Abstract
C1q is a versatile innate immune molecule that serves as the initiation subcomponent of the classical complement pathway. In addition, it is also a potent pattern recognition molecule, the versatility of which has fuelled its functional flexibility. C1q recognises an array of self, non-self and altered-self ligands. The broad-spectrum ligand-binding potential of C1q is facilitated by the modular organisation of the heterotrimeric globular head region, its ability to change its conformation in a very subtle way, and the manner in which this ancient molecule appears to have evolved to deal with the different types of ligands. Over recent years, molecules that resemble C1q have been put together to form the C1q family. In this review, we briefly summarise complement-dependent and complement-independent functions of C1q, its cognate receptors and key members of the rapidly growing C1q family.
Introduction
C1q, the first subcomponent of the classical complement pathway, is a 460-kDa protein composed of 18 polypeptide chains (6A, 6B and 6C). Each of these individual modules are composed of: (i) a short N-terminal region consisting of a half-cysteine residue that is involved in formation of inter-chain disulphide bonds; (ii) collagen-like region (CLR) consisting of ∼81 residues: and (iii) a C-terminal globular region, also called the gC1q domain, of ∼135 residues.
1
The CLR sequences within the A, B and C chains associate to give rise to a triple-helical structural unit of the composition ABC–CBA, which is held together by both covalent and non-covalent bonds. This subunit structure eventually yields a hexameric C1q molecule.
2
–
4
