations in the preceding year than the patients in the MD and LD strata. Exacerbation rates were 135, 139 and 185 per 100 patients/year in the LD, MD and HD strata, respectively (Figure 1).
Number of exacerbations per 100 patients during the year before study start according to baseline steroid dose.
Use of study medication
Mean daily doses of budesonide used during the study.

Expressed as budesonide equivalent.
Exacerbations
Exacerbations experienced during the study in the three strata are shown in Figure 2, expressed as rate per patient, per year. The fewest exacerbations were seen in the LD stratum followed by the MD and HD strata (0.094, 0.172 and 0.268 per patient, per year with 1 × 2 treatment). In the 2 × 2 treatment group, exacerbation rates were 0.076, 0.138 and 0.232 in the LD, MD and HD strata, respectively. Thus, in all three strata, there were fewer exacerbations in the 2 × 2 treatment groups than in the 1 × 2 treatment groups. The reduction in rate of exacerbations with the higher dose ranged from 13.3% (HD stratum) to 19.6% (MD stratum), but in no case was the difference between the treatment groups statistically significant (Figure 2).
Exacerbations during the study calculated as yearly rate per 100 patients.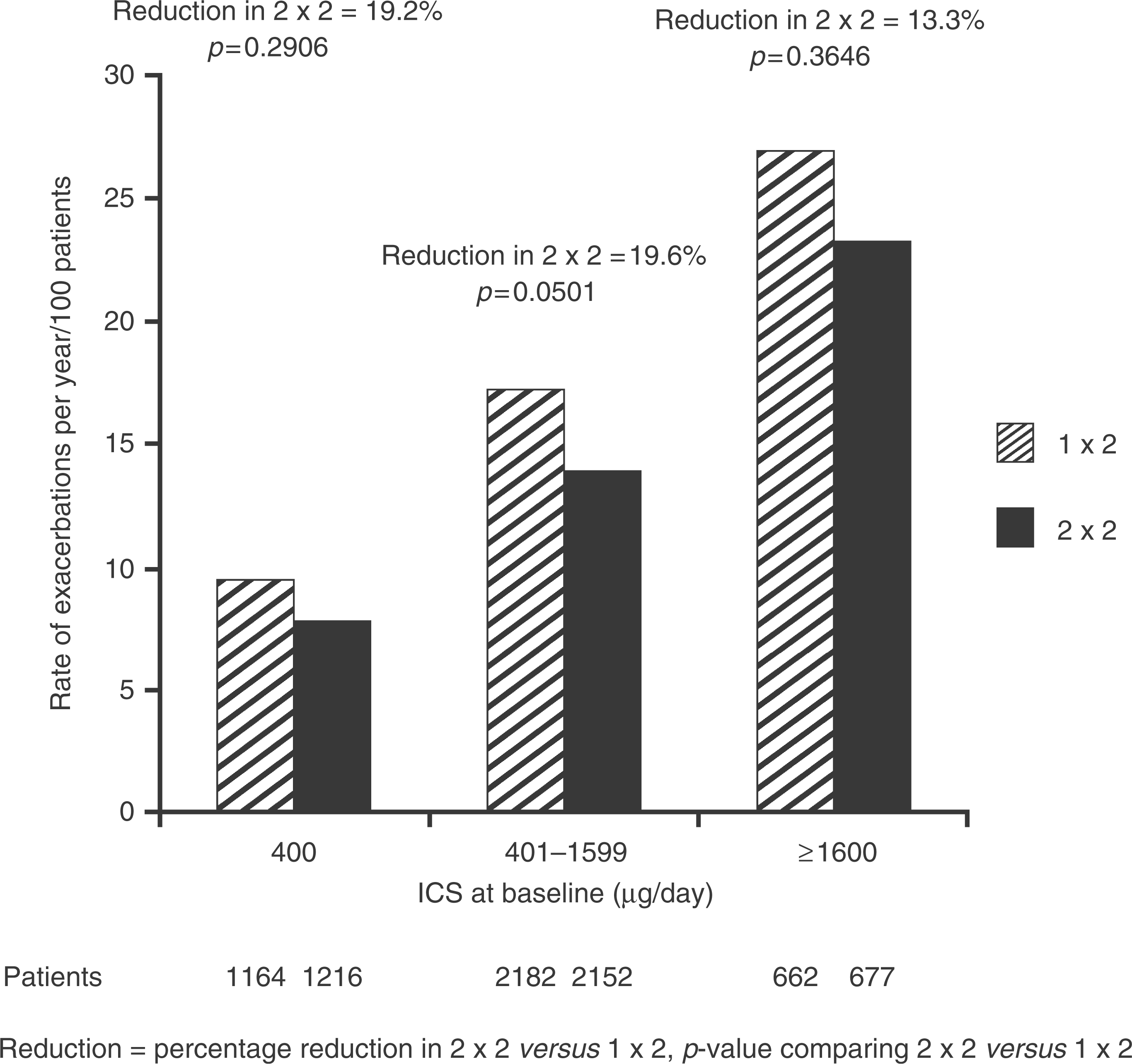
In the total study population, the time to first severe asthma exacerbation was prolonged by 18% with 2 × 2 Time to first exacerbation in high-dose ICS (≥1600 µg/day), medium-dose ICS (401 -1599 µg/day) and low-dose ICS (400 µg/day) patients.
