Abstract
Here, we describe the production and characterization of a novel p65fl/fl/LysMCre mouse model, which lacks canonical nuclear factor-kappaB member RelA/p65 (indicated as p65 hereafter) in bone marrow-derived macrophages. Cultured bone marrow-derived macrophages that lack p65 protein reveal NF-κB signaling deficiencies, a reduction in phagocytic ability, and reduced ability to produce nitrites. Despite abnormal bone marrow-derived macrophage function, p65fl/fl/LysMCre mice do not exhibit differences in naïve systemic immune profiles or colony forming units and time to death following Salmonella infection as compared to controls. Additionally, p65fl/fl/LysMCre mice, especially females, display splenomegaly, but no other obvious physical or behavioral differences as compared to control animals. As bone marrow-derived macrophages from this transgenic model are almost completely devoid of canonical nuclear factor-kappaB pathway member p65, this model has the potential for being very useful in investigating bone marrow-derived macrophage NF-kappaB signaling in diverse biological and biomedical studies.
Introduction
The nuclear factor-kappaB (NF-κB) pathway is a family of five highly conserved transcription factors (RelA/p65 (indicated as p65 hereafter), RelB, cRel, p50, and p52) that work together to promote a variety of functions, including cell survival and immune system response.1,2 There are two major NF-κB pathways: canonical and non-canonical. The canonical pathway is activated by ligands such as lipopolysaccharide (LPS) and TNF, and functions through activation of the IKK complex (IKKβ, IKKα, and IKKγ/NEMO). Active IKKβ phosphorylates the inhibitor of kappaB alpha (IκBα), which leads to its ubiquitination and subsequent proteasomal degradation.3,4 IκBα normally inhibits NF-κB family members p65/p50 by sequestration in the cytoplasm. Thus, NF-κB pathway activation and IκBα degradation leads to the release of the p65/p50 heterodimer, which then enters the nucleus and promotes transcription of a large number of target genes involved in cell survival and immune function. 5 The non-canonical NF-κB pathway, on the other hand, is activated by ligands such as RANKL and LTβ, which activates an IKKα homodimer to cause proteolytic cleavage of p100 to p52. The p52/RelB transcription factor heterodimer can then upregulate target genes involved with B cell homeostasis and differentiation. 1 NF-κB signaling is critical for many immune functions including the activation of macrophages and the production and regulation of cytokines.6–8 To better understand the role that the canonical NF-κB signaling pathway plays in myeloid cells, the canonical pathway was targeted for deletion in our model.
As full cellular p65 deletion is not compatible with life, due to massive liver apoptosis during embryogenesis,9,10 it is necessary to create conditional p65 knockout models. Other conditional p65 knockout models have demonstrated important, positive, roles for the NF-κB pathway in diverse areas such as in hematopoietic stem cell function when p65 is deleted in the hematopoietic compartment. 11 Mice with cardiomyocyte-specific deletion of p65 were protected from ischemia reperfusion injury through altered intracellular calcium homeostasis, suggesting a negative role for NF-κB. 12 Furthermore, a conditional knockout model of p65 in adipocytes suggests a dual role for NF-κB in regulating inflammation in adipose tissue, where lean conditions prompted pro-inflammation activity and obese conditions prompted anti-inflammation activity. 13 Another study describes a doxycycline inducible system, where NF-κB can be inhibited or activated in colony stimulating factor 1 receptor expressing cells. 14 It was found that during a short window of time following injection of circulating mammary tumor cells, activation of NF-κB in macrophages induced an M1 phenotype in the macrophages, which resulted in reduced lung tumor metastases. 14 This antitumor effect was no longer seen two days post tumor cell injection, indicating a dual role of macrophage NF-κB in this metastasis model.
The LysMCre animal model was initially created and characterized in 1999, 15 and has since been used extensively in various fields of biomedical research.16–18 As NF-κB has been shown to play important roles in macrophage function,19–21 we designed an animal model that lacks p65 in bone marrow-derived macrophages (BMDM). Here, we describe a conditional animal model made by crossing the LysMCre model to a p65fl/fl animal, 22 thus producing p65fl/fl/LysMCre mice which have very efficient knockdown of p65 protein in BMDMs. The efficient knockdown of p65 protein was shown to lead to altered canonical NF-κB signaling, reduced phagocytosis ability, and reduced nitrite production in BMDMs. Moreover, p65fl/fl/LysMCre mice exhibited similar circulating immune cell profiles and comparable immune response to Salmonella infection.
Here, we have specifically developed and characterized the novel p65fl/fl/LysMCre mouse model to study the role of BMDM NF-κB signaling in immune system function. As aberrant expression of NF-κB is associated with numerous diseases including autoimmune disease, neurodegenerative diseases, and many cancers, this model will provide an avenue to not only study basic immune function, but to also study many different diseases in the context of p65 loss in BMDMs.
Materials and methods
p65fl/fl/LysMCre mouse production
p65fl/fl mice were previously generated in the Baldwin laboratory. 22 Briefly, the p65 gene in these animals was manipulated to contain two LoxP sites, one near exon 4, and the other in the intron between exons 8 and 9. B6.129P2-Lyz2tm1(cre)Ifo/J animals, 15 referred to as LysMCre from here on, were purchased from The Jackson Laboratory (stock #004781). These mice contain Cre enzyme under the control of the LysM promoter, which drives Cre recombinase expression in cells of the myeloid lineage. p65fl/fl mice were bred to LysMCre mice for several generations to generate a fully transgenic p65fl/fl/LysMCre mouse. Genotypes were confirmed via PCR using the following primers: Lys Mutant Primer 5’ - CCCAGAAATGCCAGATTACG - 3’, Lys Common Primer 5’ - CTTGGGCTGCCAGAATTTCTC - 3’, Lys Wild Type Primer 5’ - TTACAGTCGGCCAGGCTGAC - 3’. Reactions included WT and common, or mutant and common primers. LysMCre PCR reaction as follows: 94°C for 3 min, 94°C for 30 s, 62°C 1 for minute, 72°C for 1 min, go to step 2 and repeat for 34 cycles, 72°C for 2 min. p65fl/fl PCR primers used are: 5’ CGACTTTGGGTTGGAGGGTTACAGAAGGC 3’ and 5’ TGGTCTGGATTCGCTGGCTAATGGC 3’. The p65fl/fl PCR reaction is: 95°C for 2 min, 95°C for 30 s, 65°C for 30 s, 72°C for 45 s, go to step 2 and repeat for 34 cycles, 72°C for 5 min.
