Abstract
Psoriasis vulgaris (PsV) is an immune-mediated inflammatory disorder with devastating psychosocial consequences. Expression of immunoregulator molecules on leukocytes in PsV remains unclear. Leukocyte-associated Ig-like receptor-1 (LAIR-1) and complement receptor-1 (CR-1) are immunoregulator receptors reported to bind complement component 1q involved in phagocytosis. We aimed to explore if altered leukocyte expression of LAIR-1 and CR-1 is associated with PsV. This case–control study included 36 PsV patients and 36 healthy controls. Neutrophils, monocytes and B and T cells were examined by flow cytometry for LAIR-1 and CR-1 mean fluorescence intensity (MFI) and positive cell percentage. Comparison between both groups revealed a significant decrease in LAIR-1 MFI on neutrophils and T cells (P < 0.001 and P = 0.003, respectively). CR-1 MFI on neutrophils, monocytes and T cells also showed a significant decrease in patients (P = 0.033, P = 0.001 and P = 0.040, respectively). There was a significant positive correlation of LAIR-1 MFI on neutrophils with CR-1 MFI on neutrophils (r = 0.503; P = 0.002) and LAIR-1 MFI on monocytes with CR-1 MFI on monocytes (r = 0.371; P = 0.026). Receiver operating characteristic curves revealed that CR-1 MFI on monocytes had the highest discrimination power to differentiate patients from controls, with 86.1% specificity and 75% sensitivity (P = 0.001). In conclusion, altered leukocytes expression of LAIR-1 and CR-1 is associated with PsV. Down-regulated CR-1 MFI on monocytes is a promising diagnostic biomarker for PsV.
Introduction
Psoriasis is a common, chronic, auto-inflammatory and debilitating disease that predominantly affects the skin, cardiovascular system and joints. Psoriasis impacts patients’ quality of life, with significant psychological co-morbidity. 1 The unpleasant skin appearance contributes to reduced employment levels, resulting in economic burden at both individual and societal levels. 2 The worldwide prevalence of psoriasis is estimated to be 1–2% of the general population. It is characterised by increased proliferation of keratinocytes and leukocytic infiltration of the papillary dermis and epidermis. 3
Autoimmune and auto-inflammatory mechanisms have been suggested to be involved in the pathology of psoriasis, along with the alteration in immunoregulatory mechanisms. 4 The most common form of psoriasis is Psoriasis vulgaris (PsV) which is characterised by well-demarcated, erythematous and raised plaques with white scales. PsV is mediated by cells and molecules of both innate and adaptive immune systems. 5
Immune de-regulation in PsV is mediated mainly by T and dendritic cells (DCs). DCs release IL-23 and IL-12 to activate IL-17-producing T cells, and Th1 and Th22 cells to produce IL-17, IFN-γ, TNF-α and IL-22. Psoriatic cytokines activate keratinocytes, promoting their hyperplasia, and switch the classes of T cells recruited to the skin to amplify the inflammatory process. 6 Cytokines also contribute to neutrophil recruitment. The degranulation of neutrophils results in generation of reactive oxygen species (ROS), activation of inflammatory mediators and the formation of auto-Ags. 2 Keratinocytes are sources of monocyte chemotactic protein-1, and monocytes differentiate into macrophages and migrate to the sites of inflammation, producing pro-inflammatory cytokines. 7
The most widely used scale to assess the severity of psoriasis is the Psoriasis Area and Severity Index (PASI). A 75% reduction in the PASI score (PASI 75) is the current benchmark of primary end points for most clinical trials of psoriasis. 3
The appropriate response of the immune system against tissue injury depends on a dynamic balance between activation and inhibitory signals. Insufficient inhibition leads to a state of hyperactivity that facilitates the development of chronic inflammation. Inhibitory receptors of the immune response play a crucial role in maintaining this balance on the cell surface. 8
Leukocyte-associated Ig-like receptor-1 (LAIR-1, CD305) is a transmembrane glycoprotein inhibitory collagen receptor involved in the regulation of leukocyte inflammatory mediator production and cytotoxicity. 9 LAIR-1 activation is reported to occur by binding to complement component 1q (C1q). 10 LAIR-1 is expressed on lymphoid and myeloid cells, monocytes and immature CD34+ progenitor cells. 9 Soluble LAIR-1 (sLAIR-1) is produced upon shedding of the extracellular region of membrane-bound LAIR-1. 11
The complement system contributes substantially to homeostasis and acts as an efficient immune surveillance system by orchestrating immune responses. 12 It is a crucial component of the host response to tissue damage and essential for fast elimination of invading pathogens mainly by opsonisation followed by phagocytosis and to a lesser extent through cell lysis by deposition of the membrane attack complex. 13 Default in complement regulation can exacerbate inflammatory and degenerative responses. 14
Complement receptor-1 (CR-1, CD35, C3b receptor) is a glycoprotein primarily expressed in erythrocytes, B and T cells, neutrophils, monocytes, microglia and neurons. 15 CR-1 also occurs in plasma as soluble CR-1 (sCR-1). 16
Membrane-bound CR-1 and sCR-1 regulate complement activity by binding cleaved C3b and C4b components and C1q deposited in altered cell components or pathogens, removing opsonised Ags and blocking the final steps of the complement cascade. Membrane-bound CR-1 also fosters Ag presentation to B cells. 17 CR-1 acts as a receptor for C1q and other defence collagens such as ficolins. C1q is involved in phagocytosis. It binds the prey through its globular regions and the phagocyte through its collagen stalks. 18
Both LAIR-1 and CR-1 are known to be involved in immune-mediated inflammatory diseases, 19 linked to phagocytic cells, and both bind to C1q which justifies the interest in studying them both on leukocytes, including peripheral blood phagocytes in PsV, which is known for its chronic auto-inflammatory process with excessive tissue damage.
