Abstract
Background:
A considerable proportion of patients have features of both asthma and chronic obstructive pulmonary disease (COPD) simultaneously, called asthma–COPD overlap (ACO).
Objectives:
The aim of this study was to identify heterogeneity of ACO from a cohort of patients with severe asthma and COPD using the same diagnostic criteria.
Design:
We used the International Severe Asthma Registry (ISAR) and the Korean COPD Subgroup Study (KOCOSS) to evaluate clinical characteristics of ACO from each cohort.
Methods:
We classified subjects into four groups: (1) pure severe asthma, (2) ACO from the severe asthma cohort, (3) ACO from the COPD cohort, and (4) pure COPD. ACO was defined by satisfying extreme bronchodilator response (BDR) >15% and 400 ml and/or blood eosinophil count ⩾300 /µL in patients aged 40 years or older and post-BD forced expiratory volume in 1 s (FEV1)/forced vital capacity (FVC) ratio <0.7.
Results:
The ACO group had 25 (23%) of 111 in the ISAR cohort and 403 (23%) of 1781 in the KOCOSS cohort. The ACO from the COPD cohort was older with more males and more smokers, but had similar degree of airflow limitation compared with the ACO from the severe asthma cohort. ICS-containing inhaler treatment was prescribed for all severe asthma subjects, but only for 43.9% of ACO subjects from the COPD cohort. Compared with patients having pure severe asthma, the risk for exacerbation was comparable in ACO either from severe asthma or COPD cohort [adjusted odds ratio (aOR): 1.54, 95% CI: 0.22–10.95 or aOR: 2.15, 95% CI: 0.59–7.85].
Conclusion:
The prevalence of ACO was similar in severe asthma and COPD cohorts applying identical diagnostic criteria. ACO from the severe asthma cohort was similar to ACO from the COPD cohort in terms of lung function and exacerbation risk.
Introduction
Traditionally, asthma and chronic obstructive pulmonary disease (COPD) were believed to be distinct diseases with different pathophysiologies. However, some patients exhibit both features of asthma and COPD simultaneously called asthma–COPD overlap (ACO). Since the first mention of the concept of ACO, 1 there have been growing interests on ACO patients. Although many studies have been published on ACO, it is difficult to obtain consistent information for those individuals because they are mutually excluded from both clinical trials of asthma and COPD. 2 In addition, it is difficult to plan a new clinical trial for ACO patients. Although inhaled corticosteroids (ICS)–containing maintenance inhaler therapy can be given to individuals with a high probability of ACO, treatment decisions for them are lacking. This is mainly attributable to the lack of unified definition of ACO. Most studies defined ACO by their home-made diagnostic criteria. Thus, prevalence of ACO varied across studies with a wide range of 9–55%.3–5 However, the prevalence of ACO was around 20% in two different nationwide COPD cohorts when the same diagnostic criteria were applied. 6
Interests in ACO continue to increase due to their unique clinical characteristics. Patients with ACO have more severe respiratory symptoms, lower quality-of-life status, lower lung function, higher risk of exacerbation, and higher mortality than patients with COPD5,7,8 or asthma9,10 alone. So far, studies on ACO have compared ACO in either the asthma or COPD cohort. A representative study showing clinical features and prognosis of ACO between asthma and COPD is the Copenhagen City Heart Study. 11 ACO was defined as a case when self-reported asthma and post-bronchodilator (BD) forced expiratory volume in 1 s (FEV1) to forced vital capacity (FVC) ratio of less than 0.7 were satisfied. ACO was then classified into ACO with early asthma onset or with late asthma onset based on whether the age at the time of asthma diagnosis was before 40 years. Mortality was the highest in ACO with late-onset asthma, followed by COPD, ACO with early-onset asthma, and asthma compared with never smokers without disease. This implies that subjects with ACO might have different clinical features compared with subjects with asthma or COPD, even within ACO. However, there are few studies comparing ACO from asthma and ACO from COPD cohort in real clinical practice. Thus, the aim of this study was to compare clinical characteristics of ACO from severe asthma and COPD cohort defined by identical diagnostic criteria.
