Abstract
Consumption of diets high in fat has been linked to the development of obesity and related metabolic complications. Such associations originate from the enhanced, chronic, low-grade inflammation mediated by macrophages in response to translocated bacteria, bacterial products, or dietary constituents such as fatty acids (FAs). Nucleotide-binding Oligomerization Domain 2 (NOD2) senses muramyl dipeptide (MDP), a component of bacterial peptidoglycan. The inability to sense peptidoglycan through NOD2 has been demonstrated to lead to dysbiosis, increased bacterial translocation, inflammation and metabolic dysfunction. Currently, it is unknown how consumption of HFDs with different FA compositions might influence NOD2-dependent responses. In this study, we subjected WT mice to a control diet or to HFDs comprised of various ratios of unsaturated to saturated fats and determined the macrophage response to TLR4 and NOD2 agonists. A HFD with equal ratios of saturated and unsaturated fats enhanced subsequent responsiveness of macrophages to LPS but not to MDP. However, a high-unsaturated fat diet (HUFD) or a high-saturated fat diet (HSFD) both decreased the responsiveness to NOD2 agonists compared to that observed in control diet (CD) fed mice. These data suggest that dietary fatty acid composition can influence the subsequent macrophage responsiveness to bacterial products.
Keywords
Introduction
Obesity affects more than a third of the US population. 1 An obese state raises the risk for the development of numerous chronic inflammatory conditions including type 2 diabetes, coronary heart disease, stroke, respiratory problems, and even cancer. In the US alone, obesity accounts for approximately ∼$150 billion dollars annually in health care costs. 1 As such, understanding the molecular and cellular consequences of an obese condition prior to development of secondary morbidities may help in testing or designing interventions which can potentially halt or slow the progression towards these more dangerous disorders.
Obesity arises from an imbalance in caloric intake, metabolism and energy expenditure. 2 Excessive caloric intake alone, particularly of high-fat “Western” diets, has been consistently associated with tipping this balance in favor of the development of obesity. 3 Experimentally, diets in which 30% or more of the energy is derived from fat is sufficient to predictably induce obesity in susceptible mouse strains. 4 Importantly, a number of the downstream physiologic consequences of obesity observed in humans such as hyperinsulinemia, hyperglycemia, and hypertension, are also reproduced in a diet-induced obesity model. 5 Over the years, closer attention has been paid not only to the amount of total fat present in the diet but also to the specific types and proportions of fat comprising the diet, and even on the dietary fat source (animal versus plant). Current recommendations and review panels suggest that lowering total fat, particularly saturated fat, as part of a reduced energy intake diet, is beneficial in this regard (generally leads to weight loss). 6
The association between obesity and the development of cardiovascular, pulmonary and metabolic disorders is believed to result from low-grade chronic inflammation brought about by increased accumulation of macrophages in various tissues including the white adipose tissue (WAT), liver, and muscle.7,8 High-fat diet (HFD) regimens have been shown to promote microbial dysbiosis leading to increased intestinal barrier permeability and translocation of bacteria or bacterial products into the blood and tissues and, in turn, to the subsequent infiltration of macrophages.9,10 It has been generally observed that in the lean state, ‘alternatively activated’ M2 macrophages predominate within tissues while obesity appears to either trigger reprogramming of resident macrophages into a more ‘classically activated’ pro-inflammatory M1 state or promote infiltration of monocytes which differentiate into M1 macrophages within the tissues.7,11,12 The most notable example for this has been the observation of an increase in infiltrating adipose tissue macrophages (ATMs) with an inflammatory profile during the development of obesity prior to the emergence of insulin resistance (IR) and the eventual development of type 2 diabetes. 7 Given these findings, there is widespread interest in studying how various inflammatory and FA sensing pathways within macrophages contribute to the development of comorbidities associated with obesity.
