Abstract
Malaria is often characterized by a complicated disease course due to multifaceted intrinsic genetic factors of the host and the parasite. This study aimed to investigate the role of interleukin-27 (IL-27) gene polymorphisms in Plasmodium falciparum malaria infection in a Saudi Arabian cohort. This case-control study obtained blood samples from 250 malaria patients with P. falciparum and 200 randomly identified healthy control subjects from the Malaria Center in the Jazan area. Malaria patients were grouped into three cohorts as follow: low (<500 parasites/µl of blood), moderate (500–1000 parasites/µl of blood), and high (>1000 parasites/µl of blood) parasitemia. The results show that the IL-27 variant rs181209 was significantly associated with malaria patients (P = 0.026). Similarly, the homozygous GG genotype of rs26528 was also associated with risk of developing P. falciparum malaria (P = 0.032). The minor allele C of variant rs181206 exhibited an association with low to moderate parasitemia (P = 0.046). Furthermore, the rs181209 AA genotype was statistically significant in age group 1–5 years (P = 0.049). In conclusion, this study suggests that variant rs181209 and rs26528 could be associated with the risk of malaria infection by P. falciparum in the population studied.
Background
Globally, malaria continues to be the leading protozoan infection, with over 219 million people in more than 100 nations currently affected by malaria infection.1,2 Plasmodium falciparum is considered the deadliest of Plasmodium species and is associated with widespread morbidity and mortality in human hosts. 3 In fact, almost 5% of the global population that suffers from malaria infection with P. falciparum has been reported to have grave clinicopathological consequences such as hemoglobinuria, severe malarial anemia (SMA), cerebral malaria (CM), and pulmonary distress. 4
Inflammatory responses are coordinated by a cohort of immune cells that modulate inflammation through the release of specific pro- and anti-inflammatory cytokines such as interferon-γ (IFN-γ), interleukins (IL) and tumor necrosis factor alpha (TNFα).5,6 A cytokine of recent interest in malaria infection is interlkeukin-27 (IL-27). IL-27 is a heterodimeric protein that belongs to the IL-12 protein family and has pro-inflammatory and anti-inflammatory properties.7,8 Physiologically, IL-27 is known to promote IL-10 production and is also involved in limiting inflammation induced by infectious diseases. Furthermore, IL-27 has been suggested to regulate T-lymphocyte activities to modulate both innate and humoral immunity, acting as a candidate cytokine to influence immunological responses. 9
Several studies have recently investigated the role of the IL-27 cytokine in malaria infection. Since IL-27 regulates the production and release of IL-10 cytokines by T-helper 1 (Th1) cells, it is assumed that enhanced IL-10 release by IL-27 could provide protection against malaria infection and associated immunopathological complications in the host.9,10 Rahiman et al. confirmed that the increase in IL-10 cytokine release and the subsequent anti-inflammatory activity after exposure to IL-27 in a mouse model infected with P. berghei, indicating the potential role of IL-27 as an immunotherapy in the resolution of malaria infection. 7 However, a study by Zhang et al. showed an increased risk of clinical malaria in children with IL-10 polymorphisms and IL-10 cytokine production. 11 Similarly, risk of severe malaria, 12 CM and SMA were prominent with IL-10 polymorphisms in a study by Ndila et al. 13 Kimura et al. also showed that in models infected with P. berghei, IL-27 produced by CD4+ T lymphocytes generated a protective immune response against malaria infection. 8 Another study showed that IL-27 regulates the expression of IL-10 cytokines by CD4+ cells in lymphoid and non-lymphoid tissues during malaria infection with P. yoelii. 14 Villegas-Mendez et al. have also reported the control of Th1 cells by IL-27 independent of IL-10 in the NK65 model of P. berghei murine malaria infection. 15 In a similar vein, Findlay et al. established that mice lacking the IL-27 receptor (IL-27R) were at increased risk of developing malaria infection by P. berghei NK65, and that the IL-27R signaling pathway regulates effector T lymphocyte function in malaria infection. 16 This evidence indicates that IL-27 appears to provide some form of protection against multiple species of Plasmodium.
