Abstract
Host response to lung infection includes coordinated efforts of multiple cell types, including the lung epithelium and macrophages. Importantly, both the lung epithelium and macrophages can internalize and clear invading pathogens. However, the mechanisms and their ability to internalize or phagocytose differ. Akt is a key cellular pathway that controls cell proliferation and survival, in addition to its role in host defense. The role of the Akt pathway was assessed using pharmacological Akt modulators in lung epithelial (A549) and macrophage (RAW 264.7) cell lines during Klebsiella bacterial infection. Our data show that the inhibition of the Akt pathway using specific Akt inhibitor MK2206 increased the phagocytic ability of lung epithelial cells but not of macrophages. In contrast, the activation of Akt using specific activator SC-79 decreased the phagocytic ability of epithelial cells, while it increased the phagocytic ability of macrophages. The altered phagocytic ability in both cell types using Akt modulators was not due to changes in bacterial adhesion to the host cell. The clinical usefulness of these Akt modulators may vary based on the type of infection and on the relative contribution of epithelial cells and macrophages in clearing the particular bacterial infection. The Akt pathway has differential roles in the internalization of Klebsiella bacteria by respiratory epithelial cells and immune cells.
Introduction
Bacterial lung infections are a leading cause of morbidity and mortality. 1 Antibiotics are the main therapy currently used to manage bacterial lung infections. However, with the emergence of new antibiotic resistance strains, the usefulness of antibiotic therapy continues to decrease.2,3 Klebsiella pneumonia (Kp) is a significant human pathogen that is inherently resistant to many of the clinically used antibiotics. Recently, new Kp strains have emerged that are resistant to carbapenem by means of expressing an enzyme known as carbapenemase.4,5 The rate of discovery of antibiotics is at an all-time low, and there continues to be an urgent need to develop new ways to boost the host defense against these bacterial pathogens. 6
Phagocytosis is a mechanism by which host cells engulf and kill invading pathogens.7,8 Although phagocytosis has been described for more than 100 yr and its usefulness in bacterial clearance remains undisputed, no clinical interventions are yet available to boost or modify this mechanism to improve the host defense against bacterial pathogens. Two major cell populations in the lungs that actively phagocytose and kill pathogens include the lung epithelium 9 and macrophages, 10 which are resident in the lung, as well as those recruited to the lung during infections.
The mechanisms of phagocytosis have been well studied, especially those in macrophages. Macrophage phagocytosis is controlled by a group of small GTPase that play a role in controlling actin cytoskeleton polymerization to form the phagocytic cup. 11 Along with the actin cytoskeleton, other organelles such as the endoplasmic reticulum participate in macrophage phagocytosis. 12 PI3K is recruited to the cell surface during phagocytosis and contributes in both pseudopodia extension and endoplasmic reticulum–mediated phagocytosis. 12 , 13 In contrast to macrophages, limited knowledge exists on the contributing pathways during phagocytosis in lung epithelium. Epithelial phagocytosis is often referred to as “internalization,” which is considered a pathogen-mediated event rather than a host-mediated event. 14 The inability of the epithelium to phagocytose is attributed to a lack of Fcγ receptors for opsonic phagocytosis. Not as much is known about how bacteria become internalized or “phagocytosed” by lung epithelial cells. In this article, the term “internalization” is interchangeable with the term “phagocytosis.” The Akt pathway, which is activated by phosphorylation by PI3K, is one of the major pathways that regulate cell survival and cell growth during the conditions of homeostasis and stress. 15 Akt has been shown to be involved in the phagocytic mechanisms in macrophages. 16 However, it is unclear how Akt relates to the phagocytic mechanisms in the lung epithelium. Here, using both macrophage and lung epithelial cell lines, we show that the Akt pathway differentially regulates phagocytic ability, depending on the cell types.
Methods
Bacterial and cell culture
GFP-Kp cells (a kind gift from Dr. Samithamby Jeyaseelan, Louisiana State University, Baton Rouge, LA) were cultured on a Luria broth (LB) agar plate overnight (16 h) at 37°C, and a single colony was transferred to liquid LB medium to culture at 37°C with shaking, which was sub-cultured for 1 h the next day, as described previously. 17 The number of bacteria was estimated by measuring OD at 600 nm and confirmed by plating serial dilutions on the agar plates. Epithelial cell line A549 (ATCC CCL-185) and macrophage cell line RAW 264.7 (ATCC® No. TIB-71™) were cultured in DMEM supplemented with 10% FBS, penicillin G (100 IU/ml), and streptomycin (100 µg/ml; all from Gibco) at 37°C under 5% CO2. Cells numbers were counted using Hematology Analyzer Coulter Ac·T diff (Beckman Coulter).
Bacterial adhesion and internalization assays
A549 cells and RAW264.7 cells were seeded at 5 × 105 cells per well on a 24-well plate and allowed to grow for 24 h at 37°C in a 5% CO2 incubator. For the adhesion assays, cells were washed three times with PBS and incubated in cell culture medium with MK2206 (Selleckchem) or SC79 (Selleckchem) for 1 h at 37°C in 5% CO2. The cells were then washed three times with PBS and infected with Kp at a MOI of 10:1 for 2 h at 37°C in 5% CO2 in the cell culture medium. After incubation, the wells were washed three times with PBS. To lyse the cells, 200 µl of lysis buffer (0.25% Triton X-100) was added to each well and incubated at 37°C for 10 min. Quantification was subsequently performed by plating serial dilutions on LB agar plates. On the second day, CFUs were counted.
