Abstract
Introduction
The autoimmune chronic demyelinating illness of the central nervous system (CNS), known as multiple sclerosis (MS), is typically accompanied by progressive impairment of varied degrees. 1 It is categorized into four major subtypes according to its clinical course with the relapsing-remitting MS (RRMS) being the most common subtype. 2 Autoreactive immune cells that penetrate the CNS and induce focal inflammation and demyelination are the primary cause of clinical exacerbations in the majority of RRMS patients. 3 Early diagnosis of MS is necessary, and investigations such as blood and cerebrospinal fluid (CSF) analyses and magnetic resonance imaging (MRI) can help in MS patients’ evaluation. 4
In MS, monocytes, macrophages, and microglia are crucial in mediating CNS inflammation and modulating both pro- and anti-inflammatory responses. 5 Macrophages originate from circulating blood monocytes, which penetrate the CNS during acute inflammation and cause disease progression and demyelination. The phenotypic polarization of monocytes depends on their surface receptor expression, functions, and cytokine production. 6 Various studies distinguished three main monocyte subsets according to the surface expression of CD14 and CD16: the classical (CD14+CD16−), intermediate (CD14+CD16+), and nonclassical (CD14−CD16+) monocyte subsets.5,7
In this context, the present study examined the presence of various monocyte subsets and their association with disease activity in Egyptian patients with RRMS.
Methodology
Study type and subjects
The present case-control study included 44 patients with RRMS diagnosed according to the 2017 MacDonalds criteria. 8 They were selected from the outpatient clinic of the Neurology Department at Ain Shams University Hospital, Cairo, Egypt. They were divided into 22 patients in relapse and 22 patients in remission. Forty-four matched controls were also included. The study did not include patients with malignancies, other neurological illnesses, chronic inflammatory diseases, or significant obesity (body mass index >40 kg/m2). The study was given ethical approval number FMASU MS 514/2020 by the Faculty of Medicine Research Ethics Committee at Ain Shams University. Before the beginning, each participant provided written, informed consent, and all study information was kept private and used exclusively for research.
Clinical evaluation
After full personal and medical history was taken from all participants, a neurological examination was done with the assessment of disease disability by the Expanded Disability Status Scale (EDSS), which measures impairment from 0 to 10 in 0.5-unit increments. Higher scores indicate more disability. People with good ambulatory ability are 1.0 to 4.5, whereas those with a loss are 5.0 to 9.5. 9
Sample collection and storage
From each participant, 2.5 ml of venous blood was withdrawn under complete aseptic conditions on an ethylenediaminetetraacetic acid dipotassium salt (K2-EDTA) vacutainer tube. Samples were processed within 2–3 h of collection in the flow cytometric analysis.
Immunophenotyping of monocyte subsets
The collected samples were used in the flow cytometric analysis of monocyte subsets by using Fluorescein isocyanate (FITC) conjugated CD 16 (Beckman coulter, France) lot number: 200109 and Phycoerythrin (PE) conjugated CD 14 (Beckman coulter, France) lot number: 200080 monoclonal antibodies. Sample acquisition was done using Navios flow cytometry (Beckman coulter, USA). The gating strategy is shown in Figure 1

Gating strategy of monocyte subsets. To distinguish monocytes from other leucocytes, the cells were chosen based on their forward and side scattering intensities (FS-INT and SS-INT). The three monocyte subsets, the classical (CD14+CD16−), intermediate (CD14+CD16+), and non-classical (CD14−CD16+) monocytes, were then represented on a CD14 vs CD16 graph.
Statistical methods
The SPSS version 23 (IBM Corp., USA, 2015) was used in the statistical analysis. After data description, the comparisons were done using the independent t-test and the One-Way ANOVA test. The correlation of quantitative nonparametric data was done using the Spearman correlation coefficient. The p-value <0.05 is significant.
Results
The clinical data of the included RRMS patients (n = 44) are included in Table 1. The percentages of classical, intermediate, and non-classical monocyte subsets were significantly higher in RRMS patients than in controls, with p-values of 0.029, 0.049, and 0.043, respectively. Table 2.
Description of RRMS patients’ (n = 44) clinical data.

CSF: cerebrospinal fluid; EDSS: Expanded disability status scale; MRI: Magnetic resonance imaging; NA: Not available; OCB: Oligoclonal bands.
Comparison between control group and RRMS patients regarding their classical, intermediate, and non-classical monocyte subset percentages.