Recently, only one study has evaluated the expression levels of IL-27 and other cytokines in infants infected with P. falciparum. 17 The study found significantly higher expression levels of IL-27 in non-infected infants with P. falciparum compared to infants with simple malaria and severe malaria. They reported very high levels of IL-10 cytokine in infants with no complications and severe malaria compared to their non-infected counterparts, 17 suggesting protection from infection through IL-27 and IL-10 pathway. However, the role played by IL-27 gene polymorphisms in influencing the dynamics of P. falciparum malaria infection is unknown. The present study thus aims to deepen and update our knowledge of the pleiotropic role of the IL-27 gene polymorphisms and the expression of cytokines in malaria infection by P. falciparum in a Saudi Arabian cohort.
Materials and methods
Study design
This case-control study obtained blood samples from 250 malaria patients with P. falciparum infection and 200 randomly identified healthy control subjects from the Malaria Center in the Jazan area, located in the southwestern of the Kingdom of Saudi Arabia. While the national malaria control program in Saudi Arabia, initiated in 1948, has accomplished a remarkable decline in malaria, it is still prevalent in southwestern regions like Jazan and Aseer. One recent study found 94.3% of the malaria cases in Jazan region to be infected with P. falciparum. 18 The level of parasitemia was measured by examining thick blood smears and counting the P. falciparum parasites based on 200 white blood cells (WBCs) count. 19 Malaria patients were grouped into three cohorts according to their parasite count as follow: Low (<500 parasites/µl of blood), moderate (500–1000 parasites/µl of blood), and high (>1000 parasites/µl of blood) parasitemia. 20
Ethical considerations
This study was granted ethical approval (registry number 041) by the Ethics Review Committee of King Fahad Hospital (KFCH), Jazan. The study was also conducted in accordance with the ethical guidelines laid out in the Declaration of Helsinki of 1975. Written informed consent was obtained from the study subjects prior to enrollment, and consent was obtained on behalf of the children from their legal guardians. Identifiers were removed from patient data and biological samples for confidentiality. The demographics and clinical characteristics of the study participants were documented and saved on a password protected computer, which can only be accessed by the research staff.
Single nucleotide polymorphism (SNP) selection
A total of six SNPs of the IL-27 gene (rs34833, rs17855750, rs181209, rs26528, rs40837, and rs181206) were included for genotyping. The following criteria were used to identify selected SNPs: (i) MAF <5%; and (ii) linkage disequilibrium (LD) threshold (r2) < 0.8, or (iii) proof of association with P. falciparum malaria. Information on the SNP marker for the IL-27 gene in this study is presented in Supplementary Table 1.
Genotyping
DNA samples were extracted from blood samples using DNeasy commercial kit (Qiagen, Valencia, CA, USA) according to the manufacturer's protocol. To identify SNPs in DNA samples, nearby primers were designed using Primer3 input software version 0.4.0 (available at: http://bioinfo.ut.ee/primer3-0.4.0/). SNP-specific primers and PCR conditions are detailed in Supplementary Table 2. Genotyping was performed using direct DNA sequencing based on polymerase chain reaction (PCR) or restriction fragment length polymorphism (RFLP).
The PCR products were analyzed using GoTaq Green Master Mix PCR (Promega, Madison, WI, USA). The PCR reaction products were analyzed by agarose gel electrophoresis to confirm the purity of the PCR products. Direct sequencing of the amplification products of SNPs (rs34833, rs17855750, rs181209, rs26528 and rs40837) was performed using the BigDyeTM Terminator v 3.1 Cycle Sequencing Kit (Applied Biosystems, Foster City, CA, USA) according to the manufacturer's protocol. Sequences were analyzed for forward and reverse reads via SeqMan Pro 15 LaserGene (DNASTAR, Inc., Madison, WI). The amplification product of rs181206 was analyzed using the RFLP method. Briefly, the amplification products of PCR were treated with the restriction enzyme (HinfI). The digested products were separated in a 2% agarose gel and visulaized under UV light. Afterwards, they were stained with ethidium bromide, examined and photographed via ultraviolet light. ImageJ software (https://imagej.nih.gov/ij/download.html) and MolWT macro (PHASE GmbH, Luebeck, Germany) were used to identify the size of restricted fragments.