The current study aimed to explore the presence of an association between PsV and altered leukocyte expression of LAIR-1 and CR-1 and if this altered expression can serve as a diagnostic biomarker for PsV. In addition, we aimed to investigate if LAIR-1 and CR-1 were linked to the disease severity indicated by the PASI.
Methods
Study participants
This case–control prospective study included 72 subjects, of whom 36 were PsV patients (23 males and 13 females) with a mean age of 37.36 ± 6.84 yr. Cases were subdivided into two groups according to their PASI score and disease severity: group 1 (severe psoriasis) with a PASI score > 12 (n = 16), and group 2 (mild and moderate psoriasis) with a PASI score ≤ 12 (n = 20). A total of 36 sex- and age-matched healthy controls (21 males and 15 females) were included, with a mean age of 36.39 ± 6.3 yr. PsV patients were recruited from the Dermatology Outpatient Clinic in Al-Zahraa University Hospital, Cairo, Egypt.
The participants were informed about the aim of the study, and written consent was obtained before enrolment in the study. The study was approved by Research Ethical Committee of the Faculty of Medicine for Girls Institutional Research Board (AFMG-IRB), Al-Azhar University (registered at the Central Administration of Research and Development; Egyptian ministry of Health, IRB number 202003223).
Inclusion criteria were adult patients aged > 18 yr of both sexes with PsV. Controls enrolled in this study were sex- and age-matched healthy individuals recruited during blood donation in Al-Zahraa University Hospital Blood Bank. Controls were included after negative viral hepatitis screening and normal laboratory results were reported. Exclusion criteria were: patients with other types of psoriasis (pustular psoriasis or erythrodermic psoriasis), PsV patients suffering from psoriatic arthritis or those under systemic steroid treatment and PsV patients with any other chronic autoimmune, inflammatory disease or known association with any oncogenic disease. PsV pregnant women, children and disabled patients were also excluded.
History taking
The patients’ personal history, including their name, age, sex, occupation and special habits of medical importance, was recorded. A history of their present illness was also taken, including onset, course, duration of the disease, the last episode, assessment of PASI and symptoms of arthritis, along with their medical history (history of other skin or systemic diseases, presence of chronic or active infections) and drug history (history of drug intake for psoriasis or any other medical problem). In addition, any history of surgical operations in the last year and a family history of psoriasis were recorded.
General and dermatological examination
A general examination was performed in order to exclude associated systemic diseases. A dermatological examination of the skin, hair, nails and mucous membrane was undertaken to exclude other dermatological diseases. Dermoscopic and histopathological confirmation was obtained for diagnosis of PsV lesions. A clinical assessment of the severity of PsV, as reported by Smith et al., was undertaken using the PASI score according to the British Association of Dermatologists. 20 The PASI score was estimated within each affected area by three clinical signs: erythema (redness), induration (thickness) and desquamation (scaling). Patients were classified according to Schmitt and Wozel by their PASI score as: mild psoriasis (PASI score < 7), moderate psoriasis (PASI score 7–12) or severe psoriasis (PASI score > 12). 21
Sample collection
A volume of 2 ml of venous blood was drawn from each subject into an EDTA tube for complete blood count and flow cytometry to be analysed within 2 h.
Study tools
A complete blood cell count using routine automated KX21N haematology cell counters (Sysmex, Kobe, Japan) was conducted at Al-Zahraa Hospital, Al-Azhar University. Flow cytometry assay was conducted at the Clinical Pathology Department, Al-Zahraa Hospital, Al-Azhar University, using four-colour FACS Calibur (BD Biosciences, San Jose, CA). Cell Quest Pro software (BD Biosciences) was used for data analysis. The compensation setting was established before acquiring the samples using colour-calibrated beads (BD Biosciences; lot no. 5093879). After adjusting the sample count for the acquisition of 50,000 events before analysis, unstained samples were acquired to detect the sample auto-florescence.
Neutrophils, monocytes, B cells (CD19+ve) and T cells (CD3+ve) were examined for the expression of LAIR-1 (CD305) and CR-1(CD35) mean fluorescence intensity (MFI) and number of positive cells.
A 50 µl of fresh blood sample after adjusting of the cell count (1 × 106 PBMCs) was incubated with 5 µl of the following: LAIR-1(CD305) PE-conjugated Ab (BD Biosciences; cat. no. 550811, lot no. 5329747), CR-1 (CD35) FITC-conjugated Ab (Immunotech; Beckman Coulter, Marseille, France; cat. no. IM1836, lot no. 30) and, for T cells, CD3PC5.5-conjugated Ab (Immunotech; Beckman Coulter; cat. no. B49203, lot no. 200025). CD19APC-conjugated Ab was used for B-cell detection (BD Biosciences; cat. no. 345791, lot no. 6281662) for 10 min at room temperature. Then, lysis reagent (BD Biosciences) was added for destruction of RBCs for 8 min before cells were washed with FACS buffer and centrifuged at 500 g.