Methods
Study population
We used data from the Korean COPD subgroup study (KOCOSS) cohort and the International Severe Asthma Registry (ISAR) cohort. KOCOSS is an ongoing, prospective observational cohort of COPD, which has been recruiting patients with COPD from 48 referral hospitals in Korea since 2012. Subjects aged ⩾40 years who were diagnosed with COPD by pulmonologist based on chronic respiratory symptoms and spirometry revealed fixed airflow limitation (post-BD FEV1 to FVC ratio less than 0.7) were enrolled. The ISAR cohort is a multi-country, observational prospective cohort on adult (18 years or older) patients with severe asthma receiving Global Initiative for Asthma (GINA) step 5 or uncontrolled asthma at GINA step 4 according to the 2018 GINA recommendation.12,13 Detailed inclusion criteria of ISAR were previously published. 13 In this study, we only included Korean patients in the ISAR cohort to avoid race and ethnic differences.
Clinical data collection
Detailed demographic, smoking status, medical history, and comorbid conditions were collected for both KOCOSS and ISAR cohorts. In terms of patient-reported symptoms and quality-of-life status, there was a difference between cohorts because these items were collected with a focus on asthma or COPD alone. The KOCOSS cohort included data on severity of dyspnea assessed by modified Medical Research Council Dyspnea Scale (mMRC), quality-of-life status expressed by both the COPD assessment test (CAT) and the St George’s Respiratory Questionnaire (SGRQ). Evaluation of exercise capacity through a 6-min walking distance (6MWD) was also included in the KOCOSS cohort. On the contrary, the ISAR cohort included item on asthma control status. For both cohorts, type of inhaler therapy was collected. Data on biologics were only restricted to a few asthmatic patients.
Clinical measurement
Pulmonary function tests (PFTs) were performed under stable conditions at enrollment. PFTs were performed according to the ATS/ERS guidelines and parameters such as FVC, FEV1, and percent-predicted values of each parameter and ratio of FEV1/FVC were collected. All the measurements were checked after the use of BD. 14 A positive bronchodilator response (BDR) was defined as having a post-BD improvement in FEV1 of ⩾12% and ⩾200%.
Definition of ACO
To compare the two cohorts, individuals aged 40 years or older who had post-BD FEV1/FVC <0.7 were included in this study. ACO was defined as who has BDR >15% and 400 ml and/or blood eosinophil count ⩾300 /μL. These criteria were equally applied to both ISAR and KOCOSS cohorts.
Risk of exacerbation
In the ISAR cohort, exacerbation was defined as acute deterioration when rescue steroid treatment was required during the past 1 year. On the contrary, in the KOCOSS cohort, the severity of exacerbation was separately collected every 6 months. Severe exacerbation was defined as acute deterioration requiring emergency department visit and resultantly hospitalization. Exacerbation requiring systemic corticosteroids and/or antibiotics treatment in outpatient clinic was defined as moderate exacerbation. In this study, we evaluated the risk of moderate-to-severe exacerbation requiring systemic steroid treatment, which could be applied equally to both cohorts within 1 year.
Statistical analysis
All data are presented as numbers (%) for categorical variables and mean ± standard deviation (SD) for continuous variables. We classified subjects into four groups: pure severe asthma, ACO from the severe asthma cohort, ACO from the COPD cohort, and pure COPD. One-way analysis of variance (ANOVA) and chi-square test were used to analyze continuous and categorical variables, respectively. The risk of exacerbation during the first year was determined by performing logistic regression analysis. Covariates including age, sex, body mass index (BMI), smoking status, post-BD FEV1 at baseline, history of exacerbation during the year prior to enrollment, and any ICS use were adjusted in the multivariable analysis. All tests were two-sided and p < 0.05 was taken to indicate statistical significance. All statistical analyses were performed using STATA software version 16 (StataCorp, College Station, TX, USA).