The most well-studied pathway and receptor found to mediate HFD-induced inflammation is NF-κB activation induced through Toll-like receptor 4 (TLR4) engagement.13,14 Initial studies seemed to indicate that dietary FAs themselves (particularly long chain SFAs) could directly signal through this receptor.15,16 More recent research instead suggests that priming macrophages through this receptor (by exposure to translocated bacterial products containing the canonical TLR4 ligand LPS) metabolically reprograms macrophages and enhances their pro-inflammatory responses to FA. 17 Regardless, HFD studies in TLR4 KO mice show that although TLR4 KO mice develop increased diet-induced obesity, activation of inflammatory signaling pathways, cytokine production, and HFD-induced IR are attenuated in the absence of this receptor. 14
Outside of TLR4, the cytosolic peptidoglycan receptors NOD1 and NOD2 have both been implicated in linking obesity-related inflammation with metabolic disease.18–20 In particular, loss of NOD2 appears to promote intestinal barrier permeability and bacterial translocation leading to increased adipose tissue and liver inflammation. 19 The dysbiosis occurring in NOD2-deficient mice also appears to impart the inflammatory cues important for the development of IR. One of the limitations with the use of genetic models is that dysbiosis is already present in NOD2 KOs prior to dietary changes and so the consequences of HFD-related inflammation on this pathway are confounded. In this study, we sought to determine how administration of HFDs with different dietary fat compositions in WT mice affects the subsequent response of macrophages to TLR4 and NOD2 ligands.
Materials and methods
Mice and diets
Eight-week old male C57BL/6J mice (000664, Jackson Laboratories) were placed on one of three high-fat diets (HFD) or a control diet (CD) for 8 weeks. Animals were used in accordance with a protocol approved by the Univ. of Central Florida IACUC. The three high-fat diets consisted of differing ratios of unsaturated (UFA) to saturated (SFA) fatty acids. The 60% HFD has a 1 UFA : 1 SFA ratio (D12492, Research Diets), the 60% high unsaturated fat diet (HUFD) has a 3.3 UFA : 1 SFA ratio (D06062303, Research Diets), the 60% high saturated fat diet (HSFD) has a 1 UFA : 10 SFA ratio (D06062302, Research Diets), and lastly the control diet (CD) contains 10% fat with a 1 UFA : 1 SFA ratio (D07020902, Research Diets). Mice were housed 3 per cage and given ad libitum access to their respective diet and water. Animal weights, food intake, and water intake measurements were collected weekly. The exact composition and the use of these diets have been described previously. 21
Immunohistochemistry
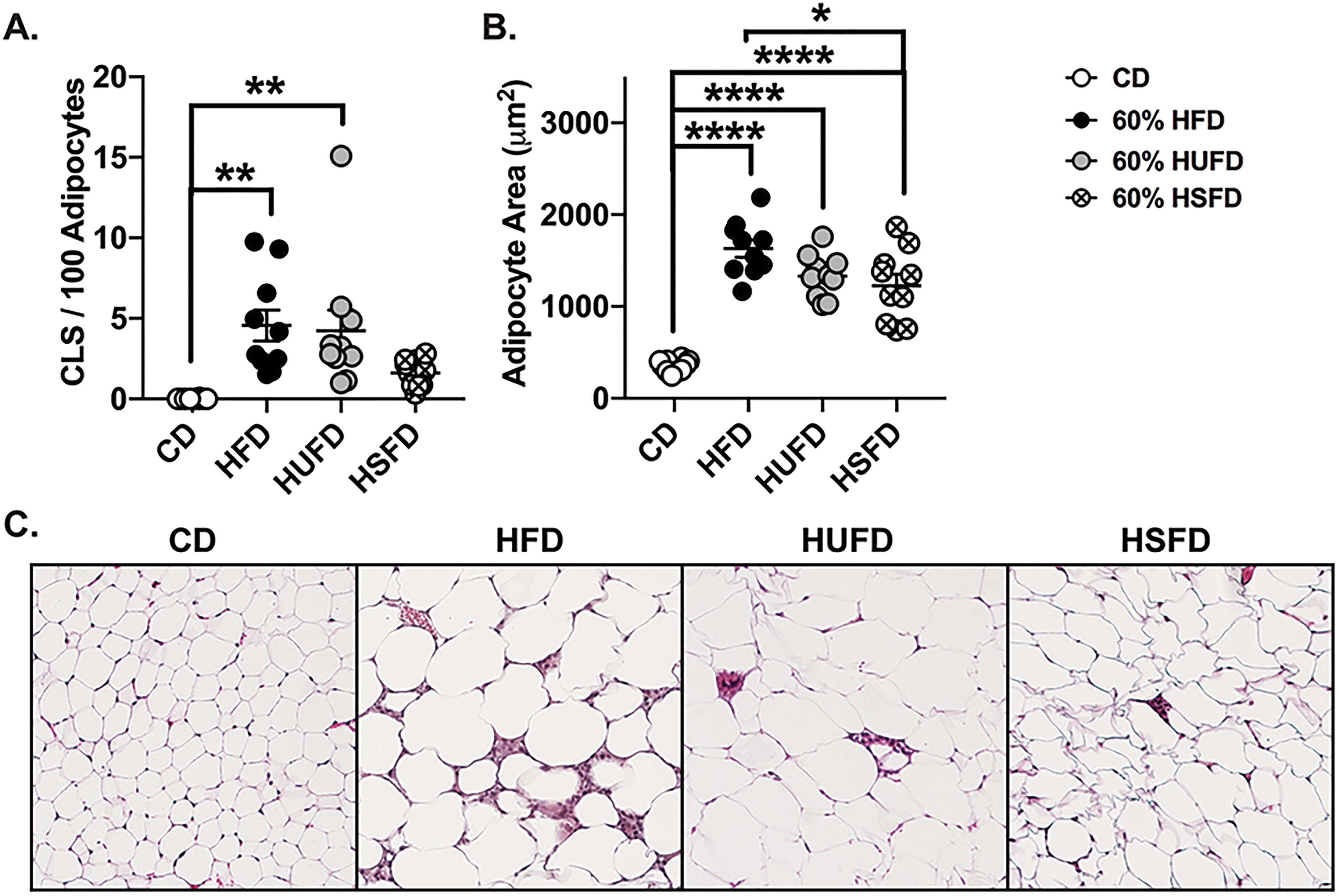
