Abstract
Recent evidence suggests a key role of dendritic cells (DC) in the immunopathogenesis of multiple sclerosis (MS). Whereas dysfunction of DC was reported in MS patients, the underlying cause for this is not fully elucidated yet. The aim of the present study was to compare the gene expression profile of molecules involved in TLR4 and TLR7 signaling in DC from patients with MS and healthy controls. For this, circulating DC subsets were purified from patients with relapsing-remitting MS (RRMS) and from healthy controls for quantitative real-time PCR analysis. Additionally, TLR responsiveness in peripheral blood was investigated. We observed an aberrant steady-state release of IL-12p70 and IFN-α in patients with RRMS compared with healthy controls. Expression of IRF1 and JUN was reduced in conventional DC from patients with RRMS. In plasmacytoid DC from patients with RRMS, expression of IRF7 and IFNGR1 was reduced, while higher expression levels of TLR4 and LY86 were found compared with DC from healthy controls. The observed alterations in the gene expression of molecules involved in the TLR4 and TLR7 signaling pathways in circulating DC subsets may underlie the impaired IL-12p70 and IFN-α secretion in patients with RRMS, thereby potentially contributing to the disease pathogenesis of MS.
Introduction
The innate immune system is central in maintaining the body's immune homeostasis by its ability to discriminate between self and non-self. This system consists of cellular and biochemical defense mechanisms that recognize and respond to pathogens. For this, innate immune cells express a variety of receptors, including pattern-recognition receptors (PRR), such as Toll-like receptors (TLR), that recognize pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMP).1–3 A unique type of innate immune cells are dendritic cells (DC). DC are professional antigen-presenting cells with the capacity to polarize the differentiation of T cells, thereby regulating the balance between inflammation and tolerance. 4 To perform this function, DC are capable of capturing antigens, processing them and presenting them on the cell surface complexed to major histocompatibility complex molecules, e.g. human leukocyte antigen (HLA)-DR. Following migration towards the draining lymph nodes, DC present these antigens to T cells. Depending on the context in which the antigen is encountered, DC will rearrange their expression profile of co-stimulatory molecules, such as CD86, and cytokines, thereby determining the outcome of the immune response. For instance, antigen presentation in steady-state conditions will initiate T cell anergy, T cell apoptosis and regulatory T cell (Treg) induction,4,5 resulting in peripheral tolerance. In contrast, antigen recognition in combination with PAMP or DAMP will induce a complex maturation process in DC resulting in effector T cell stimulation.
In human blood, two major subsets of DC have been characterized, i.e. myeloid or conventional DC (cDC) and plasmacytoid DC (pDC). 6 cDC strongly express TLR4 and secrete high levels of IL-12p70 upon TLR4 ligation. 7 However, pDC strongly express TLR7 and TLR9 and secrete high levels of IFN-α upon TLR7 or TLR9 ligation. 8 We and others have recently demonstrated impaired TLR responsiveness of both cDC and pDC in patients with multiple sclerosis (MS), as evidenced by altered secretion of IL-12p70 and IFN-α upon TLR stimulation.9–12 This may suggest a crucial role of innate immunity in the initiation as well as progression of MS.9,13,14 MS is a chronic demyelinating, inflammatory disease of the central nervous system, 15 predominantly affecting young adults in their most productive years.
In order to understand the underlying mechanisms contributing to aberrant TLR responsiveness in MS, we studied the relative gene expression level of molecules involved in TRL4 and TLR7 signaling in circulating DC from patients with relapsing-remitting MS (RRMS), in comparison with DC from healthy controls. Here, we demonstrate that an impaired steady-state release of IL-12p70 and IFN-α secretion in patients with RRMS could be mediated by changes in the gene expression level of molecules involved in the TLR-signaling pathways in circulating DC subsets.
Materials and methods
Study population
Impaired steady-state release of IL-12p70 and IFN-α in patients with RRMS.