ACQ5 scores
With both treatments (1 × 2 and 2 × 2), the mean changes in ACQ5 score exceeded 0.5 points, i.e. a clinically important change, in all three strata (Figure 4). The mean ACQ5 value improved more in the HD stratum with both 1 × 2 and 2 × 2 treatments compared with the other strata ( Change in ACQ5 score during the study.
In total, 26% of the patients in the study achieved an ACQ5 score of <0.5 points. In the LD stratum, 30% of the patients achieved this score. In the MD and HD strata, the corresponding results were 25% and 22%. In a total of 28% of the patients, ACQ5 score remained above 1.5 points. In all three strata, the percentage of patients with ACQ5 score >1.5 points at the end of the study was 22%, 30% and 35% in the LD, MD and HD strata, respectively, corresponding to an improvement from baseline of 33%, 30% and 35%, respectively.
Changes from baseline in patient-reported outcomes
Asthma symptoms
Response to treatment in high-dose ICS patients compared with the response in medium- or low-dose ICS patients at baseline.

Change from short-acting β2-agonist doses to budesonide/formoterol doses.
No difference between the 400 µg stratum and the 401–1599 µg stratum.
ICS, inhaled corticosteroid.
Awakenings
The greatest reductions in mean number of night-time awakenings per week were seen in the HD stratum but no difference was observed between 1 × 2 and 2 × 2 treatments (
With both 1 × 2 and 2 × 2 treatments, the reductions were greater in the HD stratum compared with the corresponding treatments in the MD and LD strata (
Use of as-needed medication
Before entering the study, all patients used a SABA as reliever medication. During the study, they used budesonide/formoterol (the same ICS/LABA combination as for maintenance therapy) due to the treatment concept of the study. The number of as-needed inhalations was reduced in all strata with both treatments, with greater reductions being seen with 2 × 2 treatment compared with 1 × 2 in all groups (
Safety
In the HD stratum, there were 13 (2.0%) serious adverse events in the 1 × 2 group and 10 (1.5%) in the 2 × 2 group. In the combined MD plus LD stratum, there were 64 events (1.9%) with 1 × 2 and 78 (2.3%) with 2 × 2 treatment. Eighteen patients (2.7%) discontinued the study, both in the 1 × 2 and 2 × 2 treatment groups of the HD stratum, due to adverse events. In the MD plus LD stratum, 52 patients (1.6%) discontinued treatment in the 1 × 2 group and 75 patients (2.2%) in the 2 × 2 group. There were a total of four deaths in the study; one in the HD, two in the MD and one in the LD stratum. None of the deaths were related to asthma or to the study medication.
Discussion
This is a pre-planned secondary analysis of the large pan-European study comparing two maintenance doses of budesonide/formoterol (160/4.5 µg one or two doses twice daily in addition to as-needed medication). The primary data have been published previously [Aubier et al. 2010]. It was shown that a post-bronchodilator FEV1 <80% predicted normal was the baseline factor indicating a better response to 2 × 2 compared with a 1 × 2 maintenance dose. In this study, the ICS doses used by the patients at baseline covered a wide range including a large number of patients using high doses defined as doses ≥1600 µg of budesonide.
Despite these high ICS treatment levels at baseline, patients in the HD stratum were uncontrolled on study entry, as shown by their high mean ACQ5 score (2.17) and the frequency of their daily and night-time asthma symptoms during the run-in period.
The study showed that treatment of patients using ≥1600 µg of budesonide or equivalent at baseline (with or without LABA) could be changed to budesonide/formoterol 1 × 2 or 2 × 2 (plus as-needed) without loss of asthma control measured by ACQ5 or time to first severe exacerbation, despite a reduction in overall ICS dose.