In terms of the gating strategy of lymphocytes subsets, neutrophils were distinguished according to moderate size and high side scatter (SS), and monocytes were distinguished according to their large size and moderate side scatter on the forward scatter (FS)/SS graph. T lymphocytes were identified with a CD3/CD19 histogram as CD3+ve cells, while B cells were CD19+ve, initially taken from the lymphocyte area on FS/SS. Overlay histograms were done for PsV cases versus controls for comparison (Figure 1).
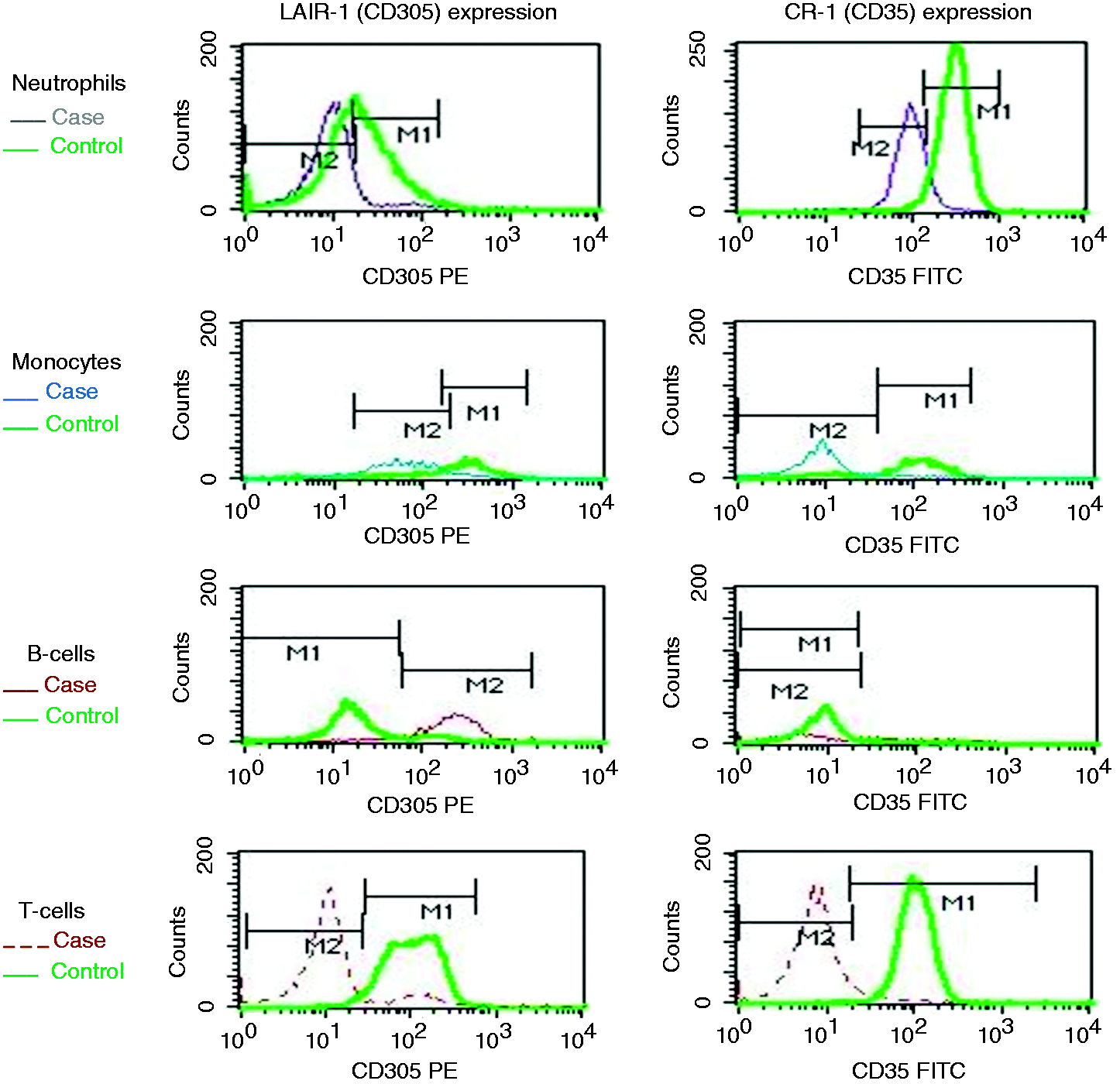
Example of overlay histograms of a Psoriasis vulgaris case versus a control introduced in the same setting. MFI of CD305 (LAIR-1) and CD35 (CR-1) were measured under marker (M) for the corresponding population (neutrophils, monocytes, CD19+ve B cells and CD3+ve T cells). M1 for controls and M2 for cases. CR-1: complement receptor 1; MFI: mean fluorescent intensity; LAIR-1: leukocyte-associated Ig-like receptor-1.
Statistical analysis
Data were coded and entered using IBM SPSS Statistics for Windows v25 (IBM Corp., Armonk, NY). Data were summarised using median, first quartile and third quartile for non-parametric quantitative data, and using frequency (count) and relative frequency (percentage) for categorical data. Comparisons between quantitative variables were done using the non-parametric Kruskal–Wallis and Mann–Whitney tests for comparing categorical data. The chi-square test was performed. Correlations between quantitative variables were done using the Spearman correlation coefficient. Receiver operating characteristic (ROC) curves were constructed, with area under the curve analysis performed to detect the best cut-off value of different parameters for detection of PsV. P Values ≤ 0.05 were considered statistically significant.