Luciferase reporter assays
IL-27 5′UTR and 3′UTR PCR fragments were amplified and subcloned in the corresponding plasmids using pGL3 Basic Vector (Promega, Madison, WI, USA) and pmirGlo Luciferase Reporter Vector, respectively. The primers were 5-’ end labeled for restriction enzymes with a specific endonuclease recognition sequence. Appropriate restriction enzymes (BglII, XhoI) and (SacI, Xbal) were used to investigate DNA fragments in the 5′UTR and 3′UTR region respectively. The primers used for amplification, reaction conditions and chromosomal position for the 5′ UTR and 3′ UTR are shown in Supplementary Table 3 and 4, respectively. The PCR reaction was performed using HotStar PCR (Qiagen) following the manufacturer's protocol. Escherichia coli DH5α-competent cell transformation with plasmid was performed for colony selection in ampicillin plates. The clones were selected, and genomic DNA was extracted using the QIAamp DNA mini kit (Qiagen) according to the manufacturer's instructions. The plasmids were then transfected into HEK293 cells (Human Embryonic Kidney 293 cells) using Lipofectamine 2000 (Qiagen), which were then collected and eventually observed for luciferase activity after 48 h.
Statistical analysis
We analyzed the SNPs in the IL-27 gene and used Haploview software version 4.0 (Broad Institute of MIT and Harvard, Cambridge, MA, USA) to calculate the haplotype frequencies and minor allele frequencies (MAF). For the genotypic association test, we used the DeFinetti program (available at: https://ihg.gsf.de/cgi-bin/hw/hwa1.pl). For possible modes of inheritance, we explored each variant by performing allele, dominant and recessive model analyses. The results of the association were presented as odds ratios (OR) with 95% confidence intervals (95% CI). The Hardy-Weinberg equilibrium (HWE) test was performed to find deviation, if any. P-values <0.05 in a two-tailed test were considered statistically significant.
Results
In the present study, we investigated the distribution of the selected SNPs within the IL-27 gene (rs34833, rs17855750, rs181209, rs26528, rs40837 and rs181206) in patients infected with the malaria parasite P. falciparum and healthy controls. The baseline characteristics of the patients included in this study are illustrated in Table 1.
Baseline characteristics of P. falciparum-infected patients.

IL-27 genotype and allele frequency distribution linked with infection with p. falciparum
The homozygous genotype of the major allele was defined as the reference genotype (Ref) in each polymorphism of the IL-27 gene. Our findings demonstrated that two polymorphisms of the IL-27 gene (rs181209 and rs26528) were significantly associated with a higher susceptibility to malaria infection with P. falciparum when compared to healthy controls. However, we did not observe significant differences between malaria patients infected with P. falciparum and healthy controls in the genotype and allele frequency distributions of the IL-27 SNPs rs34833, rs17855750, rs40837 and rs181206 (Table 2).
Genotypic distribution of IL-27 SNPs when healthy control group was compared with patients’ group.

*A p-value of less than 0.05 was considered significant.
IL-27 genotype and allele frequency distribution associated with advanced P. falciparum infection evaluated by parasite density
Of the six SNPs of the IL-27 gene, no polymorphism was found to have a significant association with low parasite density (group I). In comparison of low and moderate parasite density (groups I + II) with that of high parasite density (group III), showed statistically significant association with the minor allele C of rs181206 (OR = 2.066; 95% CI 0.999–4.271, χ2 = 3.967 and P = 0.046). The IL-27 genotype and allele frequency distribution are summarized in Tables 3 and 4.
Genotypic distribution of IL-27 SNPs when group i (low parasitemia) of P. falciparum-infected patients was compared with groups II + III (Moderate + High parasitemia).

Genotypic distribution of IL-27 SNPs when groups I + II (low + moderate parasitemia) of P. falciparum-infected patients was compared with group III (High parasitemia).

*A p-value of less than 0.05 was considered significant.
IL-27 genotype and allele frequency distribution linked to malaria infection based on age groups
A higher risk of P. falciparum infection with the IL-27 variant rs181209 was observed in malaria cases in the age groups 1–5 years compared to cases in cohorts aged 6 years and over. The frequency of occurrence of the homozygous polymorphism AA genotype rs181209 was significant in malaria infection cases in the age group 1–5 years (genotype frequency 14.3% vs 3.3%, OR = 0.131; 95% CI 0.012–1.405, χ2 = 3.85 and P = 0.049). The frequency of the minor A allele of rs181209 was significantly higher in the malaria patients in the 1–5 age group than in their counterparts in the 6 years and older age cohort (allele frequency 35.7% vs 14%, OR = 0.293; 95% CI 0.095–0.900, χ2 = 5.15 and P = 0.039) (Table 5).