Fat was excised from the peri-gonadal region and fixed in 10% buffered formalin. Tissues were processed, paraffin-embedded, and stained with Hematoxylin and Eosin using a commercial service (AML Labs, Jacksonville, FL). Five low-power fields per section were examined by a blinded observer and the number of crown-like structures (CLS) 22 per 100 adipocytes was quantified. Average adipocyte area (μm2) was also quantified by taking length and width measurements for 100 cells from five low-power fields per section.
Multiplex analysis of diabetes-associated biomarkers
After the HFD regimen, animals were euthanized for collection of tissues. Mice were subjected to cardiac puncture for collection of blood, which was then allowed to clot in serum separator tubes for 30 min at room temperature prior to centrifugation for 10 min and collection of serum. Serum was stored at −80°C until further analysis. Measurement of diabetes-associated biomarkers was performed using a Mouse Diabetes Bio-Plex assay and Bio-Plex 200 following the manufacturer's instructions (Bio-Rad).
Bone marrow-derived macrophages (BMDMs)
BMDMs were generated as previously described. 23 Briefly, bone marrow from femurs and tibias of mice undergoing control or HFD regimens were grown for 7 days in DMEM containing Ladmac-conditioned supernatant. Differentiation medium was replaced with DMEM containing 10% FBS and 1% antibiotic-antimycotic on day 7. On day 8, full serum media was replaced with low serum media (0.1% FBS) 4 h before stimulation with various agonists. Appropriate differentiation of BMDMs was verified by expression of F4/80 and CD11b by flow cytometry. Muramyl dipeptide (MDP) was purchased from Bachem and ultrapure lipopolysaccharide (LPS) was procured from Invivogen.
ELISA
BMDMs were stimulated for 16 h with the indicated agonists, after which supernatants were collected for measurement of murine CCL3, IL-6 and TNF-α by ELISA using the manufacturer's instructions (Thermofisher). Briefly, Nunc Maxisorp plates were coated overnight with a capture antibodies using the provided coating buffer. Plates were washed and then blocked for one hour using the provided assay buffer prior to washing and overnight incubation with the cell supernatants or standards. The following day, plates were washed and incubated with detection antibodies for one hour followed by washing and incubation with Avidin-HRP for 30 min. Plates were developed using the TMB substrate (Thermofisher). A Cytation5 Plate Reader (BioTek) was used for reading absorbance values at 450 nm (with background subtraction of 570 nm). Cytokine concentrations were calculated by analyzing standard and sample values using a sigmoidal dose-response curve in Prism (Graphpad).
