Abstract
Refractory asthma not only has a significant effect on quality of life, but also imposes an economic burden on society. Increasing evidence suggests that there is a pathophysiologic interaction between infection and allergic disease in patients with severe or refractory asthma. Therapeutic trials of macrolides and azoles are being utilized in some patients with refractory asthma who fail to respond to standard therapy. In this article we review the definition of refractory asthma and the potential pathophysiologic interactions between infection and allergic disease. Emerging data suggest that microorganisms and their byproducts may be a therapeutic target in the therapy of patients with severe or refractory asthma.
Introduction
Asthma is a prevalent and heterogeneous disease that markedly affects quality of life and imparts a significant economic burden on society. The pathogenesis of persistent inflammation in chronic asthma is currently not completely understood. While viral infections have been linked to asthma development and may be responsible for approximately 50% of acute asthma exacerbations, the role of other microbes (typical and atypical bacteria and fungi) has only recently been suggested. This link between microbial organisms and refractory or difficult-to-control asthma has emerged as we begin to understand more about the pathophysiology of various phenotypes of chronic asthma. In this article we review the definition of refractory asthma and the potential pathophysiologic interactions between infection and allergic disease. In addition, we present the data suggesting that some patients with refractory asthma may benefit from anti-infective therapy.
Definitions: refractory asthma
Refractory asthma (also referred to as ‘difficult-to-control’ asthma) should be distinguished from uncontrolled asthma. Uncontrolled asthma describes the condition in which the clinical manifestations of asthma have not been reduced or removed by goal-oriented therapy [Reddel et al. 2009]. Asthma control requires identification of exacerbating factors, patient education, appropriate therapy and compliance [Gaga et al. 2005]. The term refractory asthma is used for patients with persistent asthma symptoms in whom co-morbidities have been treated, trigger factors have been addressed, compliance with prescribed treatment has been evaluated, and alternative diagnoses have been excluded [Bel et al. 2010]. In addition, patients with refractory asthma have an increase in the frequency of exacerbations. There are several other definitions of refractory asthma, but perhaps the most clinically useful definition is the treatment-centered definition put forward by the European Respiratory Society (ERS) [Reddel et al. 2009; European Network for Understanding Mechanisms of Severe Asthma, 2003; American Thoracic Society, 2000]. The ERS defined refractory asthma by persistent symptoms or exacerbations and continued use of a short-acting beta agonist in subjects receiving high-dose inhaled corticosteroids (ICSs) while under the care of a respiratory specialist for a minimum of 6 months. These patients often require systemic immunomodulating therapies to control their symptoms and may have frequent severe exacerbations [Moore et al. 2007]. There is mounting evidence that infections, both acute and chronic, may play a key role in the pathogenesis of a subset asthmatic patients (both refractory and nonrefractory) in which asthma symptoms are difficult to control [Rollins et al. 2010].
Viral infections
Viral respiratory tract infections (RTI) are the most common cause of asthma exacerbations. Acute wheezing was most frequently associated with a positive viral polymerase chain reaction (PCR) assay: 80% in children and 50–75% in adults. Of the viruses identified, rhinovirus (RV) was the most common (65%) [Papadopoulos et al. 2010; Khetsuriani et al. 2007; Johnston et al. 1995]. Other frequent viruses include respiratory syncytial virus (RSV), parainfluenza virus, adenovirus, and bocavirus. In a recent study, experimental RV infection caused more asthma exacerbations and significantly worse symptomatology in asthmatic patients compared with controls [Message et al. 2008]. This study, along with several others, supports that a viral infection may modulate the immune system in susceptible hosts and cause greater morbidity in asthmatics compared with healthy individuals [Jackson and Johnston, 2010]. These studies support the concept that viral respiratory infections increase the intensity of allergic response, enhance bronchial inflammation, and increase airway hyperreactivity. It is plausible that patients with refractory asthma may be more susceptible to these types of infections and these pathogens may in turn modulate the immune response and potentiate chronic inflammation in the airways.
Several strategies to alter the natural history of viral-induced asthma exacerbations have been studied. Prevention has been a focus of great interest, but the antigenic diversity of more than 100 serotypes of RV makes it difficult to create an effective vaccine [Rosenthal et al. 2010]. Palivizumab, a monoclonal antibody against RSV fusion protein, has shown protection against recurrent wheezing in premature infants in a prospective case-controlled study [Simoes et al. 2007]. Motavizumab, a second-generation derivative of palivizumab, has a 70-fold higher affinity for the RSV fusion protein and has shown a 50% reduction in the RSV medically attended lower RTI [Carbonell-Estrany et al. 2010]. Pleconaril, an agent that targets piconavirus, is currently being investigated in the prevention of asthma exacerbations. Other therapies under investigation include inhibitors of RV protease 2 A, 3 C (pubrintivir), or both, which are enzymes required for viral replication [Jackson and Johnston, 2010]. Although these anti-infective agents are promising, more studies are needed to clarify their potential benefit in the prevention and treatment of asthma.