For the internalization or phagocytosis assay, to kill extracellular bacteria, the cells were incubated at 37°C for an additional 2 h with cell culture media containing 100 µg/ml gentamicin (Gibco). This method allowed us to count only internalized pathogens, as the antibiotic treatment for the given duration was sufficient to kill the extracellular pathogens. Intracellular Kp was released by lysing the cells using lysis buffer (containing Triton X-100), and the total number of internalized Kp was quantified after plating sample aliquots on agar plates similar to the adhesion assays.
Microscopy
Monolayers of epithelial cells and macrophages were infected as described previously. After incubation for 1 h in the cell culture medium with gentamicin (100 µg/ml), cells were washed three times using PBS and were fixed with 4% formalin. Monolayer sections were stained with fluorescein-conjugated wheat germ agglutinin (WGA-Alexa Fluor™ 594 Conjugate; 5 µg/ml; Invitrogen) for visualization of the cell membrane. Cell nuclei were stained by incubation with a DAPI solution for 10 min (Sigma–Aldrich; 5 µg/ml) in PBS. The cells were examined and imaged using a Nikon microscope.
Lactate dehydrogenase assay
Lactate dehydrogenase (LDH) release assays were performed to evaluate the toxicity of Akt inhibitor MK2206 and Akt activator SC79 in A549 and RAW 264.7 cell lines using the Invitrogen CyQUANT™ LDH Cytotoxicity Assay Kit as per the manufacturer’s protocol. Fifty µl of sample supernatant (control, time-dependent, and concentration-dependent treatments) was transferred to a 96-well plate. Absorbance was measured at 490 and 680 nm. The specific cytotoxicity was calculated using the following formula: % cytotoxicity = (treated LDH activity–control LDH activity)/(maximum LDH activity–control LDH activity) × 100.
Western blot analysis
The cells were collected and lysed in RIPA lysis buffer (Millipore) with protease/phosphatase inhibitors (Roche). The cell lysates were centrifuged at 13,800 g for 20 min at 4°C. The concentration of total protein was determined by BCA assay, as per the manufacturer’s protocol. Equal amounts of the lysate proteins were loaded into the wells of the SDS-PAGE. After electrophoresis, the proteins were transferred to a PVDF membrane using the Trans-Blot system from Bio-Rad. Membranes were then blocked with 5% nonfat milk in Tris-buffered saline solution for 1 h and incubated with primary Ab overnight at 4°C. The membrane was incubated with the HRP-labeled secondary Ab for 1 h at room temperature, and the immunoreactive bands were detected by using HRP substrate. The intensity of the bands was quantified using the Bio-Rad ChemiDoc MP Imaging System from Bio-Rad. Abs to Phospho-Akt (Ser473), total Akt (C67E7), β-actin (13E5), and anti-rabbit secondary Abs were purchased from Cell Signaling Technology.
Statistical analysis
Data were analyzed using GraphPad Prism v8 (GraphPad Software). Each experiment was performed at least three times, each in duplicate, and the data are expressed as the mean ± SEM. A two-tailed Student’s t-test or ANOVA, as appropriate, was used to assess the differences in values between the experimental groups. In all analyses, P values of < 0.05 were considered statistically significant.
Results
Macrophage and epithelial cells demonstrate time- and dose-dependent internalization ability
First, we compared the phagocytic ability of macrophages using RAW264.7 cells and lung epithelial cells using A549 cells. Our phagocytosis protocol is summarized in Figure 1. Both of these cells were incubated with either a varying number of bacteria (MOI 5–80) or a MOI of 10 at varying time points. Our data showed that there was an increase in the internalization ability of both epithelial and macrophages, in both a time- and MOI-dependent manner (Figure 2a–d). Of note, as reported earlier, the epithelial phagocytic ability was significantly lower compared to the phagocytic ability of the macrophages, which are professional phagocytes. 18 However, our data showed very interesting trends where macrophage internalization seemed to be quick and come almost to saturation by the 2-h time point (Figure 2d). In contrast, the epithelial internalization ability continued to increase until 8 h post infection (Figure 2a), suggesting a slow but steady rate of internalization of bacteria in the epithelial cells. Similarly, upon adding increasing amounts of bacteria, the epithelial internalization ability continued to increase (Figure 2b). In contrast, there was only a small increase in the internalization ability of macrophages observed at higher MOIs (Figure 2e). Together, these data suggest that while professional phagocytes are quick to respond and eliminate the pathogens, epithelial cells may play important roles by their continuous internalization ability over a prolonged period.
