Abstract
The cell envelope of pathogenic mycobacteria interfaces with the host. As such, the interaction of bacterial products localized at or released from the cell surface with the host’s immune system can determine the fate of the bacterium in its host. In this study, the effects of three different types of Mycobacterium bovis cell envelope fractions—purified protein derivative, total cell wall lipids and culture supernatant and surface extract—on bovine dendritic cells were assessed. We found that the culture supernatant and surface extract fraction induced little to no production of the pro-inflammatory cytokines TNF-α and IL-12 in bovine dendritic cells. Moreover, this muted response was associated with poor activation of ERK and NF-κB, both of which are critical for the pro-inflammatory response. Furthermore, culture supernatant and surface extract treatment increased the expression of suppressor of cytokine signaling 1 and 3, both of which are negative regulators of pro-inflammatory signaling, in bovine dendritic cells. These observations taken together suggest the M. bovis culture supernatant and surface extract fraction contain immunomodulatory molecules that may aid in M. bovis pathogenesis.
Keywords
Introduction
Bovine tuberculosis (bTB) imposes a significant financial burden on the global cattle industry through loss of productivity and in disease control costs. 1 The causative agent Mycobacterium bovis can also infect people and as such poses a serious risk to public health. 2 Although considerable effort is being spent on the research and development of vaccines for bTB disease prevention and control, a licensed vaccine for use in cattle has yet to emerge. The live attenuated M. bovis bacillus Calmette-Guerin (BCG) TB vaccine that is used to immunize people has been shown to confer protection in cattle.3,4 However, BCG is not used in cattle because the tuberculin skin test routinely used to diagnose bTB in the field does not distinguish BCG-vaccinated animals from M. bovis-infected animals. 5 Therefore, a better understanding of the effects of M. bovis cell surface-localized and secreted biomolecules on the bovine immune system will help inform bTB vaccine and diagnostics development.
Members of the M. tuberculosis (M. tb) complex, which include M. tb the human-adapted agent of TB and M. bovis, have on their cell surfaces a complex mixture of proteins, polar and apolar lipids, glycolipids and glycans with diverse biological properties.6–8 Indeed, many of these biomolecules play crucial roles in host-pathogen interactions and TB immunopathogenesis.7–11 Furthermore, M. bovis-derived cell wall polar lipids compared to apolar lipids differentially stimulate pro-inflammatoty responses in bovine monocytes and monocyte-derived dendritic cells (DCs). 12 Thus M. bovis antigens separated into fractions may be a useful approach to identify immunostimulatory Ags from non-stimulatory or perhaps even immunosuppressive Ags. To our knowledge, the effects of distinct M. bovis-derived protein, lipid and glycan-enriched fractions on bovine immune cells have never been evaluated in a head-to-head comparison. Therefore, here we sought to assess the activation of bovine DCs (bDCs) on treatment with three very different M. bovis-derived antigen fractions—bovine purified protein derivative (bPPD) enriched in polypeptides, 13 total cell wall lipid (CWL) enriched in lipids and glycolipids,14,15 and culture supernatant and surface extract (CSSE), which is enriched in easily shed glycans as well as glycolipids.16,17 We focused on bDCs because they are key professional APCs that on activation can prime and direct the immune activity of naïve and memory T cells essential for TB control.18,19 We found that M. bovis CSSE-treated bDCs exhibited little to no activation. The CWL fraction, meanwhile, induced more activation of bDCs than CSSE but less than bPPD and the LPS control. Furthermore, the muted response of CSSE was found to be associated with decreased ERK and NF-κB activation and increased expression of suppressors of cytokine signaling 1 and 3 (SOCS1 and 3).
Materials and methods
Preparation or source of M. bovis cell envelope Ag fractions
bPPD was purchased from Prionics AG (ThermoFisher Scientific Inc.). The production of bPPD, which is a purified mixture of proteins prepared from the culture filtrate of heat-killed M. bovis grown in synthetic medium, is described elsewhere. 13 bPPD was diluted in PBS before use.
Total CWL was obtained from BEI Resources (https://www.beiresources.org/). The production of total CWL, which is a complex mixture of polar and apolar lipids and glycolipids, has been described elsewhere.14,15 Briefly, M. bovis AF2122/97 was grown in glycerol-alanine-salts medium to late-log phase, pelleted and washed with PBS, gamma irradiated and lyophilized. Total lipids were then extracted from the dried cells with chloroform/methanol (2:1). Water-soluble molecules were then removed from the organic extract by biphasic partitioning in water. The organic phase enriched with total CWL was collected, dried and weighed. Before use, the CWL was resuspended in ethanol and diluted further in PBS.
CSSE, which has been shown to contain a complex mix of extracellular glycans and glycolipids, was enriched from the culture filtrates of M. bovis AF2122/97 grown in Sautons liquid media.16,17,20 Briefly, to 0.8 volumes of cell-free M. bovis culture filtrate, two volumes of methanol and one volume of chloroform was added to yield a homogenous single-phase mixture and incubated for 15 h (overnight) at 22°C (room temperature). To this mixture, water/chloroform (1:1) was added and after 30 min the organic phase was allowed to partition from the aqueous phase and recovered. After washing with 0.9% NaCl, the organic phase containing CSSE was recovered, dried and weighed. Before use, the CSSE was re-suspended in ethanol and diluted further in PBS.