Bacterial infections
Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis are common colonizers in the upper airway and have been labeled ‘usual respiratory pathogens’. In asthmatic patients these pathogens might not only be present as ‘colonizers’, but may also cause chronic inflammation and play a direct role in the pathogenesis of asthma. Recent evidence suggests that the lung microbiome may play a significant role in both the development and progression of chronic asthma. The temporal sequence of microbial colonization and/or chronic infection may impact the development and progression of chronic asthma [Horvat et al. 2010; Kraft et al. 2002]. The evidence is conflicting regarding the frequency of these pathogens and the effects on asthmatic patients. A 10-year epidemiologic study of 1200 children concluded that the presence of respiratory pathogens played no major role in asthma exacerbations. They demonstrated that the detection rate of respiratory pathogens was similar in patients with acute wheezing, recurring wheezing, or no wheezing (43%, 34%, and 39% respectively) [Nagayama et al. 2007].
In contrast to these observations, Bisgaard and colleagues showed that respiratory pathogens found in the airways of asymptomatic neonates at 1 month of age were associated with an increase by a factor of two to four in the risk of persistent wheezing, acute severe exacerbations, hospitalizations for wheezing and development of asthma by the age of 5 years [Bisgaard et al. 2007]. Even though there is a plausible relationship between the colonization of usual respiratory pathogens and asthma, studies have not yet demonstrated that antibiotics targeting these organisms change the natural course of asthma [Graham et al. 1982; Shapiro et al. 1974].
Recent investigations have identified Staphylococcus aureus as a potential contributor in the pathophysiology of severe asthma. S. aureus releases enterotoxins that have superantigenic activity that may modify B- and T-cell function. For example, S. aureus enterotoxin (SAE) B promotes T-helper-2 (Th2) cell cytokines including interleukin (IL)-2, IL-4, IL-5 and increases the production of immunoglobulin E (IgE) [Bachert et al. 2008]. In addition, SAE A can induce experimentally pro-asthmatic changes in smooth muscle cells [Veler et al. 2007]. A meta-analysis evaluated the relationship between the presence of S. aureus superantigens and the severity and persistence of asthma [Pastacaldi et al. 2010]. This study demonstrated a significantly increased prevalence of IgE to S. aureus superantigens in asthmatic patients compared with controls as well as a positive correlation with severity. A study by Kowalski and colleagues evaluated 210 patients (109 asthmatic patients and 101 controls) by measuring levels of IgE specific to SAE [Kowalski et al. 2011]. The mean serum SAE specific IgE levels was threefold higher in severe versus nonsevere asthmatics. Furthermore, serum specific IgE to SAE were significantly associated with decreased pulmonary function and increased airway reversibility. Owing to the strong evidence implicating a relationship between S. aureus superantigens and asthma, it would be reasonable to explore the use of antibiotics targeting S. aureus as a possible treatment for refractory asthma.
Atypical bacteria
Mounting evidence suggests an association between atypical bacteria (Mycoplasma pneumoniae and Chlamydophila pneumoniae) and asthma. These atypical organisms have characteristics that make them ideal candidates to implicate as having a role in the pathogenesis of asthma. M. pneumoniae is the smallest free-living bacterium (0.2 µm) and lives as a parasite, dependent on the host cell for nutrients. These atypical bacteria attach to the surface of airway epithelial cells, resulting in epithelial damage and ciliary dysfunction. M. pneumoniae has been associated with childhood wheezing [Berkovich et al. 1970] and has been detected in adults with chronic stable asthma [Kraft et al. 1998]. M. pneumoniae may survive intercellularly, but C. pneumoniae is known to be an obligate intracellular organism and causes chronic intracellular inflammation. These characteristics of the atypical bacteria make diagnosis of infection difficult with standard laboratory tests, such as cultures and standard serology. Utilizing PCR assays from the upper airway and lower airways the prevalence of M. pneumoniae and/or C. pneumoniae has been shown to be as high as 56% in moderately severe asthma patients versus 13% in controls [Martin et al. 2001]. A variety of PCR assays may be used to identify these organisms, but they vary in sensitivity. Our group has developed a molecular assay that is more sensitive in detecting M. pneumoniae by PCR [Hardy et al. 2009; Winchell et al. 2008]. We studied nasal lavage, throat swab, and sputum in 62 subjects with refractory asthma and found 30/62 (48.8%) were positive for the M. pneumoniae and 10/62 (9.7%) subjects were persistently positive [Peters et al. 2010]. These data suggest a likely role of atypical bacteria in the pathogenesis of severe/refractory asthma, but, to date, definitive proof is not available.