C1q is a versatile pattern recognition molecule that binds to a number of ligands and modulates various biological and cellular responses (Figure 1). This versatility is conferred due to the modular organisation of the individual globular heads (A, B and C).
5
In addition, the two domains, the gC1q domain and CLR, have their cognate receptor molecules (gC1qR and calreticulin, CRT, respectively) on a number of immune and non-immune cells that has also helped C1q in broadening its physiological functions beyond complement activation.
C1q-ligand interactions: the C1q molecule is divided into two domains namely, the gC1q (globular head region) and the CLR (collagen-like region). The modularity of the individual heads of C1q is also briefly explained illustratively in this diagram.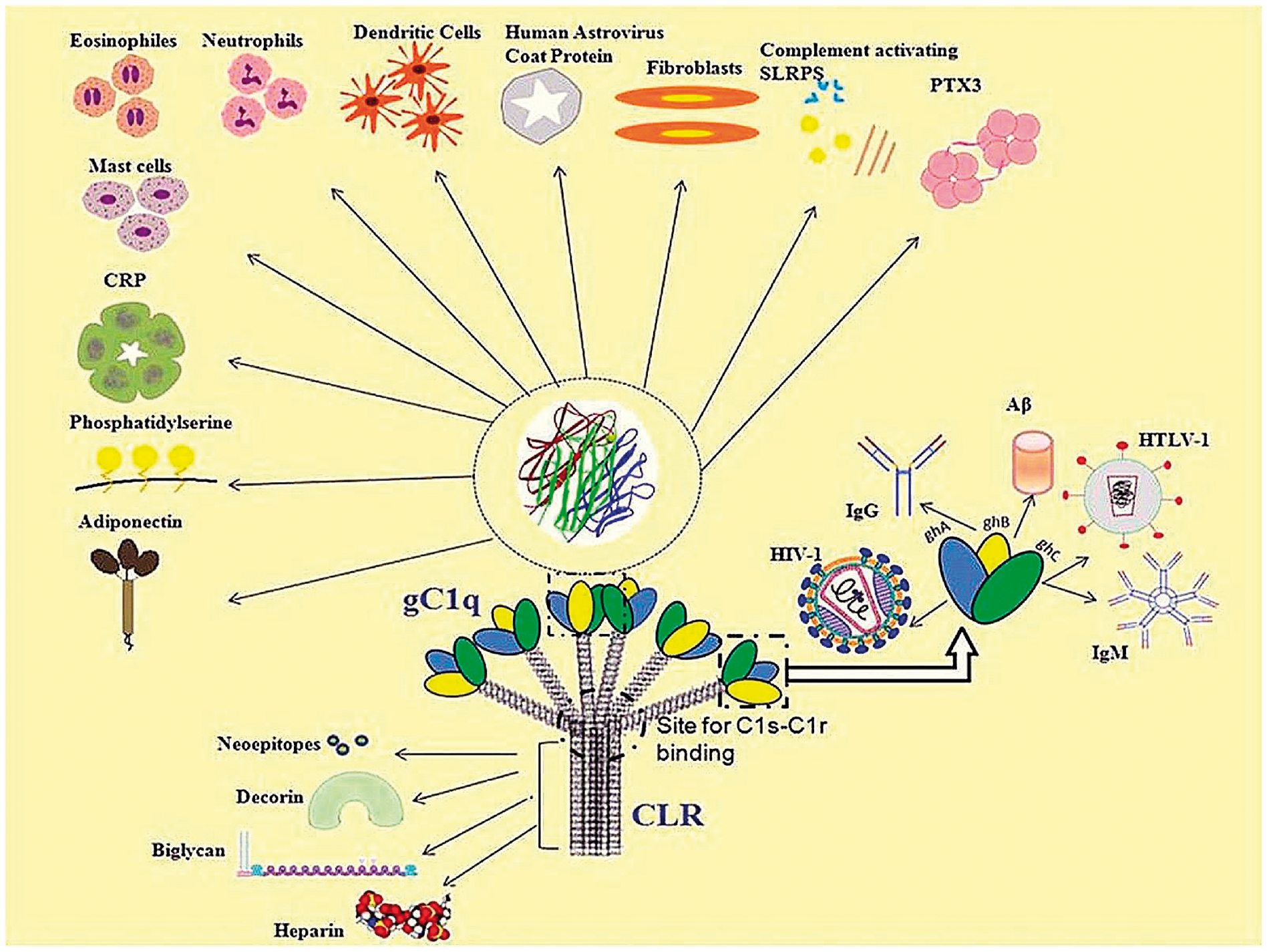
Functions of C1q that are dependent on activation of classical complement pathway
Complement activating functions of C1q


Complement-independent functions of C1q
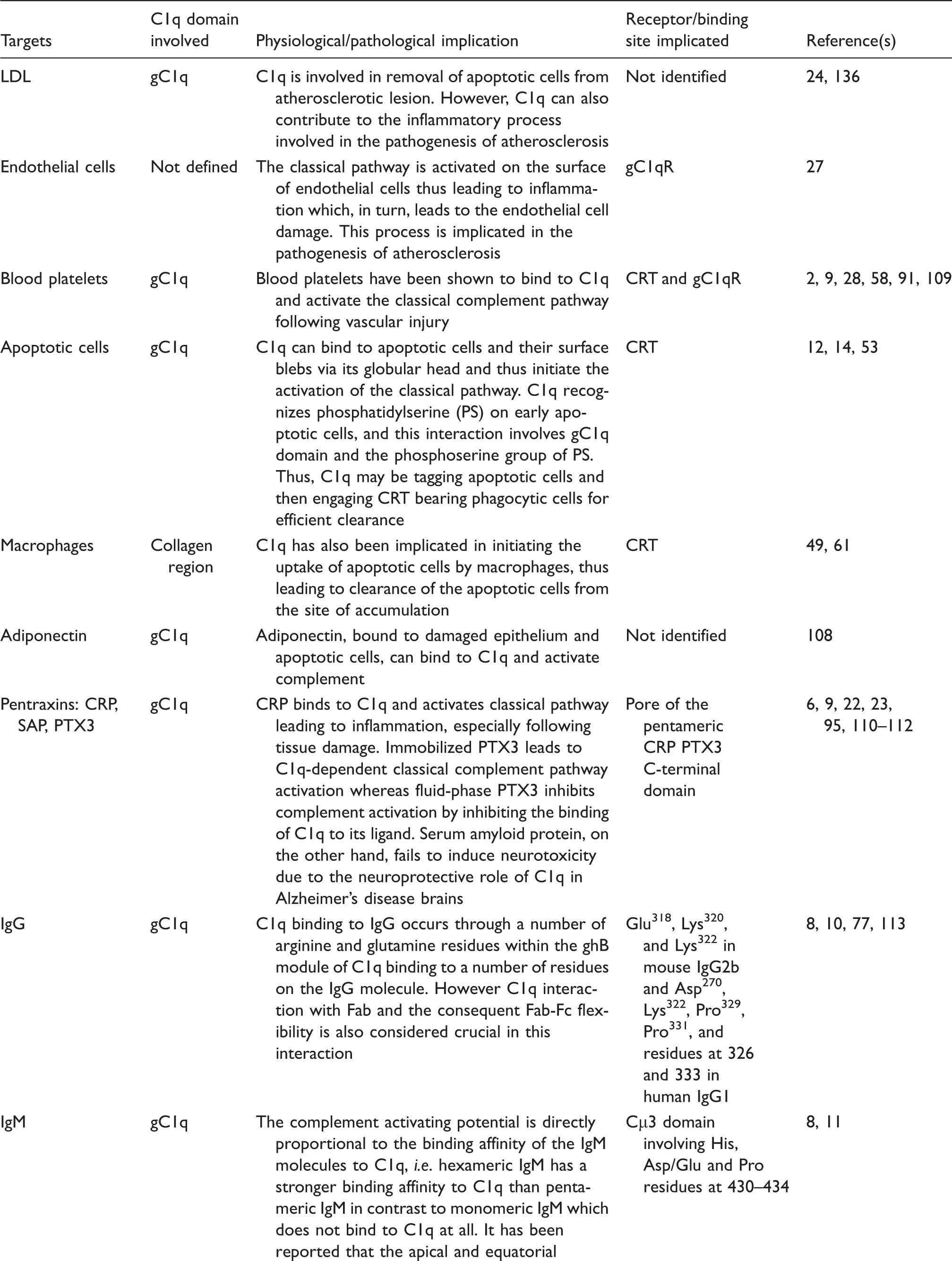
Along with binding to IgG and activating complement, C1q is also involved in various cell processes such as apoptosis and coagulation. C1q has the potential to bind to apoptotic cells as well as the surface blebs on these cells and activate complement which then leads to the removal of the apoptotic cells. 12 – 15 In late apoptotic cells, C1q binds to IgM which then opsonises the apoptotic cells and activates classical complement pathway. 16 Clearance of apoptotic cells is crucial in maintaining normal homeostasis as the inability to do so leads to accumulation of the apoptotic cells, which is a hallmark manifestation of the C1q-defiency disorder – systemic lupus erythematosus (SLE).