Results
Baseline characteristics and comorbidities
Among 161 patients in the severe asthma cohort and 2181 patients in the COPD cohort, 111 and 1781 patients were eligible for ACO analysis, respectively (Figure 1). Among eligible patients, 428 individuals were identified as ACO, of which 413 and 13 were classified according to the eosinophil count criteria and the extreme BDR criteria, respectively. Two patients satisfied both criteria. The prevalence of ACO was 22.5% (25 out of 111) in the severe asthma cohort and 22.6% (403 out of 1781) in the COPD cohort (Figure 1). Subjects in the ACO from the COPD cohort were more likely to be males (96% versus 48.0%, p < 0.01) and older (68.4 ± 7.7 years versus 57.4 ± 10.9 years, p < 0.01). They had lower BMI (23.1 ± 3.5 versus 25.5 ± 5.0, p < 0.05). They had more current or ex-smokers with higher smoking pack-years (45.7 ± 25.5 versus 22.2 ± 9.0, p < 0.05) compared with ACO from the severe asthma cohort (Table 1). Blood eosinophil count (790 ± 396.9 /µL versus 530 ± 365.7 /µL, p < 0.01) and fractional exhaled nitric oxide (FENO) level (57.3 ± 45.0 parts per billion (ppb) versus 28.7 ± 18.1 ppb, p < 0.01) were higher in ACO from the severe asthma cohort than in ACO from the COPD cohort. Difference of past exacerbation rate was not significant among the four groups (pure severe asthma, ACO from severe asthma cohort, ACO from COPD cohort, and pure COPD). Asthma control status showed no significant difference between pure severe asthma and ACO from severe asthma cohorts. Furthermore, severity of dyspnea, quality-of-life status, and exercise capacity scores did not differ between pure COPD and ACO from COPD cohorts.
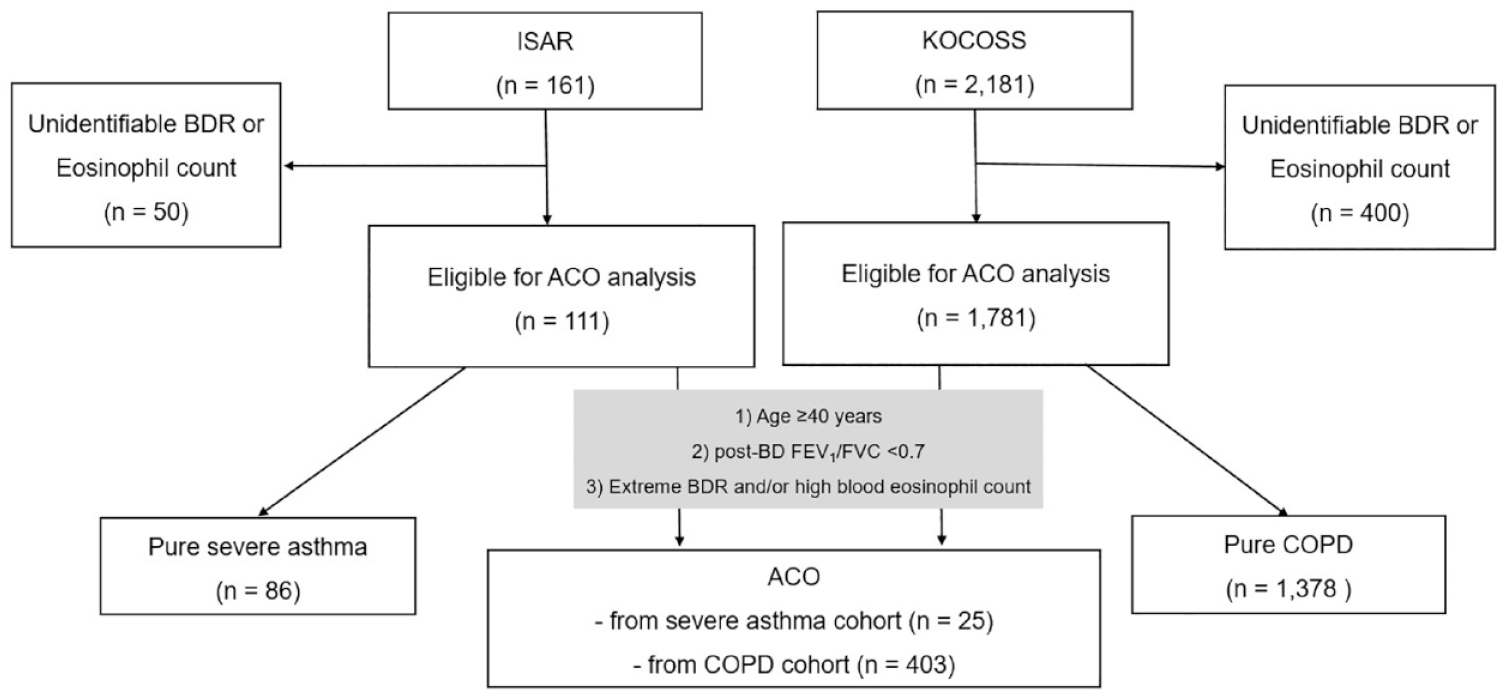
Flow of the study.