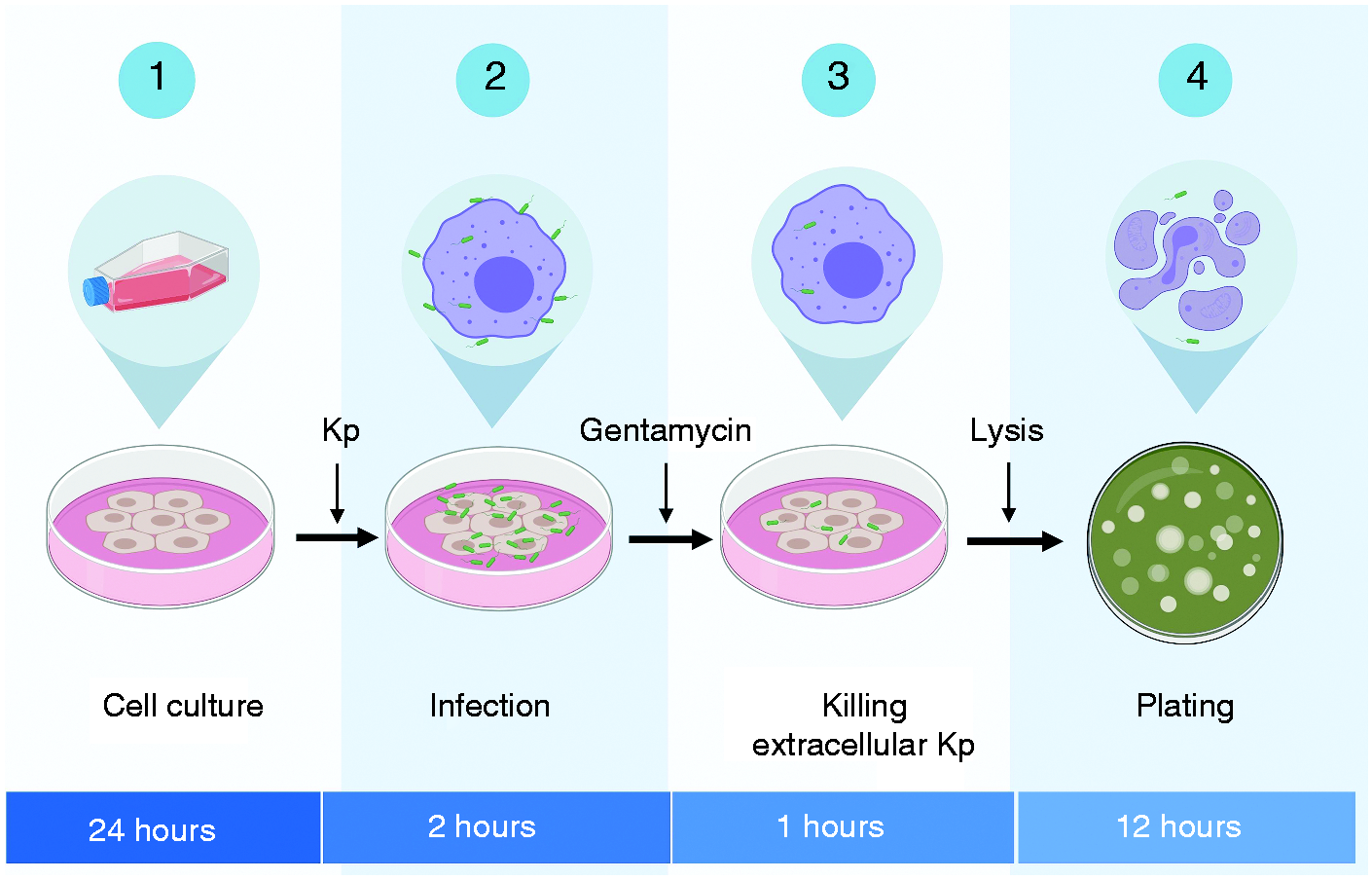
Schematic representation of phagocytosis assay. Epithelial or macrophage cell lines were seeded onto plates overnight and then infected with different MOIs of Klebsiella pneumonia (Kp) for 2 h unless indicated otherwise. Extracellular bacteria were then washed off by washing three times with PBS. To kill extracellular bacteria, cells were then treated with media containing gentamicin. Cells were washed again to remove the gentamicin using PBS. Cells were then lysed to release the intracellular bacteria, which were plated on agar plates to calculate the number of CFU.