This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. All protocols were approved by the IACUC of Augusta University (#2014-0691) and the University of North Carolina at Chapel Hill (#18-175.0). All animals were housed under barrier conditions.
Bone marrow-derived macrophage isolation
Animals between the ages of 1 and 5 months were euthanized according to IACUC protocol. Hind leg femurs and tibias were dissected and cleaned of muscle. The ends of the bones were removed, and the bone marrow was flushed out using sterile 1xPBS/2%FBS and a syringe with a 27 gage needle. The marrow was resuspended via pipetting, filtered with a 70 µm cell strainer, and cells were plated in macrophage media: DMEM (Fisher 11995-065) containing 10% FBS and 20% L929 conditioned media. 23 l929 conditioned media, containing M-CSF, was obtained by growing L929 cells (ATCC) to confluency for 1 week and collecting the culture media. 23 Cultured bone marrow monocytes were grown in the macrophage media for 1 week to allow differentiation prior to use. During the differentiation week, cells were washed with 1xPBS and media was changed twice, starting on day 3, post-harvest.
Tail vein blood collection
Mice were warmed by lamp for 5 min prior to restraining in the tailveiner restraint. Using a syringe and 30-gauge needle, one of the lateral tail veins was punctured. Between 30 µl and 70 µl of blood was collected using a heparinized micro-hematocrit capillary tube (22-362566 Fisherbrand) and was then transferred into K2 EDTA spray-coated vacutainer tubes (02-683-99C BD). Depending on the volume of blood, 300 to 500 µl of pre-warmed 1× RBC Lysis Buffer (420301 Biolegend) was pipetted into the blood samples and then all samples were incubated at room temperature for 5 min. 3.5 ml of PBS/2% FBS was added, and samples were centrifuged at 300 g for 5 min at 4°C. The supernatants were aspirated, and pellets were resuspended in 500 uL PBS/2% FBS before proceeding to antibody staining and flow cytometry analysis.
Flow cytometry
BMDM samples were fixed using the eBioscience™ Intracellular Fixation Buffer (88-8824-00 Invitrogen). Samples were analyzed, with the NovoCyte Quanteon (Georgia Cancer Center's Flow Cytometry Shared Resource Laboratory), for myeloid cell (CD11b+) and macrophage (CD11b+, F4/80+) populations. Zombie Violet (423113 Biolegend) was used as a viability dye. Antibody conjugations were as follows: CD11b-PE-Cyanine7 (25-0112-82 Invitrogen) and F4/80-FITC (123108 Biolegend).
Blood samples from six 15 week-old LysMCre control and six 15 week-old p65fl/fl/LysMCre mice were processed (described in the tail vein blood collection method) and analyzed with the NovoCyte Quanteon (Georgia Cancer Center's Flow Cytometry Shared Resource Laboratory), for various cell populations: Macrophages (CD11b+, F4/80+); G-MDSCs (CD11b+, Ly-6G+); M-MDSCs (CD11b+, Ly-6C+); T Cells (CD3+); T Helper cells (CD3+, CD4+); Cytotoxic T cells (CD3+, CD8α+). DAPI (564907 BD) was used as a viability dye. Antibody conjugations were as follows: CD11b-PE-Cyanine7 (25-0112-82 Invitrogen), F4/80-FITC (123108 Biolegend), Ly-6G-APC-Cyanine7 (127624 Biolegend), Ly-6C-PE (12-5932-82 Invitrogen), CD3-PE (12-0032-82 Invitrogen), CD4-APC (17-0042-82 Invitrogen), CD8α-FITC (11-0081-82 Invitrogen).
Western blotting
Tissues/cells were lysed in RIPA buffer containing protease (Roche #11697498001) and phosphatase (Sigma #P0044-1ML) inhibitors, and lysates were run on 4-12% precast protein gels (Invitrogen #NP0231Box). Standard immunoblotting methods were followed. Antibodies used for western blotting include: p-p65 (Cell Signaling Technologies #3033), total p65 (Cell Signaling Technologies #8242), GAPDH (Santa Cruz #25778), and β-actin (Cell Signaling Technologies #4970).
Immunofluorescent staining
BMDMs were plated in 6-well (1.0 × 106 cells/well) cell culture plates containing cover glasses and allowed to adhere overnight. Wells were washed 1× with PBS and media was refreshed. Adherent cells were stimulated with LPS (50 ng/ml) for 3 h and were then washed once with PBS and fixed with 4% formaldehyde for 10 min at room temperature. Fixative was removed from wells and cells were washed 3× with PBS prior to immunostaining. Cells were incubated with PBS containing 5% goat serum and 0.3% triton x-100 for 60 min at room temperature followed by monoclonal rabbit anti-mouse NF-κB p65 (1:400 dilution) (Cell Signaling Technologies #8242) overnight at 4°C. Cells were washed 3× with PBS and then incubated with goat anti-rabbit Alexa Fluor 555 (1:300 dilution) (Cell Signaling Technologies #4413) for 60 min at room temperature, washed again, and mounted in ProLong Gold medium (Thermofisher #P36930). Cells were examined using a Zeiss Axiovert 200 fluorescence microscope and captured and analyzed with a Zeiss AxioCam MRm camera and Axiovision software, respectively.
