Abstract
Introduction
Repeated injections of low-dose lipopolysaccharide (LPS preconditioning) augment the antibacterial activity of liver macrophages. In this study, a mouse model of acute kidney injury (AKI) induced by Staphylococcus aureus (S. aureus) bacteremia was used to investigate the effects of LPS preconditioning on renal macrophages.
Methods
Eight-week-old C57BL/6J mice were preconditioned with either low-dose LPS (5 μg/kg) or the vehicle for three consecutive days. Kidney immune cells were isolated, and the antibacterial activity of renal macrophages was assessed by pHrodoTM-labeled S. aureus in vitro. Twenty-four hours after the last LPS injection, the mice were intravenously challenged with S. aureus (2 × 107 CFU) and their renal function was evaluated to identify the changes.
Results
Mouse renal macrophages exhibited a weak antibacterial activity against S. aureus compared with the liver and spleen macrophages. LPS preconditioning elevated the count of F4/80low CD11bhigh bone marrow-derived macrophages (BMDM) and augmented their antibacterial activities in the mouse kidney. It also enhanced the antibacterial activity of F4/80high CD11blow tissue-resident macrophages (TRM) without altering their abundance. LPS preconditioning lowered the bacterial propagation in the kidney in the challenged mice and ameliorated sepsis-associated AKI compared with the control. LPS preconditioning upregulated the CD80/CD206 expression (M1/M2) ratio in BMDMs in the kidney before bacterial challenge and reduced their M1/M2 ratio following S. aureus challenge compared with the control.
Conclusion
LPS preconditioning enhanced the antibacterial activity of the renal macrophages against S. aureus and suppressed the excessive activation of M1 macrophages following S. aureus challenge, resulting in the amelioration of AKI caused by S. aureus bacteremia.
Introduction
According to the Acute Kidney Injury-Epidemiologic Prospective Investigation (AKI-EPI) study, more than half of the ICU patients suffer from an acute kidney injury (AKI). An increase in AKI severity is associated with increased mortality. According to the AKI-EPI study, the primary cause of AKI was sepsis. 1 In the pathogenesis of sepsis-associated AKI, infection triggers systemic proinflammatory cytokine production and increases the concentration of pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPs) in the kidney. The toll-like receptors (TLRs) present on the proximal tubular cells recognize the PAMPs and DAMPs that have been filtered by the glomeruli, triggering a localized excessive immune response. 2 Apoptosis and necrotic cell death caused by tubular epithelial injury leads to the release of cytokines, chemokines, and reactive oxygen species (ROS), which triggers an inflammation cascade in the kidneys. Inflammatory cells, primarily neutrophils and mononuclear cells damage the tubular epithelium by infiltrating the peritubular capillaries and locally spread the inflammatory response. 3
LPS preconditioning
In the normal inflammatory response to infection, lipopolysaccharides (LPS) activate macrophages and enhance their ability to defend against infection, heal wounds, and regulate metabolism. 4 lPS preconditioning is a phenomenon in which low doses of LPS are administered to cause downregulation of the inflammatory response without negatively influencing the innate immune response. The most important feature of this effect was thought to be the marked reduction of TNF production in macrophages.5,6 In addition, LPS tolerance was observed to reduce the number of viable bacteria in the liver in E. coli-challenged mice and dramatically augment the bacterial clearance in the liver. 7 Several studies have previously explored the induction of LPS tolerance through deliberate repeated exposures to low-level LPS through a procedure called LPS preconditioning. LPS preconditioning protects the host from the damage caused by secondary exposure to LPS and also from the damage caused by ischemia-reperfusion injury or stimulation of other TLR agonists (cross-resistance). 8
Renal protection by LPS preconditioning
The prevention of sepsis-associated AKI is vital for reducing mortality and preserving renal function in ICU patients. A recent report introduced the possibility that LPS preconditioning exerts a renoprotective effect through the involvement of macrophages and tubular cells. 9 Our hypothesis is that the nonspecific renoprotective effects of LPS preconditioning are at least partially caused by the functional changes in macrophages caused by the exposure to low-dose LPS. The two types of macrophages include tissue-resident macrophages (TRM) and bone marrow-derived macrophages (BMDM), and they differ in their phagocytic- and cytokine-producing capabilities. 10 The changes in the population dynamics and function of kidney macrophages were examined in this study to explore the effect of LPS preconditioning on these two types of macrophages. Staphylococcus aureus (S. aureus) is the most common pathogen in patients suffering from nosocomial sepsis-associated AKI.11,12 Therefore, the effect of LPS preconditioning in mice challenged with S. aureus was also investigated in this study.
Materials and methods
Animals
Male C57BL/6J mice (age 8–12 weeks) were procured from CLEA Japan (Tokyo, Japan). The mice were housed in a pathogen-free environment with a standard 12-h light/dark cycle, controlled temperature (25 °C) and humidity (40%), and were provided ad libitum access to food and water. The Ethics Committee of Animal Care and Experimentation of the National Defense Medical College, Japan (approval number 20018), approved the experimental procedures.
LPS preconditioning protocol
LPS derived from Escherichia coli (E. coli) O111:B4 was procured from Sigma-Aldrich. LPS preconditioning was performed as reported in a previously conducted study. 7 In brief, the procedure consisted of intraperitoneal administration of low-dose LPS (5 µg/kg) that was dissolved in 500 µL of saline, for 3 consecutive days. Control mice received 500 µL of vehicle saline intraperitoneally for a 3-day period.
Model of mouse sepsis-associated AKI
The mice were injected with 2 × 107 CFU of S. aureus (ATCC6538P) suspended in 200 µL saline through the tail vein using a 29 G needle, 24 h after the last injection of LPS or control vehicle. Food and water were provided ad libitum with a 12-h light/dark cycle. Three days after the bacterial challenge, the mice were euthanized under deep anesthesia using 4% isoflurane and their blood, urine, kidneys, liver, and spleen samples were harvested.
Measurement of the blood samples
Blood (500–700 µL per mouse) was collected from the inferior vena cava under deep anesthesia. After collection, the blood samples were centrifuged in microcentrifuge tubes at 4 °C, 900 × g, for 10 min, and the supernatant plasma was retained. Enzymatic assay (SRL, Tokyo, Japan) was performed to measure the blood urea nitrogen (BUN) and plasma creatinine (Cr).
Preparation of the kidney single-cell suspension
Kidney single-cell suspensions were obtained by following a previously established protocol.13–15 In brief, the kidneys were perfused thoroughly with 10 ml of saline and harvested immediately following sacrifice and exsanguination. After mincing the whole kidneys into small pieces, they were placed into 15 ml of Hanks’ Balanced Salt Solution (HBSS) modified with calcium and magnesium. The samples were incubated at 37 °C for 40 min following the addition of Collagenase (124 ug). Following centrifugation, the cells were resuspended in Roswell Park Memorial Institute (RPMI) medium with 1% fetal bovine serum (FBS) and HEPES buffer, passed through a 63 µm stainless steel mesh, and separated using Percoll-based gradient centrifugation. The leukocyte-rich layer was harvested and filtered through a 40 µm cell strainer following resuspension in the erythrocyte lysate.