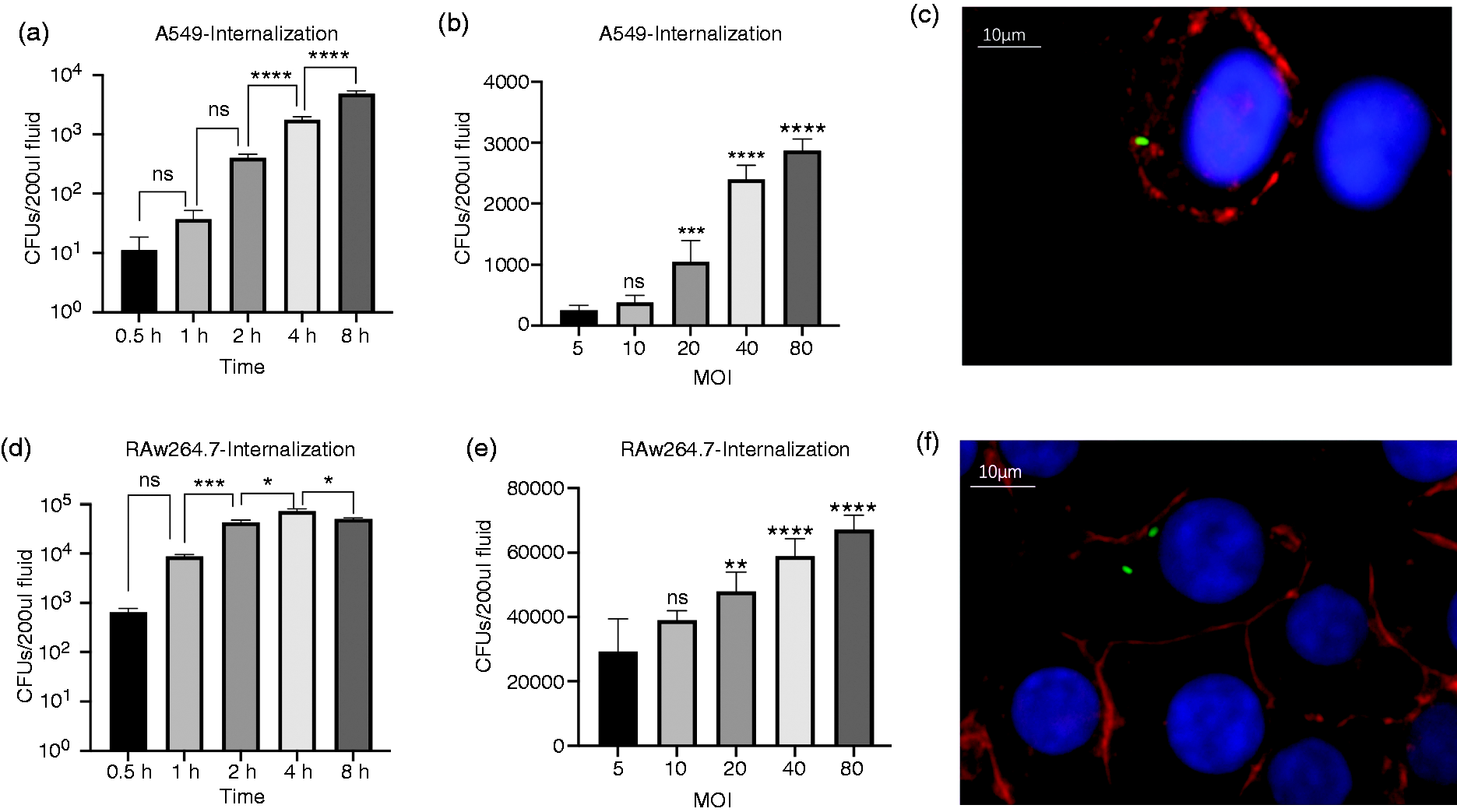
Time- and dose-dependent phagocytic ability of macrophage and epithelial cells. The internalized bacteria numbers in A549 (a) and RAW264.7 (d) after infection with Klebsiella pneumoniae (Kp) at MOI of 10 at indicated time points. The internalized bacteria number in A549 (b) and Raw264.7 (e) after 2 hours of incubation with Kp at MOI from 5 to 80 (c and f) Microscopy showing internalized Kp (GFP) in A549 (c) and RAW264.7 (f) at the 400 X after staining with wheat germ agglutinin (Red) and DAPI stain to localize the nucleus (blue). Scale bars represent 10μm. The data are expressed as the means ± SEM of three individual experiments. *P < 0.05 vs control.
Kp differentially activates the Akt pathway in epithelial cells and macrophages
The Akt pathway has been shown to play an important role in gut epithelial cells, where Akt regulates bacterial translocation across the gut epithelial monolayer. 19 Similarly, overexpression of constitutively active phosphorylated Akt has been shown to promote phagocytosis in macrophages. 20 We first sought to determine if the Akt pathway is activated during the phagocytic process in epithelial cells. We performed a time-course experiment of Akt activation in the lung epithelial cell line A549 (Figure 3a) during Kp infection. Our data showed that epithelial cells had a rapid Akt activation, which was evident by 15 min post infection. Akt phosphorylation remained stably high until 4 h post infection, followed by a decrease at 8 h post infection. In contrast, in the macrophage cell line RAW264.7 cells, infection with Kp led to a deactivation of the Akt pathway at the early time point of 5 min and continued to be down-regulated until 4 h post infection, at which point Akt phosphorylation was restored to baseline (Figure 3b). These data demonstrate that Kp activates the Akt pathway in epithelial cells but deactivates the Akt pathway in macrophage cells.