Results
The present study included 72: 36 PsV patients and 36 sex- and age-matched healthy controls. PsV patients were further subdivided into two groups: group 1 with a PASI score > 12 (severe cases; n = 16), and group 2 with a PASI score ≤ 12 (mild to moderate cases; n = 20). Demographic and clinical data of the PsV cases are illustrated in Table 1.
Demographic data of Psoriasis vulgaris patients.
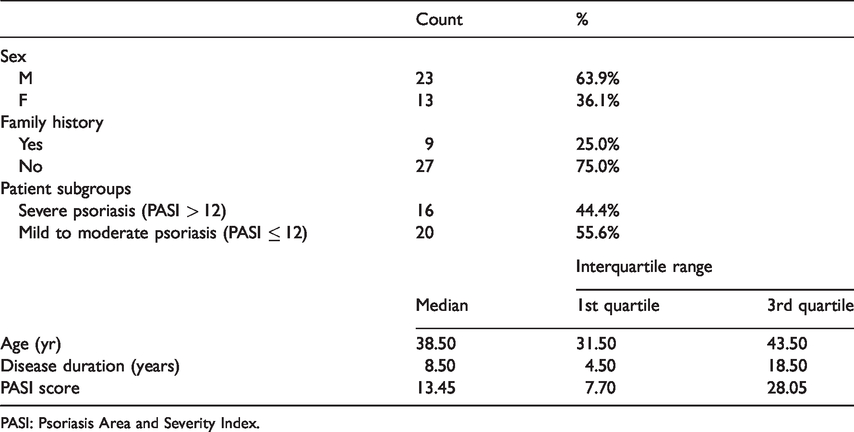
PASI: Psoriasis Area and Severity Index.
Comparisons between PsV cases and controls are illustrated in Table 2. Cases revealed a significant decrease in LAIR-1 MFI on neutrophils and T cells (P < 0.001 and P = 0.003, respectively) and a significant decrease in the percentage of neutrophils expressing LAIR-1 (P = 0.001). B cells showed a significant increase in the percentage of cells expressing both LAIR-1 and LAIR-1 MFI (P < 0.001).
Comparison of the studied markers on leukocyte subsets between PsV patients and controls.
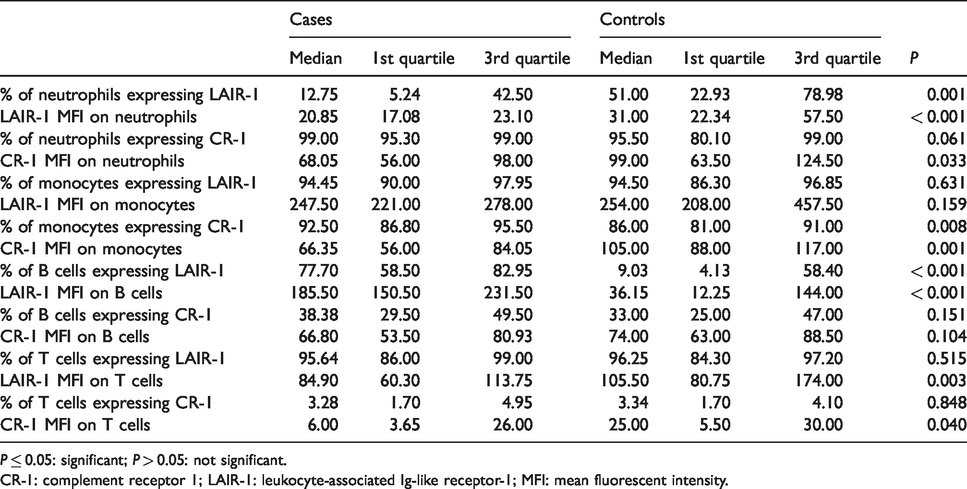
P ≤ 0.05: significant; P > 0.05: not significant.
CR-1: complement receptor 1; LAIR-1: leukocyte-associated Ig-like receptor-1; MFI: mean fluorescent intensity.
The comparison also revealed a significant decrease in CR-1 MFI on neutrophils, monocytes and T cells (P = 0.03, 0.001 and 0.040, respectively). In addition, there was a significant decrease in the percentage of monocytes expressing CR-1 in PsV cases (P = 0.008).
The comparison between PsV subgroups (groups 1 and 2) revealed a significant decrease in the percentage of neutrophils expressing LAIR-1 (P = 0.033) and LAIR-1 MFI on monocytes (P = 0.007) in group 1.
The correlation of the PASI score with the expression of the studied parameters in severe cases (n = 16) revealed a significant negative correlation of the PASI score with the percentage of T lymphocytes expressing CR-1 (r = –0.612; P = 0.012; Figure 2), while no significant correlations were obtained between the PASI score and all studied parameters in all PsV participants (n = 36).