Isolation of bovine CD14+ monocytes and culture of monocyte-derived mature DCs (bDCs)
Blood was collected from healthy cattle in 60 ml syringes containing 7.5% EDTA. PBMCs were obtained from these blood samples using the Ficoll (GE Healthcare Bioscience) method of isolation. 21 CD14+ monocytes were isolated from PBMCs by positive selection using MACs columns (Miltenyi Biotec Inc.). Mature bDCs were subsequently derived from CD14+ monocytes after culturing in a complete Roswell Park Memorial Institute-1640 (RPMI-1640) medium supplemented with 10% heat-inactivated FBS and 4-(2-hydroxyethyl)-1piperazineethanesulfonic acid (HEPES) (Hyclone Laboratories Inc.), 2-mercaptoethanol (Sigma-Aldrich), recombinant bovine GM-CSF at 100 ng/ml (Bio-Rad) and IL-4 at 50 ng/ml (Biorad) as previously described. 22 bDCs were assessed by flow cytometry and the expression of DC-specific marker CD206 was found to be greater than 97%.
In vitro cell culture and treatments
bDCs generated from CD14+ monocytes described above were seeded in multi-well plates and incubated in serum-free RPMI 1640 culture medium for 24 h. The serum-free medium was then replaced with 1 ml of complete medium containing LPS (1 µg/ml; Sigma-Aldrich), bPPD (1 µg/ml), CWL (1 µg/ml), or CSSE (1 or 10 µg/ml as indicated). In all experiments, the treated cells were incubated for 15 h at 37°C, 5% CO2 before supernatant collection. Untreated cells (culture medium only) served as a negative control. Culture supernatants were collected for ELISA. At 5, 15, 30, 60, 120 and 240 min, treated bDCs were lysed after removal of media with RIPA lysis buffer containing protease inhibitors. The resulting cell lysate was used for Western blot analysis.
ELISA
ELISAs were performed to determine the concentrations of IL-12 and TNF-α in bDC supernatants. Briefly, high-binding Immulon-2 (Thermofisher Scientific Inc.) ELISA plates and Maxisorp (Thermofisher Scientific Inc.) ELISA plates were respectively coated with bovine-specific TNF-α mAbs produced in-house and bovine-specific IL-12 mAbs (AbD Serotec) 23 in coating buffer (sodium carbonate, sodium bicarbonate and distilled water at pH 9.6) and incubated for 15 h at 4°C. Following incubation, the plates were washed five times with a wash buffer (TBST) and blocked for 1 h at 22°C. Then 200 µl/well of 0.1% gelatin (Sigma-Aldrich) and 0.1% casein (Sigma-Aldrich) were used for both TNF-α and IL-12 blocking respectively. The micro-well plates were rinsed five times with wash buffer and appropriately diluted recombinant IL-12 and TNF-α standards were titrated in a two-fold dilution to generate the standard curve. Samples were added undiluted (100 µl/well) and incubated at 22°C for 2 h. Thereafter, the plates were washed 5 times with washing buffer and 100 µl of biotinylated detection Ab at 4 µg/ml was added to all wells. After 1 h incubation at 22°C, the plates were washed five times and 100 µl of streptavidin (1/5000 dilution) was added to the wells then incubated for 1 h. Then, 100 µl of p-nitrophenyl phosphate (1 mg/ml) was added to all the wells and incubated for 1 h at 22°C. Plates were read at 405 nm using a SpectraMax plus microplate reader (Molecular Devices).
Western blot analysis
Protein concentrations of lysates from bDCs treated with LPS, bPPD, CWL and CSSE were determined using Bicinchoninic acid assay (Thermofisher Scientific Inc.). Next 15 µg of total protein were loaded in each lane and resolved by SDS-PAGE. Following electrophoresis, the protein bands were transferred to a nitrocellulose membrane using the iBlot 2 dry blotting system (Thermofisher Scientific Inc.). The membranes were blocked with 5% powdered skimmed milk in TBS and incubated for 1 h at 22°C. Thereafter, the membranes were incubated for 15 h at 4°C with primary Abs diluted in TBST. Immunoblotting of total and phosphorylated ERK, total and phosphorylated NF-κB (p65 subunit), SOCS1 and 3 and β-actin was performed as described previously. 24 The Abs used were: anti-p44/42 MAPK (ERK1/2) rabbit mAb (clone 137F5) and anti-phospho-p44/42 (ERK1/2) (Thr202/Tyr204) rabbit mAb XP® (clone D13.14.4E) (Cell Signaling Technology), anti-NF-κB p65 rabbit mAb (clone C22B4) and anti-phospho-NF-κB p65 (Ser536) rabbit mAb (clone D13.14.4E) (Cell Signaling Technology), anti-SOCS1 (middle region) rabbit polyclonal Ab (Antibodies-online.com), anti-SOCS3 (internal region) rabbit polyclonal Ab (LSBio) and anti-β-actin mouse mAb (clone mAbcam 8226) (Abcam). After washing with TBST, the membranes were incubated for 1 h at 22°C with Alexa Fluor® conjugated secondary Ab, at a dilution of 1:10,000 in dilution buffer. Signals were acquired using LI-COR imaging system (LI-COR Biosciences). Densitometry was performed using the band analysis tools of the Image Lab software version (LI-COR Biosciences).