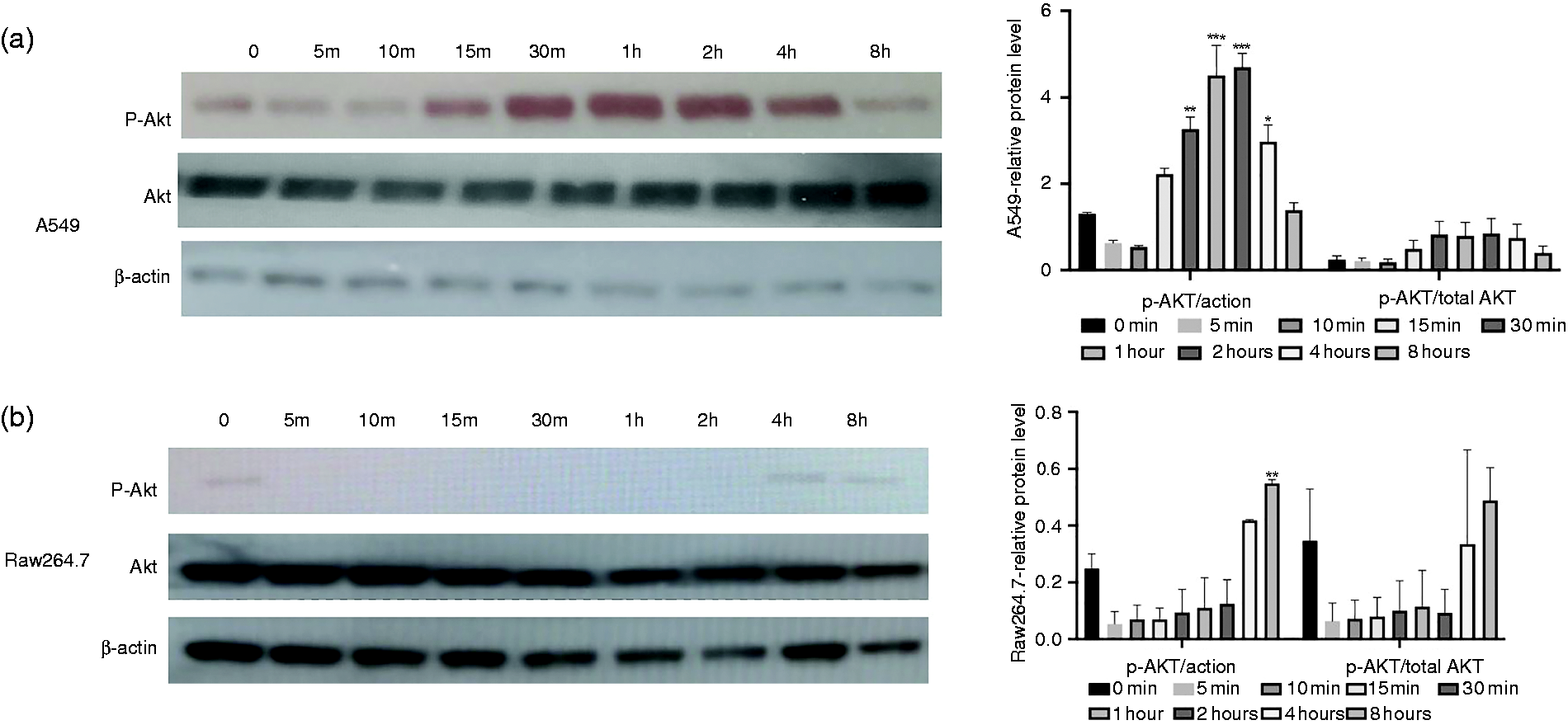
Western blot analysis of the Akt pathway in both epithelial cells and macrophages. Cell lysates were collected after Kp infection at a MOI of 10 at different time points and subjected to immunoblot with anti-phospho Akt (Ser473) (top panel) and anti-total Akt (middle panel). (a) A549 cells show a rapid Akt activation, which was visible from 15 min to 4 h post infection, and a decreasing Akt activation was observed by 8 h. The right bar graph shows the quantification or phosphorylated Akt normalized to either β-actin or total Akt. (b) RAW264.7 cells demonstrate a deactivation of the Akt pathway at early time points, which was restored to baseline by 4 h post infection. The right bar graph shows the quantification or phosphorylated Akt normalized to either β-actin or total Akt. *P < 0.05; **P < 0.01; ***P < 0.005 compared to 0 h control group using one-way ANOVA.
Pharmacological inhibition of the Akt pathway increased phagocytic ability of epithelial cells but not macrophages
Next, we sought to determine whether inhibition of the Akt pathway alters the phagocytic ability of macrophages and epithelial cells. Phagocytosis/internalization assay was performed as depicted in Figure 1. We used MK2206, a well-characterized specific inhibitor of the Akt pathway, to treat these cells. First, we determined the nontoxic concentrations of MK2206 that can be used to study its impact on phagocytosis in epithelial and macrophage cells. Our data showed that there was no significant LDH release by MK2206 at concentrations up to 100 μM (Figure 4a). Our phagocytosis data demonstrated that treatment with MK2206 at 1 and 10 μM concentrations significantly increased the phagocytic ability of epithelial cells as measured by a gentamicin isolation assay (Figure 4c). These effects are specific to phagocytosis, as no difference was observed on the adhesion of bacteria to the A549 monolayer by MK2206 (10 µM; Figure 4b). In contrast to epithelial cells, macrophage phagocytosis was not affected by treatment with MK2206 at similar concentrations (Figure 4e). Again, no changes in the adhesion of bacteria to the cell surface were observed in the presence of MK2206 in a macrophage cell line (Figure 4d). Overall, these data demonstrate that the inhibition of the Akt pathway may contribute differentially in phagocytosis of Kp.