p-value <0.05: Significant.
•: Independent t-test.
There was no significant difference in the percentages of the classical and intermediate monocyte subsets with any of the clinical parameters of all RRMS patients (p-values >0.05). At the same time, the non-classical monocyte subset percentages in RRMS patients were significantly higher in those with evidence of activity in MRI than those without (20.77 ± 8.42 vs. 9.09 ± 4.59; p-value = 0.002). No other significant differences in the percentages of the non-classical monocyte subset with the other clinical parameters were found. Table 3.
Comparison of the classical, intermediate, and non-classical monocyte percentages according to the clinical data of the ms patients.
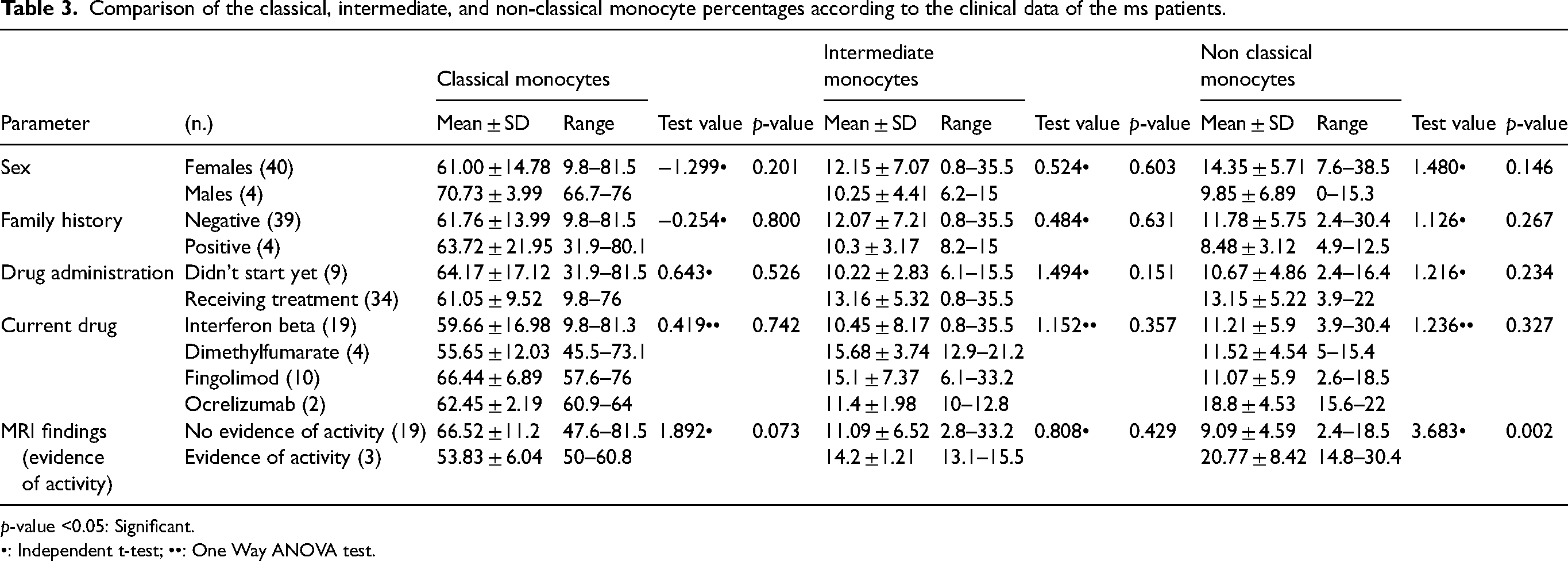
p-value <0.05: Significant.
•: Independent t-test; ••: One Way ANOVA test.
No statistically significant correlations (p-values >0.05) were found between the percentages of the different monocyte subsets and RRMS patients’ age, EDSS scores, duration of the disease, and the number of relapses in the last year. Table 4.
Correlation between the percentages of the different monocyte subsets and other clinical data of the RRMS patients.

p-value <0.05: Significant.
EDSS: Expanded disability status scale; r: Spearman correlation coefficient.
In addition, the percentage of each monocyte subset did not show any significant differences between RRMS patients in remission and those in relapse (p-values >0.05). Table 5.
Comparison of the monocyte subset percentages between remission and relapse groups among the included RRMS patients.