Statistical analysis
Cytokine data are presented as means ± standard error of means whereas densitometry data are presented as means ± standard deviation. A one-way ANOVA was used to compare differences in cytokine production using GraphPad Prism software. Significance was considered if P < 0.05.
Results
bDCs treated with CSSE produce little TNF-α and IL-12
APCs such as macrophages and DCs produce the pro-inflammatory cytokines TNF-α and IL-12 in response to infection with members of the M. tb complex.25,26 In turn both TNF-α and IL-12 help to trigger naïve T-cells to mediate a Th1 response critical for the control of intracellular pathogens such as mycobacteria.19,27 Here we assessed production of TNF-α and IL-12 by bDCs on treatment with 1 µg/ml each of M. bovis-derived bPPD, CWL and CSSE, with LPS serving as positive control. Relative to no treatment, LPS at 1 µg/ml was found to induce the strongest response followed by bPPD and CWL, which were also at 1 µg/ml (Figure 1a and b). In contrast CSSE at 1 µg/ml induced very little to no production of TNF-α and IL-12 (data not shown). Strikingly, even when the concentration of CSSE was increased to 10 µg/ml, it failed to induce appreciably higher levels of TNF-α and IL-12 compared to no treatment (Figure 1a and b). These results show that M. bovis-derived bPPD is best at stimulating bDCs followed by CWL, whereas CSSE is the poorest inducer of the pro-inflammatory response in these cells.
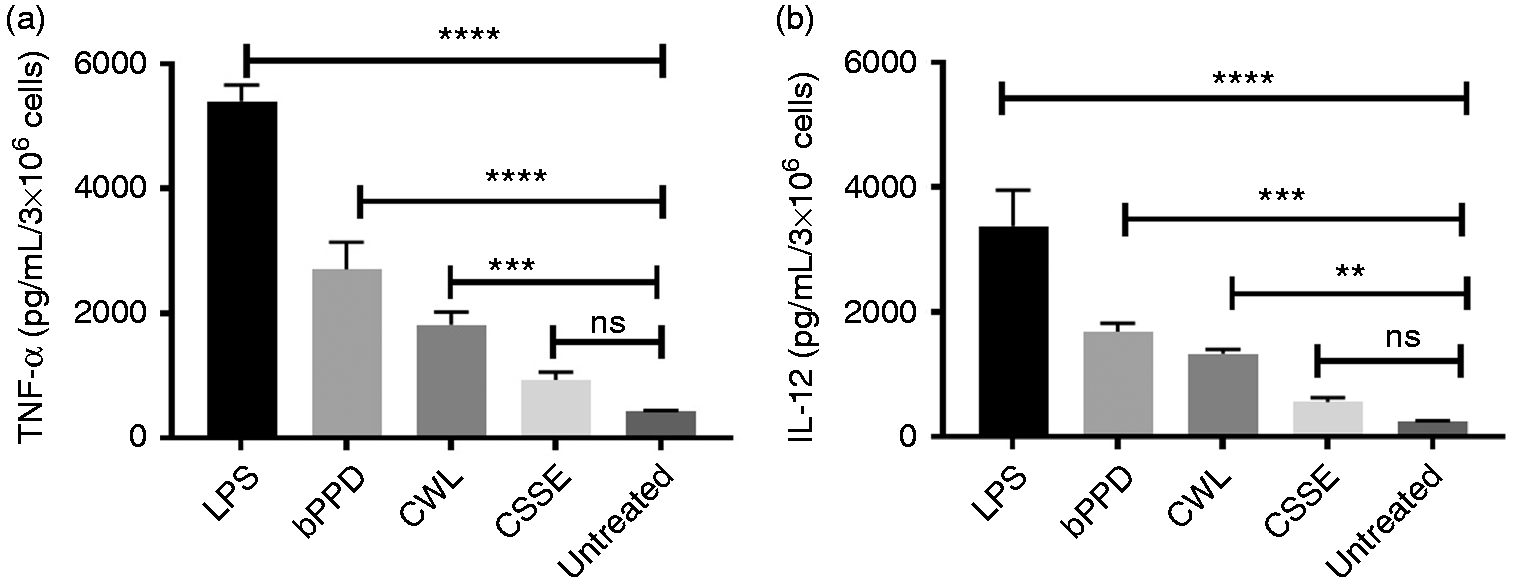
TNF-α and IL-12 production by treated bDCs. bDCs were treated with bPPD (1 µg/ml), CWL (1 µg/ml) and CSSE (10 µg/ml) for 24 h and the production of TNF-α (a) and IL-12 (b) were determined by ELISA. In all experiments, LPS (1 µg/ml) and unstimulated cells served as positive and negative controls respectively. Results are presented as means (+/− SEM) of signal from replicate wells and are representative of three independent experiments (**P < 0.01; ***P < 0.001; ****P < 0.0001).