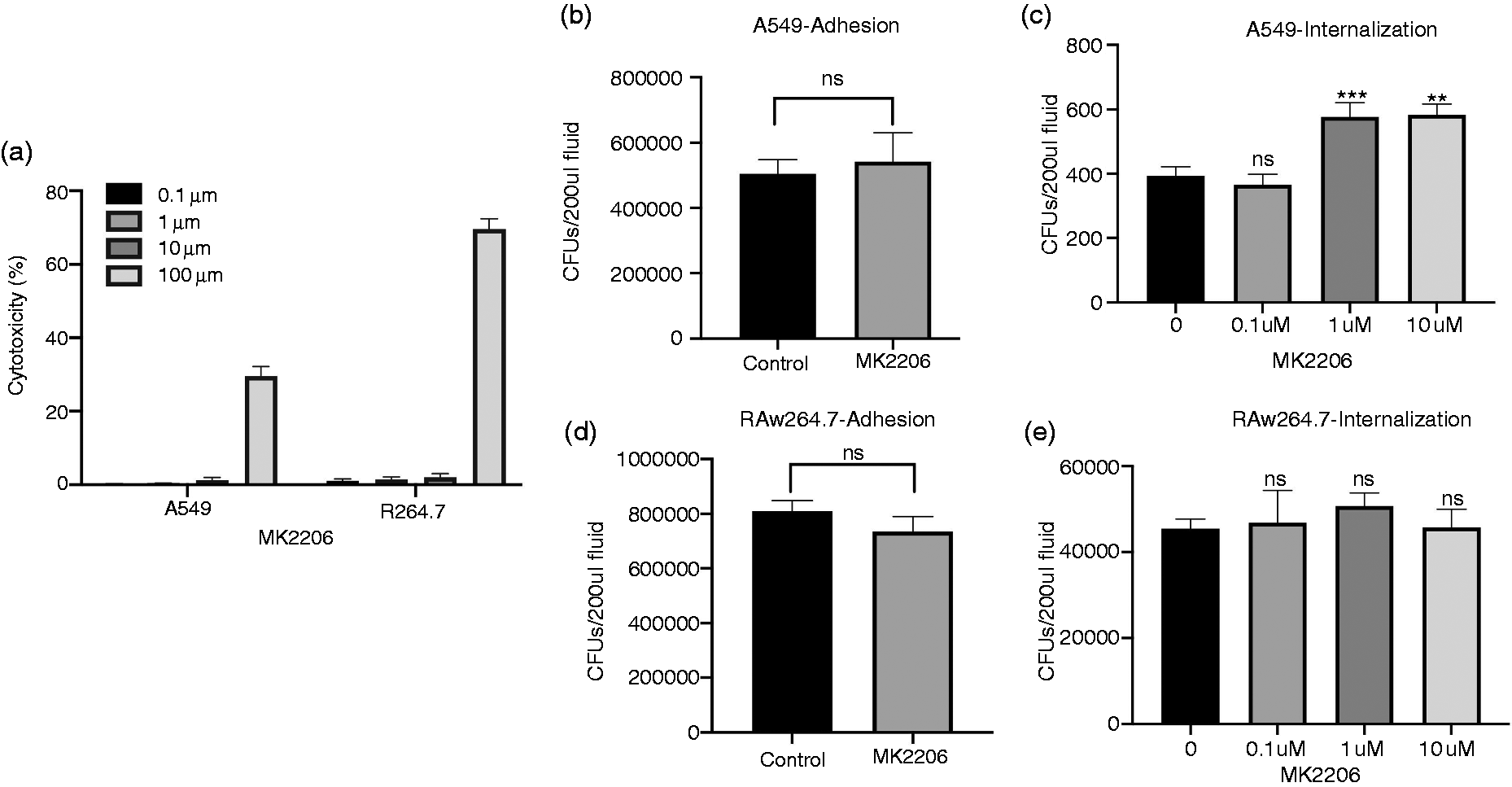
Effects of pharmacological inhibition of the Akt pathway on the phagocytic ability of epithelial cells and macrophages. (a) Levels of lactate dehydrogenase (LDH) release were measured in both of the cell lines in the presence of indicated concentrations of MK2206. The number of adhered bacteria was counted in the presence or absence of MK2206 in A549 (b) and RAW264.7 (d) cells at a MOI of 10. Bacterial internalization was measured in the presence or absence of MK2206 in A549 (c) and RAW264.7 (e) cells at a MOI of 10. Results are shown as the mean ± SEM of three separate experiments. *P < 0.05 vs. control. ns: not significant.
Pharmacological activation of the Akt pathway decreased phagocytosis in lung epithelium but increased phagocytosis in macrophages
Next, we sought to determine if pharmacological activation of Akt pathway contributes to the alteration in the phagocytic ability of lung epithelium and macrophages. We used Akt activator SC-79 to activate Akt in both these cell types. First, we performed LDH release assays to determine the nontoxic doses of SC-79 on macrophages and epithelial cells, where doses up to 10 μM were found to be nontoxic to both cell types (Figure 5a). Next, we used nontoxic doses of SC-79 to understand its effects on phagocytosis in epithelial cells and macrophages. Our data showed that Akt pathway activation led to a dose-dependent decrease in the phagocytic ability of epithelial cells (Figure 5c), which was independent of the adhesion of bacteria to epithelial cells, as similar adhesion was observed in groups that were treated or not treated with SC-79 (Figure 5b). In contrast to the epithelial cells, we did not observe any decrease in the phagocytic ability of macrophages in the presence of SC-79. Indeed, an increase in the phagocytic ability was observed at 1 μM concentration of SC-79 (Figure 5e). These effects on phagocytosis were not due to altered adhesion of Kp to macrophages or epithelial cells, as similar adhesion was observed in the presence of SC-79 in macrophages (Figure 5d). Together, these data suggest that Akt activator potentiates the phagocytic ability of macrophages. However, in epithelial cells, Akt activation leads to decreased phagocytic ability.