A few proteins in the short leucine-rich glycoproteins (SLRPs) subclass II and IV exhibit the potential to activate the classical complement pathway by binding to C1q. These proteins include fibromodulin (which is involved in assembly of the extracellular matrix), 17 osteoadherin, and chondroadherin (proteins found in the territorial matrix of articular cartilage). Fibromodulin and osteoadherin bind to the gC1q with a very high affinity when compared to chondroadherin with binds C1q with a lesser affinity and hence exhibits lesser extent of complement activation. It has been suggested that, in diseases such as rheumatoid arthritis and osteoarthritis, the activation might be driven by the speculative presence of free floating complement activating SLRPs binding to serum C1q. 18
Complement activation is one of the major factors causing inflammation in pathological conditions such as autoimmune and neurodegenerative diseases, as well as atherosclerosis. C-Reactive protein is an acute-phase reactant protein that is a strong activator of the classical complement pathway and is thus an important clinical marker of inflammation, especially in cardiovascular diseases and cancer.
19
–
21
Binding of CRP to its respective ligands (such as phosphorylcholine; PC) exposes its binding sites for C1q,
22
and hence subsequent complement activation.
23
This process contributes to inflammation leading to tissue damage in atherosclerosis as well as cancer to some extent. Several studies in recent years have suggested the role of C1q in activating the complement system in atherosclerosis24,25 and thus contributing to the inflammation observed in the disease. C1q recognises deposited low density lipoprotein (LDL) on atherosclerotic lesions and activates complement which consequently leads to inflammation.