p-value <0.05: Significant.
•: Independent t-test.
Discussion
The progression of MS and its disability risk is unpredictable. Despite several studies, few biomarkers can monitor MS progression and therapy responses. 10 Microglia, macrophages, and monocytes all contribute significantly to the CNS inflammation observed in MS. 7 Numerous investigations have demonstrated that the pathogenesis of MS is promoted by the inflammatory cytokines secreted by monocytes, which could contribute to blood-brain barrier breakdown, especially in active demyelinating lesions. Monocytes may also play a role in the resolution phase of inflammation and repair processes.7,11
Characterizing monocyte subsets in MS can help understand disease pathogenesis and treatment targets. The most recent successful MS treatments depend on either depleting peripheral blood B cells or preventing T cells from entering the CNS. 12 MS may be treated with a similar strategy targeting monocytes or myeloid lineage cells. 10
The present study showed an increase in the pan-monocyte subsets in RRMS patients compared to healthy controls. Several studies investigated the percentages of different monocyte subsets. Still, they gained conflicting results, possibly due to how the patient cohorts were assembled with varying disease stages and activity and differences in treatment plans or methods used for monocyte purification before analysis. In addition, it is widely believed that the different blood monocyte subsets correspond to different maturation phases, with non-classical monocytes being the most developed. 13
Fischer et al. in 2019 1 reported that classical monocytes were significantly more abundant in MS patients independently of the disease activity than in healthy controls. However, the non-classical monocytes were less frequent in MS patients, and the intermediate monocytes were unaltered. Also, Waschbisch et al. in 2016 14 showed that RRMS patients had lower peripheral blood non-classical monocyte percentages than healthy controls. They explained the decrease in circulating non-classical monocytes by insufficient bone marrow recruitment, unbalanced monocyte differentiation, and CNS compartmentalization.
On the other hand, Chuluundorj et al. in 2014 11 found that the proportion of CD14+ monocytes was decreased in their MS patients compared to the controls, while the proportion of CD16+ monocytes, which are more differentiated form of CD14+ monocytes, increased and exhibited a highly proinflammatory activity being more effective in antigen presentation and activation of T cells than the CD14+ monocytes. Similarly, several studies reported that the peripheral blood proinflammatory CD16+ monocytes displayed higher percentages in MS patients when compared to healthy controls.5,10,15 Chuluundorj et al. in 2014 11 also found that CD16+ monocytes from MS patients displayed higher inflammatory markers (HLA-DR and CCR2) and produced significantly more interleukin (IL)-6 than CD14+ monocytes after activation. They stated that the role of CD14+ monocytes in MS might be mainly clearance of neuronal debris following the inflammation and regeneration of neurons rather than the generation of inflammation being the primary producer of IL-10.
Although animal studies imply that non-classical monocytes play a protective function in MS, it is still unclear whether the elevated frequencies of non-classical monocytes seen in humans with MS are compensatory or disease-causing. 7 Narasimhan et al. in 2019 7 reported that non-classical monocytes maintain vascular endothelial homeostasis and are considered protective as they can help identify and eliminate infections and malignant cells. However, in chronic disease, their action is unclear; many investigations have linked them to disease burden.
The present study found that the monocyte subtype percentages in RRMS patients did not correlate with the degree of disability by EDSS nor the number of relapses during the last year. This finding goes along with several previous studies.5,10,16 In addition, although there were no significant differences in the percentages of monocyte subsets regarding clinical disease activity in the current study as monocyte subset percentages did not differ significantly between patients in relapse and those in remission, similar to Fischer et al., 2019, 1 the included RRMS patients with evidence of activity in MRI had a significant increase in the non-classical monocyte percentage than those without evidence of activity in MRI. This finding could be explained by the close resemblance between the CD16+ monocytes and the neurotoxic monocytes found in demyelinating MS lesions. Additionally, during MS, CD16+ monocytes, but not CD14+ monocytes, play a critical role in triggering inflammatory T cells that damage nerves.11,17,18 As CD14+ monocytes are not detected in active MS lesions but in surrounding tissue, they are unlikely to damage neurons directly. 3 In contrast to our results, Haschka et al. in 2020 16 found a significant expansion in classical and non-classical monocytes in inactive RRMS patients compared to those in clinical activity. Gjelstrup et al. in 2018 5 found that the monocyte subsets were not correlated with MRI findings.
Treatment with disease-modifying therapy (DMT) can result in an intense M2 polarization of myeloid cells, ameliorating MS symptoms. 1 The current study found no difference in monocyte subset percentages according to the DMT administered. Similarly, Haschka et al. in 2020 16 found no effect of DMT administration on the monocyte subset distribution pattern. Also, Fischer et al. in 2019 1 found that monocyte subsets remained unaltered by glucocorticoid treatment concerning short- and long-term effects. They stated that it was challenging to remove monocytes with DMT because of the idea that they have a short half-life.
The current study's limitations including the small sample size and lack of comparing patients according to their different treatment regimens, point out the need for more extensive, multicenter studies with more extended follow-up periods, enrollment of patients with various types of MS and levels of activity, and use of different treatment protocols to better understand the function of monocytes in MS pathogenesis.
In conclusion, the current study found a pan-monocyte expansion, including the classical, intermediate, and non-classical subsets in RRMS patients, regardless of the disease activity or the DMT administration. The non-classical monocytes were associated with MRI evidence of disease activity. These results denote the vital role of monocytes, especially the non-classical, and innate immunity, in RRMS pathogenesis and suggest monocytes as a promising therapeutic target and a possible diagnostic tool.
Footnotes
Acknowledgments
The authors thank all patients who participated in this study and the research assistants who helped collect data.
Authors contribution
The study design was done by all authors. S.I.T. shared in clinical evaluation and sample collection and reviewed the manuscript. H.G.M. shared in clinical evaluation and reviewed the manuscript. R.M. shared in clinical evaluation and reviewed the manuscript. N.E.K. did the investigations and wrote the manuscript. S.S.K shared in clinical evaluation and sample collection and wrote the manuscript.
Consent for publication
Not applicable.
Data availability
All data are available in the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Human ethics and Consent to participate
The study was given ethical approval number FMASU MS 514/2020 by the Faculty of Medicine Research Ethics Committee at Ain Shams University. Before the beginning, each participant provided written, informed consent, and all study information was kept private and used exclusively for research.