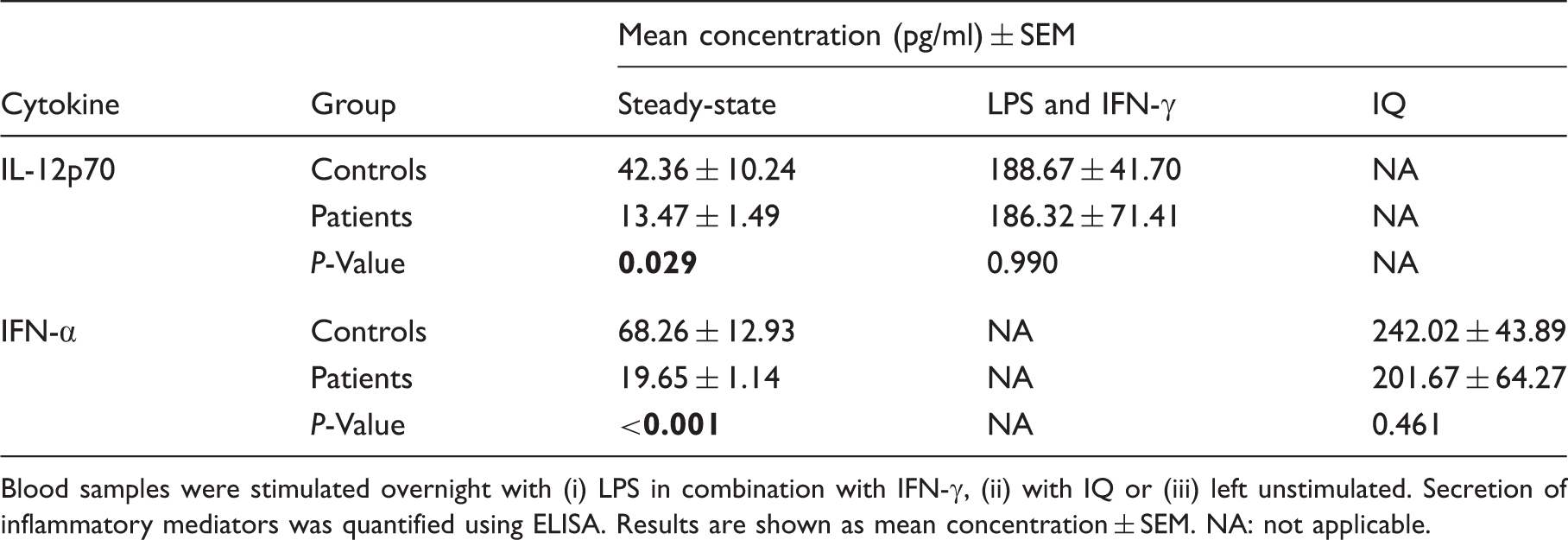
Blood samples were stimulated overnight with (i) LPS in combination with IFN-γ, (ii) with IQ or (iii) left unstimulated. Secretion of inflammatory mediators was quantified using ELISA. Results are shown as mean concentration ± SEM. NA: not applicable.
Isolation and stimulation of leukocytes
For flow cytometric cell sorting of DC subsets, leukocytes were isolated from patients with RRMS (n = 10) and healthy controls (n = 10) using density gradient centrifugation (Ficoll Paque PLUS; GE Healthcare, Chalfont St. Giles, UK). Additionally, in order to evaluate TLR responsiveness, 1 ml peripheral blood from patients with RRMS (n = 6) and healthy controls (n = 18) was stimulated overnight (16–20 h) with (i) 2 μg/ml LPS, a TLR4 ligand (Invivogen, Toulouse, France) and 50 µg/ml IFN-γ (Immunotools, Friesoythe, Germany), or (ii) with 10 μg/ml imiquimod (IQ), a synthetic TLR7 ligand (Invivogen), or (iii) left untreated as a control. Plasma was collected and stored at –20℃ for batch analysis of TLR-mediated cytokine production. Next, leukocytes were enriched after red blood cell lysis (0.155 M NH4Cl, 0.01 M KHCO3, and 0.1 mM Na2-EDTA) for immunophenotyping of DC.