Baseline clinical characteristics.

ACO, asthma–chronic obstructive pulmonary disease (COPD) overlap; BMI, body mass index; CAT, COPD assessment test; COPD, chronic obstructive pulmonary disease; FENO, fractional exhaled nitric oxide; mMRC, modified Medical Research Council; SGRQ, St. George’s Respiratory Disease Questionnaire; 6MWD, 6-min walk distance.
ACO from severe asthma cohort versus ACO from COPD cohort.
p < 0.05; **p < 0.001.
Comparison of comorbid conditions are shown in Table 2. Allergic rhinitis and atopy were more frequent in ACO from the severe asthma cohort than in ACO from the COPD cohort. Only 40.9% of ACO from the COPD cohort were found to have self-reported asthma diagnosis. Age of asthma diagnosis was more likely to be older in the COPD cohort (p for trend < 0.01). However, there was no significant difference in age of diagnosis between ACO from severe asthma cohort and COPD cohort (44.4 years versus 51.4 years, p = 0.109). Diabetes and hypertension were more frequent in the COPD cohort, whereas prevalence of heart failure, ischemic heart disease, and osteoporosis did not show statistical significance.
Comparison of comorbid conditions.
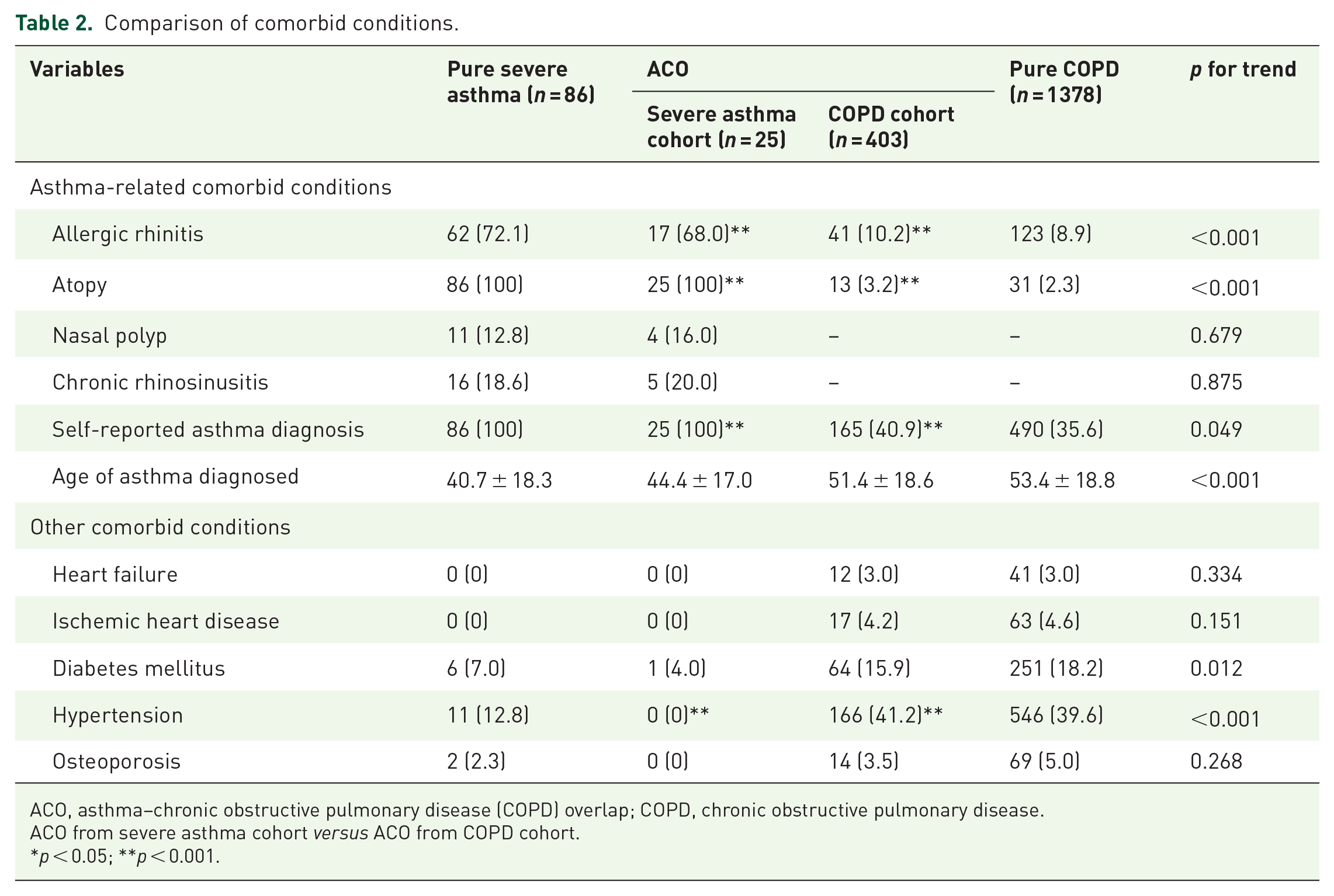
ACO, asthma–chronic obstructive pulmonary disease (COPD) overlap; COPD, chronic obstructive pulmonary disease.
ACO from severe asthma cohort versus ACO from COPD cohort.
p < 0.05; **p < 0.001.
Lung function test results
Post-BD FEV1 tended to be poorer in patients in ACO from the severe asthma cohort than that in the pure severe asthma group, whereas there were no differences between ACO from the COPD cohort and pure COPD (p for trend < 0.01). There was no significant difference in FEV1 between ACO from severe asthma and COPD patients (61.1 ± 16.9 versus 58.4 ± 18.4, mean % of predicted value, respectively; Table 3). FVC was higher in the ACO from severe asthma cohort than in the ACO from the COPD cohort (90.6 ± 20.1 versus 80.3 ± 16.6, mean % of predicted value, respectively). The FEV1/FVC ratio showed a tendency to be lower in the COPD cohort. However, it showed no significant difference between ACO from severe asthma and COPD cohort.