Other evidence suggests that there is a relationship between the presence of M. pneumoniae and/or C. pneumoniae and patients with difficult-to-control asthma and with acute asthma exacerbations [Cosentini et al. 2008; Betsou et al. 2003; Bjornsson et al. 1996]. In addition to the epithelial and cilliary damage that infection with M. pneumoniae and C. pneumoniae induces, hypersecretion of IgE, modulation of the immune responses, and cytokine secretion to a dominant Th2 response also have been observed [ten Brinke et al. 2001]. In addition, there is ciliostasis, augmented mucus production, and chronic inflammation in the airways [Kraft, 2000]. This immune response may vary between different susceptible hosts [Wright, 2002]. Asthmatics with M. pneumoniae infection determined by PCR contained higher levels of tumor necrosis factor alpha (TNF-α), IL-4, and IL-5 in the airways compared with PCR-negative asthmatic patients [Kraft et al. 1998].
Our group identified the 68-kDa protein unique to M. pneumoniae called the community-acquired respiratory distress syndrome (CARDS) toxin [Hardy et al. 2009]. Exposure of the bronchial surface to the CARDS toxin causes an intense lymphocytic response and marked cytokine response in both mouse and baboon models, subsequently causing airway hyperreactivity as well as prolonged airway obstruction [Hardy et al. 2009]. This inflammatory response may contribute to refractory asthma in some patients and increase the risk of asthma exacerbations. Evidence obtained from murine models demonstrated that the timing of the infections with these atypical bacteria in relation to the age of the host plays a role in the way the host responds to the infection [Horvat et al. 2010]. Compared with controls, the serologic evidence of these bacteria in asthma exacerbations is significantly higher (M. pneumoniae: 18–20% vs. 2–5%, C. pneumoniae: 9–11% vs. 3–5%) [Biscardi et al. 2004; Lieberman et al. 2003; Miyashita et al. 1998; Emre et al. 1994]. The presence of these inflammatory markers supports the notion that these atypical bacteria may play an important role in difficult-to-control asthma. Defining the role of Mycoplasma infection in refractory asthma may depend on utilizing newer techniques to identify the organism as well as the ability to measure production of CARDS toxin.
Anti-inflammatory properties of macrolides
Macrolides have been shown to concentrate intracellularly and modulate cell functions at doses below those known to have antimicrobial effects [Labro, 2004]. Studies have demonstrated that macrolides significantly affect host–pathogen interactions, cellular signaling pathways, cytokine responses, oxidative stress pathways, innate immunity, mucus secretion, and cellular chemotaxis [Friedlander and Albert, 2010]. These effects are known to be beneficial in several pulmonary conditions including cystic fibrosis [Clement et al. 2006], diffuse panbronchiolitis [Kudoh et al. 1998], chronic obstructive pulmonary disease (COPD) [Seemungal et al. 2008], posttransplant bronchiolitis obliterans [Benden and Boehler, 2009], and bronchiectasis [Yalçin et al. 2006]. In addition to the anti-inflammatory and immune-modulating effects of macrolides in other respiratory conditions, macrolides have been shown to have similar effects in patients with asthma. Clarithromycin was shown to decrease blood and sputum eosinophils and sputum eosinophilic cationic protein levels compared to placebo in an 8-week, double-blind, randomized, crossover trial. [Amayasu et al. 2000]. This macrolide was also shown to significantly and persistently lower cytokines such as TNF-α, IL-1, and IL-10 compared with placebo in a randomized, double-blinded, placebo-controlled trial in subjects with acute asthma exacerbations [Fonseca-Aten et al. 2006]. Interestingly these effects were greater in subjects with a positive PCR for M. pneumoniae and C. pneumoniae. Other beneficial effects of macrolides in asthmatic patients include a decreased production of superoxide anion by neutrophils [Kamoi et al. 1995], IL-8 [Simpson et al. 2008], and significant reductions in levels of IL-5, IL-12, TNF-α in bronchoalveolar lavage fluid as well as TNF-α in airway tissue [Kraft et al. 2002]. Therefore, macrolides have been shown to have anti-inflammatory and immune-modulating effects in many respiratory diseases, including asthma.