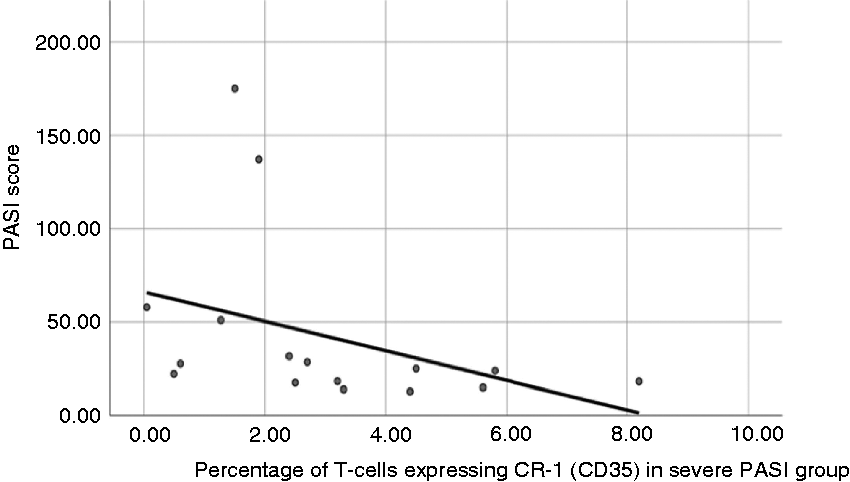
Correlation of PASI score with percentage of T cells expressing CR-1 in severe cases. Correlation of PASI score with percentage of T cells expressing CR-1 in severe cases (n = 16) showed a significant negative correlation (r = –0.612, P = 0.012). PASI: Psoriasis Area and Severity Index.
There was a significant positive correlation of LAIR-1 MFI on neutrophils with neutrophilic CR-1 MFI and monocytic CR1-MFI (r = 0.503, P = 0.002; r = 0.389, P = 0.019, respectively). There was also a significant positive correlation of LAIR-1 MFI on monocytes with monocytic CR1-MFI (r = 0.371, P = 0.026; Figure 3), while no significant correlations were observed in B cells and T cells concerning both study markers with each other.
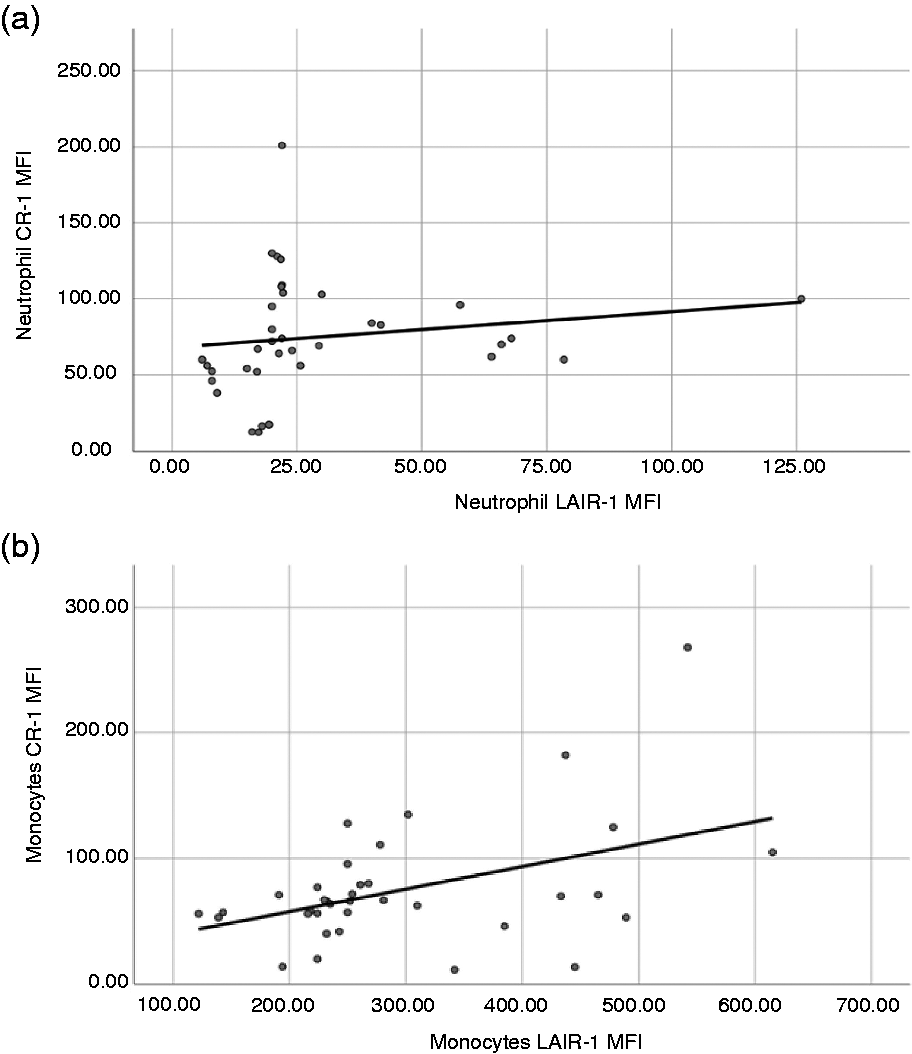
Correlation studies of study markers. (a) A significant positive correlation of neutrophilic LAIR-1 MFI with neutrophilic CR-1 MFI (r = 0.503, P = 0.002) was identified, as well in (b), showing a positive correlation of monocytic LAIR-1 MFI with monocytic CR1-MFI (r = 0.371, P = 0.026).
ROC curves were done to determine the sensitivity and specificity of the studied markers, focusing on their ability to discriminate between cases and healthy controls. The suggested cut-offs are illustrated in Table 3, Figure 4 and Supplemental Figure S1.