Flow cytometry
Immunophenotyping of DC was done by direct immunofluorescence staining using the following fluorochrome-labeled mouse anti-human monoclonal antibodies: anti-blood dendritic cell antigen (BDCA)-1 phycoerythrin (PE) (Miltenyi Biotec, Leiden, the Netherlands), anti-BDCA-2 allophycocyanin (APC) (Miltenyi Biotec), anti-lineage I (Lin I; anti-CD3, anti-CD14, anti-CD16, anti-CD19, anti-CD20 and anti-CD56) fluorescein isothiocyanate (FITC) (BD Biosciences), anti-CD86 V450 (BD Biosciences), anti-CCR5 PE-cyanine (Cy)7 (BD Biosciences) and anti-HLA-DR APC-H7 (BD Biosciences) antibodies. Moreover, dead cells were excluded by addition of violet live/dead stain (Life Technologies, Carlsbad, CA, USA) to the antibody mixture. cDC and pDC were identified as Lin I− BDCA-1+ and Lin I– BDCA-2+, respectively. Fluorescence minus one in combination with non-reactive isotype-matched Abs was used as control. For analytical flow cytometry, at least 105 events were measured using a Cyflow ML flow cytometer (Partec, Münster, Germany). All results were analyzed using Flowjo software version 9.5.3 (Tree Star, Inc, Ashland, OR, USA).
Fluorescence-activated cell sorting (FACS) of DC was done by direct immunofluorescence staining using the following fluorochrome-labeled mouse anti-human monoclonal antibodies: anti-CD3 APC (BD Biosciences), anti-CD14 APC (Life Technologies), anti-CD19 APC (BD Biosciences), anti-CD203c APC (BD Biosciences), anti-HLA-DR PE-Cy7 (BD Biosciences), BDCA-1 APC-Cy7 (Biolegend, San Diego, CA, USA) and BDCA-2 FITC (Miltenyi Biotec). DC were identified as negative for lineage (i.e. CD3, CD14, CD19 and CD203c) and positive for the antigen-presentation molecule HLA-DR. Next, two DC subsets were sorted: (i) cDC identified as Lin− HLA-DR+ BDCA-1+ and (ii) pDC as Lin– HLA-DR+ BDCA-2+. Labeled cells were sorted on a FACS Aria II (BD Biosciences). Data acquisition was done using FACSDiva software version 6.1.2 (BD Biosciences).
Soluble analyte secretion assays
For quantitative detection of IL-12p70 and IFN-α, collected plasma samples were analyzed using the following commercially available ELISA kits: IL-12p70 (eBioscience) and IFN-α (PBL InterferonSource, Piscataway, NJ, USA), according to manufacturer's instructions.
RNA isolation and qPCR
Sorted DC subsets were immediately disrupted and homogenized with guanidine thiocyanate-containing lysis buffer. Total RNA was extracted from the cell lysates using a RNeasy Micro Kit (Qiagen, Düsseldorf, Germany), according to the manufacturer's instructions. The RNA concentration was determined by measuring the absorbance at 260 nm (A260) using a Nanodrop device (Wilmington, DE, USA). RNA integrity number values were used as a measure for RNA quality as assessed by means of an Agilent RNA 600 Pico chip (Agilent Technologies, Santa Clara, CA, USA) using a 2100 Agilent Bioanalyzer. Thereafter, RNA was converted to cDNA by reverse transcription (RT) using the iScript Advanced cDNA Synthesis Kit for RT-qPCR (Bio-Rad, Hercules, CA, USA). qPCR for relative mRNA quantification was performed using SYBR Green technology. All qPCR was conducted at 95℃ for 2 min, and then 40 cycles of 95℃ for 5 s and 60℃ for 30 s. The specificity of the reaction was verified by melt curve analysis. Validation data of the gene-specific primers for the target genes and reference genes can be found in Supplementary Table 2. Samples were pooled according to the 3 × 3 pooling strategy. For this, three pools of samples from patients with RRMS and three pools of samples from healthy controls were generated. Three replicates were used per pool. Relative mRNA levels for all target genes were normalized to levels of the following reference genes: GAPDH, ACTB and PGK1. Fold changes were calculated using the comparative Cq method (ΔΔCq method), as compared with healthy controls. Experiments were performed using a CFX96 thermal cycler (Bio-Rad) and data processing was performed using Bio-Rad CFX Manager version 3.1.