25
However,
Complement activation that follows binding of C1q invariably leads to inflammation. However, inflammation is not always harmful, as it leads to host defence against pathogens and several other threats. For instance, C1q can bind to outer membrane proteins of Gram-negative bacteria and lead to complement susceptibility of the invading pathogen. 30 But some pathogens, like the human astrovirus in particular, have special coat proteins that bind to C1q and inhibit the complement activation and hence evade the host immune response.31,32 Failure to activate the complement pathway can also be protective; for example, low molecular mass heparin, inhibits complement activation in pregnancy and hence averts pregnancy loss. 33
Functions of C1q that are not dependent on complement activation
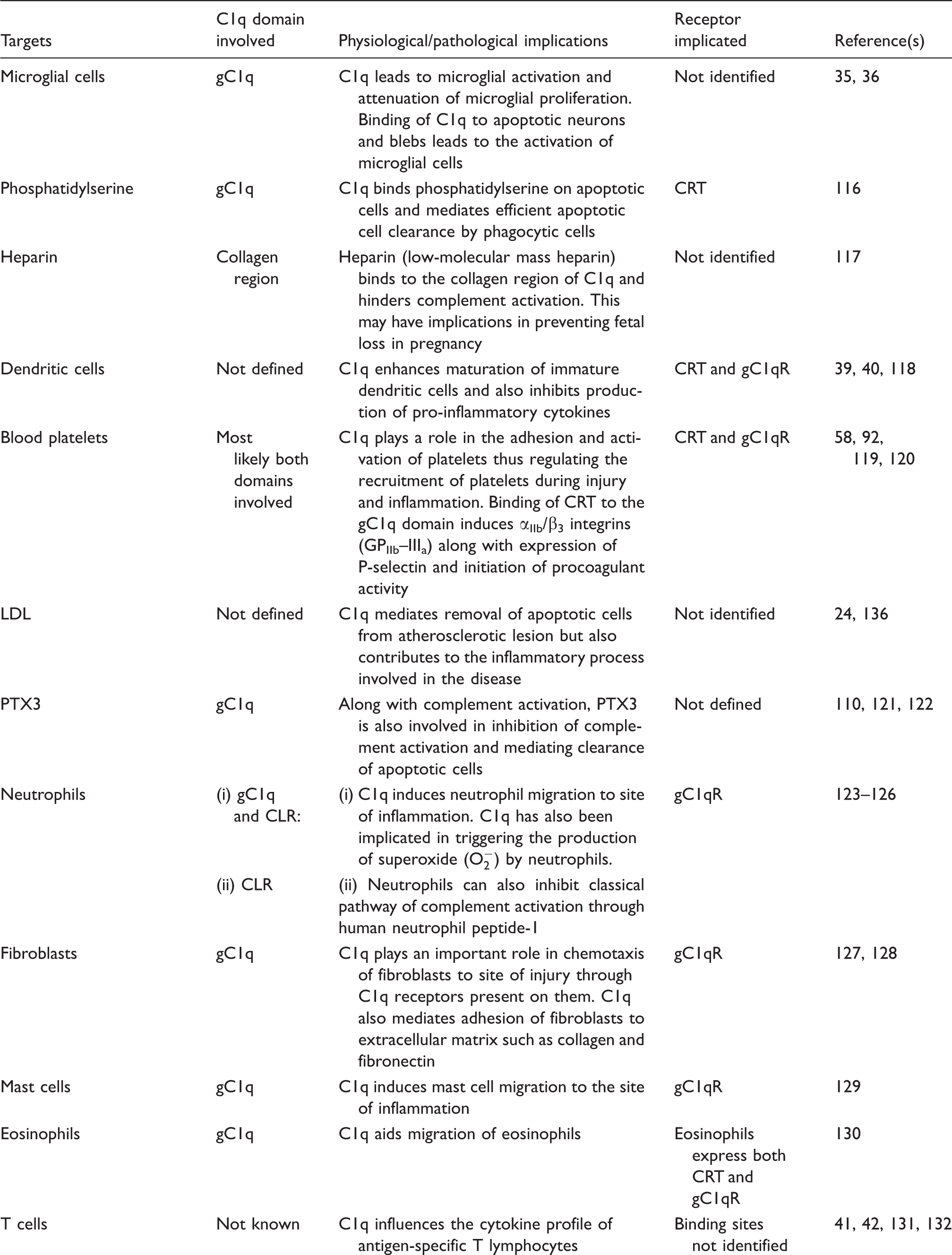
Although C1q is a complement initiating molecule, it is also engaged in an array of processes that is completely independent of the complement activation. 34 This is evident in the case of microglia activation in the central nervous system (CNS). Microglial cells are the phagocytic cells of the CNS and are found in abundance in neurodegenerative diseases that have impaired blood–brain barrier like Alzheimer’s disease. Microglial cells try to dispose of, in this case, the deposited amyloid peptides and tangles. These phagocytic cells are also known to synthesize C1q. However, the activated microglia cells have been shown to be highly dependent on extrinsic C1q for their activation. 35 Extrinsic C1q leads to activation of microglia cells which, in turn, attempts to phagocytose the opsonised targets, for example; apoptotic neurons and amyloid peptides. 36 The locally synthesized C1q has been suggested to act in an autocrine/paracrine manner to control microglial activation and recruitment. 35 C1q also down-regulates the production of the pro-inflammatory cytokines IL-1α, IL-1β, IL-6 and TNF-α. 36 Thus, within the CNS, C1q has a neuroprotective role in the form of activating microglial cells in order to ingest apoptotic neurons and by suppressing pro-inflammatory cytokine production. On the other hand, C1q also plays a neurodegenerative role by triggering complement activation in the CNS. 37 C1q is also implicated in non-inflammatory synapse pruning during fetal brain development.34,38