ROC curve data of the studied markers.
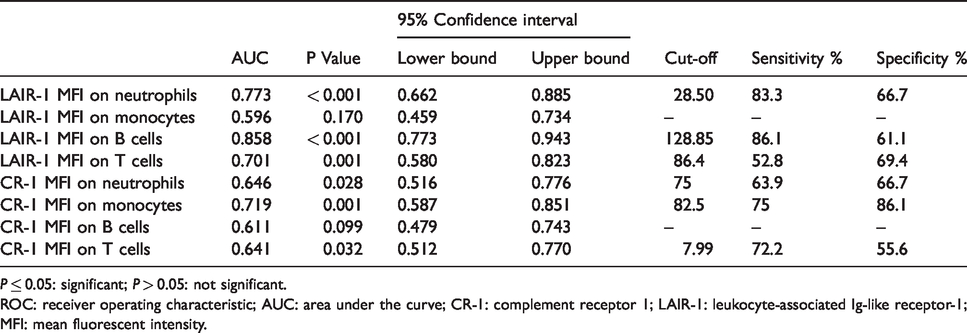
P ≤ 0.05: significant; P > 0.05: not significant.
ROC: receiver operating characteristic; AUC: area under the curve; CR-1: complement receptor 1; LAIR-1: leukocyte-associated Ig-like receptor-1; MFI: mean fluorescent intensity.
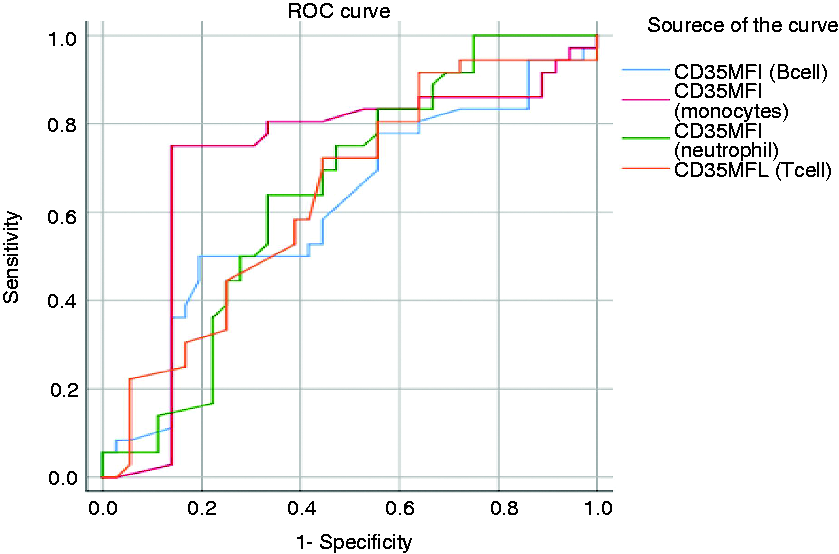
Data of ROC curves for CR-1 MFI in different leukocyte subsets. CR1 MFI on monocytes showed the highest discriminating ability to differentiate Psoriasis vulgaris patients from the control at a cut-off of 82.5. The specificity was 86.1%, while the sensitivity was 75%, with an AUC of 0.719 and P = 0.001. ROC: receiver operating characteristic; AUC: area under the curve.
Discussion
PsV is a disease in which autoimmune and auto-inflammatory processes intersect. Exploring the expression of CR-1 and LAIR-1 on leukocytes in PsV can provide a better understanding of the disease pathology and may help in introducing new avenues for therapy.
In the current study, there was a significant decrease in the percentage of neutrophils expressing LAIR-1 and its MFI in PsV patients when compared to controls (Table 2). Phosphorylated LAIR-1 immuno-tyrosine inhibition motifs serve as docking sites for SH2-domain-containing phosphatases (SHP-1 and SHP-2), which negatively regulate the intracellular signalling pathways required for neutrophil activation. 22 Accordingly, a decline in membrane expression of LAIR-1 would diminish this inhibitory effect, leading to continuous immune activation.