The genomic distribution of IL-27 SNPs was compared in patients aged 1–5 years with patients aged 6 years and older.

*A p-value of less than 0.05 was considered significant.
IL-27 haplotype frequency distribution
Haplotypes consisting of the C allele of rs181209, the T allele of rs17855750, the A allele of rs26528, and the G allele of rs34833 (C-T-A-G) were significantly associated with a lower risk of malaria infection by P. falciparum (Frequency 72.7%, P = 0.013). On the contrary, the A allele of rs181209, the T allele of rs17855750, the G allele of rs26528 and the G allele of rs34833 (A-T-G-G) were significantly related with ahigh risk of malaria infection (Frequency 11.3%, P = 0.045) (Table 6). None of the haplotype blocks were found to be significant upon comparison of malaria patient cohorts based on malaria parasite density. Lastly, the haplotype block of the A allele of rs181209, the T allele of rs17855750, and the G allele of rs26528 (A-T-G) had a significantly higher frequency in cases with malaria-infected for age group 1–5 years when compared to 6 years and older (Frequency 13.3%, P = 0.013) (Table 7).
Haplotype analysis of IL-27 SNPs was performed when the control group was compared with patients infected with P. falciparum.

*A p-value of less than 0.05 was considered significant.
Haplotypes of the IL-27 gene were compared when group 1–5 years of parasite was compared with 6 years and older.
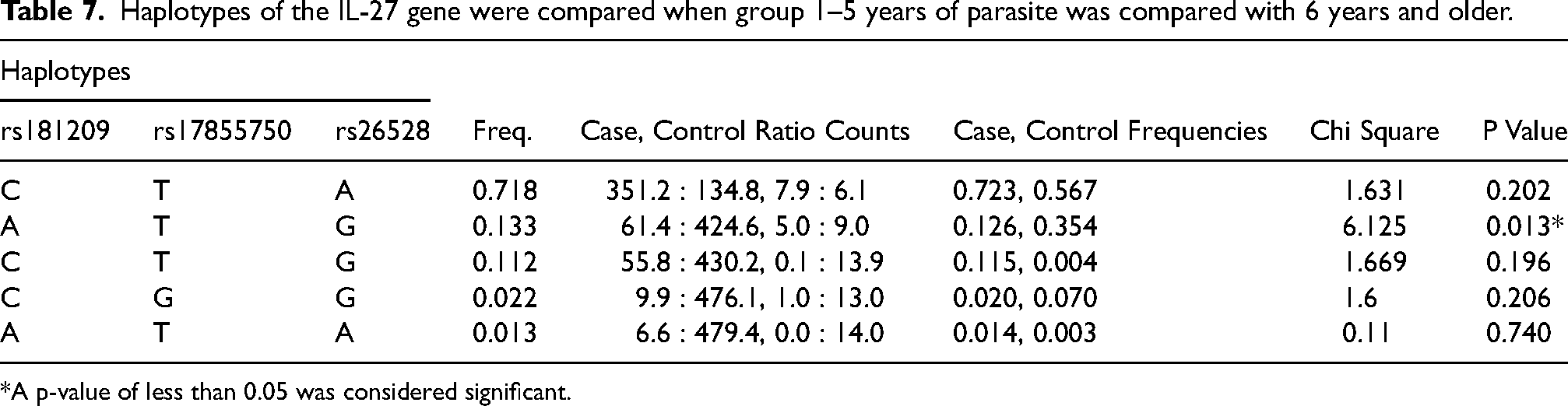
*A p-value of less than 0.05 was considered significant.
Deletion analysis of the 5′ UTR and 3′ UTR regions of the il-27 gene
For 5′ UTR, a significantly high mean luciferase activity (relative luciferase unit, RLU) was observed in constructs F4R1 (P = 0.001) and F3R2 (P = 0.001) in the 5′ UTR region of the IL-27 gene. However, a significant reduction in the expression of the IL-27 gene was observed for group F3R1 (P = 0.045). While group F1R2 (P < 0.0001) demonstrated significantly higher mean luciferase activity, indicating high gene expression, group F1R1 (P < 0.0001) exhibited significantly lower mean luciferase activity, and therefore low expression in the 3′UTR region (see Figures 1 and 2).