C1q is associated in the maturation of dendritic cells (DCs), and immature DCs are also good sources of C1q on their own, an association that is similar to that of C1q and microglial cells and macrophages.34,39,40 C1q is also involved in modulating T-cell and B-cell activity. C1q regulates T-cell proliferation and also influences the cytokine profile of antigen-specific T-cells.41,42 This is proven by defective production of IFN-γ by T cells in C1q-deficient mice. C1q stimulates the production of IgG by B-lymphocytes and also plays a role in the negative selection of autoreactive B cells. 43 Defective B-cell tolerance checkpoints have been observed in individuals suffering from SLE. 44 C1q is also involved in the migration of eosinophils, mast cells, neutrophils and fibroblasts, 34 thus acting as a potent chemotactic molecule (Table 2). Receptors for gC1q and CLR have been localised on the surface of these immune cells.
C1q binds to various extracellular matrix proteins such as decorin, biglycan, lumican, laminin and fibronectin. C1q binds strongly to decorin and biglycan, which are leucine-rich proteoglycans involved in matrix assembly and bone mineralisation, respectively.45,46 The binding of these extracellular matrix proteins hampers activation of complement cascade. 46 C1q also binds to lumican, laminin and fibronectin with a lower affinity.18,47,48 This binding has been implied in deposition and retention of immune complexes in the basement membrane. However, an interesting feature of this interaction is the non-complement dependent binding of C1q to extracellular matrix proteins, which probably involves the CLR domain. 18 Wherever the interaction involves the gC1q domain, complement activation mostly results. 18 The nature of C1q involvement into non-complement associated functions is extended to pregnancy and development too. 34
Apoptotic cell clearance is mediated by C1q in a complement-dependent and complement-independent manner. The non-complement activating way of clearing apoptotic cells including apoptotic neurons and neuronal blebs, is through C1q binding to CRT on macrophages which, in turn, leads to apoptotic cell phagocytosis through CD91-induced macropinocytosis by phagocytic cells like macrophages and microglial cells.36,49,50 C1q also has the ability to recognise self DNA on apoptotic cells, thus leading to clearance by macrophages. 51 Further research into the ligand binding capacity of C1q can lead to a better understanding of the process of inflammation along with many more other cellular processes including apoptosis and cell adhesion.