A significant decrease was also noticed in neutrophilic CR-1 MFI in PsV patients. Neutrophils are functionally versatile cells with critical roles in chronic inflammation. 23 Functional defects of neutrophils with down-regulated CR-1 were reported. CR-1 participates in mediating the signals involved in particle uptake in the process of phagocytosis. Accordingly, down-regulation of CR-1 negatively impacts the ability to engulf opsonised pathogens. Also, reduced cellular adhesion was reported with neutrophils with down-regulated CR-1. 24
A significant decrease in the percentage of monocytes expressing CR-1 and CR-1 MFI was detected in PsV patients (Table 2). Antagonising CR-1 inhibits monocytes derived macrophage phagocytosis of pathogens and inhibits particle adherence to phagocytic cells. 25 Dysregulation of complement regulatory proteins was noticed in autoimmune disorders.2627 Oliveira et al. suggested an anti-inflammatory role for CR-1, claiming CR-1 as a potential new therapeutic target. 28
Furthermore, it was reported that CR-1 expression on phagocytes is involved in the binding of C3b/4b-opsonised elements, which are then ingested, preventing the formation of C5 convertase. That would in turn block complement activation and the formation of the membrane attack complex. CR-1 also has decay-accelerating activity and serves as a cofactor for the degradation and inactivation of C3b/C4b by factor I. Thus, CR-1 is considered as a potent inhibitor of complement activation, serving to protect from the inflammatory consequences. 29
CR-1 was reported to be responsible for the clearance of cellular debris by circulating phagocytes, which is important for tissue remodelling and the regeneration needed for the resolution of inflammation. 30 This may explain why it was claimed that analysing the expression of immunomodulatory receptors on circulating phagocytes could provide new diagnostic markers for inflammatory diseases. 31
A significant decrease in LAIR-1 MFI on T cells in PsV cases was obtained when compared to controls (Table 2). This was in line with Zhang et al. who reported that LAIR-1 expression was significantly decreased in T cells in rheumatoid arthritis patients compared to healthy individuals. LAIR-1 modulates cytokine generation in CD4+ T cells, down-regulates IL-2 and IFN-γ production and induces TGF-β. 32
There was also a significant decrease in CR-1 MFI on T cells in PsV cases. Wagner et al. observed that blocking CR-1 inhibited the proliferation of T cells by reducing the synthesis of proliferating cell nuclear Ag (PCNA) and cyclins. This may add a role for CR1 in the pathophysiology of PsV, as PsV is known as a T cell-mediated disease. 33 Török et al. observed that CR-1, when expressed on activated T cells in the presence of IL-2, generated Tregs in the secondary lymphoid organs, where they may interact with B cells. CR-1 ligation inhibits B cell receptor-mediated activation, and differentiation to plasma cells, performing a protective function. 34
Interestingly, B cells showed a significant increase in both the percentage of cells expressing inhibitory LAIR-1 and their LAIR-1 MFI in PsV cases when compared to controls. Martínez-Esparza et al. reported that the cross-linking of LAIR-1 with its ligands inhibited the differentiation and activation of B cells induced by B cell receptor cross-linking. 35 However, Liang et al. demonstrated that adaptive immune responses predominated in mild chronic forms of psoriasis such as PsV, while innate and auto-inflammatory responses dominated in acute severe forms such as pustular psoriasis. 5 This controversy could be explained by the fact that our 36 PsV cases included 16 severe cases with a PASI score > 12, which could explain the extreme inhibition of the B cell adaptive response. 5
The actual inhibition of B cells with the up-regulated membrane-bound LAIR-1 must be carefully studied. Reduction of LAIR-1 expression profiles in other leukocyte subsets in the current study can be associated with elevated levels of sLAIR-1 which antagonise membrane-bound LAIR-1 activity through competition for ligands. 36 Furthermore, experiments demonstrated a role for C1q in modulating LAIR-1 activation. 10 Lastly, it was suggested by Zhang et al. that LAIR-1 may exert different functions on different cells. 37
A significant decrease in the percentage of neutrophils expressing LAIR-1 was observed in the severe cases in group 1 compared to the mild and moderate cases in group 2. A significant decrease was also obtained in monocytic LAIR-1 MFI in group 1 (Table 2). These results are consistent with more defective self-tolerance in severe cases. LAIR-1 suppresses neutrophilic tissue activation and migration. LAIR-1 also acts as a negative regulator of neutrophil-driven inflammation which explains the significant down-regulation on neutrophils in severe cases. 39
Son et al. reported that a reduction in LAIR-1 expression in lupus erythematosus on monocytes was correlated with inflammation. 38 The decline of LAIR-1 diminishes the cross-linking with its ligands. This cross-linking is responsible for the inhibition of inflammatory type I IFN production by monocytes. 35 Type I IFN play an indispensable role in initiating psoriasis through enhancing the development of monocyte-derived DC (moDC), which contributes to Th1 and Th17 differentiation and keratinocyte proliferation. Th1 and Th17 interact with monocytes and instruct monocytes to differentiate into Th1- and Th17-promoting moDCs, leading to the formation of more specialised moDC subsets.4041
Correlation studies in severe cases revealed a significant negative correlation of the PASI score with the percentage of T lymphocytes expressing CR-1 (Figure 2). This could be explained by Török et al., as they demonstrated that the engagement of surface CR-1 expressed on activated CD4+ve T cells augmented Treg proliferation, which suppressed the inflammation induced by Th17 cells. 34 The presence of impaired Tregs in PsV patients was confirmed by Shi et al. 40 These data are in line with Arbore et al. who stated that CR-1 influenced immune modulation and prevented self-damage caused by uncontrolled inflammation, which impact the pathophysiology of chronic inflammatory diseases. 42
There was a significant positive correlation of neutrophilic LAIR-1 MFI with neutrophilic CR-1 MFI and with monocytic CR1-MFI. There was also a significant positive correlation of LAIR-1 MFI on monocytes with CR1-MFI on monocytes (Figure 3). These associated down-regulations of both receptors could be attributed to a single mechanism controlling the proteolytic shedding of receptors from the membrane to create soluble forms. Those soluble forms might act as decoy receptors by competing with membrane-bound receptors for ligand binding, preventing subsequent signal transmission and C1q-dependent clearance of damaged cell which may add to the pathological feature of the disease.