Macrolides in the treatment of refractory asthma
Owing to the plausible role of atypical bacteria in the development of refractory asthma, the use of antibiotics that target these pathogens is attractive; however, data are conflicting regarding the risks versus benefits of antibiotics in this subset of asthmatics. In addition to the potential independent anti-inflammatory and immune-modulating effects of macrolides described above, these agents are one of the main therapies for both M. pneumoniae and C. pneumoniae [Black, 1997]. These characteristics make macrolides an ideal choice of antibiotic to study for therapy in refractory asthmatics. Early studies showed positive outcomes when asthmatics were treated with macrolides [Spector et al. 1974; Itkin and Menzel, 1970]. Even though there was a possible association with its intended antimicrobial properties, the effects were initially thought to be more strongly associated with a decrease in the clearance of both corticosteroids and theophylline [Weinberger et al. 1977]. Later studies showed that this ‘steroid-sparing’ effect of macrolides did not significantly alter the course of asthma in corticosteroid-dependent patients [Evans et al. 2001]. Therefore, macrolides were studied in more targeted populations of patients with asthma to evaluate their effects on outcomes such as symptoms, asthma control, lung function, quality of life, and exacerbations. In a double-blind, randomized, controlled trial (RCT) of 6 weeks of clarithromycin versus placebo in 55 subjects with chronic stable asthma, the presence of M. pneumoniae and C. pneumoniae in the airway was determined by PCR via bronchoscopy before and after treatment [Kraft et al. 2002]. The prevalence of these atypical bacteria was over 55% by PCR, but the serology and cultures were negative for M. pneumoniae in all patients. In this study, only subjects with a positive PCR who received clarithromycin had a significant improvement in the forced expiratory volume in 1 second (FEV1). Black and colleagues studied 232 subjects with asthma and serological evidence of C. pneumoniae in a double-blind, placebo-controlled RCT evaluating 6 weeks of treatment with roxithromycin 150 mg twice a day [Black et al. 2001]. The roxithromycin group demonstrated a significant improvement in peak expiratory flow rates immediately after the course of treatment, but this effect diminished at 6 months follow-up. Another multicenter, blinded (allocation concealed), placebo-controlled, parallel group trial was performed by Hahn and colleagues in stable asthmatic patients and evaluated an initial course of 3 days, followed by weekly azithromycin for a total of 6 weeks [Hahn et al. 2006]. The azithromycin group had a favorable clinical response in patients with a positive C. pneumoniae serum IgA, but no significant difference in the Juniper Asthma Quality of Life Questionnaire. Johnston and colleagues conducted a multicenter, double-blind, placebo-controlled RCT to evaluate the efficacy of 10 days of telithromycin in 278 adults with acute asthma. The results showed that the subjects in the treatment group had a significant improvement in FEV1 by day 10 and in asthma symptoms at day 10 and at the end of the 6-week trial [Johnston et al. 2006]. More recently, a trial evaluated the effectiveness of clarithromycin for the treatment of suboptimally controlled mild–moderate asthma [Sutherland et al. 2010]. In this double-blind, placebo-controlled, RCT, 253 asthmatic adults were treated for four weeks with ICSs, and then a subgroup of 92 patients were classified as suboptimally controlled based on a score of 15 or less on the Asthma Control Questionnaire. This subgroup underwent bronchoscopy for PCR detection of M. pneumoniae and C. pneumoniae and were randomized to receive clarithromycin or placebo. Subjects who received clarithromycin added to the ICSs did not have a beneficial effect in asthma control or lung function compared with the control group. Although there was an improvement in airway hyperresponsiveness with clarithromycin, this benefit was not accompanied by improvements in other secondary outcomes. Finally, a Cochrane review to determine whether macrolides are effective in the management of patients with chronic asthma evaluated RCT of children and adults with chronic asthma treated with macrolides versus placebo for more than 4 weeks [Richeldi et al. 2005]. The authors included a total of 416 participants in seven studies and the quality of methodology was classified as low for most trials. Some trials demonstrated significant differences in eosinophilic inflammation and symptoms, but no differences in FEV1; therefore, the authors concluded that there is insufficient evidence to support or refute the use of macrolides in patients with chronic asthma. In summary, macrolides may be helpful in improving symptoms in select patients with refractory asthma, but the routine use cannot be supported based on the current evidence in the literature.