Preparation of the liver single-cell suspension
The following procedure was used for preparing the liver single-cell suspension, with reference to previously conducted studies.7,10 The organs were perfused with saline and the livers were harvested and shredded with a scalpel. Livers weighing less than 1.5 g were subjected to collagenase treatment, which involved addition of 15 ml of HBSS modified with HEPES, calcium, and magnesium, 124 µg of collagenase, followed by shaking it in a water bath at 37 °C for 30 min. After collagenase treatment, the tissue was passed through a 63 µm stainless steel mesh, collected in a petri dish, and was finally dissolved in 50 ml of RPMI with 1% FBS. The supernatant solution was collected after centrifuging the samples at 70 × g for 3 min. The collected supernatant solution contained liver mononuclear cells, which were precipitated and collected by repeat centrifugation at 430 × g for 5 min. After centrifugation, the liver suspension was placed in a 15 ml conical tube, adjusted to 7 ml with 1% RPMI, mixed with 7 ml of 50% Percoll, which finally resulted in a 25% Percoll solution. This solution was centrifuged at 15 °C, 430 × g for 30 min. The impurities were removed. Erythrocyte lysis treatment was performed and the cells were collected in 1% FBS-added RPMI through a 40 µm cell strainer. After two repeat centrifugations at 4 °C, 430 × g for 5 min to purify the cells, the cells were counted.
Preparation of the spleen single-cell suspension
The following procedure was used in accordance with previously conducted studies. 16 A 60 µm mesh was used to detach the mouse spleen from the cells. The spleen was subsequently dissolved in RPMI with 1% FBS, collected in 15 ml conical tube, centrifuged at 430 × g for 5 min at 4 °C, and the supernatant was discarded. After adding three times the volume of erythrocyte lysate to the cell suspension and allowing it to react for 1 min for breaking the cell membrane of the erythrocytes, the cells were purified through a 40 µm cell strainer and counted.
Flow cytometric analysis
The single cell suspensions were incubated with Fc blocker (93, eBioscience, San Diego, CA) for 15 min at 4 °C after washing, after which the cells were incubated with one of the following antibodies (Abs) for 15 min at 4 °C: APC-conjugated anti-CD45 Ab (30-F11, eBioscience), FITC-conjugated anti-F4/80 Ab (BM8, eBioscience), PE-cyanine 5-conjugated anti-CD11b Ab (M1/70, eBioscience), PE-conjugated anti-Ly6C Ab (HK1.4, eBioscience), PE-conjugated anti-Ly6G Ab (1A8, eBioscience), PE-conjugated anti-CD80 Ab (B7-1, eBioscience), PE-conjugated anti-CD206 Ab (MR6F3, eBioscience), PE-conjugated anti-CD282 (TLR2) Ab (6C2, eBioscience), PE-conjugated anti- CD64 Ab (X54-5/7.1, eBioscience) and/or PE-conjugated anti- MHC classII Ab (M5/114.15.2, eBioscience). The cells were fixed and permeabilized using the Cytofix/Cytoperm™ fixation/permeabilization kit (BD Bioscience) just before incubating them with the CD206 Abs. Isotype Abs corresponding to each fluorescently conjugated Ab were used as controls.
Measurement of intracellular cytokines in renal macrophages
After harvesting the kidney mononuclear cells using the procedure described above, the cells were cultured with S. aureus (ATCC6538P, 1.0 × 104 CFU/well) and GolgiStop™(a protein transport inhibitor containing monensin, BD Bioscience) in Up Cell® (CellSeed Inc. Tokyo, Japan) for 3 h at 37 °C, 5% CO2. APC-conjugated anti-CD45 Ab (30-F11, eBioscience), FITC-conjugated anti-F4/80 Ab (BM8, eBioscience), and PE-conjugated anti-TNFα Ab(MAb11, eBioscience) or anti-IL-10 Ab(JES5-16E3, eBioscience) were used to measure intracellular cytokines by flow cytometry.
Evaluation of the antibacterial activity using pHrodo-conjugated S. aureus
Macrophages from the kidneys, liver, and spleen of normal mice were obtained to examine the differences in the antibacterial activities of the macrophages from the kidneys and other organs (liver and spleen). Mononuclear cell suspensions were prepared from the kidneys, livers, and spleens of untreated normal mice in accordance with the method described above. Using culture medium, the cells from the collected mononuclear cell suspensions were adjusted to a cell count of 2.5 × 106 /ml and placed in Up Cell® (CellSeed Inc. Tokyo, Japan) plates at 250 μL per well. Following the addition of 30 μL of pHrodoTM (acidic pH-dependent fluorescence)-conjugated S. aureus (heat-killed)(pHrodoTM Red S. aureus BioParticlesTM Conjugate for Phagocytosis; Invitrogen Life Technologies A10010, Carlsbad, CA, USA) to each cell, they were allowed to react for 60 min at 37 °C and 5% CO2. For the negative control, pHrodoTM-conjugated S. aureus was added and reacted at 4 °C (on ice) for 60 min. The histograms of the samples and the negative control sample were overlapped to determine the cutoff value (fluorescence intensity), and the pHrodoTM-positive rate of F4/80 positive cells in each tissue was determined. We used the pHrodo to measure bacterial uptake in a low pH compartment. We did not measure real bacterial killing but measured a phenomenon indicative of bacterial killing (uptake and low pH), because most (but not all) bacteria will be killed in such low pH phagolysozomes.
Counting of viable bacteria
The mice were euthanized under deep anesthesia with isoflurane three or seven days following the intravenous administration of bacteria, the organs were perfused with 10 ml of saline, and the kidney, liver, and spleen samples were harvested. Around 0.1–0.05 g of tissue was randomly excised from each organ and crushed with 250 µL of saline for 1 min using a Biomasher II (NIP, Tokyo, Japan). After addition of 250 µL of saline, a total of 500 µL of tissue homogenate solution was obtained. After overnight incubation at 37 °C on BHI agar medium (Eiken Chemical Co. Tokyo, Japan), the bacteria were counted. Counts were calculated as the number of bacteria per gram of organ.
Kidney histology and AKI score
The kidney tissue was short-axis sectioned after perfusion with saline. It was then fixed in 4% phosphate-buffered formalin, embedded in paraffin, and stained with periodate-Schiff's base (Cosmo Bio, Co., Tokyo, Japan) with hematoxylin nuclear staining. The AKI score criteria developed by Nomura et al. were used for semiquantitative scoring of the tissue sections from each mouse for tubular damage. 17 In brief a score ranging from 0 (no damage) to 3 (severe/extensive damage) was assigned by two blinded observers, for each of the three different variables: tubular dilation/flattening, tubular cast, and tubular degeneration/vacuolation. After the random examination for each variable of 10 cortical high-power fields (HPF) for each mouse, a score of 0 was assigned if less than 5% of the tubules were affected; 1, if 5%–33% were affected; 2, if 34%–66% were affected; and 3, if 66% or more were affected (total score 0–9).