Quantitative RT-PCR
BMDMs were stimulated for 4 h with the indicated agonists, after which, cells were harvested and RNA was isolated. A commercial RNA isolation kit and Reverse Transcription kit were used following manufacturer's instructions (Qiagen). Quantitative RT-PCR was performed using the generated cDNA, SybrGreen (BioRad), and the following mouse primers:

All primers were synthesized by IDT (Coralville, IA). Relative gene expression was determined using the ΔΔCt method.
Statistical analysis
Statistical analysis was performed using GraphPad Prism. Significance level was fixed at 5% for each response measured. Figure legends indicate specific tests used for analysis of each dataset, number of animals per group, and number of times the experiment was repeated. Reported error bars indicate Mean +/- SEM.
Results
Isocaloric high-fat diets with differing fat compositions result in similar weight gain but differential effects on diabetes-related biomarkers
All three HFDs used in this study were comprised of calorically equivalent amounts of fat but differing in the ratio of unsaturated (UFA) to saturated (SFA) fats. The control diet (CD) had 10% total fat and a 1 UFA : 1 SFA ratio, the HFD had 60% total fat and a 1 UFA : 1 SFA ratio, the high-unsaturated fat diet (HUFD) had a 60% total fat and a 3.3 UFA : 1 SFA ratio and the high-saturated fat diet (HSFD) had a 60% total fat and a 1 UFA : 10 SFA ratio. As seen in Figure 1A, at the end of the 8-week feeding period, all 3 high-fat diets (HFD, HUFD, HSFD) resulted in a significant weight gain of ∼40–55% and a significant increase in food but not water intake compared to mice receiving the CD (Figure 1B and 1C).

Isocaloric high-fat diets with differing fat compositions result in similar weight gain but differential effects on diabetes-related biomarkers. C57BL/6 mice were subjected to 4 different diets for 8 weeks: a control diet (CD), a high-fat diet (HFD), a high-unsaturated fat diet (HUFD) or a high-saturated fat diet (HSFD). (A) Weight gain of mice receiving different diets expressed as the percentage of starting weight. (B) Food consumption of mice receiving different diets expressed as kilocalories. (C) Water intake of mice receiving different diets. (D) Levels of serum biomarkers related to obesity and diabetes in mice receiving different diets. Bars within graphs indicate means ± SEM. Data are representative of n = 15 mice/group for A.) - C.) and n = 10 mice/group for (D). AUC analysis followed by unpaired t-test was performed for data in (A)–(C) while One-way ANOVA analysis followed by Dunnett's test was performed for data in (D). *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
To determine whether other metabolic abnormalities were already evident by this time, we also collected serum from the mice at the end of the study and analyzed the levels of a variety of diabetes-related biomarkers (Figure 1D). These biomarkers are comprised of various hormones and peptides associated with regulating food intake, regulating energy metabolism and mobilization and influencing glucose uptake or secretion. Despite the seeming equivalency of the diets in inducing weight gain, the HFD diet (primarily lard) induced the greatest changes in serum biomarker levels, significantly increasing the levels of insulin, leptin, PAI-1 and resistin; and lowering the levels of ghrelin (Figure 1D). The HUFD diet (safflower oil and lard-based) mimicked the effect of the HFD but to a lesser extent while the HSFD (coconut oil-based), with the exception of lowering ghrelin levels, had minimal effects on diabetes-related serum biomarkers (Figure 1D) compared to the CD.