Hypersensitivity reaction: allergic bronchopulmonary aspergillosis
The respiratory disorders caused by the Aspergillus species can be classified as saprophytic (aspergilloma), allergic (sinusitis, allergic bronchopulmonary aspergillosis [ABPA], hypersensitivity pneumonia), and invasive (invasive aspergillosis, necrotizing pulmonary aspergillosis) [Segal, 2009]. ABPA is caused by a hypersensitivity to Aspergillus fumigatus manifesting as chronic asthma, pulmonary infiltrates, and bronchiectasis. It is believed that 1–2% of asthmatic patients have this condition [Greenberger, 2003]. The clinical features include: low-grade fever, bronchospasm, airway hyperreactivity, hemoptysis, or productive cough. Expectoration of dark mucus plugs is common, but patients can also be asymptomatic. Patients with ABPA have a positive skin test to A. fumigatus, elevated total serum IgE (typically above 1000 IU/ml), and the presence of serum IgG and IgE specific to A. fumigatus [Agarwal, 2009].
The treatment involves the use of glucocorticoids to control inflammation. Although there have been several reports in which itraconazole has demonstrated a positive clinical outcome in the treatment of ABPA [Wark et al. 2004], there are only two RCTs testing this effect. In a multicenter, randomized study, 55 patients with ABPA received either itraconazole or placebo [Stevens et al. 2000]. In the treatment group, 46% of the patients showed a significant effect in one or more of the following: decrease in oral corticosteroid use, decrease in total IgE level, and an improvement in pulmonary function tests. In the placebo group, only 19% met any of these criteria. Wark and colleagues showed in a single-center RCT that patients with stable ABPA had a reduction in sputum and blood eosinophils, serum total IgE, and IgG levels to A. fumigatus when itraconazole was used [Wark et al. 2003]. In addition, there were fewer exacerbations in the treatment group. Both studies showed a clear clinical benefit in patients with ABPA after treatment with itraconazole. Currently, it is recommended to consider itraconazole as a corticosteroid sparing drug or if corticosteroids alone are ineffective [Patterson and Strek, 2010]. Voriconazole and posaconazole are promising new drugs with improved bioavailability and safety profiles, but have not adequately been tested in ABPA.
Fungal infections/colonization
There is abundant evidence that atopy, especially to mold allergens, is associated with asthma severity [Matsuoka et al. 2009; Niedoszytko et al. 2007]. Mold sensitivity has been linked with asthma severity, hospital admissions, and increased bronchial hyperreactivity [Agarwal and Gupta, 2010]. A cross-sectional study of more than 1300 asthmatic patients showed that sensitization to Alternaria alernata or Cladosporium herbarum was a significant risk factor for severe asthma [Neukirch et al. 1999]. A study in which 181 asthmatic patients were classified based on the number and recurrence of hospital admissions showed that that frequency of skin reactivity to molds was more common in the patients with more frequent admissions (76% vs. 16–19%) [O’Driscoll et al. 2005]. Similar findings were reported in a study of 121 patients with severe asthma in which sensitization to one or more molds were detected in 66% of the cases [O’Driscoll et al. 2009]. Based on the above observations the concept of a distinct asthma phenotype of severe asthma with fungal sensitization (SAFS) was developed [Denning et al. 2006]. SAFS describes patients with severe asthma, positive testing for fungal sensitization, and in which ABPA has been excluded. It is important to note that compared with other allergens and organic exposures, molds have the additional capability of germinating and infecting susceptible tissues or colonizing the respiratory tract [Denning et al. 2006]. It may be difficult, in a sensitized patient, to determine whether the patient is reacting to an environmental exposure, respiratory tract colonization, or both.
Antifungal therapy for non-ABPA
Because asthmatic patients frequently have positive skin testing for different fungal pathogens, the use of antifungal agents to control asthma symptoms have been explored outside the spectrum of ABPA. In a double-blind, controlled trial, asthmatic patients with late-onset asthma and positive skin testing for three Trichophyton species received either fluconazole or placebo for 5 months [Ward et al. 1999]. Afterward, both groups were treated with fluconazole for 5 months. Although the number of studied patients was small (n = 11), there was significant improvement in bronchial reactivity, steroid requirements, and symptom scores. These findings were followed by the Fungal Asthma Sensitization Trial (FAST) [Denning et al. 2009]. FAST was a multicenter, RCT in which patients with SAFS were randomized to receive either itraconazole for 32 weeks or placebo and were then followed for another 16 weeks. Although there was a significant improvement in quality of life in the majority of patient in the treatment group (60%), patients returned to baseline after cessation of therapy. At this time, our group evaluates refractory asthmatics for fungal sensitivity, aggressively treats their dermatomycosis, and attempts to reduce environmental exposures. Given the cost and side effects of systemic antifungal therapy, we are awaiting future studies evaluating both systemic and inhaled antifungal treatment in subjects with SAFS.