Immunohistochemistry
Thin paraffin sections of the kidney (3 µm) were hydrophilized after paraffin thawing, immersed in a pH 9.0 activation solution, and heated at 121 °C for 5 min to activate the antigen. After rehydration with PBS, Dako-REAL peroxidase blocking solution was used to perform endogenous peroxidase blocking for 10 min. This was followed by nonspecific blocking using Blocking One for 30 min. The cells were incubated overnight with primary Ab targeting F4/80 (M-300, Santa Cruz Biotec, sc.25830, 200x dilution) at 4 °C . After washing, the cells were treated with secondary Ab (antirabbit IgG, HRP-linked Ab(Cell Signaling TECHNOLOGY, #7074, 1000× dilution) for 60 min at 20 °C and stained with DAB (diaminobenzidine) for 8 min. Keyence BZ-X710 (KEYENCE Co. Osaka, Japan) was used for image acquisition.
Measurement of plasma TNF, IL-1β and IL-10 levels and urine KIM-1 levels
TNF Mouse (Invitrogen BMS607-2INST), IL-1β Mouse (BioLegend 432606) and Mouse IL-10(BioLegend 550613) ELISA kits were used to measure the plasma TNF, IL-1β and IL-10 levels. Mouse KIM-1 ELISA Kit (Abcam, ab216792) was used to measure the urine KIM-1 (kidney injury molecule 1) levels, which were then normalized to urine Cre levels (measured by SRL, Tokyo, Japan).
Measurement of the kidney homogenate MPO
Neutrophil activity of the renal homogenates was examined by measuring the myeloperoxidase (MPO) activity . Approximately 20–30 mg of renal tissue pieces were dissolved in 100 μL of protein extraction buffer (radio-immunoprecipitation assay buffer: RIPA buffer) (Wako, Osaka, Tokyo) with 1% protease inhibitor (Nacalai Tesque, Kyoto, Japan) to perform protein extraction. Biomasher III (NIP, Tokyo, Japan) was used for 1 min for crushing the samples. Using the MPO Mouse ELISA Kit (Abcam, ab155458, Cambridge, UK), the MPO activity was measured. The PrinceTM BCA Protein Assay Kit (Thermo Scientific, Waltham, Massachusetts, USA) was used to quantify the proteins in the solution by performing the bicinchoninate assay (BCA) method, and MPO per gram of protein was calculated.
Statistics
Results have been presented as mean ± standard error (SE). Turkey's post hoc test or two-way ANOVA were applied as appropriate to assess the differences among the three or four groups. Differences between the two groups were assessed using the Wilcoxon test. The JMP software (version 15; SAS Institute Inc., Cary, NC) was used for performing all statistical analyses with values of p < 0.05 considered statistically significant.
Results
Mice showed signs of AKI after S. aureus challenge with substantial bacterial load in the kidneys
Three days after the S. aureus challenge, the mice demonstrated significant elevations in plasma Cr, BUN, and urine KIM-1 levels (Figure 1(a)–(c)), indicating the development of AKI after the induction of S. aureus bacteremia. Only half of the mice survived seven days after bacterial challenge in this model, with substantial propagation of bacteria in the kidney on the 7th day as compared to the liver and spleen (Figure 1(d)). These findings suggest that the potent propagation of challenged S. aureus in the kidney causes AKI in mice.
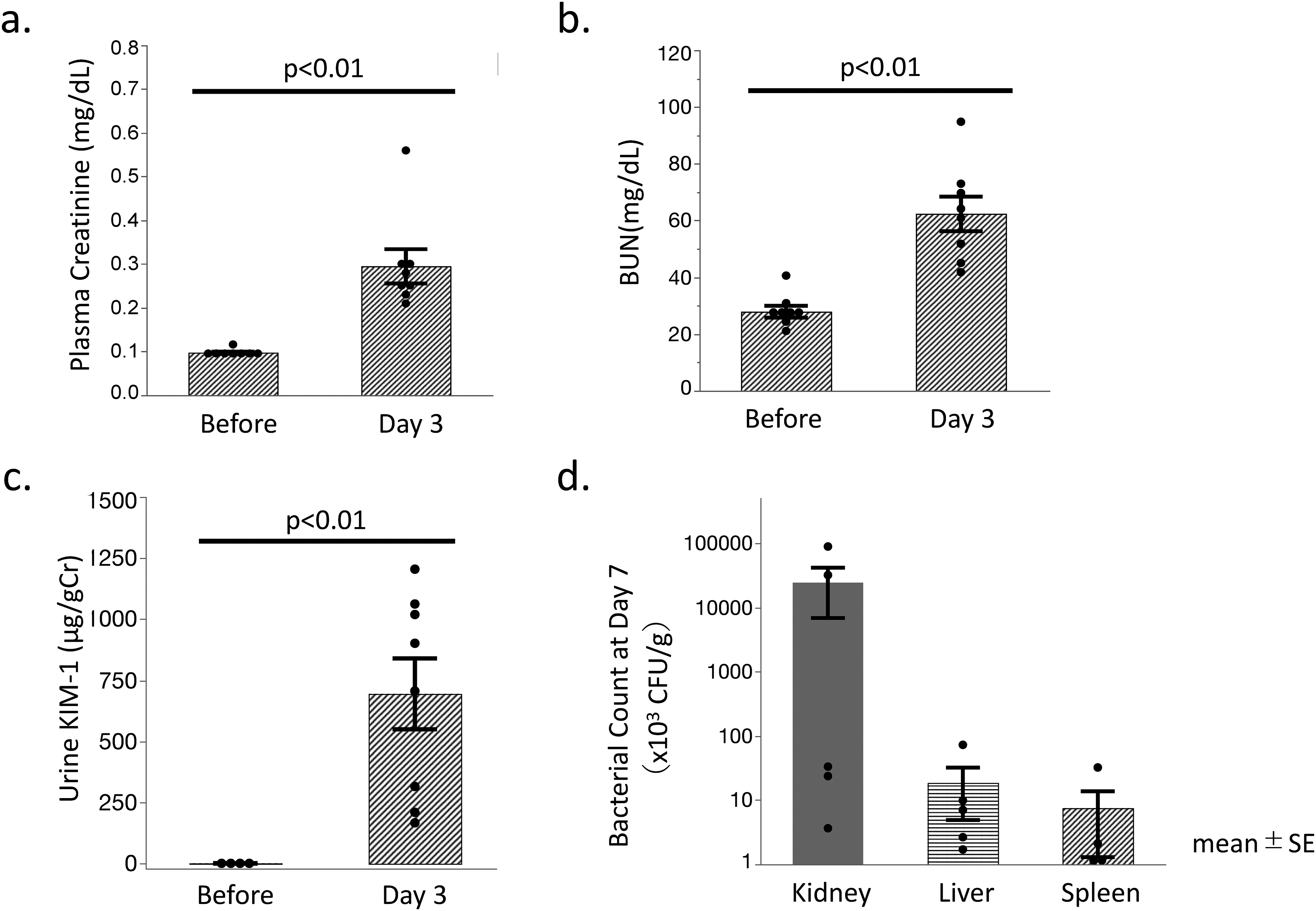
Renal dysfunction and bacterial propagation in each organ after S. aureus challenge.