C1q receptors
C1q receptors comprise a diverse group of proteins, cell surface and intracellular, that recognise and bind to C1q and either trigger or enhance various cellular responses. gC1qR (previously known as p33 or C1q BP) 52 and calreticulin (CRT; previously known as cC1qR) are two well-studied and most ubiquitous C1q receptors involved in mediating biological processes.53,54 The 33-kDa protein gC1qR has been shown to recognise and bind to the gC1q domain of C1q with high affinity interactions 52 and is found in the cytoplasm, especially mitochondria and also on fibroblasts, neutrophils and endothelial cells, whereas the 60-kDa CRT interacts with the collagenous region of C1q with a lower binding affinity. 55
In terms of secretion, different studies have challenged various findings of how gC1qR is secreted. Peterson et al. 56 demonstrated that gC1qR is secreted in a soluble form from cytoplasmic vesicles on the plasma membrane of Raji cells and peripheral blood lymphocytes. However, yet another study 57 has shown that gC1qR is localised in the vesicular compartment of endothelial cells concluding that its primary location is intracellular with only a minority located on the surface. Along with binding to C1q, gC1qR has also been shown to bind to other various plasma proteins such as thrombin, prothrombin and vitronectin, respectively. 57 Although expressed on the cell surface, both CRT and gC1qR interact with CD91 and α2β3-integrin to allow transmission of signals to the cell surface. 58 A recent study reports that gC1qR on the surface of CD4+ cells can also bind to the 3S motif of the HIV gp41 and thus play a role in the PI3K activation that leads to NKp44L translocation to the plasma membrane. 59 This, in turn, can render uninfected bystander cells susceptible to lysis by natural killer cells which can lead to the CD4 depletion observed in HIV-1 infection. The observations are interesting as C1q is also seen to bind to the HIV gp41 motif between residues 601–620 and the 3S motif that gC1qR binds is located between 618–623.5,59 However, binding of C1q leads to activation of the classical complement pathway and binding of gC1qR leads to PI3K activation. In addition to being a receptor for C1q and recognising a range of bacterial and viral pathogens, gC1qR is also seen offering a platform for variety of pathogens. 60
CRT, being able to bind to collagen-like structures, is also referred to as the ‘collectin receptor’ due to its ability to bind to the collagen-like regions of various collectin, such as mannose-binding lectin (MBL), surfactant protein (SP)-A and SP-D.54,61 It is an intracellular expressed protein located in the lumen of the endoplasmic reticulum and belongs to the family of heat shock proteins, which are the most abundant soluble intracellular proteins. Calreticulin consists of a globular N-domain with carbohydrate and polypeptide binding sites along with the C1q binding sites, a P-domain and a C-terminal domain that has a high binding specificity to Ca2+. 62 The interactions between CRT and C1q are crucial in apoptotic cell clearance via macrophages with C1q opsonizing on the surface of apoptotic cells and CRT linked to CD91 over the phagocyte cell surface. 63
The chemotactic properties of gC1qR and CRT have also been observed. Dendritic cells treated with anti-gC1qR and anti-CRT antibodies show reduced chemotactic responses to C1q. 64 Dendritic cells treated with anti-cC1qR (CRT) antibodies completely abolish DC chemotaxis, whereas anti-gC1qR treatment reduces cell migration. This suggests that each of these receptors hold different chemotactic properties.
C1qRp also called CD93, a type I membrane glycoprotein, is a speculative C1q receptor involved in elimination of pathogens and immune complexes by phagocytosis. 65 This particular protein binds to the CLR of C1q as well as collectins, such as MBL and SP-A, 66 and is found on myeloid cells, cultured endothelial cells and platelets. 67 However, this receptor is not abundant on lymphocytes, fibroblasts and smooth muscle cells, which suggests that C1qRp may not have a versatile role as a C1q receptor and is of limited significance. The ability of C1qRp to bind to C1q has been speculated by McGreal et al., 68 who demonstrated that cells infected with C1qRp failed to bind to C1q, and that C1qRp is involved in C1q-mediated enhancement of phagocyte function by monocytes, as a co-receptor rather than direct binding. Thus, C1qRp does not seem to bind directly to C1q under physiological conditions. C1qRp has recently been renamed as CD93 and has been recognised as a molecule involved in cellular adhesion. This was demonstrated when engineered C1qRp–Fc chimera intact with the C domain of C1qRp was found to bind to vascular endothelial cells of inflamed human tonsil, suggesting evidence of a C1qRp ligand, a feature of which is true to a cellular adhesion molecule. 68