Post-bronchodilator spirometry results.

ACO, asthma–chronic obstructive pulmonary disease (COPD) overlap; BDR, bronchodilator response; COPD, chronic obstructive pulmonary disease; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; PC20, provocation dose causing a 20% decline in FEV1.
ACO from severe asthma cohort versus ACO from COPD cohort.
p < 0.05; **p < 0.001.
Use of inhalers
Most patients in the severe asthma cohort (94.2% in pure severe asthma and 80% in ACO) were prescribed with ICS-containing inhalers, whereas 36.7% of those in the ACO from COPD cohort used ICS-containing inhalers (Figure 2). Fifty-six percent of ACO from the severe asthma cohort and less than 30% of ACO from the COPD cohort were prescribed with triple therapy (ICS/LABA/LAMA). There was no significant difference in the use of ICS-containing inhaler between ACO from COPD cohort and pure COPD (36.7% versus 34.5%, p = 0.698). Biologics including anti-IgE, anti-IL4, and anti-IL5) were used only for asthmatics, showing no difference between pure severe asthma and ACO (15.1% versus 24%, p = 0.299).

Prescription status of inhaler treatment.
Risk of exacerbations
Incidence of moderate-to-severe exacerbation in pure severe asthma, ACO from severe asthma cohort, ACO from COPD cohort, and pure COPD were 22.7%, 37.5%, 47.0%, and 38.9%, respectively (Table 4). Compared with patients with pure severe asthma, the risk for exacerbation had a tendency to be higher in ACO either from severe asthma or COPD cohort [adjusted odds ratio (aOR): 1.54, 95% confidence interval (CI): 0.22–10.95 or aOR: 2.15, 95% CI, 0.59–7.85], although the difference did not reach statistical significance. Compared with the ACO from severe asthma cohort, the risk of exacerbation in ACO from COPD cohort was not significantly different (Supplementary Table 1).