Statistical analysis
Data were analyzed using SPSS statistics version 21 (IBM, Brussels, Belgium) by independent samples t-test. Response variables were logarithmically transformed when necessary. Graphs were generated in GraphPad version 5 software (Prism, La Jolla, CA, USA) or Excel 2007 (Microsoft, Redmond, WA, USA). All data are presented as mean ± SEM.
Results
Impaired steady-state release of IL-12p70 and IFN-α in patients with RRMS compared with healthy controls
In order to assess the TLR responsiveness of circulating DC, blood samples were stimulated with a TLR4 ligand, LPS in combination with IFN-γ, or a TLR7 ligand, IQ. Both ligands are known to activate cDC and pDC, respectively.17,18 Using flow cytometry, we measured the fold change in the expression level of CCR5, CD86 and HLA-DR on cDC and pDC upon TLR stimulation. In both patients and controls, CD86 and HLA-DR expression is up-regulated on cDC and pDC following TLR stimulation, while CCR5 expression on cDC, but not on pDC, is down-regulated following TLR stimulation. No significant differences regarding the fold change of surface expression of migratory and co-stimulatory molecules upon TLR stimulation could be demonstrated between DC from patients with RRMS and DC from healthy controls (data not shown).
Next, the secretion of inflammatory mediators upon TLR stimulation of peripheral blood was assessed. In patients and controls, IL-12p70 and IFN-α secretion is up-regulated following TLR stimulation. Although no difference in IL-12p70 and IFN-α secretion following TLR ligation could be demonstrated between patients with RRMS and healthy controls, we observed a lower steady-state release, i.e. in the absence of stimulation with TLR ligands, of both IL-12p70 (P = 0.029) and IFN-α (P < 0.001) in patients with RRMS compared with healthy controls (Table 1).
Circulating cDC display reduced IRF1 and JUN gene expression in MS
In order to gain insight in the mechanisms potentially contributing to the observed aberrant cytokine release, we evaluated the relative gene expression of molecules that are involved in TLR signaling. For this, RNA was isolated for qPCR analysis from circulating DC purified using FACS. We observed lower relative expression levels of JUN (P = 0.015) and