CR1 is another protein that acts as a C1q receptor with a lower binding affinity and is involved in targeting immune complexes (IC). 69 This transmembrane protein is expressed on erythrocytes, monocytes, neutrophils and lymphocytes and serves as a binding protein by attaching to IC bound by C3b. C3b enhances opsonisation of IC by attaching itself to receptors on macrophages and neutrophils making them more susceptible to CR1 for clearance. 70 C1q, C3b and C4b all serve as opsonins of the complement system. Thus, CR1 binds to all the three molecules. 71 CR1 is targeted to the collagen region of C1q, and its cellular functions differ depending upon which cell it is expressed on. 72 For instance, CR1 on B-cells plays a role in cellular activation, 73 whereas its appearance on erythrocytes play a role in the cleavage of C3b and C4b, initiating the activation of the complement cascade. 74 CR1 present on phagocyte cells is involved in mediating phagocytosis of complement pathogens. 75 The role of CR1 in mediating IC elimination through C1q binding has, however, been challenged as it failed to uptake IC for removal in patients deficient for C3b or C4b fragments, which clearly states that the interaction between CR1 and C1q is not vital in complement-mediated clearance of ICs. 76
A recent study has also supported evidence for the possibility of CD91 on its own (α2-macroglobulin receptor, or low-density lipoprotein receptor-related protein) also being a receptor of C1q through direct recognition of C1q thus playing a role in the clearance of C1q bound material such as apoptotic cells. 77
C1q family proteins
Over 30 proteins, both collagenous and non-collagenous, have been identified to contain the gC1q signature domain and hence have collectively been termed as the C1q family.5,78,79 Some members of this family also exhibit the typical N-terminal secretion signal peptide and collagen triple-helical repeats. 80 All proteins of the C1q family, except precerebellin and multimerin, show the gC1q domain located at the C-terminus of the collagen domain. As all the proteins possess a gC1q domain, analysis has shown sequence similarity between these proteins to vary from 21% (shown between C1qA and MMRN1) and 96% (shown between C1QTNF9 and LOC38791). Eight invariable residues are conserved in all the members, which are crucial for maintenance of the structural integrity of the gC1q domain.
The C1q family is further divided into two subgroups: a larger family that consists of C1q-like and cerebellin-like proteins, and smaller family consisting of EMILINs and multimerins. 79 C1q-like proteins include C1QA, C1QB, C1QC, COL8A1, COL8A2, COL10A1, C1QL1, C1QL2, C1QL3, C1QL4 and adiponectin. Cerebellin-like proteins of this family include Cbln1, Cbln2, Cbln3 and Cbln4.78,79 Adiponectin is a C1q family protein expressed in adipose tissue and low levels of this protein have been reported to be associated with the development of type 2 diabetes. 81 Adiponectin has been widely studied due to its implications in several other diseases including hypertension and atherosclerosis. 82 Cbln1, Cbln2, Cbln3 and Cbln4 are all members of the cerebellin-like group of the C1q family as they are expressed in the CNS system. 83 Cbln1 plays a role in forming and stabilising synaptic contacts as well as controlling the postsynaptic endocytosis pathway 84 whereas Cbln2–4 are expressed in the regions of developing and mature brains. The C1q-like subgroup is comprised of C1QL1, and C1QL2, C1QL3 and C1QL4. C1QL3 is expressed in glial cells and is similar to another C1q family protein ‘C1q-related factor’ (C1RF). This protein is highly localized in the regions of the brain engaged in motor function such as the accessory olivary nucleus, the pons and the red nucleus. 85 The collagens, COL8A1, COL8A2 and COL10A1, also belong to the C1q-like subgroup. COL8A1 and COL8A2 encode collagen V11 and are involved in maintaining the integrity of blood vessels and vascular tissue development. 86
Multimerin 1 and 2 are C1q domain family members found on endothelial cells. Multimerin 1 is a large soluble protein stored in platelets α-granules and endothelial cell bodies; 87 once released, it binds to the cell surface of platelets, endothelial cells and the extracellular matrix where it plays a role in platelet factor storage and stability. 88 Multimerin 2 has been found to play a role in vasculogenesis, angiogenesis and cell matrix adhesion.
EMILIN-1 is an extracellular matrix glycoprotein that is found in abundance in tissues that are rich in elastin. EMILIN-2 is involved in the assembly of components of the basement membrane. EMILIN-1 is also involved in cell adhesion,
The nCLP2 is a novel C1q family protein that is expressed in the CNS and is suggested to play a role in synaptic function and maintenance of the CNS.