Mouse renal macrophages exhibited weak antibacterial activity against S. aureus compared with the liver or spleen macrophages
In order to examine the antibacterial activity of nontreated normal mice using pHrodoTM-S. aureus, F4/80+ macrophages were obtained from their kidney, liver, and spleen. Interestingly, renal F4/80+ macrophages exhibited weak antibacterial activity against S. aureus, while hepatic and splenic F4/80+ macrophages exhibited potent antibacterial activity against S. aureus (Figure 2(a)). Renal macrophages as well as the liver macrophages in mice can be divided into the F4/80low CD11bhigh BMDM and the F4/80high CD11blow TRM (Figure 2(b)).7,10,13 BMDM as well as TRM in the mouse kidney failed to exhibit marked antibacterial activities against S. aureus. The proportions of pHrodoTM-S. aureus-positive cells were less than 10% in both BMDM and TRM (BMDM 4.95% ± 1.48%, TRM 5.18% ± 1.07%).

Bactericidal activity of F4/80-positive cells in each organ against S. aureus.
LPS preconditioning augmented the antibacterial activity against S. aureus in both BMDM and TRM of the mouse kidney
Immunohistochemical analysis demonstrated that LPS preconditioning enhanced the levels of F4/80+ cells in the kidney (Figure 3(a) and (b)). A diffused increase in these cells was observed in the renal tubular interstitium (Figure 3(a)). Additionally, flowcytometric analysis demonstrated that LPS preconditioning significantly enhanced the count of BMDM, but not TRM, in the kidney tissue (Figure 3(c)), while it augmented the antibacterial activity against S. aureus in renal BMDM and TRM (Figure 4). With regards to the cytokine production by renal macrophages, significantly higher positive percentages of intracellular TNF-α staining by in vitro S. aureus stimuli were exhibited by BMDMs compared with that of TRMs in control mice (BMDMs: 5.0 ± 1.6 vs. TRMs: 0.9% ± 0.1%, p < 0.05 in control mice). LPS preconditioning did not affect the intracellular TNF-α staining in both BMDMs as well as TRMs (BMDMs: 6.8 ± 1.8, TRMs: 0.9% ± 0.3%, in LPS-pretreated mice). The apparent intracellular IL-10 by in vitro S. aureus stimuli could not be detected in either BMDMs or TRMs in mice, with as well as without LPS preconditioning (Supplemental Figure 2). Intracellular IL-1β (active form) could not be examined since it is difficult to distinguish the IL-1β (active form) from the IL-1β precursor.
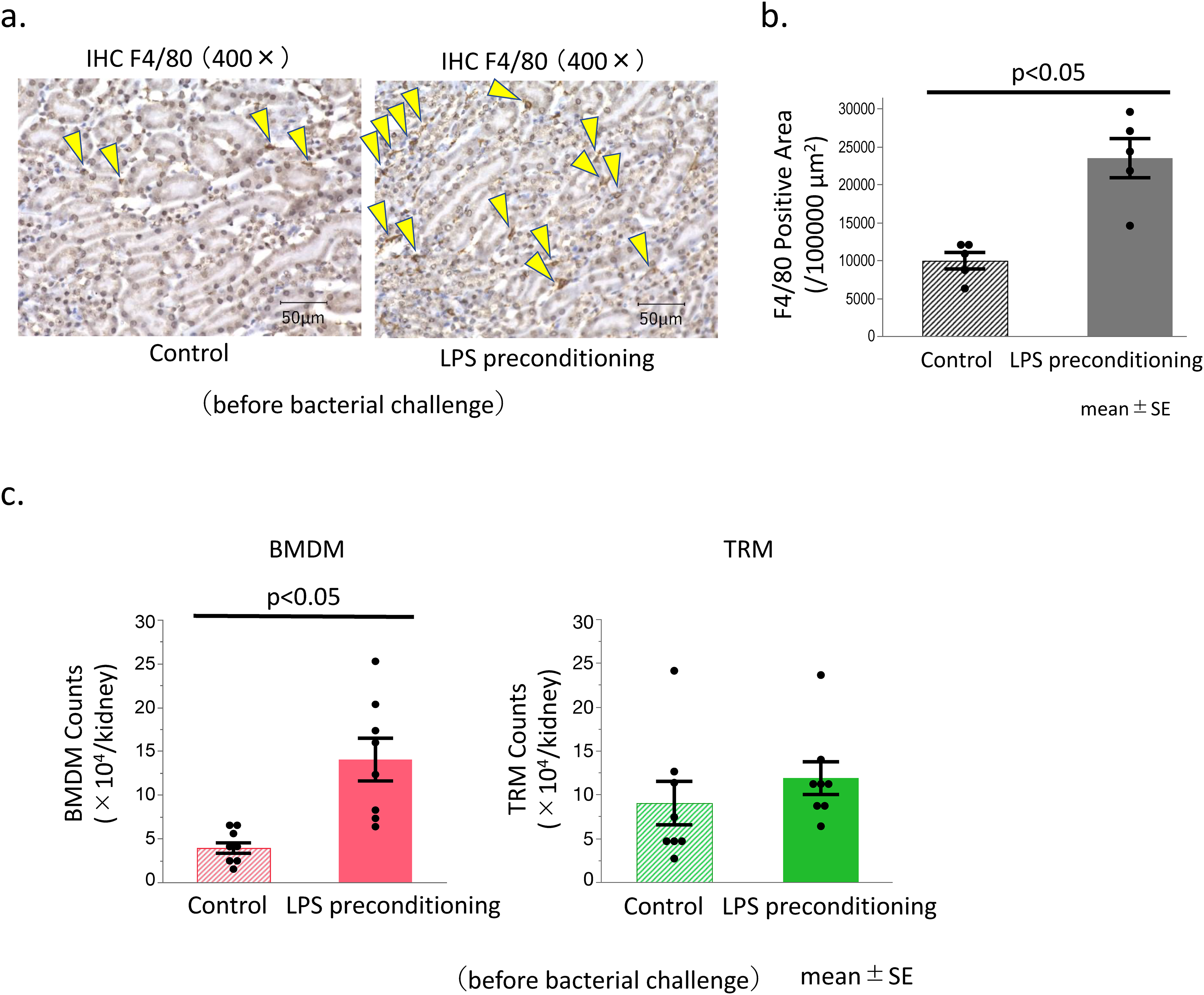
LPS preconditioning induced the accumulation of BMDM in the kidney.