IRF1 (P = 0.006) in circulating cDC from patients with RRMS compared with cDC from healthy controls (Figure 1A). No significant differences could be demonstrated for the relative expression levels of TLR4, LY96, TIRAP, MYD88, TICAM2, TICAM1, NFKB1, IRF3, TLR7, IRF7, CD180, LY86, TOLLIP, SIGIRR, IFNGR1 and STAT1 between cDC from patients with RRMS and cDC from healthy controls (Figure 1A).
Aberrant gene expression levels of molecules involved in TLR signaling in DC from patients with RRMS. The relative gene expression of molecules involved in TLR4 and TLR7 signaling in cDC (A) and pDC (B) from patients with RRMS and healthy controls was measured using qPCR. Fold changes were calculated using the comparative Cq method (ΔΔCq method) compared with healthy controls. Black bars: P < 0.05. LY: lymphocyte antigen; TIRAP: Toll-IL-1 receptor domain containing adaptor protein; MYD88: myeloid differentiation primary response gene 88; TICAM: TLR adaptor molecule; JUN: jun proto-oncogene; IRF: IFN regulatory factor; TOLLIP: toll interacting protein; SIGIRR: single immunoglobulin and Toll-IL-1 receptor domain; IFNGR: IFN-γ receptor; STAT: signal transducer and activator of transcription.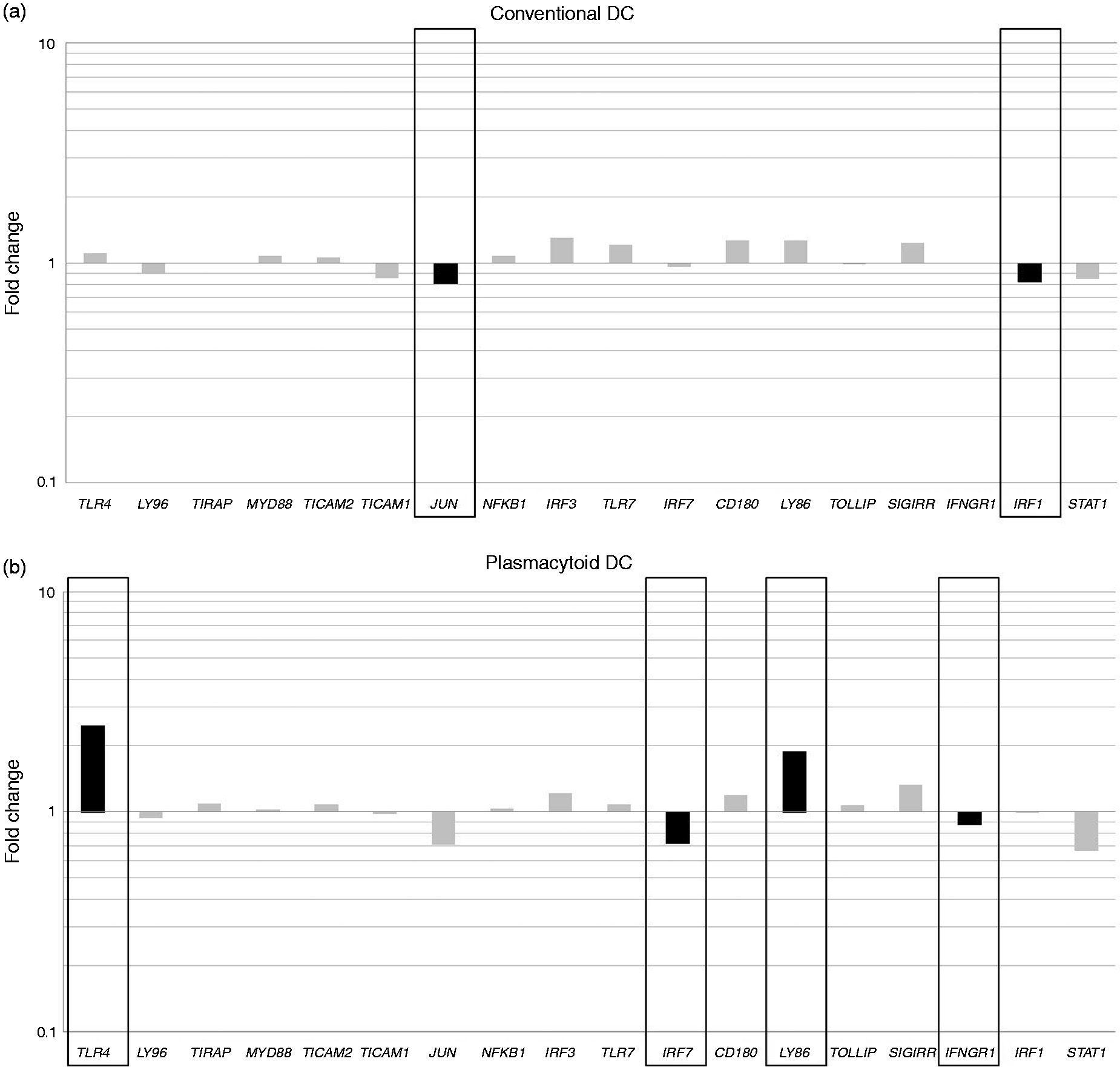
Circulating pDC from patients with RRMS demonstrate aberrant gene expression of IRF7, IFNGR1, TLR4 and LY86
The relative gene expression of molecules involved in TLR signaling was investigated in pDC purified using FACS. Our observations demonstrate that the relative expression levels of IRF7 (P = 0.016) and IFNGR1 (P = 0.032) in pDC from patients with RRMS was lower compared with the gene expression level in pDC from healthy controls (Figure 1B). Although pDC from both patients and controls displayed a much lower expression level of TLR4 (P < 0.001) and LY86 (P < 0.001) than cDC (Supplementary Figure 1), pDC from patients with RRMS showed an increased relative expression level of TLR4 (P = 0.017) and LY86 (P = 0.020) in comparison with healthy controls (Figure 1B). No significant differences could be demonstrated for the relative expression levels of LY96, TIRAP, MYD88, TICAM2, TICAM1, JUN, NFKB1, IRF3, TLR7, CD180, TOLLIP, SIGIRR, IRF1 and STAT1 between pDC from patients with RRMS and pDC from healthy controls (Figure 1B).