CSSE-treated bDCs exhibit low phosphorylation of ERK
The pro-inflammatory response in APCs is mediated by signal-transduction events involving multiple MAPKs. 28 Activation of a MAPK protein called ERK via phosphorylation plays a key role in pro-inflammatory cytokine production in response to products of bacterial origin such as LPS.24,28 Therefore, we sought to examine the phosphorylation of ERK in bDCs treated with bPPD (at 1 µg/ml), CWL (at 1 µg/ml), and CSSE (at 10 µg/ml), with LPS (at 1 µg/ml) serving as positive control. LPS consistently induced phosphorylation in 22–28% of total ERK within 5 min post-addition, which decreased subsequently thereafter (Figure 2a to f). In contrast, bPPD induced phosphorylation in 28% of total ERK later at 15 min post-addition, and subsided shortly thereafter (Figure 2a and b). CWL treatment induced phosphorylation in ERK to the same level as LPS (∼20% of total ERK) by 5 min post-addition but maintained the same rate of phosphorylation longer than LPS treatment, which had waned after 5 min, before also decreasing (Figure 2c and d). In contrast, CSSE induced phosphorylation in no more 8% of total ERK throughout the duration of the experiment (Figure 2e and f). These results show ERK in CSSE-treated bDCs is poorly phosphorylated and activated and is consistent with the poor induction of pro-inflammatory response in these cells.
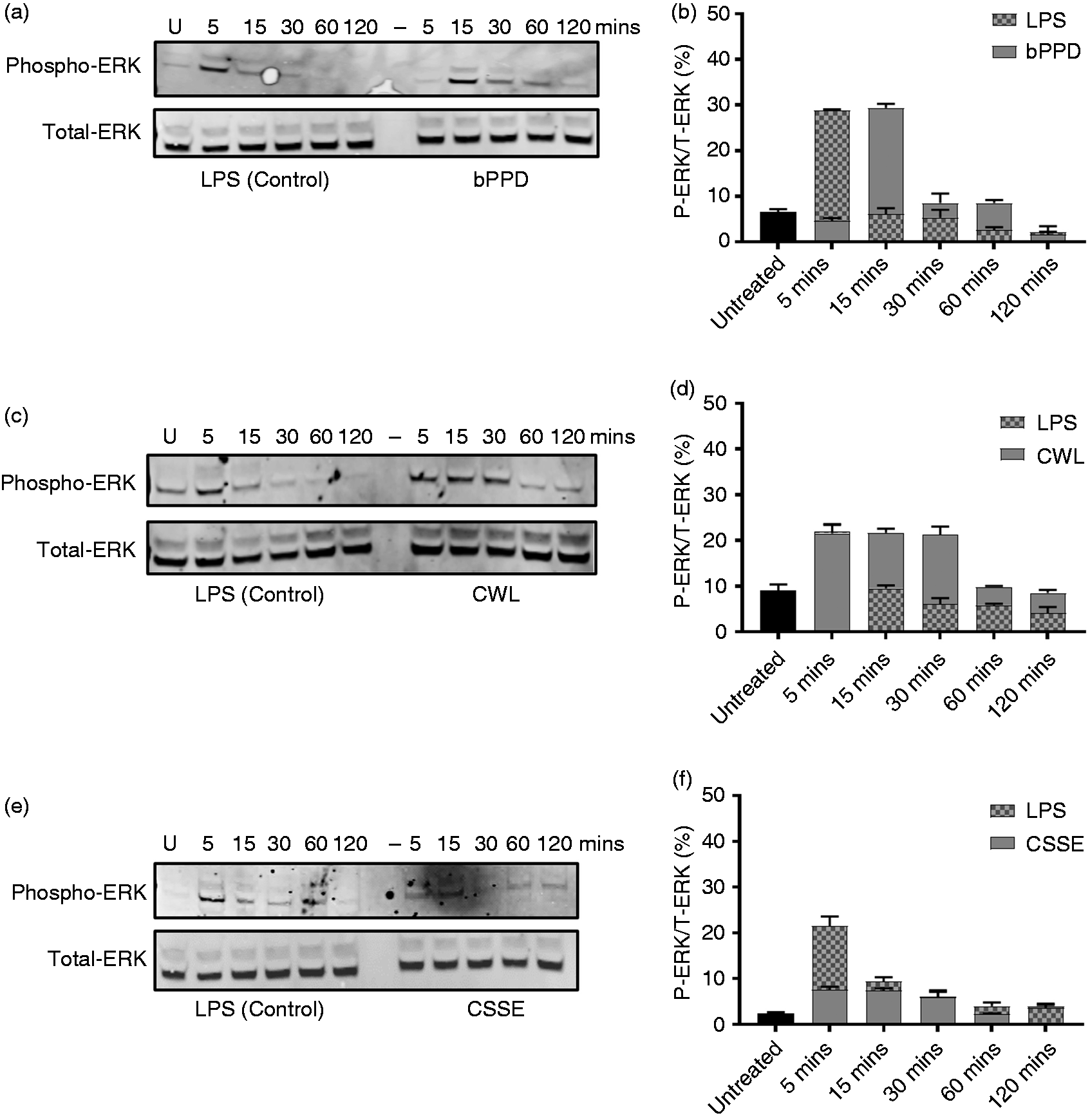
Phosphorylation of ERK protein in treated bDCs. bDCs cells were treated with bPPD (1 µg/ml), CWL (1 µg/ml) and CSSE (10 µg/ml) with LPS (1 µg/ml) as a positive control. Cell lysates were collected at different intervals post-addition of indicated M. bovis-derived fraction or LPS. Total cell lysate was assessed for phosphorylation of ERK protein (a, c and e) by Western blot. The percentage of phosphorylated ERK relative to total ERK was calculated by densitometry and plotted as bar graphs (b, d and f). The blots are representative of three independent experiments whereas the bar graphs show the means (+/− SD) of the three different blots.