83
Yet another recent study identified and characterized AmphiC1q, a primitive C1q molecule from
A group of proteins registered as C1q and TNF-related proteins (CTRP1–9) have been recently shown to have a diverse range of functions that appear to act as link between the prototypical functions of C1q and TNF members. Most of these functions have their origin in the adipose tissue (adiponectin paralogs).79,92 CTRP1, as an adipokine, stimulates aldosterone production and thus may have implications in obesity-associated metabolic alterations. 93 CTRP-3 has been assigned functions ranging from angiogenesis, regulation of adipokine secretion, and smooth cell proliferation. 94 – 97 CTRP9 has recently been shown to reduce glucose levels in mice. 98 Further identification of functions of CTRPs may reveal their level of influence on adipose tissue mediated metabolic and inflammatory processes.
The crystal structure of the gC1q domain of adiponectin/ACRP30 revealed a symmetrical trimer of β-sandwich subunits, each having a 10-strand jelly-roll-folding topology. 99 This structural scaffold is also present in the conserved C-terminal TNF homology domain within the members of the TNF family (reviewed by Kishore et al. 78 ). Thus, the existence of super-imposable β-strands, their identical positions and lengths, similar gene structures has led to the foundation of a C1q–TNF superfamily that probably diverged from an evolutionarily conserved and common precursor molecule. Curiously, the jelly-roll structural scaffold present within C1q–TNF superfamily resembles capsid proteins of picornaviruses including foot-and-mouth and poliovirus. It is speculated that C1q and TNF superfamily members may have originated via horizontal gene capture from a primordial viral genome.
Evolution of C1q and its putative lectin activity
The evolution of the human complement system has long been a subject of discussion and debate. All vertebrates excluding the agnathans share a similar complement system. The classical complement pathway has been suggested to have evolved from the lectin pathway although it adapted to become an immunoglobulin recognising system with the advent of the adaptive immunity in the evolutionary history. In jawless fish, especially lamprey, the only complement systems present appear to be the alternative and lectin pathways, along with a total lack of known major components of the adaptive immunity. In lower vertebrates and invertebrates, the role of C1q as a classical complement initiation molecule is challenged as there is no classical pathway present in these organisms. However, a C1q-like molecule has been discovered with the ability to participate actively in the lectin pathway. Lamprey C1q has 35% similarity to mammalian C1q at the amino acid level and exhibits lectin activity,
Conclusions and perspectives
C1q is a versatile charge pattern recognition molecule of innate immunity that has also evolved to link innate and adaptive immunity via its ability to bind target bound IgG and IgM and activate classical pathway of complement activation. C1q binds to a diverse range of self, non-self and altered-self ligands in an Ig-dependent and Ig-independent manner. Studies in the last decade have used a variety of strategies to explain the structural and functional basis of the versatility of C1q molecule. These include understanding the modular nature of heterotrimeric organisation of the gC1q domain via structure–function and crystallographic studies, mutational studies using individually expressed globular head regions (ghA, ghB and ghC), and molecular modelling using a selected range of C1q ligands. Although there is limited evidence to suggest that versatility of ligand recognition by the gC1q domain is also facilitated by flexibility within the three chains, the apex and side chains and their scattered surface charge residues appear to dictate recognition and avidity of interactions. Since C1q has adapted to deal with a range of ligands, a number of its functions are not defined by its ability to trigger complement activation. Again, the range of non-complement related functions is so broad that C1q can act as a generic prototypical molecule for the rest of the C1q family. More than 30 proteins containing a gC1q signature domain have been identified in the human genome hinting at gene duplication and functional multiplications. The amino acid similarity (ranging between 20–96%) between family members is interesting in view of the fact that up to eight invariant residues are highly conserved. These residues, however, are central to structural maintenance of the gC1q domains, highlighting the importance of jellyroll topology throughout evolution. The most pertinent question and dilemma associated with C1q and its functions remains the receptor aspects of this molecule. CRT/CD91 complex, gC1qR, CR1,
Footnotes
Abbreviations
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. UK is supported by BRIEF and Brunel University Infrastructure and strategic funding.
Acknowledgements
We thank Dr Janez Ferluga for interesting discussions, Asif Qaseem for help at the proof stage of the manuscript.