Diets high in saturated fats cause the least amount of adipose tissue inflammation
Accumulation of ATMs has been shown to be coincident with the increase in pro-inflammatory cytokine and chemokine production by the ATMs themselves and an increase in adipokine production by adipocytes which seems to precede the onset of metabolic dysfunction. 7 Precipitating factors in ATM accumulation are numerous and varied and include: direct sensing by ATMs of free FA (FFAs) induced by increased FFA flux in adipocytes during states of overnutrition, 24 ATM response to increased apoptosis of adipocytes during the development of obesity, 25 and/or a result of sensing translocated bacteria or bacterial products within the WAT. 20 In order to determine the effect of the different isocaloric HFDs on adipose tissue inflammation, we isolated peri-gonadal WAT from mice on each diet for immunohistochemical analysis. We quantified the numbers of crown-like structures (CLS) - accumulations of macrophages surrounding apoptotic adipocytes, within the WAT for each diet, as well as measured the changes in adipocyte cell size. Closely mirroring the metabolic abnormalities observed in Figure 1D, administration of a HFD or HUFD led to similar significant increases in the presence of CLSs within the WAT compared to CD-fed mice while no such increase was observed for mice administered a HSFD (Figure 2A and 2C). In line with this, hypertrophy or adipocyte size were also significantly increased by all HFDs compared to CD with HSFD promoting the least size increase (Figure 2B and 2C). During the development of obesity, expansion of adipose tissue is a result of both hyperplasia and hypertrophy. Hyperplastic adipose tissue has been reported to be metabolically normal while hypertrophic AT is associated with adipocyte dysfunction, overproduction of pro-inflammatory cytokines and IR. 26 Our findings indicate that the HFD and HUFD equally promoted adipocyte inflammation and led to remodeling of AT in a manner consistent with metabolic dysfunction while the findings for the HSFD group was suggestive of a more metabolically healthy obese (MHO) state.
High-fat diets comprised of both saturated and unsaturated fat promote enhanced pro-inflammatory cytokine production by macrophages in response to TLR4 but not NOD2 agonists while high-fat diets comprised of primarily saturated or primarily unsaturated fats resulted in decreased sensitivity to NOD2 agonists
It has been demonstrated that macrophages which infiltrate into the WAT and which promote inflammation and metabolic dysfunction are derived from the bone-marrow. 