Dual down-regulation of CR-1 and LAIR-1 could also be attributed to combined genetic alterations affecting CR-1 and LAIR-1 genes. Genetic variability was reported to influence CR-1 expression levels and molecular mass. 18 Oliveira et al. reported that the CR-1 haplotype with the major rs6656401*G allele of the Knops blood group was associated with increased susceptibility to Pemphigus foliaceus. 28 Sandri et al. reported that the distinct CR-1 exon 29 variant affected CR-1 expression levels and increased susceptibility to Chagas disease. 19 Kretzschmar et al. and Malik et al. suggested a regulatory role for CR1 polymorphisms on mRNA and sCR1 levels. It was claimed that single nucleotide polymorphisms present in the regulatory and coding regions in CR-1 genes were involved in the down-regulation of CR-1 expression.4344
Camargo et al. also suggested that two LAIR-1 variants (rs56802430 G and rs11084332 C) and one LAIR-2 (rs2287828 T+) were associated with altered mRNA expression levels of the receptors and contributed to differential susceptibility to Pemphigus foliaceus. 45 Previous data suggested the presence of associating genetic variables causing down-regulation of LAIR-1 and CR-1, which could increase susceptibility to PsV.
Tackling CR-1 and LAIR-1 immunomodulators in targeted therapy can open doors in addressing incurable PsV. All given PsV treatments depend on steroids and immunosuppressive drugs known for their devastating side effects. Recently, some advanced biological anti-cytokines were introduced for PsV treatment targeting IL17, IL12 and TNF-α. Although they are safer and more tolerated, they are not concerned with phagocytosis through C1q, which is a crucial process for tissue growth, flagging apoptotic cells, debris clearance and resolution of inflammation. In the current study, neutrophilic and monocytic CR-1 MFI values were found to be down-regulated in PsV cases. De-regulation of CR-1 expression was confirmed to reduce opsonisation-mediated phagocytosis. 24
Moreover, down-regulation of CR-1 MFI on T cells disturbed T lineage differentiation and interrupted the crosstalk between adaptive and innate immunity. 46 Hence, CR-1 MFI stimulation in PsV by targeted therapy could reverse all the inflammatory processes and cut the cycle of auto-Ag production which could modify the course of PsV. The stimulation of LAIR-1 by targeted therapy would also regain its inhibitory protective function that leads to the suppression of the production of inflammatory cytokines and modulates the suppressor T-cell differentiation. 47
Regarding the data of the ROC curves for LAIR-1 MFI and CR-1 MFI in different leukocyte subsets, CR-1 MFI on monocytes showed the highest discriminating ability to differentiate PsV patients from controls (Figure 4). Interestingly, LAIR-1 MFI on B cells showed a different sensitivity and non-satisfactory specificity (Table 3 and Supplemental Figure S1).
Conclusion
Altered leukocyte expression of LAIR-1 and CR-1 is associated with PsV, highlighting a potential role in its pathophysiology which needs more evidence. The study highlights the potential role of LAIR-1 and CR-1 as therapeutic targets for this incurable disease. Moreover, down-regulation of the percentage of T cells expressing CR-1 could serve as an indicator of PsV severity. Marked down-regulation of LAIR-1 in severe cases suggests a role in disease progression. Down-regulation of CR-1 MFI on monocytes can serve as a diagnostic biomarker for PsV.
Our study sheds light on a novel regulatory mechanism involved in leukocyte-induced inflammation in PsV. Further studies are recommended to investigate cellular activation and ligand availability in order to clarify the reasons for the discrepancy in LAIR-1 expression of B cells in PsV, explore the effect of reduced CR-1 MF1 and LAIR-1 MFI on neutrophils functionality in PsV and measure plasma sLAIR-1 and sCR-1 to correlate their levels in plasma with the leukocyte surface expression. However, this expansion was not possible in the current study due to financial limitations. Still, one of the initial findings in the current study was proving the altered expression of LAIR-1 and CR-1 on leukocytes with the involvement of circulating phagocytic cells rather than being enclosed by lymphocytes, as seen in most previous research.
Supplemental Material
sj-pdf-1-ini-10.1177_1753425920942570 - Supplemental material for Immunoregulatory complement receptor-1 and leukocyte-associated Ig-like receptor-1 expression on leukocytes in Psoriasis vulgaris
Supplemental material, sj-pdf-1-ini-10.1177_1753425920942570 for Immunoregulatory complement receptor-1 and leukocyte-associated Ig-like receptor-1 expression on leukocytes in Psoriasis vulgaris by Reham Hammad, Mervat Hamdino, Asmaa M El-Nasser, Ali Sobhy, Noha Abdel-Rahman Eldesoky, Alya Mohammad Mashaal and Hany F Ali in Innate Immunity
Supplemental Material
sj-jpg-2-ini-10.1177_1753425920942570 - Supplemental material for Immunoregulatory complement receptor-1 and leukocyte-associated Ig-like receptor-1 expression on leukocytes in Psoriasis vulgaris
Supplemental material, sj-jpg-2-ini-10.1177_1753425920942570 for Immunoregulatory complement receptor-1 and leukocyte-associated Ig-like receptor-1 expression on leukocytes in Psoriasis vulgaris by Reham Hammad, Mervat Hamdino, Asmaa M El-Nasser, Ali Sobhy, Noha Abdel-Rahman Eldesoky, Alya Mohammad Mashaal and Hany F Ali in Innate Immunity
Footnotes
Acknowledgements
We thank Dr Claude Lambert, International Federation of Clinical Chemistry and Laboratory medicine (IFCC), Flow Cytometry Working Group Coordinator, Director of Immunology Laboratory, Saint-Etienne University Hospital, France, for continuous support and guidance through development of the proposal.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