Peritoneal macrophage isolation
Animals were euthanized according to IACUC approved protocols and cold 1xPBS/5 mM EDTA was injected into the peritoneal cavity via syringe with a 27 gage needle. The peritoneal area was gently massaged and the PBS/EDTA was collected via syringe and 25 gage needle. 24 Cells from six LysMCre control mice were combined, and cells from six p65fl/fl/LysMCre mice were combined. The isolated cells were plated for 4 h, after which, floating cells were aspirated. The remaining, adherent cells were then further selected for macrophages by using CD11b+ microbead/magnetic column selection (Miltenyi # 130-093-636). Purified peritoneal macrophages were then processed for western blotting.
qPCR analysis
BMDMs grown in culture for 1 week (described above) were treated with 100 ng/ml LPS (Invitrogen #tlrl-eblps) or vehicle control for 4 h or 50 ng/ml LPS and 150 U/ml IFNγ for 24 h. Cells were harvested, and RNA was isolated using RNeasy Mini Plus Kit (Qiagen #74134). cDNA was then produced using the SuperScript II First-Strand Synthesis System (Invitrogen 11904018). TaqMan primer/probe systems (ABI/Life/Fisher) and 2× Master Mix (Roche 06402682001) were used for quantitative PCR. NF-κB target genes IL-6 (Mm00446190_m1), IL-1β (Mm00434228_m1), and TNFα (Mm00443258_m1) were normalized to GusB (Mm01197698_m1) expression. For polarization studies, iNOS (Mm00440502_m1), IL-12 (Mm01288989_m1), and IL-10 (Mm00439614) were normalized to GAPDH (Mm99999915_g1). Each sample was run in triplicate and statistical analysis was determined using GraphPad Prism software.
Cytokine array
Mouse Cytokine Array Panel A (R&D Systems ARY006) was used to detect cytokines secreted into the media by LysMCre and p65fl/fl/LysMCre BMDMs. 1 × 106 BMDMs were seeded into 6 well cell culture plates with 4 ml of media, which was collected for the array after 24 h. R&D systems directions were followed, and the film was exposed for 1.5 h. ImageJ software was used to analyze the spots, and GraphPad Prism software was used for graphing and statistical analysis (unpaired t test).
Salmonella infection and splenic cell population analysis
LysMCre and p65fl/fl/LysMCre mice were infected (IP) with 700 CFUs of Salmonella typhimurium bacteria (4–5 mice each group) or sterile PBS (2 mice each group). Spleens and mediastinal lymph nodes were collected after 48 h and analyzed for bacterial colony forming units. Data is representative of two experiments. GraphPad Prism software was used for graphing and statistical analysis.
Splenomegaly analysis
Non-infected 7 week old LysMCre and p65fl/fl/LysMCre mice were examined for splenomegaly. Following euthanasia, spleens were dissected and weighed. GraphPad Prism software was used for graphing and statistical analysis (unpaired t test). A minimum of 6 mice were used per group.
Phagocytosis assay
Per directions, (Vybrant Phagocytosis Assay Kit -Thermo/Fisher #V6694), 50,000 LysMCre control or p65fl/fl/LysMCre BMDMs were plated into a 96 well tissue culture treated plate (Costar 3610). The BMDMs were allowed to settle overnight, and were then treated with 150U of IFNγ (Fisher PMC4031) for 6 h, or vehicle control. The media was then removed, cells were washed, and 0.2 µg of LPS was added for 18 h. The fluorescent E. coli particles were added to the cells and incubated for 2 h to allow for phagocytosis to occur. Fluorescence activity was determined using a BioTek Synergy HT plate reader, analyzing at 480 nm excitation and 520 nm emission conditions. Each group, including positive and negative controls, was run in quadruplicate, and statistical analysis was determined using GraphPad Prism software (two-tailed student's t test).
Nitric oxide assay
Day 6 BMDM were plated in a 24 well tissue culture plate at a concentration of 3 × 105 cells per well, and were allowed to adhere overnight. Cells were washed with 1× PBS and 500ul of culture media was added to each well. BMDMs were treated with either vehicle control (sterile water), 50 ng/ml LPS for 24 h, 150 U/ml IFNγ for 24 h, 50 ng/ml LPS and 150 U/ml IFNγ for 24 h, or 150 U/ml IFNγ for 6 h followed by 50 ng/ml LPS for 18 h. Supernatants were collected and analyzed for nitric oxide response, per directions, using the QuantiChrom Nitric Oxide Assay Kit (BioAssay Systems D2NO-100). Colorimetric activity was determined using a BioTek Synergy HT plate reader with analysis at 540 nm. Each group, including positive and negative controls, was run in quadruplicate, and statistical analysis was determined using GraphPad Prism software.
Results
p65fl/fl/LysMCre bone marrow cells can differentiate into macrophages
p65fl/fl animals, previously generated in the Baldwin lab, 22 were crossed with LysMCre mice 15 purchased from The Jackson Laboratory. LysMCre mice contain Cre enzyme, which has been placed under the control of the LysM promoter, thus driving Cre recombinase expression in cells of the myeloid lineage. 15 p65fl/fl mice were bred to LysMCre mice for several generations to generate fully homozygous p65fl/fl/LysMCre mice (Figure 1a), which were confirmed by PCR based genotyping. It was first determined via flow cytometry that bone marrow cultured monocytes from age matched control and p65fl/fl/LysMCre mice would yield a high purity, and that these monocytes could differentiate into macrophages (CD11b+/F4/80+) (Figure 1b). In fact, bone marrow from LysMCre and p65fl/fl/LysMCre mice produced equivalent percentages of bone marrow-derived macrophage (BMDM) populations following 1 week in culture. Cells were allowed to differentiate for one week and were stained with the CD11b monocyte marker (Figure 1b top panel), and CD11b and F4/80 antibodies to detect macrophages (Figure 1b bottom panel). There were no significant differences in percentages of CD11b+ myeloid cells (∼99%) or CD11b+/F4/80+ macrophage populations (∼93%) between control and p65 deficient BMDMs (Figure 1b).