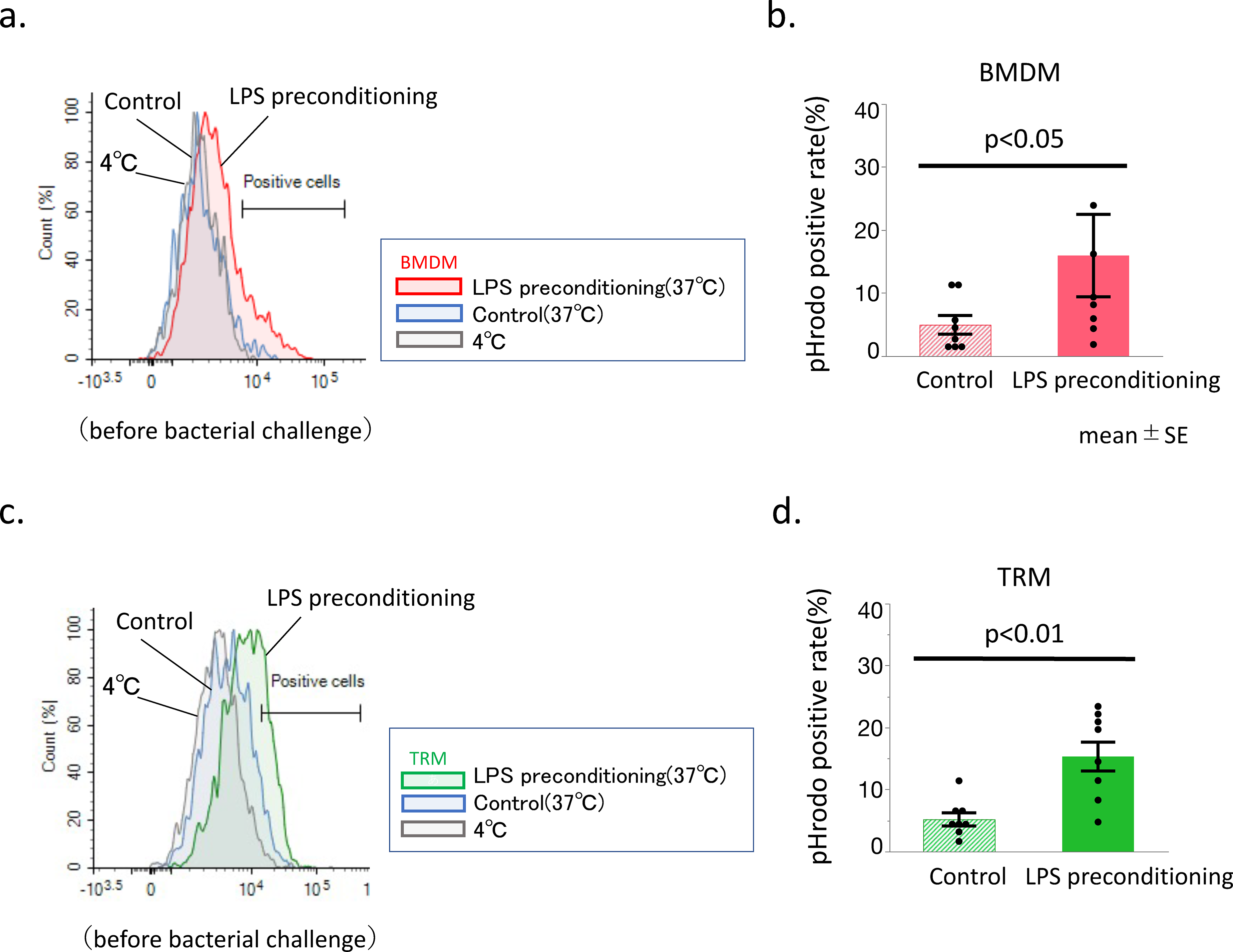
Change in the phagocytosis rate of renal macrophages after LPS preconditioning.
LPS preconditioning reduced the bacterial load in the kidneys of S. aureus-challenged mice and improved survival rates
Substantial bacterial load was observed in the kidneys of the untreated mice three days after the S. aureus challenge. LPS preconditioning significantly reduced the bacterial load 3 days after the S. aureus challenge in not only the kidneys but also in the liver and the spleen (Figure 5(a)). Consistent with these findings, LPS preconditioning significantly upregulated the survival rates of mice following S. aureus challenge and mitigated the weight loss observed 3 days after S. aureus challenge (Figure 5(b) and (c)).

Effect of LPS preconditioning on bacterial propagation and mouse survival after S. aureus challenge.
LPS preconditioning ameliorated AKI in mice after S. aureus challenge
Untreated mice demonstrated biochemical signs of AKI in the form of elevated plasma BUN, creatinine, and urine KIM-1 levels 3 days following S. aureus challenge. LPS preconditioning caused a significant reduction in the levels of these biomarkers (Figure 6(a)–(c)), thereby confirming that the LPS preconditioning ameliorated AKI in mice following S. aureus challenge. S. aureus challenge also elevated plasma proinflammatory cytokine levels, such as TNF and IL-1β, measured at 3 days in untreated mice after, while LPS preconditioning markedly reduced these elevation of plasma TNF and IL-1β (Figure 6(d) and (e)). No significant increase was observed in the plasma IL-10 levels after the S. aureus challenge in both the LPS preconditioning group as well as the control group (data not shown).