8 Previous studies have also shown that macrophages derived from the bone marrow of mice administered HFDs, despite a short period of in vitro culture, retain the characteristics of being exposed to high levels of dietary fat.27,28 Given that TLR4 and NOD2 have previously both been implicated in mediating and regulating inflammation in response to dietary FA, we generated bone marrow-derived macrophages (BMDMs) from mice which had been administered the CD or various HFDs and stimulated them in vitro with the TLR4 agonist LPS (Figure 3A) or the NOD2 agonist MDP (Figure 3B) to assess the production of pro-inflammatory cytokines and chemokines. Interestingly, only HFD administration (with equal ratios of unsaturated and saturated fats) significantly enhanced inflammatory cytokine production in response to stimulation with LPS when compared to the CD (Figure 3A). No such enhancement was observed when macrophages from the different diets were restimulated with MDP (Figure 3B). In fact, there was a significant reduction in responsiveness to MDP in BMDMs from HUFD and HSFD-fed mice when compared to CD (Figure 3B).
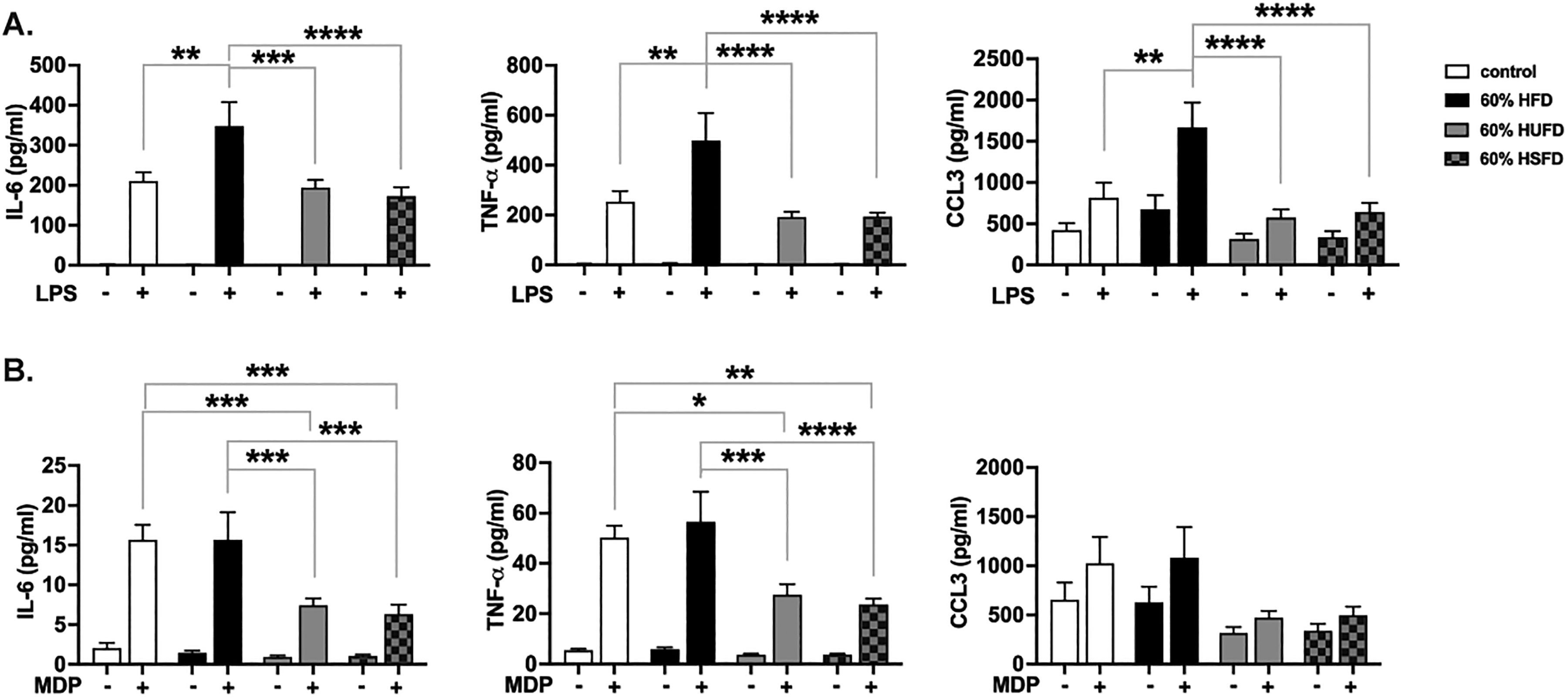
High-fat diets comprised of equal ratios of saturated and unsaturated fat increase the sensitivity of macrophages to TLR4 but not to NOD2 agonists. C57BL/6 mice were subjected to 4 different diets for 8 weeks: a control diet (CD), a high-fat diet (HFD), a high-unsaturated fat diet (HUFD) or a high-saturated fat diet (HSFD). At the end of the 8 week period, mice were euthanized and bone marrow from femurs and tibias were collected for generation of BMDM cultures. (A) ELISA analysis of pro-inflammatory cytokine and chemokine response of BMDMs from each diet regimen to overnight stimulation with 10 ng/ml LPS. (B) ELISA analysis of pro-inflammatory cytokine and chemokine response of BMDMs from each diet regimen to overnight stimulation with 10μg/ml MDP. Bars within graphs indicate means ± SEM. Data are representative of n = 10 mice/group for (A) and (B). Statistical analysis was performed using a two-way ANOVA followed by Tukey's multiple comparison test. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
A high saturated fat diet (HSFD) had the most significant effect on M2 skewing
Early studies examining the phenotypic changes in ATMs during the development of obesity indicated a switch from a predominantly M2 phenotype in lean conditions to a more M1 state in obese conditions. We sought to determine if we could detect diet-induced changes in the expression of markers associated with an M1 or M2 phenotype in response to restimulation of macrophages with TLR4 or NOD2 agonists. Using qRT-PCR, we measured the expression of two M1 associated markers: inducible nitric oxide synthase (iNOS) and CXCL1 (KC) as well as two M2 markers: Mannose Receptor 1 (Mrc1/CD206) and Ym1/Chi3l3 (lectin chitinase-like 3). None of the diets resulted in the expected M1 skewing, either basally, or upon restimulation with LPS or MDP (Figure 4). However, the HSFD led to decreased expression of KC and increased expression of MR in response to LPS stimulation when compared to the CD, suggesting the HSFD enhances M2 skewing (Figure 4). An increase in MR expression was also present basally (without further restimulation) in HSFD macrophages compared to those derived from CD (Figure 4).