p65fl/fl/LysMCre BMDMs lack p65 protein
We next confirmed via western blotting whether BMDM from mice lacked p65 protein. Protein analysis revealed that 7 day old p65fl/fl/LysMCre BMDMs lack p-p65 and have dramatically reduced total p65, as compared to LysMCre and p65fl/fl control mice (Figure 2a, left panel and Figure 2b). This has been consistently seen in BMDMs from male and female mice, from younger mice to older mice, and over multiple generations. However, it was found that peritoneal macrophages still retained residual amounts of p65 protein, indicating a leaky Cre system (Figure 2a, right panel). This is not surprising, as the original LysMCre model was described as having less than 100% knockdown in mature macrophages. 15
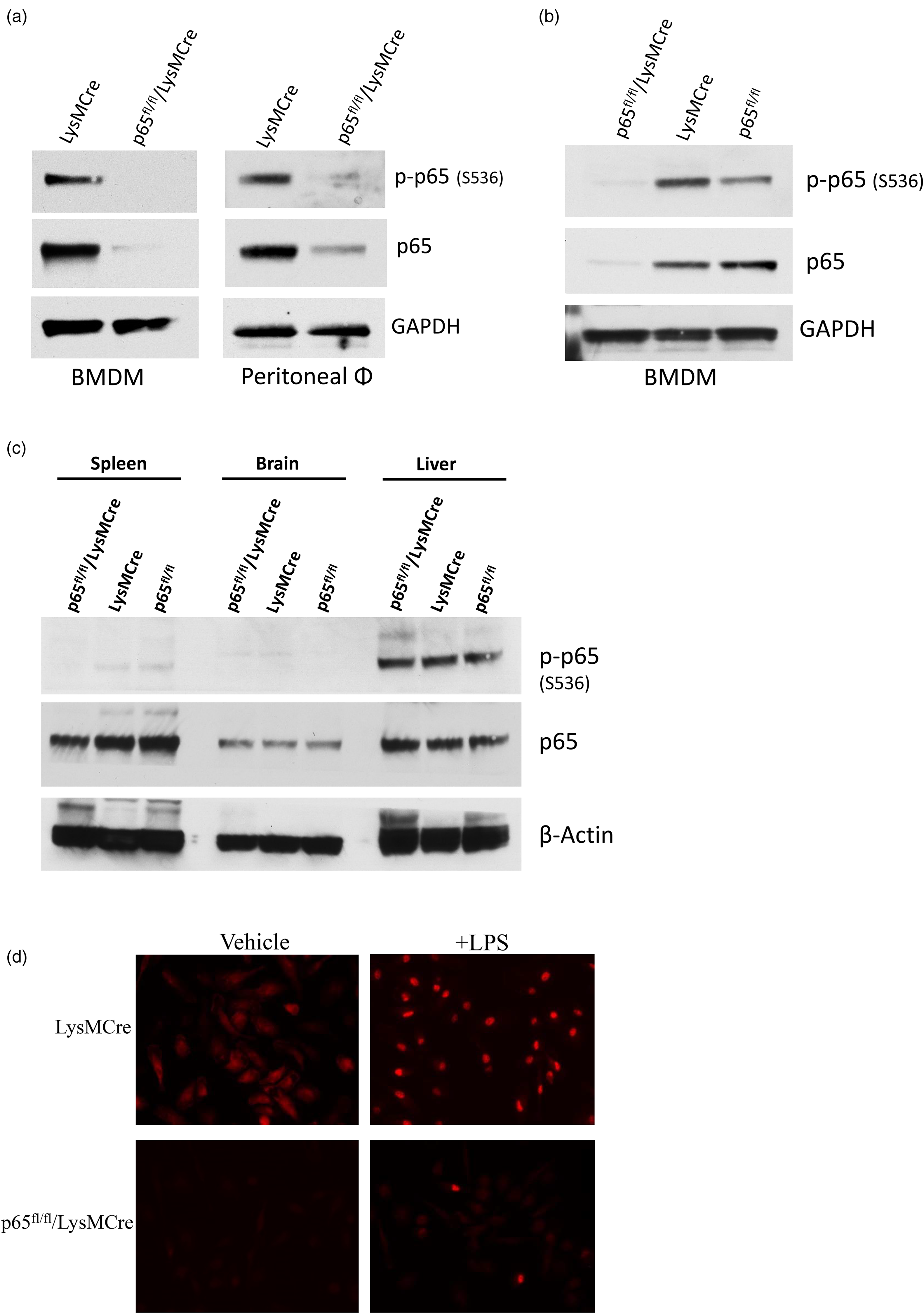
To determine any differences in phosphorylated or total p65 protein levels in various tissues, spleen, brain, and liver was analyzed via western blotting (Figure 2c). p65/p-p65 protein levels did not vary much between the brain and liver from control and the p65fl/fl/LysMCre mice, but there was a small reduction in p65 protein between the p65fl/fl/LysMCre spleen and control spleens (16.5% reduction when normalized to β-actin). No p-p65 protein was detected in the p65fl/fl/LysMCre spleen, while a very small amount was present in the control spleens (Figure 2c).
Immunohistochemistry was also performed to verify that p65fl/fl/LysMCre BMDMs lack p65 protein following lipopolysaccharide (LPS) stimulation (Figure 2d), as compared to LysMCre control BMDMs. LysMCre and p65fl/fl/LysMCre BMDMs were treated with LPS or vehicle control for 4 h to stimulate canonical NF-κB pathway activity, and cells were stained with p65. The staining clearly shows the lack of p65 staining in unstimulated p65fl/fl/LysMCre BMDMs and drastically reduced nuclear localization of p65 upon LPS treatment (Figure 2d).
p65fl/fl/LysMCre mice have altered canonical NF-κB signaling
Canonical NF-κB signaling pathway activity was next analyzed. LysMCre and p65fl/fl/LysMCre BMDMs were treated with LPS or vehicle control for 4 h or 6 h to stimulate canonical NF-κB pathway activity. IL-6, IL-1β, and TNFα, all downstream target genes of canonical NF-κB, were analyzed via qPCR to determine expression. LysMCre control BMDMs were able to efficiently upregulate the NF-κB target genes after 4 h of LPS exposure, while the p65fl/fl/LysMCre BMDMs had significantly reduced expression of IL-6, IL-1β, and TNFα, indicating defective canonical NF-κB signaling (Figure 3).