Luciferase reporter assay to monitor the activity of the IL-27 5′ UTR promoter region. Different parts of this region were generated and tested for their activities as detected by the luciferase activities. The constructs were transfected into HEK293 cells that were lysed after 6 h and luciferase activity was determined. Each experiment was repeated three times in triplicates ± SEM. Statistical significance was established using the unpaired one-way Analysis of Variance (ANOVA) test (p < 0.05).

Luciferase reporter assay to monitor the activity of the IL-27 3′ UTR region. Different parts of this region were generated and tested for their activities as detected by the luciferase activities. The constructs were transfected into HEK293 cells, the cells were lysed after 6 h and the luciferase activity was determined. Each experiment was repeated three times in triplicates ± SEM. Statistical significance was established using the unpaired one-way ANOVA test (p < 0.05).
Discussion
In this study, we investigated six polymorphisms (rs34833, rs17855750, rs181209, rs26528, rs40837, and rs181206) in the IL-27 gene that produces a pleiotropic IL-27 cytokine. The study evaluated the association of SNPs with the risk of development or protection against P. falciparum malaria infection, the level of P. falciparum parasite density, or the age-related distribution of P. falciparum malaria infection. Of the six SNPs genotyped for the IL-27 gene, our findings demonstrated that the frequency of the AA genotype of variant rs181209, the GG genotype, and the minor allele G of variant rs26528 were significantly associated with the risk of malaria infection with P. falciparum.
Cytokines have been shown to play a key role in the immunoprotection and immunopathology of malaria infection by P. falciparum. 21 IL-27 has been reported to induce proliferation and survival of activated effector CD4+ T lymphocytes during malaria infection, which helps in reducing the morbidity and mortality associated with Plasmodium malaria infection.10,16 However, unlike the healthy control in this study, we found that two SNPs of the IL-27 gene (rs181209 and rs26528) were significantly associated with a high risk of malaria infection by P. falciparum. This indicates that such variations could influence the function of effector CD4 + T lymphocytes whose function is modulated by IL-27 22 and could consequently result in severe malaria complications. Related to these results, Ayimba et al. reported that children who had a decrease in IL-27 cytokine levels were prone to severe malaria. 23 In addition, Ayimba et al. showed that severe malaria was caused by the reduced expression of the immunoregulatory IL-27 cytokine among infants infected with the parasite P. falciparum. 17
IL-27 is considered a potent blocker of Th17 cell differentiation, thus attenuating inflammatory responses by inhibiting Th17 response after successful alleviation of the parasite P. falciparum in malaria infection.24,25 Here, the two variants of the IL-27 gene, rs181209 and rs26528, could possibly reduce the expression of IL-27 cytokines, thus improving persistent responses of Th17 cells and increasing the risk of P. falciparum malaria infection. However, this finding contradicts with those of El-behi et al., who suggested that IL-27 has no effect on differentiated Th17 cells. 26 This suggests that Th17 cells exhibit functional differences, depending on their stage of development, with developing Th17 cells being vulnerable and differentiated Th17 cells could be resistant to IL-27 inhibition.26,27 A study by Armah et al. in the Ghanaian children population cited that cerebral malaria-related mortality was not related to IL-27 cytokine. 28 This evidence stands in stark contrast to the findings of our study, which has highlighted that IL-27 does not mediate the inflammatory response of Th17 cells during malaria infection with P. falciparum.