Risk of exacerbation.

ACO, asthma–chronic obstructive pulmonary disease (COPD) overlap; AE, acute exacerbation; BD, bronchodilator; BMI, body mass index; CI, confidence interval; COPD, chronic obstructive pulmonary disease; FEV1, forced expiratory volume in 1 s; ICS, inhaled corticosteroid; OR, odds ratio.
p < 0.05.
Model 1: Adjusted for age, sex, and BMI.
Model 2: Adjusted for age, sex, BMI, and any ICS use.
Model 3: Adjusted for age, sex, BMI, smoking status, post-BD FEV1 % of predicted value, past AE history.
Model 4: Adjusted for age, sex, BMI, smoking status, post-BD FEV1 % of predicted value, past AE history, and any ICS use.
Discussion
Since Gibson and Simpson 1 first introduced disease entity of ACO in 2008, a large number of studies have been performed to reveal its clinical characteristics and optimal management plan.1,15 The concept of ACO is a step further from the Dutch hypothesis, which states that various chronic obstructive diseases including asthma and COPD have overlapping pathophysiology and clinical features. 16 Despite efforts to define ACO, there has been no unifying definition of ACO, resulting in heterogeneity of clinical implication depending on the definition of ACO used. In our study, we focused on the cohort with ACO defined with the same diagnostic criteria to compare clinical features of ACO from severe asthma cohort (ACO-asthma) and ACO from COPD cohort (ACO-COPD). ACO from each cohort may imply predominant features of either disease. Those with ACO-asthma were found to be more likely to be females and younger with high BMI, higher blood eosinophil count, and higher FENO level. ACO-asthma presented more asthma-related comorbid conditions including allergic rhinitis, atopy, and self-reported asthma diagnosis, whereas ACO-COPD presented more non-allergic comorbid conditions such as hypertension. There was no significant difference in severity of airflow limitation between the two groups except that the ACO-asthma group had higher FVC than the ACO-COPD group. In terms of risk of exacerbation, both ACO groups showed an increasing risk compared with pure severe asthmatics, although there were no significant differences in risk of exacerbation between the two ACO groups.
Numerous studies have extracted ACO patients from either severe asthma or COPD cohort. Patients with ACO extracted from severe asthma cohort showed poor FEV1 with more frequent exacerbation than the pure severe asthma group.9,10,17 However, inconsistent results were reported in the COPD cohort, especially on lung function and the risk of exacerbation7,18–23 These differences in outcomes between studies might have resulted from different study populations and criteria used to define ACO. Few studies have been conducted on differences between asthma- and COPD-predominant ACO. The Copenhagen City Heart Study classified chronic airway disease into six groups according to smoking status and age at asthma onset and reported lung function trajectories with 18 years follow-up and mortality of 22 years. FEV1 in the COPD and ACO with late asthma onset groups showed faster decline (39.5 ml/year and 49.6 ml/year, respectively) than the asthma group (25.6 ml/year) and the ACO with early asthma onset group (27.3 ml/year). 11 Furthermore, COPD and ACO with either early- or late-onset asthma had higher all-cause and respiratory mortality than asthma group (HRs for all-cause mortality were 2.76, 2.55, and 3.72, respectively; HRs for respiratory mortality were 10.45, 8.17, and 31.86, respectively). Bourdin et al. 24 have used French national Bronchial Obstruction and Asthma Cohort (COBRA) of both asthma (n = 877) and COPD patients (n = 228) and reclassified patients into asthma, ACO, and COPD with K-means clustering algorithm. ACO patients who were originally enrolled as asthma were named as ACO-asthma and those enrolled as COPD were named as ACO-COPD. Although reclassification was mainly originated from the asthma group, reclassifying asthma as ACO had no clinical significance in terms of prognosis, whereas reclassifying COPD to ACO more often encountered better outcomes. Interestingly, there were no significant differences baseline characteristics between the ACO from asthma and the ACO from COPD groups except that the ACO-asthma group had better lung volume parameter compared with ACO-COPD group, consistent with our results. Risk of exacerbation were comparable between groups. Our results showed that even asthma patients in our study were primarily enrolled from severe asthma cohort. Type 2 inflammation is an important molecular mechanism of asthma. Elevated eosinophil in blood or sputum and higher FENO level are well-known biologic markers of type-2 T helper (Th2) cell-mediated airway inflammation. 