CSSE-treated bDCs exhibit low phosphorylation and transactivation of the NF-κB p65 subunit
The multi-subunit NF-κB protein is a master transcriptional regulator that plays a vital role in immune activation.29,30 Moreover, transactivation of the p65 subunit of NF-κB by phosphorylation has been shown to up-regulate pro-inflammatory gene expression in APCs.24,29 Therefore we also sought to assess the phosphorylation status of the NF-κB p65 subunit over time in bDCs on treatment with bPPD (at 1 µg/ml), CWL (at 1 µg/ml) and CSSE (at 10 µg/ml), with LPS (at 1 µg/ml) treatment serving as positive control. LPS induced phosphorylation in 20–42% of total NF-κB p65 by 5 min post-addition and remained at that level for the rest of the experiment (Figure 3a to f). bPPD also induced phosphorylation in 20–42% of total NF-κB p65 within 5 min post-addition, with these levels remaining constant for the remaining duration of the experiment (Figure 3a and b). Phosphorylation of NF-κB p65 upon treatment with CWL appeared to be more variable, with 20% of total NF-κB p65 being phosphorylated within 15 min post-addition followed by a drop to 10% for the next 45 min before increasing again to 35% of total NF-κB p65 by 120 min post-addition (Figure 3b and c). In striking contrast, CSSE treatment induced phosphorylation in no more than 8% of total NF-κB p65 for the entire duration of the experiment (Figure 3e and f). These results clearly show that NF-κB p65 in CSSE-treated bDCs are poorly phosphorylated and activated. The results of these experiments are also consistent with the poor phosphorylation of ERK and the low production of pro-inflammatory cytokines by bDCs after CSSE treatment.
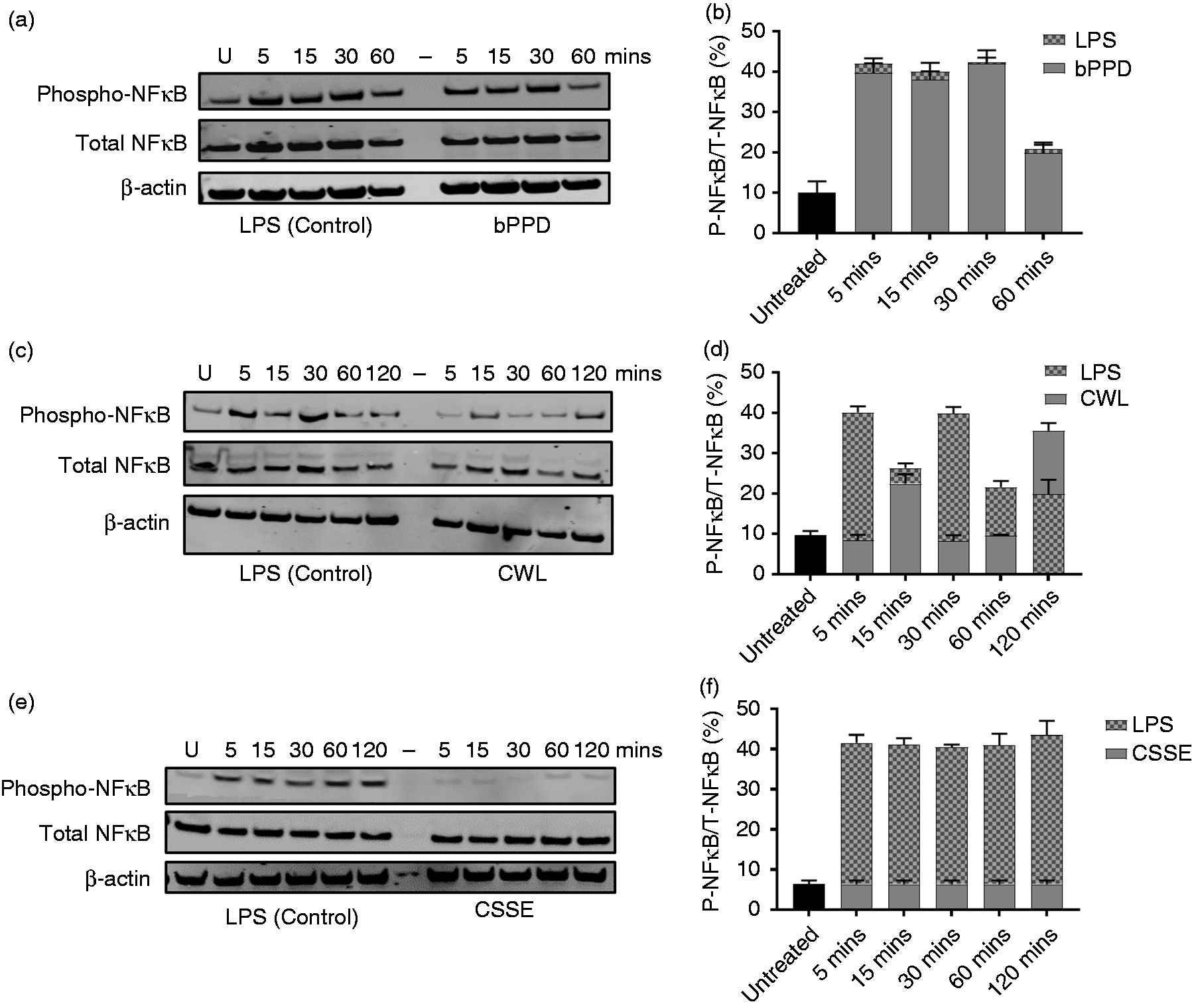
Phosphorylation of NF-κB p65 in treated bDCs. bDCs were treated with bPPD (1 µg/ml), CWL (1 µg/ml) and CSSE (10 µg/ml) with LPS (1 µg/ml) as positive control. Cell lysates were collected at different intervals post-addition of indicated M. bovis-derived fraction or LPS. Total cell lysate was assessed for phosphorylation of NF-κB p65 (a, c and e) by Western blot. The same blots were stripped and re-probed with Abs against β-actin. The percentage of phosphorylated NF-κB p65 relative to total NF-κB p65 was calculated by densitometry and plotted as bar graphs (b, d and f). The blots are representative of three independent experiments whereas the bar graphs show the means (+/− SD) of the three different blots.