Discussion
The consensus on the relationship between FA saturation and the development of obesity, inflammation, and metabolic dysfunction (IR) is mixed. In truth, this is likely to be a combination of multiple direct and indirect factors. Indirect effects include the ability of FAs to modulate gut permeability and the microbial flora (make more or less colitogenic) which, in turn, can enhance or reduce inflammation-induced metabolic dysfunction.29,30 Direct effects include the inherent ability of different FAs to be readily oxidized versus stored, 31 to directly induce or reduce pro-inflammatory signaling in immune cells and adipocytes15,32 and even the ability of different FAs to incorporate into the lipid membrane, displace proteins from membrane rafts and affect the responsiveness and activation of immune cells. 33 The generally held belief that SFAs are more detrimental than UFAs in terms of promoting obesity and metabolic dysfunction was not observed in our study. In fact, in terms of metabolic markers, when compared to the 1 UFA:1 SFA lard-based HFD, both HUFD and HSFD seemed to display a beneficial effect with the HSFD group more consistently behaving similar to CD-fed mice. HUFD and HSFD dietary fat substitutions were both primarily derived from plant (safflower oil for HUFD and coconut oil for HSFD), making the source of dietary fat an improbable cause for the observed effects. The outcome was also not likely a result of FA composition alone as coconut oil (HSFD), comprised of primarily lauric acid, has been regarded as pro-inflammatory among the SFAs yet had the least detrimental effect.15,32 Conversely, safflower oil (HUFD) comprised of primarily linoleic acid, an n-6 PUFA with established beneficial effects, 34 had a more unfavorable shift in the direction of metabolic markers compared with HSFD. We did not assess the microbiome shifts or effects on barrier permeability induced by the different diets in this study and these as well may contribute to our unexpected findings. Nonetheless, we find that despite a similar obesogenic effect, dietary FAs can differentially modulate markers of metabolic dysfunction, and not necessarily in a manner expected of the predominant FA type.
Administration of palmitic acid to macrophages has been shown to increase expression of TLR4 35 which would lead us to expect that diets high in this FA would be most responsive to subsequent TLR4 ligation. Among the 3 HFDs (HFD, HUFD, HSFD), the lard-based HFD had the greatest level of palmitic acid and did, in fact, result in the highest inflammatory response in BMDMs stimulated with LPS. Lowered responses with the HUFD safflower oil-based diet are also in accordance with randomized controlled trials indicating a beneficial (anti-inflammatory) effect of diets high in linoleic acid. 36 Given previous reports that SFAs could bind TLR4 and promote inflammatory responses, 15 we expected that the HSFD among the 3 HFDs would have the most profound effect on cytokine and chemokine production. Interestingly, the HSFD coconut oil-based diet (lauric acid) was the least inflammatory of the 3 HFDs. While this is in contrast to the in vitro studies using purified FAs, this unexpected finding of lowered inflammation for coconut oil-based HSFD was also observed by other groups.37,38 Such discrepancies might be accounted for by SFA chain length given that another study demonstrated protection from HFD-induced type 2 diabetes and non-alcoholic steatohepatitis (NASH) when the ratio of medium chain fatty acids (MCFAs) to long chain fatty acids (LCFAs) was increased. 38
Administration of lauric acid to HCT116 colonic epithelial cells was previously shown to activate NF-κB and promote IL-8 secretion which was reduced by a dominant negative form of NOD2 and by NOD2 knockdown, suggesting a potential interplay between fatty acid sensing and peptidoglycan sensing. 39 Lauric acid (the primary FA constituent of HSFD) is sensed through GPR4040,41 and GPR8442,43 while linoleic acid (the primary FA constituent of HUFD) is sensed by GPR4040,41 and GPR120. GPR84 is considered pro-inflammatory and is highly expressed in macrophages. 43 However, in the present study, both HUFD and HSFD blunted subsequent macrophage responsiveness to MDP. Interestingly, there have been some in vivo studies looking into the use of NOD2 agonists as “postbiotics” which were found to sensitize to insulin and lower adipose tissue inflammation in a mechanism requiring IRF4. 44 Follow up work by this same group using bone marrow chimeras indicated the requirement for RIP2, the kinase downstream of NOD2, within the non-hematopoietic compartment as important for mediating this effect. 45 Thus, in the future, it would be interesting to understand how these particular FA receptors modulate the responsiveness of cells towards NOD2 agonists.