p65fl/fl/LysMCre BMDMs secrete altered levels of select cytokines
To examine the effects of altered canonical signaling in the p65fl/fl/LysMCre BMDMs on cytokine secretion, a cytokine profiler array was utilized. 24-h conditioned media from LysMCre or p65fl/fl/LysMCre BMDMs was subjected to the Mouse Cytokine Array Panel A (R&D Systems) blot to determine whether there was a difference in cytokine secretion (Figure 4). While there was an unmeasurable amount of some cytokines analyzed in the panel (Supplemental Figure 1), LysMCre and p65fl/fl/LysMCre BMDMs did release varying levels of multiple cytokines into their culture media. Significantly less CXCL10, TNFα, GM-CSF, and IL-1α, and significantly more TIMP-1 and M-CSF was secreted over 24 h by the p65fl/fl/LysMCre BMDMs, as compared to control BMDMs (Figure 4). Some cytokines were detected but did not have significantly different levels between the two groups, including CXCL1, IL-16, IFNγ, and CCL2. This indicates that altered NF-κB signaling in the p65fl/fl/LysMCre BMDMs is in fact leading to altered secretion of some soluble factors.

p65fl/fl/LysMCre BMDMs have altered innate immune function
A phagocytosis assay was used to determine whether the lack of NF-κB signaling reduced phagocytosis efficiency in p65fl/fl/LysMCre BMDMs. LysMCre control and p65fl/fl/LysMCre BMDMs were plated into a 96 well plate, and were treated with vehicle control or with a pre-treatment of IFNγ for 6 h, followed by LPS treatment for 18 h to induce phagocytosis activity. The Vybrant assay used for this experiment provides fluorescein-labeled E. coli (K-12 strain) bio-particles, which should be efficiently engulfed by BMDMs. Macrophages that engulf these particles will fluoresce, which can be quantified using a plate reader. The p65fl/fl/LysMCre BMDMs engulfed significantly fewer E. coli particles than the LysMCre control BMDMs in both the vehicle and IFNγ/LPS treated groups (Figure 5a). Interestingly, treatment of IFNγ/LPS had only negligible effects on phagocytic activity in either LysMCre control or p65fl/fl/LysMCre BMDMs compared to vehicle treated BMDMs (Figure 5a).
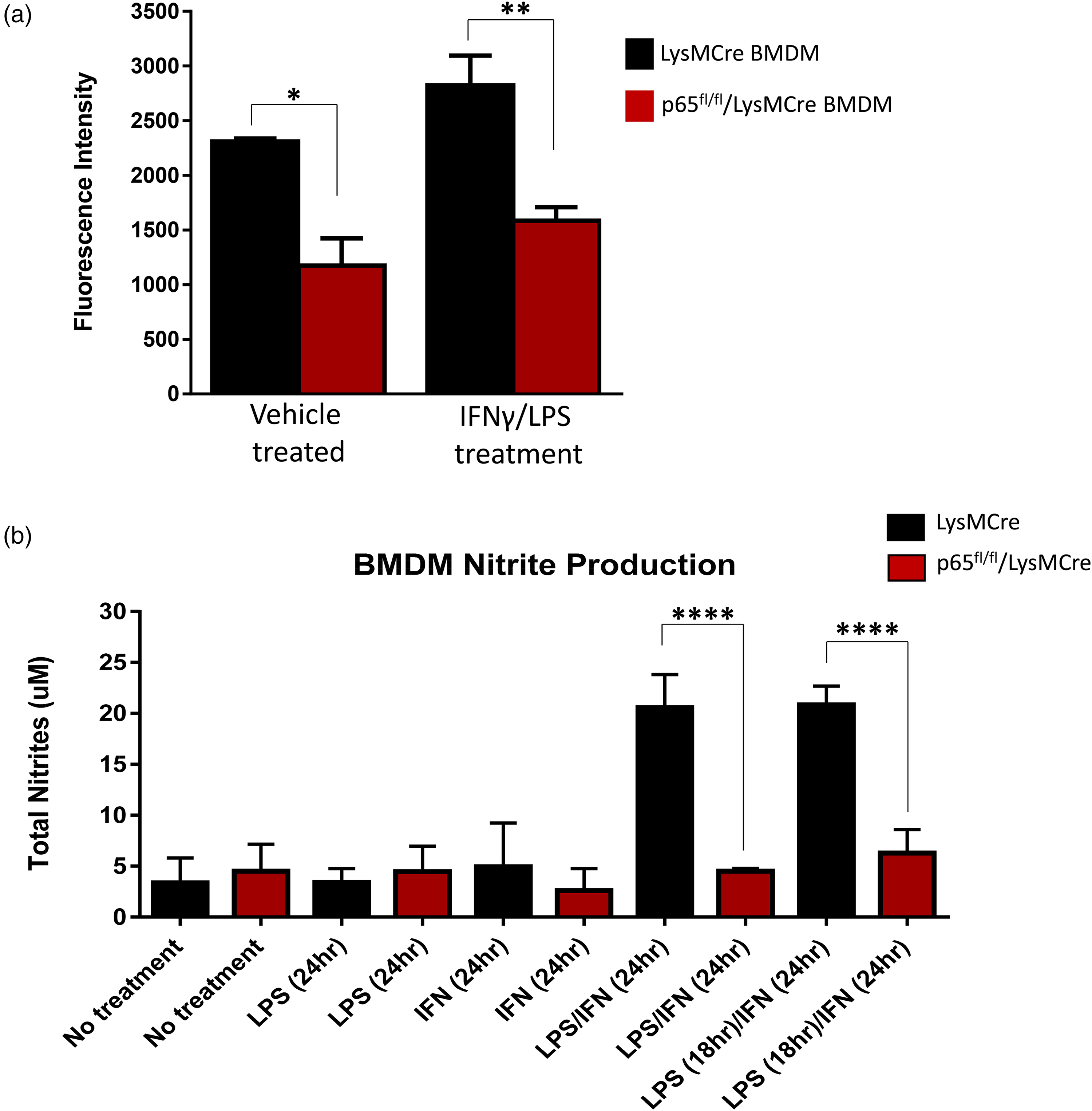
LysMCre control and p65fl/fl/LysMCre BMDMs were subjected to a nitrite production assay following 24 h of treatment with LPS, IFNγ, or LPS and IFNγ combined, to determine the ability of these cells to secrete nitric oxide, a common characteristic of stimulated macrophages that helps them kill pathogens. 25 The activity of this assay was based on the total amount of nitrite secreted into the media, as nitric oxide is a very short-lived molecule and nitrite is a much more stable metabolite of nitric oxide. The nitric oxide assay showed very little difference in nitrite levels among cells that had been stimulated by either LPS or IFNγ alone. However, cells that had been stimulated by LPS and IFNγ showed significant differences in the total amount of nitrites (Figure 5B). BMDMs lacking the canonical NF-κB signaling pathway produced much lower concentrations of nitrates than the control macrophages, further indicating innate immune activity dysfunction.