Treatment of bDCs with CSSE up-regulates SOCS 1 and 3 expression
Our results suggest CSSE is a poor inducer of inflammation in bDCs. However, to determine whether this fraction simply lacks sufficient amounts of the bacterial products needed to activate pro-inflammatory responses or if it contains immunomodulatory biomolecules that actually suppress activation of pro-inflammatory responses in bDCs, we decided to look at the status of negative regulators of inflammation such as the SOCS proteins. SOCS proteins prevent hyper-inflammation following interaction with microbial products and thus play a key role in ensuring an appropriate and balanced immune response. 31 Moreover, SOCS proteins exert their regulatory effects by blocking pro-inflammatory signal transduction through a negative feedback loop to control the activation of immune cells as well as cytokine production. 32 Specifically, the initial steps of TLR signaling in macrophages is inhibited by SOCS 1 whereas other studies have implicated SOCS 1 and 3 in the blockage of NF-κB p65 activation.33,34 In light of the poor activation of NF-κB observed in CSSE-treated bDCs, we assessed the expression of SOCS 1 and 3 in bDCs treated with either LPS or CSSE. We found that by 5 min post-addition, CSSE caused a 22% increase in the expression of SOCS 1 relative to β-actin. This increased to approximately 40% by 60 min then stayed constant for the remainder of the experiment (Figure 4a and b). In contrast the expression of SOCS 1 was not increased substantially in LPS-treated bDCs (Figure 4a and b). SOCS 3 expression was increased to 10% of β-actin in CSSE-treated bDCs by 5 min post-addition, remained at this level for an additional 25 min before increasing to 40% by 60 min and stayed at this level for the remainder of the experiment (Figure 4c and d). In contrast, LPS appeared to increase SOCS 3 expression by no more than 10% of β-actin for the duration of the experiment (Figure 4c and d). These results clearly show that CSSE treatment increases SOCS 1 and 3 expression and may underlie the poor activation of NF-κB and pro-inflammatory cytokine production in bDCs.