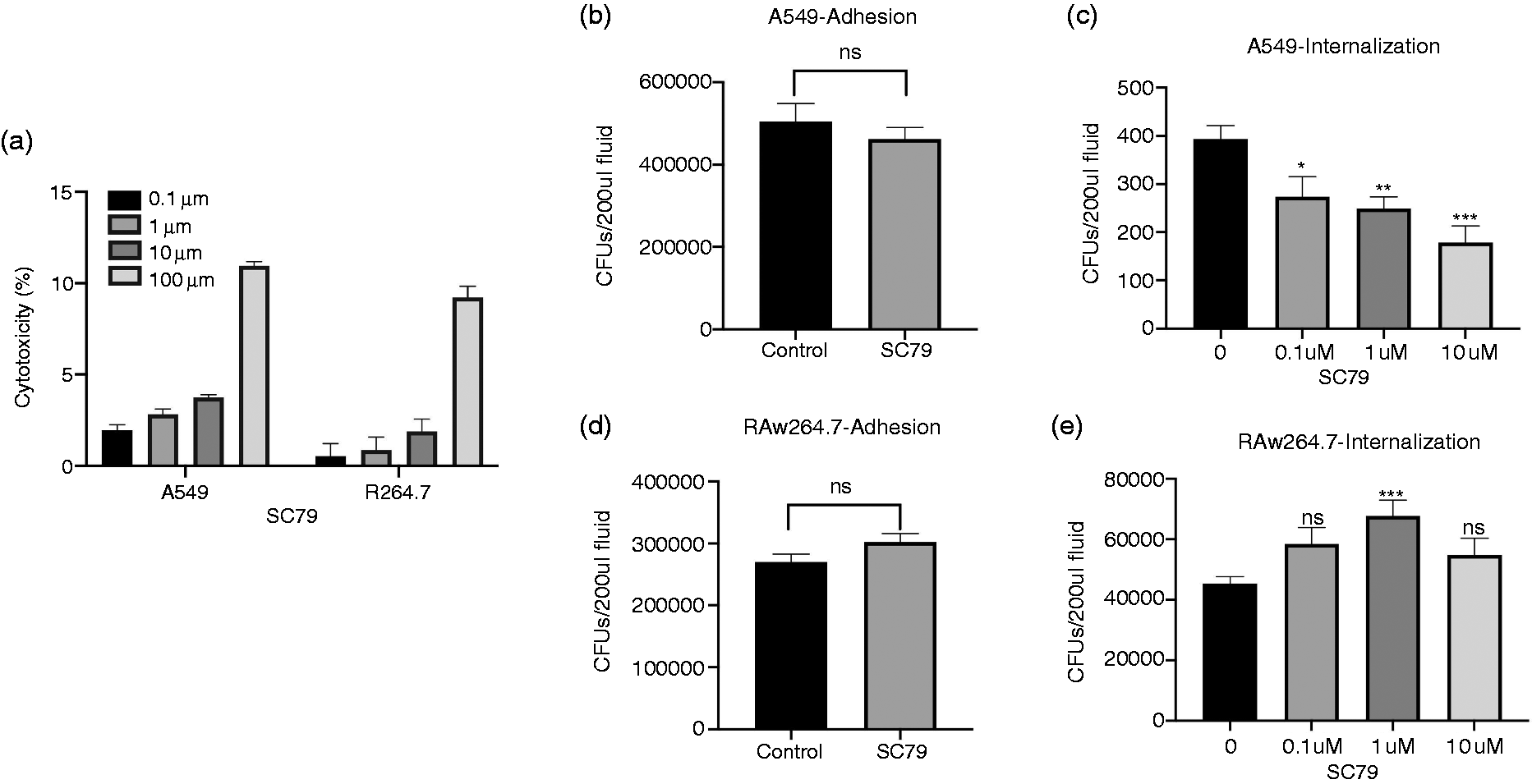
Effects of pharmacological activation of the Akt pathway on the phagocytic ability of epithelial cells and macrophages. (a) Levels of LDH release were measured in both of the cell lines in the presence of indicated concentrations of SC79. Bacterial adhesion in the presence or absence of SC-79 at a 10 μM concentration in A549 (b) and RAW264.7 (d) cells at a MOI of 10. Bacterial internalization was measured at a MOI of 10 at indicated concentrations of SC-79 in A549 (c) and RAW264.7 (e) cells. Results are shown as the mean ± SEM of three separate experiments. *P < 0.05 vs. control.
Discussion
Phagocytosis is a key mechanism by various cells that supports a vast variety of functions from the fertilization of eggs to the clearance of bacterial, viral, and fungal pathogens from the organs. 21 , 22 In the lung, phagocytosis remains a key mechanism to maintain homeostasis, as the lung is persistently exposed to environmental threats such as particles and pathogens. 23 , 24 Various structural cells such as lung epithelium, or immune cells such as macrophages that are both resident and recruited during lung infection, can perform the phagocytic function in the lung. In epithelial cells, this process has also been termed “internalization.” Although the end outcome of phagocytosis is bacteria being engulfed within the epithelial cell, the process can be significantly different. 9 Immune cell–mediated phagocytosis is an active process where epithelium recognizes the pathogen by its PRRs and initiates the phagocytosis process. 25 Epithelial phagocytosis may be pathogen initiated, where various pathogens use it to hide within the epithelial cell. 14 Our data reveal interesting and novel differential roles of Akt pathways in these different cell types.