p65 deficiency inhibits p65fl/fl/LysMCre BMDMs from appropriately polarizing
As p65fl/fl/LysMCre BMDMs showed reduced innate immune activity, we next determined whether these cells could polarize to an M1, classically activated, phenotype. BMDMs from LysMCre control and p65fl/fl/LysMCre mice were treated with vehicle or a combination of 50 ng/ml LPS and 150 U/ml IFNγ for 24 h. qPCR analysis revealed that p65fl/fl/LysMCre BMDMs have a statistically significant reduction in iNOS and IL-12 mRNA expression, indicating a reduced ability to polarize to a pro-inflammatory (M1) phenotype (Figure 6a and 6b). p65fl/fl/LysMCre BMDMs also had a significant increase in IL-10 expression, indicating that the lack of p65 potentially pushes these cells into an alternatively polarized (M2-like) phenotype (Figure 6c).

Lysmcre and p65fl/fl/LysMCre mice have similar systemic immune profiles
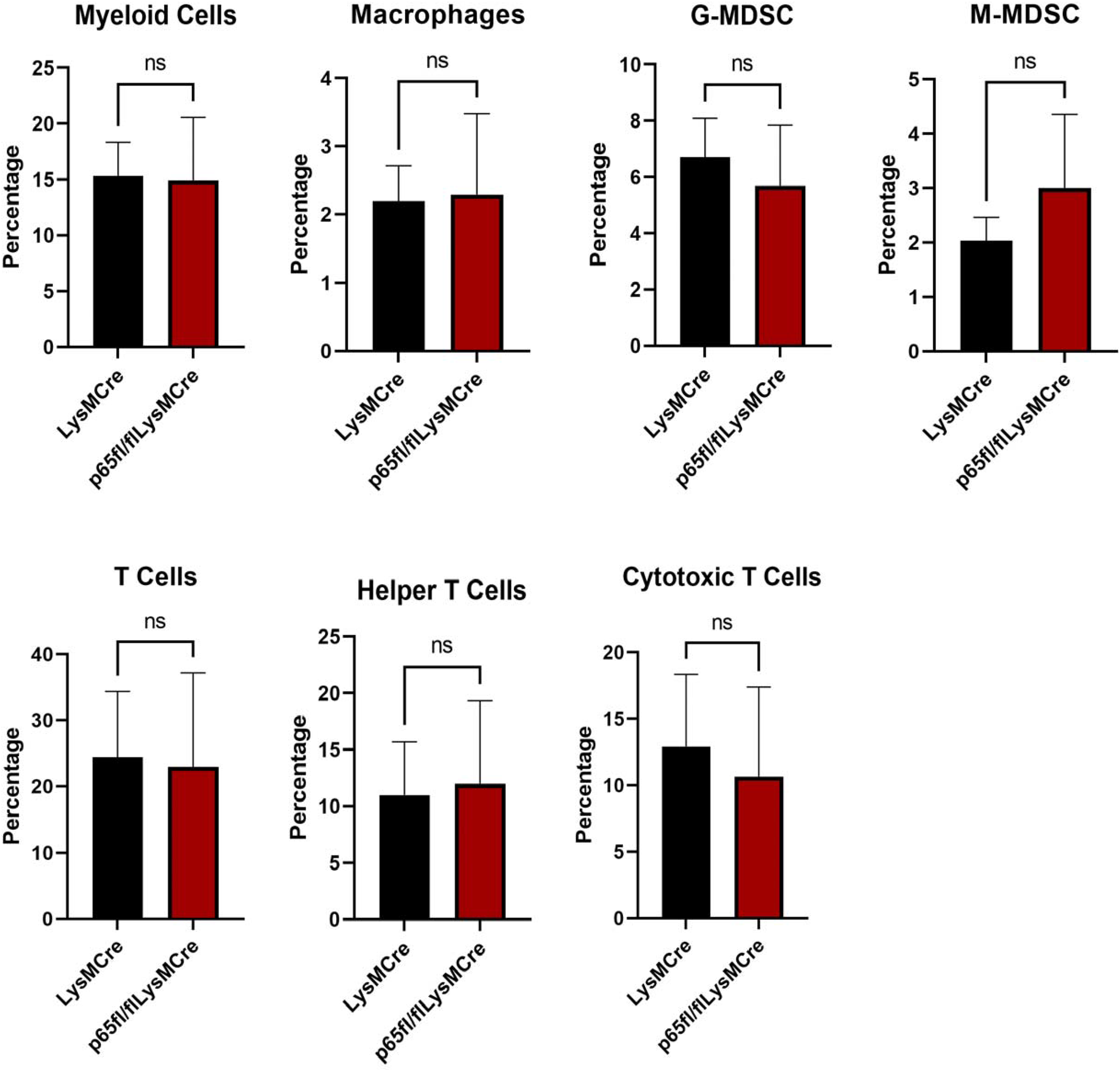
We next decided to characterize the systemic immune cell populations of the p65fl/fl/LysMCre mice. In order to assess baseline differences between p65fl/fl/LysMCre mice and LysMCre control mice, blood serum from 6 age-matched LysMCre mice and 6 age-matched p65fl/fl/LysMCre mice was evaluated through flow cytometry. Collected blood samples were treated with RBC lysis buffer and stained with common immune cell marker antibodies. No significant differences were seen between the controls and p65 deficient mice in percentages of myeloid cells (CD11b+), macrophages (CD11b + F4/80+), granulocytic-MDSCs (CD11b + Ly-6G+), monocytic-MDSCs (CD11b + Ly-6C+), T cells (CD3+), T helper cells (CD3 + CD4+), and cytotoxic T cell (CD3 + CD8+) populations. However, there are noticeable trends, with fewer G-MDSCs and cytotoxic T cells, but more M-MDSCs in p65fl/fl/LysMCre mice (Figure 7). Flow cytometry histograms are in the supplemental data section (Supplemental Figure 2).