Peroxisome Proliferator-Activated Receptors (PPARs) are nuclear hormone receptors which function as transcription factors. PPARs have been demonstrated to be important regulators of metabolism through their ability to sense dietary FAs (or their metabolites) and exert an anti-inflammatory effect. 46 Among the PPARs, PPARγ is known as a master regulator of adipogenesis. 47 It has been shown to increase insulin sensitivity by enhancing fatty acid storage and promoting release of adiponectin from adipocytes. PPARγ can also promote M2-skewing of macrophages. 48 While we were not able to test involvement of PPARγ (or other PPARs) in our system, it is possible that sensing of dietary FAs (HUFD and HSFD conditions) could promote PPAR-mediated transrepression of inflammatory genes and blunting of subsequent MDP induced cytokine responses. Likewise, this receptor may also be involved in the observed HSFD-induced skewing to an M2 phenotype basally and upon restimulation with LPS. However, these will need to be empirically confirmed.
It is now appreciated that macrophages exhibit a spectrum of activation states in vivo. In particular, a recent study using single cell transcriptomics highlighted the functional heterogeneity within ATMs, demonstrating that populations which tracked with adiposity and disease (CD9+ ATM) did not fall neatly into the classical M1 category. 49 However, the authors did emphasize the inflammatory nature of the CD9+ ATM subset, which tracks with the markers we measured for our M1 classification. We do recognize that use of BMDMs rather than ATMs is a limitation of our study and that analysis of recently identified populations such as CD9+ ATMs in future studies may be more relevant. Nonetheless, we demonstrate that use of BMDMs may be a useful surrogate which have been shown by other groups to retain the epigenetic changes associated with exposure to different diets in vivo.27,28
We are very interested in the potential for intervention in settings of chronic inflammation such as during the chronic low-grade inflammation induced by HFDs. Our laboratory has previous experience with using various pharmacologic inhibitors for in vitro and in vivo inhibition of NOD2:RIP2 signaling.50,51 While the data presented in this work (no enhancement of inflammatory responses towards NOD2 agonists by dietary fats) as well as work from other independent groups, 52 suggest that use of RIP2 inhibitors to prevent diet-induced metabolic dysfunction might have limited clinical utility, it is worth noting that specific RIP2 inhibitors have not yet been tested in this context. Furthermore, the fact that RIP2 also lies downstream of NOD1 whose agonism has been shown to drive IR and hyperglycemia, 53 suggests that additional studies using specific RIP2 inhibitors may still be worthwhile.
Overall, our data suggest that chronic exposure to diets with differing fatty acid composition can influence the subsequent macrophage responsiveness to bacterial products in a manner that does not correlate with the degree of fatty acid saturation. In this context, NOD2 agonism appears to have a neutral (HFD) or beneficial (HSFD/HUFD) effect on inflammation.
Footnotes
Abbreviations
Acknowledgements
The authors would also like to thank Dr Griffith Parks and his laboratory for the use of their RT-PCR machine and the Imaging Core for the use of the Leica histology microscope. This work was supported in part by a UCF Vice President of Research Advancement of Early Career Researchers (AECR) Award and start-up funds from the Univ. of Central Florida to J.T.T-A and in part by an NIH R01DC013318 to T.A.G. We would also like to take this opportunity to acknowledge all the authors whose work we failed to cite due to space and reference limitations.
Authorship
M.G.S. performed a number of the experiments, collected some of the data, and contributed to the writing of the manuscript.
M.H.M. performed a number of the experiments, collected some of the data, and contributed to the writing of the manuscript.
A.N.C performed a number of the experiments and collected some of the data.
T.A.G. designed the high-fat diet compositions, supervised the feeding experiments and contributed to the writing of the manuscript.
J.T.T-A and was responsible for the conception and design of the study. She also contributed to the writing of the manuscript, approved the final version of the manuscript to be submitted, and was involved in the interpretation of the data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a UCF Vice President of Research Advancement of Early Career Researchers (AECR) Award and start-up funds from the Univ. of Central Florida to J.T.T-A and in part by an NIH R01DC013318 to T.A.G.