Conclusion
The heterogeneous nature of asthma makes it difficult to treat. Fortunately, most asthmatics respond to standard pharmacotherapy; however, a subgroup of asthmatics has persistent symptoms even after optimal therapy, treatment of comorbidities, and avoidance of triggers. Increasing evidence suggests that there is a pathophysiologic interaction between infection and allergic disease in these refractory asthmatic patients. Currently, therapeutic trials of macrolides and azoles are being utilized in some patients with refractory asthma. As we move forward in our understanding of the interactions between infection (Figure 1) and allergic inflammation, we will be able to provide individualized and evidence-based care to patients with refractory asthma.
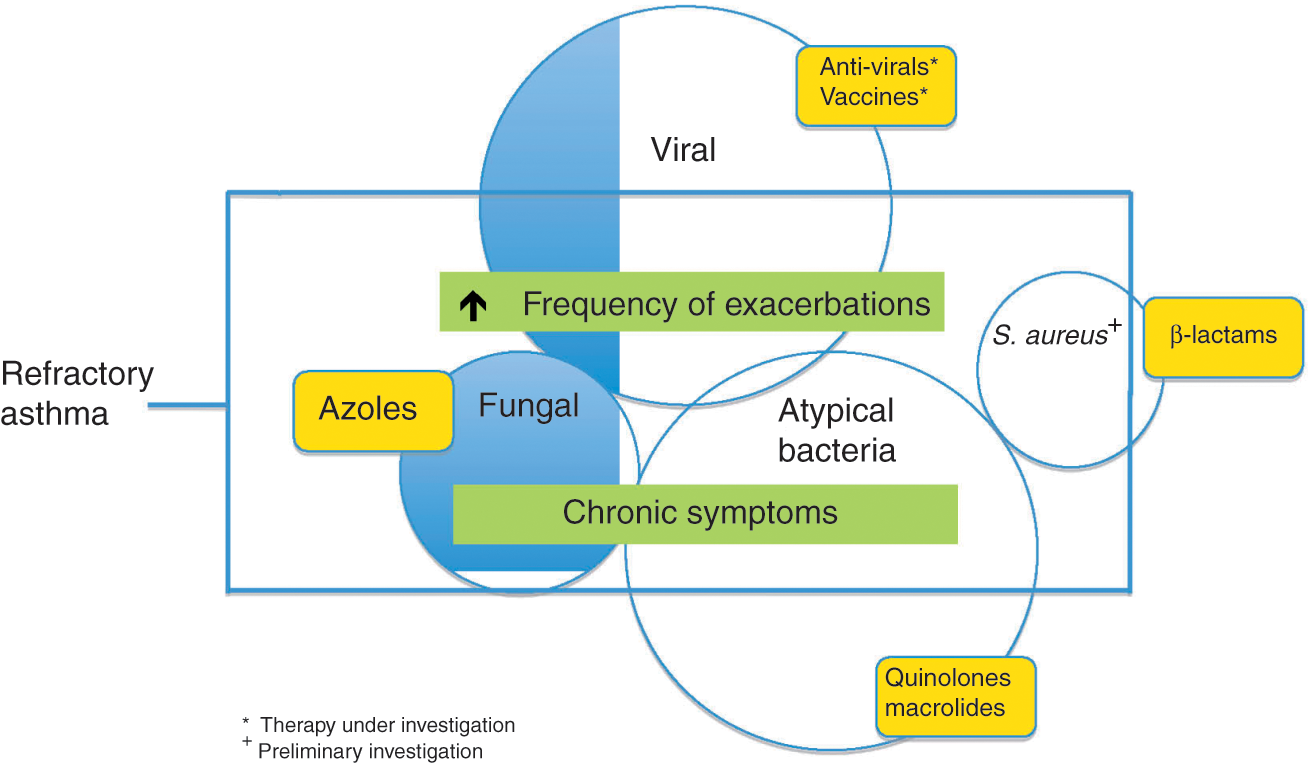
Interaction and treatment of infections in refractory asthma.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
None declared.