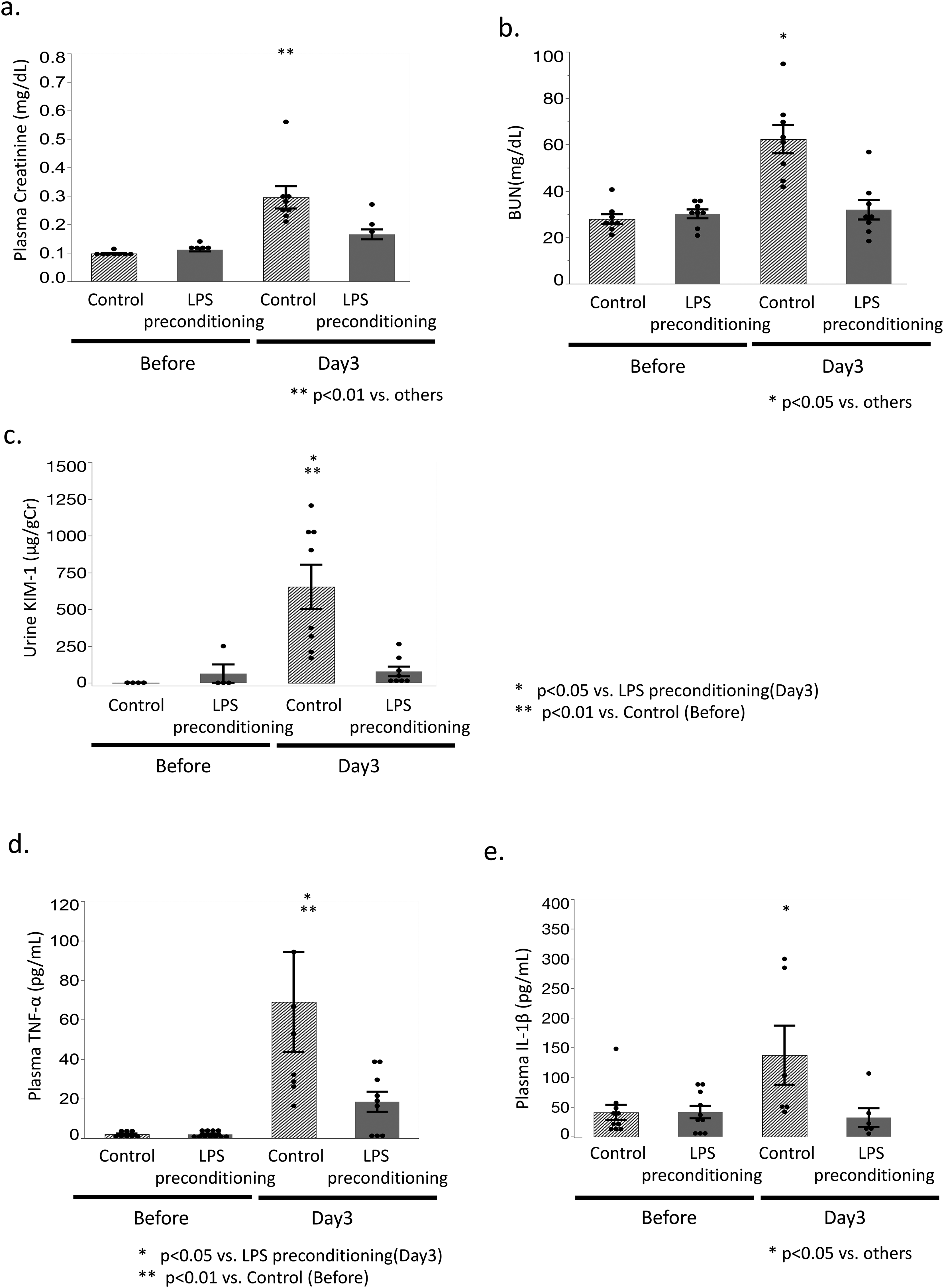
Effect of LPS preconditioning on renal function and plasma cytokine levels.
No abscess formation was found in the kidneys of LPS-preconditioned mice after S. aureus challenge
While the LPS-preconditioned mice exhibited no abscess formation in the kidney at 3 days after the S. aureus challenge, approximately 43% (3/7) of the untreated mice exhibited grossly visible abscess (Figure 7(a)). Histologically, the control mice exhibited the presence of neutrophils karyomegaly, exudates, fibrin precipitation, and abscess formation, accompanied by bacterial growth and necrosis in the kidney. Although localization varied among individuals, a tendency for abscess formation near the renal pelvis and around the urothelium was observed. Quantifying the exact area of the abscesses was difficult since the area varied depending on the section of the renal specimen. Therefore, the number of bacteria in the renal tissue homogenate was counted rather than those confined to just the abscess area, to evaluate the total bacterial counts in the tissue (Figures 1(d), 2(c), 4(a)). Besides the kidney, abscess formation was not observed in any other organs including the liver, spleen, lung etc., 3 days after the S. aureus challenge in untreated mice. Tubular dilation, tubular cast, and tubular degeneration were observed in the control mice, 3 days following the S. aureus challenge, while LPS preconditioning reduced the extent of tubular damage, thereby resulting in an improved AKI score (Figure 7(b) and (c)). LPS preconditioning exhibited the tendency of increasing the number of BMDM (but not TRM) in the kidney 3 days after the challenge (Figure 7(d)). In contrast, tissue myeloperoxidase (MPO) levels (a marker of neutrophil content) in the kidney were similar in LPS mice as well as the untreated mice, indicating that LPS preconditioning did not affect neutrophil accumulation in the mouse kidney after S. aureus challenge (Figure 7(e)).
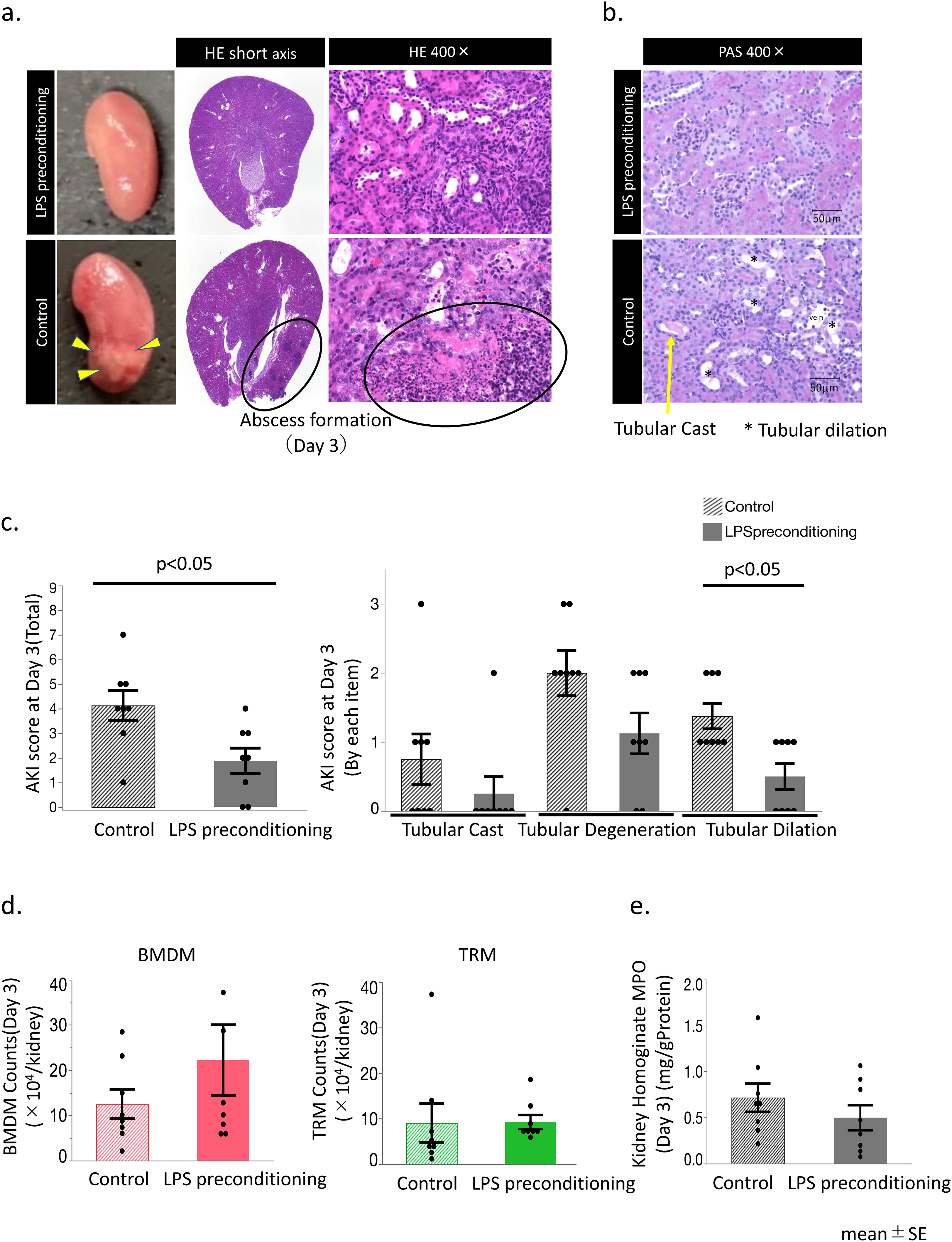
Histological findings and leukocyte accumulation in the kidneys following the S. aureus challenge.