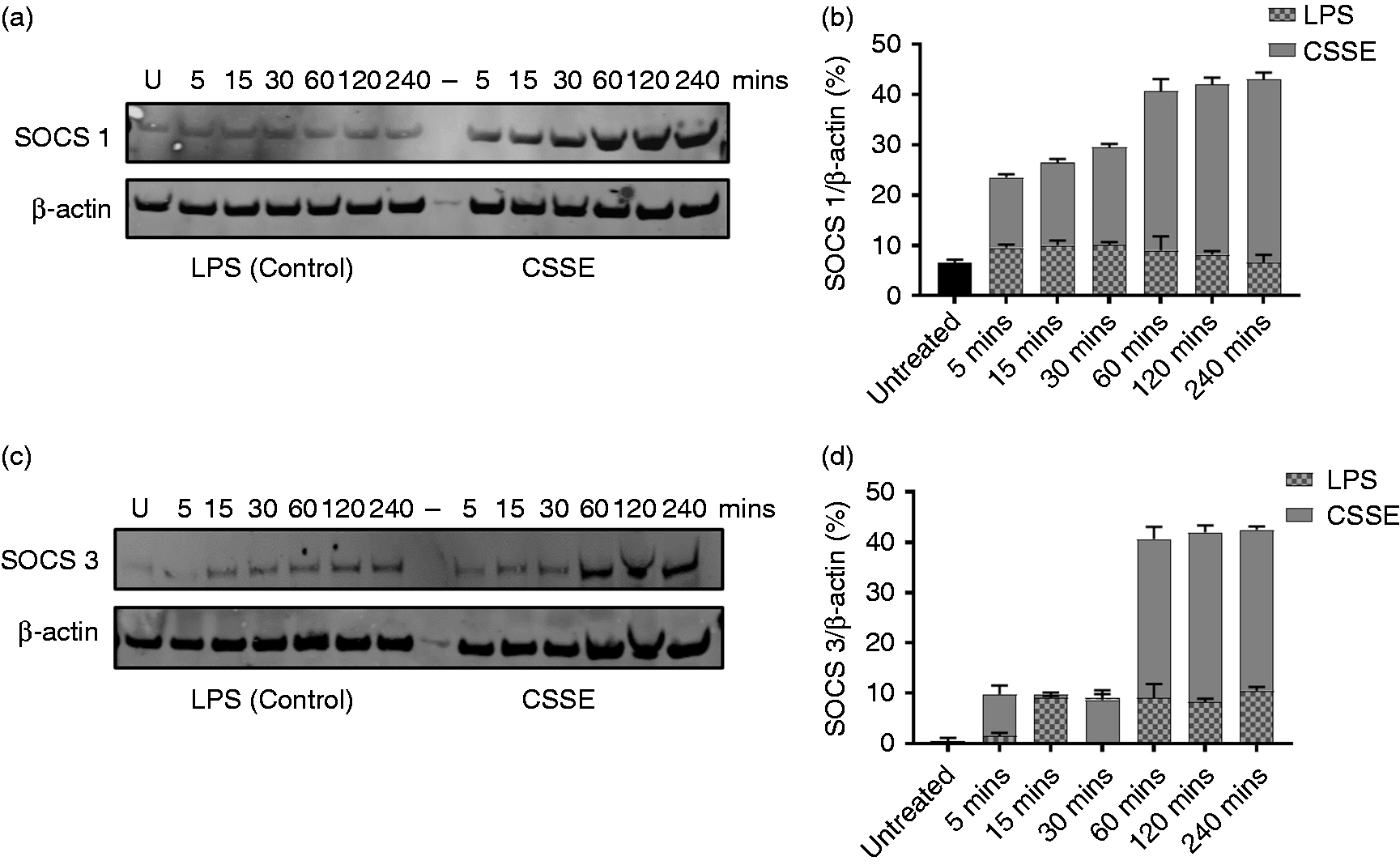
Expression of suppressors of cytokine signaling 1 and 3 (SOCS 1 and 3) proteins in treated bDCs. bDCs were treated with LPS (1 µg/ml) or CSSE (10 µg/ml). Cell lysates were collected at indicated intervals post-addition of M. bovis-derived fraction or LPS. Total cell lysate was assessed for SOCS 1 and 3 protein (a and b) by Western blot. The same blots were stripped and re-probed with Abs against β-actin. The percentage of SOCS 1 and 3 relative to β-actin was calculated by densitometry and plotted as bar graphs (c and d). The blots are representative of three independent experiments whereas the bar graphs show the means (+/− SD) of the three different blots.
Discussion
During mycobacterial infection, pro-inflammatory cytokines such as TNF-α and IL-12 produced by DCs help drive the induction of Th1 immunity.18,19 However, numerous studies show that pathogenic mycobacteria can modulate pro-inflammatory signaling in macrophages and DCs, thus enabling the bacterium to survive and persist within its host.7–11 Given that distinct lipidic components of the M. bovis cell surface appear to induce differential pro-inflammatory responses in bovine innate immune cells, 12 the purpose of this study was to perform a direct head-to-head comparison of the effect of three different types of M. bovis cell envelope fractions on bDCs to help determine which of these might be suitable bTB vaccine components.
We found that unlike bPPD, M. bovis-derived CWL and CSSE are relatively poor inducers of TNF-α and IL-12 production by bDCs, with the latter being the poorest of them all. Further analysis revealed that the activation of ERK and NF-κB in CSSE-treated bDCs were significantly reduced, suggesting the meagre production and secretion of TNF-α and IL-12 may be due to reduced activation of the ERK/NF-κB signaling pathway. Alternatively, it is conceivable that decreased phosphorylation of ERK protein directly or indirectly affects phosphorylation of NF-κB. It is also possible that CSSE inhibits phosphorylation of NF-κB by directly blocking the ability of the inhibitor of Kappa B kinase complex to degrade IκB proteins, thus preventing the activation and translocation of NF-κB to the nucleus. Further studies to delineate the mechanisms by which CSSE affects NF-κB phosphorylation in DCs are warranted.
Previous studies have shown that SOCS 1 suppresses DC maturation and functions to prevent the development of systemic autoimmunity.35,36 Most importantly, pathogenic mycobacteria have been reported to induce expression of SOCS 1 and 3 proteins as part of their virulence strategy.37,38 As such, we hypothesized that SOCS 1 and 3 might be involved in blocking IL-12 and TNF-α production in CSSE-treated bDCs. Indeed, we found CSSE treatment increased the expression of SOCS 1 and SOCS 3 proteins in bDCS. As such, the CSSE-mediated hypo-activation of ERK and NF-κB and concomitant lack of TNF-α and IL-12 production may also be due to increased production of SOCS 1 and 3.