25 However, some COPD patients exhibit eosinophilic airway inflammation and respond to corticosteroid therapy.26,27 Not surprisingly, the ACO-COPD group showed higher eosinophil count and FENO than the pure COPD group. The ACO-asthma group also presented higher Th2 inflammation markers than the pure asthma group. As higher Th2 biomarkers were associated with poor asthma control and frequent exacerbations, more poorly controlled patients were assigned to ACO rather than pure asthma in the asthma cohort.28,29 However, previous studies have shown opposite 9 or insignificant10,17 results between pure asthma and ACO-asthma. Further investigations are needed. On the contrary, higher eosinophil levels can increase the risk for COPD exacerbation, although the relationship and mechanism remain controversial.30,31 Furthermore, the asthma-predominant ACO group showed higher levels of Th2 biomarkers than the COPD-predominant group. This implies that ACO does not represent a single disease. The multifaceted nature of the spectrum might be related to different clinical features between the two groups.
This study has some limitations. First, all patients included in our study are Koreans, which might not represent patients with other ethnic or race. Patients enrolled in KOCOSS or ISAR are symptomatic patients. Most of them have medical care in a referral hospital. This implies that our data might not represent patients with mild disease. Second, although smoking history (⩾10 pack-years) is frequently used in diagnostic criteria of previous studies,6,32,33 we did not use it as an ACO component since the number of patients categorized as ACO-asthma was too small. However, recent guidelines, including GOLD 2023 guideline, have announced that a substantial proportion of COPD is associated with non-smoking risk factors. 3 Furthermore, several studies including the Copenhagen study did not use smoking in the diagnostic criteria either6,11,32 Third, ACO was defined by lung function and blood eosinophilic and this simple criteria has possibility of overestimation ACO especially in severe asthma cohort although it is easy to apply in real practice. Finally, the severe asthma cohort had a relatively smaller number of patients than the COPD cohort. Non-significant result in exacerbation risk between pure severe asthma and ACO-asthma might have resulted from the small number of asthma patients. Previous studies have consistently shown an increased exacerbation risk in those with an ACO phenotype.9,10,17 Moreover, we could not perform subgroup analysis on heterogeneity itself of ACO from either severe asthma or COPD cohort due to relatively small number of ACO in severe asthma cohort.
In conclusion, we enrolled patients from severe asthma and COPD cohorts, categorized ACO and non-ACO (pure) patients with simple criteria, and analyzed different clinical features between pure severe asthma, ACO-asthma, ACO-COPD, and pure COPD patients. Although there were some clinical differences according to the nature of the cohort in both ACO groups, the risk of exacerbation during a year did not differ between ACO-asthma and ACO-COPD groups. Treatment options were quite different between the two ACO groups, particularly ICS use, although they did not influence the outcome. Interests and efforts to identify ACO could be meaningful when prognosis and treatment response become clearly evaluated. Of note, ACO might show different phenotypes, making it difficult to approach and decide appropriate treatment plan. Regardless of originally enrolled cohorts, using simple criteria allows us to screen for ACO patients and enable personalized medicine. Further studies are needed to investigate optimal treatments for those who have high probability of ACO.
Supplemental Material
sj-docx-1-tar-10.1177_17534666231169472 – Supplemental material for Heterogeneity of asthma–chronic obstructive pulmonary disease (COPD) overlap from a cohort of patients with severe asthma and COPD
Supplemental material, sj-docx-1-tar-10.1177_17534666231169472 for Heterogeneity of asthma–chronic obstructive pulmonary disease (COPD) overlap from a cohort of patients with severe asthma and COPD by Joon Young Choi, Chin Kook Rhee, Kwang Ha Yoo, Ki-Suck Jung, Jae Ha Lee, Hyoung Kyu Yoon, Seung Won Ra, Myung Goo Lee and Yong Suk Jo in Therapeutic Advances in Respiratory Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