In our study, we found a significant association between the minor allele C of the IL-27 gene variant rs181206 and low to moderate parasitemia, indicating an association with a lower risk of advanced malaria infection with P. falciparum. Findlay et al. confirmed that wild-type mice (control) developed high parasitemia when infected with P. berghei NK65; however, mice knocked out of the IL-27 receptor (WSX-1- / -) were more susceptible to malaria infection much faster than wild-type mice regardless of small malaria parasite counts. The etiology of death in WSX-1−/− mice infected with P. berghei was secondary to liver pathology aggravated by Th1 immune responses. 16 Consistent with this finding, Villegas-Mendez et al. reported that the IL-27 receptor (WSX-1) mediates Th1 immune responses during malaria infection, independent of IL-10 and Foxp3+ T-regulatory cells. 15
Polymorphisms of the IL-27 gene rs34833, rs181209 and rs26528 are located in the intronic region, which could regulate the expression of the IL-27 gene. 29 Furthermore, the polymorphism of the IL-27 gene rs17855750 is located in the exonic region, which is a missense mutation, and could influence the coding or structure of the IL-27 cytokine, 29 and subsequently the function of the protein product. Several research studies have presented a link between the variants of IL-27 that were found in this study and also show a significant association or frequency of occurrence with the status of the disease. Previous studies have shown an association between the IL-27 variant rs17855750 and several pathologies such as allergic rhinitis, preeclampsia, dilated cardiomyopathy, atrial fibrillation, increased total abdominal fat, HT, ALL, and renal cell carcinoma.29–35 In addition, Posadas-Sánchez et al. found that the IL-27 rs26528 T allele is associated with a lower risk of premature coronary artery disease, 36 and Vargas-Alarcon et al. showed that the rs26528 T allele is associated with hypoadiponectinemia in subjects resistant to insulin and hypertension in subjects not resistant to insulin. 35 To our surprise, the literature search did not reveal any disease process associated with the IL-27 polymorphism rs181209.
Analysis of haplotypes of variants of the IL-27 gene revealed that the frequency of occurrence of two haplotypes, C-T-A-G and A-T-G-G, was significant in patients infected with the malaria parasite P. falciparum and the healthy control group. The C-T-A-G haplotype (rs181209, rs17855750, rs26528 and rs34833 respectively) was associated with protection against malaria infection by P. falciparum, while the A-T-G-G haplotype (rs181209, rs17855750, rs26528 and rs34833 respectively) was found to be related to a high vulnerability to malaria infection by the parasite P. falciparum. Furthermore, we found a haplotype was found to be significantly predominant in the P. falciparum malaria population at 1–5 years in our study; A-T-G (rs181209, rs17855750 and rs26528 respectively) in this study. All haplotypes found to be statistically significant had at least one risk allele present.
We also performed a luciferase reporter assay to examine the contribution of IL-27 variants to the expression of the IL-27 gene in the 5′UTR and 3′UTR regions. The finding confirmed that the IL-27 variants significantly enhanced luciferase activity in HEK293 cells and therefore could influence the transcription of the IL-27 gene and the stability of RNA transcripts. Our data allowed us to draw the conclusion that the genetic variation of IL-27 could influence the risk of P. falciparum malaria infection by increasing and/or decreasing its expression.
Taking into account the limitations of the study mentioned below, we emphasize that the findings of our study should be interpreted with caution: 1) the causal relationship cannot be established owing to the retrospective nature of the study; 2) false negative associations could be anticipated due to the small number of participants in the study; 3) there is potential selection bias due to convenient selection of subjects restricting the extension of the findings of our study to the general population; 4) since we studied only variants tagged in the functional region of the IL-27 gene, variants of the IL-27 gene in the noncoding region or between exons that could influence IL-27 cytokine expression could have been ignored; and 5) the preliminary findings of this study with respect to the polymorphisms of the IL-27 gene and its association with P. falciparum malaria infection observed in other ethnic populations.
Conclusions
In summary, our study findings suggest that the AA genotype of variant rs181209 and the GG genotype and the minor allele G of variant rs26528 could be associated with the risk of malaria infection by P. falciparum by influencing the protein expression of the IL-27 gene and subsequent alteration of the immune response related to Th1, Th2 and Th17. We stress that future research on this specific area is needed as our study is not a true reflection of the immunopathological mechanisms underlying the malaria infection by P. falciparum. However, it does offer a well-directed route for potential work.
Supplemental Material
sj-docx-1-ini-10.1177_17534259231178594 - Supplemental material for Association between interleukin-27 gene polymorphisms and Plasmodium falciparum Malaria
Supplemental material, sj-docx-1-ini-10.1177_17534259231178594 for Association between interleukin-27 gene polymorphisms and Plasmodium falciparum Malaria by Nada H. Aljarba, Mashael R. Al-Anazi, Tahani M. Al-Hazani, Mohammed I. Shafeai, Fuad H. Rudiny, Ali M. Motaen, Saad M. Bin Dajem, Hani Alothaid, Jahad Alghamdi, Saad Alkahtani and Ahmed A. Al-Qahtani in Innate Immunity
Footnotes
Acknowledgment
This work was funded by Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2023R62), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Princess Nourah Bint Abdulrahman University Researchers Supporting Project Number (PNURSP2023R62), Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