It is interesting to note that patients on high-dose ICS medication at study entry did not have more exacerbations in the study, even when treated with the 1 × 2 regimen, a much lower total daily dose of ICS. A reason for this could be that the patients were overtreated prior to the study and could have their ICS dose lowered without resulting in an exacerbation. However, this appears unlikely as the patients were symptomatic at entry, had experienced more exacerbations during the preceding year despite their high-dose ICS use compared with the MD and LD patients, and had a reduction in their ACQ5 scores throughout the study. Two possible explanations remain: improved adherence with medication during the study, or an additional effect of the as-needed use of a LABA and an ICS, rather than inhaled SABA, when deemed necessary by the patient. In the MD and LD strata, it was not expected that there would be large changes when these patients were switched from their previous ICS dose plus LABA to the budesonide/formoterol maintenance and reliever therapy concept. However, these patients did benefit from this therapy, and more so from the 2 × 2 treatment compared with the 1 × 2 treatment, in terms of changes in ACQ5 scores and reductions in exacerbations, confirming the effectiveness of the treatment concept. It is also interesting to note that there was no difference in reduction in ACQ5 score seen in the HD stratum between patients randomized to 1 × 2 or 2 × 2 treatment. We have no explanation for this. The total daily doses of ICS were quite different in the two treatment groups: 498 µg and 764 µg in the 1 × 2 and 2 × 2 groups, respectively. One explanation may be the number of patients in these dose strata: in the LD stratum, there was a larger number of patients and a small but statistically significant difference was found between the dose groups. With more patients in the HD stratum, we may have seen a similar difference there.
It is known that a dose–response for ICS in asthma exists but that the dose–response curve flattens as dose increases [Miyamoto et al. 2000; Busse et al. 1998]. In unselected asthma populations, the greatest benefit from ICS therapy is usually achieved with doses ≤ 500 µg per day [Powell and Gibson, 2003], but the individual response varies considerably [Szefler et al. 2002]. It might also be that some patients have been prescribed unnecessarily high doses and no subsequent attempts have been made to reduce the dose. It is known that it may take more than a year to determine the lowest effective ICS dose in the individual patient [Selroos et al. 1994]. In our naturalistic study, we have no information about what dose the patients actually needed but suppose that most of the patients were using a clinically appropriate dose. The total ICS doses used during the study were much lower than the doses prescribed at baseline. The most extreme change in ICS dose, in the 1 × 2 group of the HD stratum, did not result in asthma deterioration or lost asthma control. In contrast, asthma control improved in the majority of the patients, perhaps due to improved adherence to maintenance medication seen in clinical trials. It may also be that as-needed use of the LABA and the ICS contributed to the improvement in asthma control as reported in previous studies [Rabe et al. 2006].
Limitations of the study include the retrospective assessment of exacerbations in the previous year, although they were, in most cases, based on patient records. Another limitation is the uncertainty about the minimum effective dose of ICS at baseline. We cannot exclude the suggestion that some patients were overtreated when entering the study. However, since all patients had to be symptomatic at baseline, this seems unlikely.
No safety concerns were raised in the study. There were no further serious adverse events or discontinuations due to adverse events in the group of patients using high doses of ICS at baseline who switched to much lower total daily doses of ICS.
We conclude that patients selected for management with the budesonide/formoterol maintenance and reliever therapy can be started on the standard 1 × 2 regimen, regardless of their baseline steroid dose. However, the level of post-bronchodilator lung function should always be considered [Aubier et al. 2010].
Footnotes
Acknowledgements
We thank Professor Olof Selroos, MD, PhD, of SEMECO AB, who drafted the manuscript for which he received financial support from AstraZeneca, and Dr Manda Gent of MediTech Media, who provided editorial assistance funded by AstraZeneca.
Funding
This study was funded by AstraZeneca.
Conflict of interest statement
M. Aubier has participated as a speaker in scientific meetings or courses organized and financed by various pharmaceutical companies (GlaxoSmithKline, AstraZeneca, Novartis, Pfizer, Nycomed).
J. Haughney has received reimbursements for attending symposia, fees for speaking and organizing educational events, funds for research, and fees for consulting from AstraZeneca (the sponsor of this study). He has received support from other pharmaceutical companies: Boehringer Ingelheim, GlaxoSmithKline, Merck Sharp & Dohme, Mundipharma, Novartis, Nycomed, sanofi-aventis and Teva.
O. Selroos has, during the last 5 years, received consultation fees from AstraZeneca and Galenica, fees for speaking at medical meetings organized by AstraZeneca and fees for drafting publications for AstraZeneca, Orion Pharma and Schering-Plough. O. Selroos has shares in AstraZeneca.
O. van Schayck has received a fee for consulting from AstraZeneca.
R. Buhl has received reimbursement for attending scientific conferences, and/or fees for speaking and/or consulting from AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Novartis, Nycomed and Pfizer. The Pulmonary Department at Mainz University Hospital received financial compensation for services performed during participation in clinical trials organized by various pharmaceutical companies.
T. Ekström is a retired employee of AstraZeneca and holds stocks in the company. J. Ostinelli is a full-time employee at AstraZeneca and holds stocks in the company.
1
Neither the Symbicort SMART posology nor the dry powder formulation, Turbuhaler, is currently approved in the US.