LPS preconditioning increased the CD80/CD206 (M1/M2) ratio of renal BMDM before bacterial challenge, whereas it tended to decrease the M1/M2 ratio 3 days after S. aureus challenge
Prior to the bacterial challenge, the LPS-preconditioned mice exhibited elevated CD80-positive (M1 macrophage) population in renal BMDM without any change in the CD206-positive (M2 macrophage) population, compared with the control mice (Figure 8(a), Supplemental Figure 1a and b). These population changes resulted in an elevated CD80/CD206 (M1/M2) ratio (Figure 8(b)). However, the LPS-preconditioned mice exhibited significantly low CD80-positive counts in BMDM 3 days following the S. aureus challenge without any alteration in the CD206-positive population as compared to untreated mice (Figure 8(a), Supplemental Figure 1a and b). This resulted in a lower CD80/CD206 (M1/M2) ratio (Figure 8(b)). LPS preconditioning did not affect the CD80/CD206 (M1/M2) ratio in the renal TRM either before or after bacterial challenge (Figure 8(c), Supplemental Figure 1c and d).

CD80/206 ratio (M1/M2 ratio) of the renal BMDM and TRM.
LPS preconditioning did not alter TLR2 expression on BMDM or TRM in the kidney, but it decreased the level of MHC class II expression
The expression of TLR2, which recognizes peptidoglycan, a component of Gram-positive cocci bacteria including S. aureus, was examined in this study. The level of TLR2 expression remained the same despite LPS preconditioning in BMDM as well as TRM (Figure 9(a)). Nonspecific phagocytosis was assessed by examining CD64. CD64 (FcγRI) expression was similar in LPS-preconditioned as well as control mice, although CD64 expression elevated both BMDM and TRM in both groups following the S. aureus challenge (Figure 9(b)). In contrast, MHC class II expression was significantly reduced by LPS preconditioning, not only on BMDM but also TRM in the kidney, although these decreases in MHC class II expression disappeared 3 days after the S. aureus challenge (Figure 9(c)). MHC class II is an indicator of antigen presentation capacity.

Expression of TLR2, CD64 (FcγRI), and MHC class II on renal macrophages.
Discussion
Our research has demonstrated the lower antibacterial activity of renal F4/80+ cells against S. aureus, compared with liver-resident Kupffer cells and spleen F4/80+ Cells. This factor could be attributed to the greater susceptibility of the kidneys to local abscess formation compared with the other organs, although several other factors could possibly be involved. Notably, LPS preconditioning significantly augmented the antibacterial activity against S. aureus in both types of macrophages (BMDM and TRM) in the kidney, thereby increasing the rate of survival of the mice after S. aureus infection and ameliorating S. aureus-induced AKI due to the rapid clearance of the bacterial load. Consistent with these findings, LPS preconditioning resulted in reduced pathological findings of renal abscess after S. aureus challenge in mice. In a mouse AKI model associated with rhabdomyolysis, the predominance of the renal macrophages F4/80low CD11bhigh Ly6bhigh CD206low was observed 2 days after the onset of AKI. After 8 more days, the macrophages F4/80high CD11bint Ly6blow CD206high were reported to be the most abundant. 18 These two types of renal macrophages (presumably BMDM and TRM) were highly involved in the pathogenesis of AKI in a time-dependent manner in this model. 18
LPS preconditioning significantly enhanced the number of renal BMDM with an increase in CD80 expression, a canonical marker of proinflammatory (M1) polarization. However, after challenging with S. aureus, signs of M1 suppression were observed in the BMDMs in preconditioned mice, but not in the untreated mice. Conversely, the number or the M1/M2 (CD80/CD206) ratio in renal TRM was not significantly affected by the LPS preconditioning. According to literature, TRM is a phenotype with strong M2 polarity. Consistent with this understanding, a high rate of polarization of TRMs to M2 was observed in LPS-preconditioned mice and control mice. TRMs being inherently highly M2-polarized, the M1/M2 ratio may not significantly be converted into M1 suppression by LPS preconditioning after S. aureus challenge, indicating that BMDM are the major contributors to the LPS preconditioning effect.
Previous studies on the effects of LPS preconditioning in the kidney have been conducted on ischemia-reperfusion and LPS-induced sepsis models. Most of these reports have focused on the alterations in the tubular structure and function. During LPS preconditioning, the CD14/TRIF signaling pathway in myeloid cells, particularly macrophages, has been reported to be vital for tubular protection. 18 After LPS preconditioning, the macrophages accumulate around the S1 proximal tubule, polarize toward M2, increase the expression of redox and iron handling molecules, decrease renal tubular peroxisomal damage, and reduce the oxidative stress. 18 lPS preconditioning was also observed to eliminate S2 and S3 tubular damage. 18 The present study is novel since sepsis-associated AKI was induced with S. aureus bacteremia and the analysis focused on investigating changes in two types of kidney macrophages, particularly the changes in their phagocytic capacity and inflammatory response. This study demonstrated that the macrophages BMDM and TRM had different responses to LPS preconditioning, with BMDM exhibiting greater changes in population number while TRM exhibited greater changes in phagocytosis capabilities. This analysis has provided a more granular analysis of the changes in renal macrophages during LPS preconditioning and suggested a mechanism of the pathogenesis of sepsis-associated AKI resulting from the changes in the dynamics of these two subpopulations.