Mycobacterial cell surface components in the CWL and CSSE fractions, especially lipids, glycolipids and glycans, are known to play prominent roles in the evasion of host immune responses by pathogenic mycobacteria.6–11,39 As such, our observations in this study with respect to lipid, glycolipid and glycan-enriched M. bovis CWL and CSSE fractions certainly appear consistent with the immunomodulatory effects reported for these molecules. Indeed, a number of mycobacterial lipids, glycolipids and glycans with immunomodulatory properties have been identified and characterized, and these include the phosphatidylinositol mannosides (PIMs), lipomannan (LM), lipoarabinomannan (LAM), mannose-capped LAM (ManLAM), trehalose-containing glycolipids and phenolic glycolipids (PGL).3,9,10 For instance PIMs from M. tb have been shown to inhibit LPS activation of human DCs by reducing the up-regulation of MHC II molecules and pro-inflammatory cytokine production. 10 Moreover, M. tb-derived LM reportedly blocks the biosynthesis of TNF-α in human macrophages by destabilizing TNF mRNA transcripts, thereby allowing M. tb to evade host immune response and potentially increase its virulence. 40 Furthermore, ManLAM has been shown to block IL-12 production by human DCs previously stimulated with LPS 41 and inhibit LPS-induced dendritic cell maturation. 42 PGL-1 and PGL-tb produced by clinical isolates of M. tb were shown to block TLR2 agonist-driven activation NF-κB and cytokine production43,44 and decrease TRIF-dependent TLR4 signaling in macrophages, resulting in limited pro-inflammatory and bactericidal responses. 45 More recently, it was reported that di-O-acyl-trhalose promotes a tolerogenic phenotype in bone marrow-derived murine DCs activated with mycobacterial Ags and TLR agonists, resulting in low expression of Ag presentation and co-stimulatory molecules and altered cytokine production. 46 It has also been shown that the M. tb-complex CSSE is enriched in phenolic glycans called para-hydroxy benzoic acid derivatives (p-HBADs), with M. tb producing two types p-HBAD I and II, and M. bovis producing the p-HBAD I type only.6,17 Both p-HBAD I and II are required by M. tb to dampen the pro-inflammatory response in vivo—mutants unable to produce these molecules induce stronger inflammatory responses in the murine model of infection compared to the wild type strain. 47 Moreover, chemically synthesized p-HBAD-I and II have been shown to suppress T-cell proliferation and activation, and block the production of pro-inflammatory cytokines by irradiated M. tb-stimulated murine bone marrow-derived macrophages in vitro. 48 Given the overwhelming evidence that many individual mycobacterial glycolipids and glycans individually display immunomodulatory properties, their overall effect on the host immune system when immunostimulatory mycobacterial Ags are also present, remain to be ascertained and warrant further study.
Our results also raise questions as to whether M. tb CSSE might have the same effect on human DCs as M. bovis CSSE does on bDCs or if M. tb CSSE is just as poorly stimulatory towards bDCs as M. bovis CSSE. It is tempting to speculate that perhaps because M. tb produces p-HBAD-I and II whereas M. bovis produces p-HBAD I, this may underlie the host-specific adaptation of M. tb and M. bovis to humans and cattle respectively. For example, comparative studies of an M. bovis mutant strain that produces both p-HBAD I and II or an M. tb mutant strain that produces only p-HBAD I with their isogenic wild type counterparts will be enlightening and is certainly warranted. At the same time, one also cannot rule out the possibility that the absolute amount, composition or combination of glycans, glycolipids and even protein Ags might have a role to play in M. tb-complex host adaptation.
Nonetheless, based on our data we posit that the M. bovis-derived CSSE fraction and perhaps even the CWL fraction, albeit to a lesser extent, are enriched with immunosuppressive glycolipids and/or glycans that contribute to the overall survival and proliferation of the bacteria within its host. We further speculate that the mycobacterial products in these fractions would probably be detrimental to include or have present in any next generation TB vaccine preparations. We are investigating these hypotheses and performing studies to identify and define the roles of immunomodulatory CSSE components in the context of M. bovis pathogenesis.
In conclusion, our study clearly shows that the M. bovis-derived CSSE fraction is a very poor immune stimulator. Furthermore, our data suggest this fraction contains molecules that actually contribute to the virulence of M. bovis by inhibiting the generation of effective host immune responses.
Footnotes
Acknowledgements
We thank the VIDO-InterVac Animal Care Unit for bovine blood sampling and senior research technician Ze Long Lim for help with the Western blot experiments. The following reagent was obtained through BEI Resources, NIAID, NIH: Mycobacterium bovis strain AF2122/97 total lipids. This manuscript was published with permission from the Director of VIDO-InterVac as journal series number #888.
Animal ethics approval
Approval for this work was obtained from the University of Saskatchewan Animal Care Committee. All the cattle from which the blood was obtained were maintained at the Central Animal Care Services under specific pathogen-free conditions, and used according to guidelines stipulated by the Canadian Council on Animal Care.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Genome Canada, Genome Prairie, Genome British Columbia, and the Saskatchewan Ministry of Agriculture to AAP, and the Banting Research Foundation to JMC.