p65fl/fl/LysMCre mice do not show increased Salmonella burden following infection
Even though systemic immune cell populations did not significantly vary between control and p65fl/fl/LysMCre mice (Figure 7), it was determined that the p65fl/fl/LysMCre mice consistently displayed splenomegaly (Figure 8a). Female p65fl/fl/LysMCre mice seem to be even more susceptible to enlarged spleens than male p65fl/fl/LysMCre mice of the same age (Figure 8a). Due to this observation, and because there was a clear decrease in the ability of p65fl/fl/LysMCre BMDMs to engulf E. coli particles and produce nitrite products, it was next determined whether p65fl/fl/LysMCre mice had increased vulnerability to bacterial infection as compared to controls. Salmonella typhimurium was chosen for this study as this pathogen targets macrophages for intracellular infection. p65fl/fl/LysMCre and LysMCre control mice were infected (IP) with 700 colony forming units (CFUs) of Salmonella bacteria or vehicle control, and mediastinal lymph nodes and spleens were harvested 48 h post infection to determine CFU burden. Results indicate that the p65fl/fl/LysMCre mice did not have a significant difference in Salmonella burden, as compared to LysMCre control mice in either the mediastinal lymph nodes or spleens (Figure 8b). A higher dose (4 × 104 CFUs) of Salmonella was also injected (IP), with no difference in CFU burden between LysMCre control and p65fl/fl/LysMCre mice after 48 h (data not shown). Latency to death following Salmonella infection was also assessed, and no difference was found between the two groups (data not shown).

p65fl/fl/LysMCre mice exhibit splenomegaly but do not show increased Salmonella burden following infection when compared to control mice. Spleens were weighed from 7 week old animals (A). Animals were injected with 700 CFUs of Salmonella typhimurium bacteria or vehicle control. CFUs of Salmonella in spleen and mediastinal lymph nodes were counted in control LysMCre and p65fl/fl/LysMCre animals 48 h post infection (B). Data is representative of two experiments. * indicates a p value of < 0.04, **** indicates a p value of < 0.0001.
Discussion
Here, we describe the creation and characterization of an animal model that efficiently lacks canonical p65 in bone marrow-derived macrophages (BMDMs). We examined the effects of knocking down p65 in both BMDM expression and functionality, and on the ability of the p65fl/fl/LysMCre mouse model to respond to Salmonella infection. The p65fl/fl/LysMCre model was created by crossing the p65fl/fl mouse model 22 to the LysMCre mouse model. 15 In theory, the LysMCre animal should provide deletion of floxed genes in all cells of the myeloid lineage, but in reality, full deletion is not usually seen in all myeloid cell types. Full myeloid deletion was not seen in the p65fl/fl/LysMCre model, as p65 is not fully knocked down in peritoneal macrophages, however, there is excellent p65 knockdown in BMDMs (Figure 2). Lack of full myeloid deletion is certainly a limitation of this model for in vivo assessment of total myeloid NF-κB activity, however, this model still provides a useful method to determine the role of BMDM NF-κB signaling in various in vitro and in vivo roles. This model is also simple to use compared to many conditional animal models because no additional inducing agent, such as tamoxifen administration, is needed to induce p65 deletion. While robust p65 knockdown was seen in male and female mice of all ages, mice around 3 months of age were frequently used for BMDM studies, as these primary cells grew best from animals of this age. As mice age, bones tend to become more brittle and it is harder to collect the bone marrow. Otherwise, no mice were excluded from these studies.
As part of the initial characterization of the p65fl/fl/LysMCre mouse model, we examined p-p65/p65 protein levels in three different tissues; spleen, brain, and liver. As the spleen is the largest lymphatic organ and contains a large number of monocytes, 26 we were interested in seeing whether p-p65/p65 levels were altered in splenic tissue of the p65fl/fl/LysMCre mice. In fact, we saw a slight decrease in both total and p-p65 in the spleens of p65fl/fl/LysMCre mice (Figure 2c). The resident macrophages of the central nervous system are microglia, which are derived from embryonic yolk sac progenitors 27 and not hematopoietic progenitors. 28 Thus, we did not expect much p-p65/p65 change in the brain tissue of the p65fl/fl/LysMCre mice as compared to controls, which was demonstrated in the western blot (Figure 2c). The liver has a large population of resident macrophages called Kupffer cells, even during times of health. 29 As most Kupffer cells are also yolk sac-derived30,31 and not bone-marrow derived, we did not anticipate p-p65/p65 differences, which was supported by the western data (Figure 2c).
Phospho-p65 and total p65 knockdown in p65fl/fl/LysMCre BMDMs corresponded to a reduction in common canonical NF-κB target gene expression levels (Figure 3). Changes in production and secretion of some cytokines was also seen in naïve (untreated) p65fl/fl/LysMCre BMDMs, with less CXCL10, TNFα, GM-CSF, IL1α, and more TIMP-1, and M-CSF being detected, as compared to LysMCre BMDMs (Figure 4). LPS treatment would probably increase the differences seen in secreted cytokines between the p65fl/fl/LysMCre and control BMDMs. Phospho-p65 and total p65 protein levels were also examined in several organs in control and p65fl/fl/LysMCre model mice, including the spleen, brain, and liver (Figure 2c). Phospho-p65 and total p65 protein levels were slightly decreased in the spleens of p65fl/fl/LysMCre mice, and the p65fl/fl/LysMCre mice, especially females, exhibited splenomegaly (Figure 8a). Liver and brain p65 protein levels were very similar between p65fl/fl/LysMCre model and control mice, indicating these organs are largely unaffected by LysMCre action.