Phagocytosis is one of the major roles of macrophages. After recognizing dead cells, pathogens, and toxins, the macrophages uptake them by phagocytosis followed by subsequent processing and degradation. During in vitro stimulation with pHrodo S. aureus, LPS preconditioning was observed to enhance the phagocytotic and bactericidal capacity of renal macrophages. The process of digestion requires phagosome acidification, which is regulated by the NLRP3 inflammasome and caspase-1 in phagosomes containing Gram-positive bacteria. 19 After accumulating in phagosomes, active caspase-1 locally regulates the pH by modulating the buffering strength by the NADPH oxidase NOX2. 20 The NLRP3 inflammasome of macrophages is reportedly activated by the in vitro stimulation with LPS. 19 These mechanisms might be possibly involved in enhanced phagocytosis by renal macrophages in LPS-preconditioned mice. However, the inflammatory response is drastically reduced by the LPS preconditioning,5,6 which indicates the inactivation of the inflammasome. Future studies are warranted to examine the possible relationship between LPS preconditioning and NLRP3 inflammasome activation.
Yet another major role of macrophages is antigen-presentation. Exogenous antigens (bacteria, viral particles, etc.) are absorbed into macrophages by endocytosis or phagocytosis. Subsequent fusion of the endocytic vesicles with lysosomes generates extracellular antigen-derived peptides. Vesicular transport carries the MHC class II molecules bound to antigenic peptides to the cell surface and present the antigen to helper T lymphocytes. Macrophages recognizing trace amounts of LPS are normally expected to present the LPS-derived peptides as antigens on the cell surface, but their MHC class II expression was reduced in the current study. Further studies are needed to examine whether LPS preconditioning altered the actual antigen presentation.
Cytokine production is the third major role of macrophages. MyD88- and TRIF-dependent intracellular signaling in macrophages that is promoted by TLR activation, enables the recognition of PAMPs via TLRs, ultimately resulting in NF-κB activation and inflammatory activation. In accordance with previously conducted studies, LPS preconditioning was previously reported to reduce TLR4 expression; 21 however, changes in TLR4 expression in macrophages could not be detected in this study. TLR2 recognizes peptidoglycans from Gram-positive cocci, including S. aureus. However, TLR2 expression did not change after LPS preconditioning, indicating that LPS preconditioning did not “prime” the innate immune system to recognize S. aureus. In the present experiment, the Control and LPS preconditioning groups demonstrated comparable production of inflammatory cytokines (TNF-α) by renal macrophages. Anti-inflammatory cytokine (IL-10) levels were low in both groups. This indicates the possible limited effect of LPS preconditioning on cytokine production by renal macrophages. However, one reason for the lack of differences could be the fact that intracellular cytokines were measured with in vitro stimulation.
There are several limitations to this study. First, the current study demonstrated a marked reduction in the bacterial load in the mouse kidney following S. aureus bacteremia; however, this cannot be attributed to the effect of kidney macrophages alone. Indeed, there is a high likelihood that the systemic effects were caused not only by the kidney but also by the macrophages resident in different organs (such as the liver). However, LPS preconditioning was confirmed to enhance the bactericidal activity against S. aureus in macrophages extracted from kidneys using the in vitro pHrodo assay. Another limitation in considering future applications of LPS in clinical settings is that the biological responses in mice and humans to the LPS doses might differ. LPS, which is a causative agent of organ damage and sepsis, is also a potent, dose-dependent pyrogen. Finally, one key limitation was that the observation period of AKI was limited to 3 days following infection. Approximately half of the control mice died during the 7-day observation period; therefore, long-term observations could not be conducted with the current model system. Therefore, kidney fibrosis and also the transition to chronic kidney disease after AKI could not be examined. In addition, limitations regarding the animal models existed. AKI associated with S. aureus bacteremia included the pathogenesis of septic AKI as well as the pathogenesis of AKI caused by renal infection/abscess. Considering the availability of limited animal models that are appropriate for septic AKI, further investigation of an appropriate septic AKI model is imminent.
Perspectives and significance
LPS preconditioning augmented the phagocytic capacity of renal macrophages and reduced AKI following S. aureus challenge. LPS preconditioning is a preventive measure and not a treatment for sepsis, which limits the scope of its clinical application. Appropriate indications for LPS preconditioning could include compromised patients at high risk of bloodstream infection owing to catheter placement and/or diabetic patients prone to severe skin and soft tissue infections. LPS preconditioning might be a useful measure to prevent severe infections and complications of AKI as an adjunctive therapy for such patients.
Supplemental Material
sj-docx-1-ini-10.1177_17534259251335770 - Supplemental material for Lipopolysaccharide preconditioning augments the antibacterial activity of renal macrophages and ameliorates acute kidney injury caused by Staphylococcus aureus bacteremia in mice
Supplemental material, sj-docx-1-ini-10.1177_17534259251335770 for Lipopolysaccharide preconditioning augments the antibacterial activity of renal macrophages and ameliorates acute kidney injury caused by Staphylococcus aureus bacteremia in mice by Keiko Tanoue, Manabu Kinoshita, Bradley M Kearney, Seigo Ito1, Hiroyasu Goto, Aoi Yamashiro, Tsugumi Fukunaga, Hiroki Sato, Kazuma Mori, Koji Kuwata, Hidehito Matsubara, Azusa Kato, Masahiro Nakashima, Hiroyuki Nakashima, Toshihiko Imakiire and Naoki Oshima in Innate Immunity
Footnotes
Acknowledgments
We would like to thank Hiroo Kumagai, Emeritus Professor, of the National Defense Medical College (NDMC)(Tokorozawa, Saitama, Japan), for useful discussions. We are also thankful to Professor Emeritus Shuji Seki for discussing the results. We are grateful to all the staff in the Department of Immunology and Microbiology and the Department of Nephrology and Endocrinology of NDMC, including laboratory assistants and secretaries.
Ethical considerations
The protocols for the animal experiments were approved by the Ethics Committee of Animal Care and Experimentation of the National Defense Medical College, Japan(approval number 20018) on May 27, 2021, in compliance with the NDMC guidelines for the care and use of laboratory animals.
Consent to participate
Not applicable
Consent for publication
Not applicable
Author contributions/CRediT
KT and MK designed the study, the main conceptual ideas, and the proof outline. KT, SI, HG, KK, and KM collected the data. TF, HS, and AY aided in interpreting the results and worked on the manuscript. HM, AK, MN, and HN provided support on how to conduct the experiment and analyze the results. NO supervised the project. KT wrote the manuscript with support from MK and B.M.K. All authors discussed the results and commented on the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by JSPS KAKENHI (grant number 21H3038), advanced research of defense medicine, and the TERUMO LIFE SCIENCE FOUNDATION under Grant [number 23-III5005], and the funding agency had no role in the design, data collection, data analysis, and reporting of this study.
Conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