Consistent with previous notions, epithelial cells possess significantly lower per cell ability to phagocytose than macrophages (Figure 2a–d). The second important insight provided by our data is that the macrophage phagocytic ability process is relatively quick and reaches saturation within the first hour (Figure 2d and e). However, the epithelial phagocytic ability is slower but steady over the time studied (Figure 2a and b). It is possible that the phagocytic function of the epithelial cells is instead pathogen mediated, which continues to invade the epithelium cell over time. Another explanation may be that macrophages can effectively kill the phagocytosed pathogen, which limits its accumulation in these cells. The lysosomal maturation in macrophages can happen within a few minutes, while the same process may take hours in epithelial cells. 26 , 27 However, these measurements were not made for lung epithelial cells or a respiratory pathogen such as Kp. In the absence of effective killing mechanisms, epithelial internalized pathogens may keep accumulating over time. Bacterial invasion of epithelial cells has been reported previously, where it contributes toward the pathogenicity of the invading bacteria. 28 , 29
Akt is a central pathway that controls cell growth and metastatic invasion—a well-characterized phenomenon in cancer biology. However, the role of the Akt pathway in infection biology has only recently emerged. Pathogens are known to activate the Akt pathway, 30 including Kp. Our data confirm the earlier findings in the A549 cell line 31 that Kp induces early activation of Akt, which was evident by 15 min post infection in these cells (Figure 3a). However, in contrast, we did not see a strong induction of Akt activation in the macrophage cell line RAW264.7 (Figure 3b). These data show a differential response to Akt in epithelial and macrophage cell lines, possibly due to the differential response mounted by these two types of cells.
Similar to activation patterns, we observed dramatic differences in the effects of Akt modulators on the phagocytic ability of macrophages and epithelial cells. While specific inhibitors of Akt increased the phagocytic ability of epithelial cells (Figure 4c), they did not alter the phagocytic ability of macrophages (Figure 4e). In contrast to Akt inhibitor, our data show that Akt activator SC-79 inhibited the phagocytic ability of lung epithelial cells in a dose-dependent manner (Figure 5c). However, in contrast to epithelial cells, we observed an increased phagocytic ability of macrophages when they were treated with SC-79 (Figure 4e), suggesting opposing effects of the Akt pathway in epithelial and immune cells.
Similar to our finding, the genetic overexpression of activated Akt in the RAW264.7 cell line led to an increase in phagocytic ability. 20 It seems that the Akt pathway may be conserved to enhance the phagocytic ability of macrophages. At the same time, pathogens have developed mechanisms to inhibit the Akt to decrease the phagocytic ability of macrophages to evade the host defense. For example, capsular proteins from Streptococcus has been shown to inhibit the Akt pathway, which limits the ability of macrophages to phagocytose the bacteria. 32 It is unknown how Akt modulators lead to differential effects on the phagocytic ability of immune and structural cells, but it may be possible due to the different phagocytic mechanisms, as shown in these two types of cells in our study. While phagocytosis by macrophages may represent a host defense mechanism, the invasion of epithelium by bacteria may represent pathogen mechanisms to invade the tissue. 29 The overall contribution of these opposite effects needs to be studied using in vivo experiments.
Further studies are also required to understand how cell-specific alteration of the Akt pathway can be achieved to enhance host resistance against pathogens. Limiting epithelial invasion by Akt activators may ultimately be beneficial to the host if epithelial invasion provides a safe niche for replication and avoiding killing by immune cells and antibiotics. In such cases, boosting macrophage phagocytic function may provide an added advantage in more effectively eliminating pathogens that fail to enter epithelial cells. On the other hand, limiting macrophage invasion using Akt inhibitors can be beneficial for lung infections with intracellular pathogens such as Legionella species, which replicate within macrophages. 33 However, the effect of Akt activation on macrophage and epithelial phagocytic function needs to be ascertained against other bacterial pathogens, including clinical isolates of Kp, to understand if the presented paradigm applies in a broader extent.
This article has focused mainly on a representative structural cell (epithelium) and immune cells (macrophages) to study the effect of the Akt pathway in understanding the host immune response. However, other immune cells such as neutrophils play a key role in bacterial clearance from the lung, including during Kp lung infections. 34 In neutrophils, Akt has been demonstrated to be essential in the generation of reactive oxygen species—a key process by which neutrophils kill the engulfed pathogens. 35 However, further studies are needed to confirm their role during Kp infection in neutrophils using pharmacological activators and inhibitors. Furthermore, it remains unclear how Akt modulation affects other functions of epithelial cells and macrophages, such as those related to pathogen sensing and the secretion of cytokines. Further detailed studies, including in vivo studies, are required to understand the full spectrum of modulation of the Akt pathway during bacterial lung infections.
Footnotes
Acknowledgements
We really appreciate the efforts of all the researchers whose articles are included in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Beijing Nova Program Interdisciplinary Cooperation Project (DC; No. Z191100001119021), China Scholarship Council (DC; No. 201809112037), and NIH NHLBI (CDC; No. R01HL126094), ALA (LS; No. 513385) and ATS (LS; No. 2018-18).