Functionally, p65fl/fl/LysMCre BMDMs were unable to perform innate immune functions as well as control BMDMs, as was seen in the reduced ability to engulf bacterial products and produce nitrites (Figure 5). It is well known that canonical NF-κB plays a role in macrophage polarization,20,32–34 so expression of markers associated with classically activated M1 macrophages (iNOS, IL-12) and alternatively activated M2 macrophages (IL-10) was analyzed (Figure 6). The p65fl/fl/LysMCre BMDMs lacking p65 were skewed towards an alternative phenotype, which is logical based on their reduced ability to efficiently perform innate immune functions.
Because p65fl/fl/LysMCre BMDMs are defective in engulfment ability and production of nitrites (Figure 5), and due to the fact that p65fl/fl/LysMCre animals exhibit splenomegaly (Figure 8a), we determined whether p65fl/fl/LysMCre mice would be able to clear bacterial infection as efficiently as LysMCre control mice. Salmonella typhimurium was chosen for this study as this pathogen targets macrophages for intracellular infection, and it is one of the most commonly used model pathogens to study the macrophage/pathogen interaction. Our results demonstrate that while p65fl/fl/LysMCre BMDMs are defective in innate immune functions, the p65fl/fl/LysMCre mice have a normal enough overall immune system to combat Salmonella infection (Figure 8b). This result was somewhat surprising, as we figured the dysfunctional BMDMs in the p65fl/fl/LysMCre animals would have negatively impacted their ability to fight bacterial infection. However, naïve p65fl/fl/LysMCre mice do not show significant differences in blood leukocyte populations, as compared to controls (Figure 7), suggesting that the target myeloid cells that Salmonella would infect are present. It may be that in wild type mice, the many Salmonella type III secretion effectors that block NF-κB signaling prevent a strong NF-κB response in infected cells. 35 Thus, p65 depletion has no effect because the response is normally inhibited. It could be that Salmonella strains carrying deletions in specific effectors would have their attenuation restored in p65fl/fl/LysMCre animals. It is still surprising that uninfected cells that are not intoxicated by these effectors do not require p65 to mount a normal response.
The p65fl/fl/LysMCre model has very efficient knockdown of canonical NF-κB p65 in BMDMs, which could render this model very useful in determining the role of canonical myeloid NF-κB signaling in a wide variety of applications. To that end, our lab has successfully used this model in an immunology study 36 as well as in a glioblastoma study, 37 which implicates the importance of canonical NF-κB signaling in peripheral macrophages in the growth and development of glioblastoma tumors. These studies underscore the idea that the p65fl/fl/LysMCre model can be valuable not only in cell culture studies but also in a wide range of in vivo applications that implicate canonical NF-κB signaling in BMDMs.
Supplemental Material
sj-pdf-1-ini-10.1177_17534259231205993 - Supplemental material for Production of a p65fl/fl/LysMCre mouse model with dysfunctional NF-κB signaling in bone marrow-derived macrophages
Supplemental material, sj-pdf-1-ini-10.1177_17534259231205993 for Production of a p65fl/fl/LysMCre mouse model with dysfunctional NF-κB signaling in bone marrow-derived macrophages by Ahmet K. Korkaya, Jeffrey Fischer, Anthony Peppers, Sean M. Crosson, Manira Rayamajhi, Edward A. Miao, Albert S. Baldwin and Jennifer W. Bradford in Innate Immunity
Supplemental Material
sj-pdf-2-ini-10.1177_17534259231205993 - Supplemental material for Production of a p65fl/fl/LysMCre mouse model with dysfunctional NF-κB signaling in bone marrow-derived macrophages
Supplemental material, sj-pdf-2-ini-10.1177_17534259231205993 for Production of a p65fl/fl/LysMCre mouse model with dysfunctional NF-κB signaling in bone marrow-derived macrophages by Ahmet K. Korkaya, Jeffrey Fischer, Anthony Peppers, Sean M. Crosson, Manira Rayamajhi, Edward A. Miao, Albert S. Baldwin and Jennifer W. Bradford in Innate Immunity
Supplemental Material
sj-docx-3-ini-10.1177_17534259231205993 - Supplemental material for Production of a p65fl/fl/LysMCre mouse model with dysfunctional NF-κB signaling in bone marrow-derived macrophages
Supplemental material, sj-docx-3-ini-10.1177_17534259231205993 for Production of a p65fl/fl/LysMCre mouse model with dysfunctional NF-κB signaling in bone marrow-derived macrophages by Ahmet K. Korkaya, Jeffrey Fischer, Anthony Peppers, Sean M. Crosson, Manira Rayamajhi, Edward A. Miao, Albert S. Baldwin and Jennifer W. Bradford in Innate Immunity
Footnotes
Acknowledgements
The authors would like to thank Mr Aaron Ebbs and Mr Jose Roques from the University of North Carolina at Chapel Hill for technical assistance. We thank Dr Rebekah Tritz, Director of the Augusta University Flow Cytometry Core Facility for assistance with the flow cytometry experiments. We also thank Dr Fulya Alkan, Mr Hilmi K. Alkan, and Dr Hasan Korkaya at the Georgia Cancer Center for their assistance on this project. We thank Dr Richard Griner from the Augusta University Department of Biological Science for support. We thank Mr Scott Bradford for graphic design assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics statement
The authors attest that the work presented in this manuscript is original and has not been previously published. Strict adherence to the scientific research ethics code has been maintained. This work was carried out in accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. All animal protocols were approved by the IACUC of Augusta University and the University of North Carolina at Chapel Hill.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for JWB was provided by the NIH National Research Service Award (1-F32-CA162628-01), Augusta University startup funds, the Augusta University Internal Grants Program, and by the Augusta University Center for Undergraduate Research and Scholarship program. Funding for EAM provided by the National Institutes of Health (AI139304).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