Discussion
In this study, we aimed to provide clues at a molecular level for the reported dysfunction of DC in patients with RRMS. In line with previous findings, 9 our observations demonstrate reduced plasma levels of both IL-12p70 and IFN-α in steady-state conditions in patients compared with healthy controls. However, we could not demonstrate significant differences between patients and controls for IL-12p70 and IFN-α secretion upon TLR stimulation, which is in contrast with reports by others.9,11,12 Previously, we demonstrated that circulating DC populations are the main producers of the cytokines detected. 9 Here, we observed lower absolute numbers of circulating cDC and pDC in MS compared with the healthy situation (data not shown). As others have suggested that changes in cytokine levels are most likely attributed to changes in absolute cell number than to a change in the amount of cytokine produced per cell,19–21 reduced cDC and pDC numbers could have contributed to the reduced baseline IL-12p70 and IFN-α secretion in patients with MS.
Interestingly, we found that the expression of JUN and IRF1 was reduced in cDC from patients. JUN encodes for c-Jun, a protein that together with c-Fos forms the transcription factor activator protein 1 (AP-1). In turn, AP-1 induces the expression of genes encoding for co-stimulatory molecules and cytokines, such as IL-12p70, upon TLR4 stimulation (Figure 2).
22
IRF1 encodes the transcription factor IRF1. By increasing and prolonging the recruitment of TLR4-induced transcription factors, such as AP-1, IRF1 synergizes the effect of TLR4 on the expression of cytokines, such as IL-12p70.
23
Reduced expression of IRF1 was previously demonstrated in PBMC from untreated patients with active MS in comparison with PBMC from healthy controls.
24
Moreover, lack of IRF-1 in knockout mice causes low IL-12 production by macrophages.
25
Altogether, we hypothesize that impaired AP-1 activity due to reduced IRF1 and JUN expression in cDC from patients with RRMS may contribute to the reduced steady-state IL-12p70 secretion.
Lessons at molecular level of molecules involved in TLR pathways for the function of DC. Binding of LPS to the TLR4/MD2 complex results in downstream activation of the MyD88-dependent pathway. This leads to the nuclear translocation of the transcription factors AP-1 and NF-κB, mediated by the adaptor proteins, MyD88 and TIRAP. The MyD88-independent pathway is dependent on TRIF and TRAM, and leads to the nuclear translocation of IRF3. The MyD88-independent pathway involves late-phase AP-1, NF-κB and IRF7 activation. RP105, a TLR4 homologue complexed with MD1, and TOLLIP, targeting phosphorylation of kinases in the MyD88-dependent pathway, inhibit TLR4 responses. Binding of viral single-stranded RNA or the synthetic ligand, IQ to TLR7 is MyD88-dependent and leads to the nuclear translocation of IRF7. TLR7 ligation involves late-phase AP-1 and NF-κB activation. SIGIRR inhibits the activation of TLR4 and TLR7 signaling pathways by interfering with the association of adaptor proteins to the receptor complex. The downstream effect of TLR signaling is the initiation of a complex maturation process in DC, i.e. AP-1 and NF-κB-mediated up-regulation of the expression of CD86, HLA-DR and IL-12p70 and MyD88-mediated down-regulation of the expression of CCR5. By increasing and prolonging the recruitment of TLR4-induced transcription factors, AP-1 and NF-κB, STAT1 and IRF1 synergize the effect of TLR4 on the expression of cytokines, e.g. IL-12p70. Activation of IRF3 induces IFN-β production, while activation of IRF7 induces both IFN-α and IFN-β production. Underlined molecules are up- or down-regulated in untreated patients with RRMS compared with healthy controls. IFN-γR: IFN-γ receptor; SIGIRR: single immunoglobulin IL-1 receptor-related molecule; Tollip: Toll interacting protein; MyD88: myeloid differentiation primary-response protein 88; TIRAP: Toll/IL-1 receptor (TIR)-domain-containing adaptor protein; TRIF: TIR-domain-containing adaptor protein inducing IFN-β; TRAM: TRIF-related adaptor molecule; STAT: signal transducers and activators of transcription; IRF: IFN-regulatory factor; AP: activator protein; CCR: C-C chemokine receptor; HLA: human leukocyte antigen.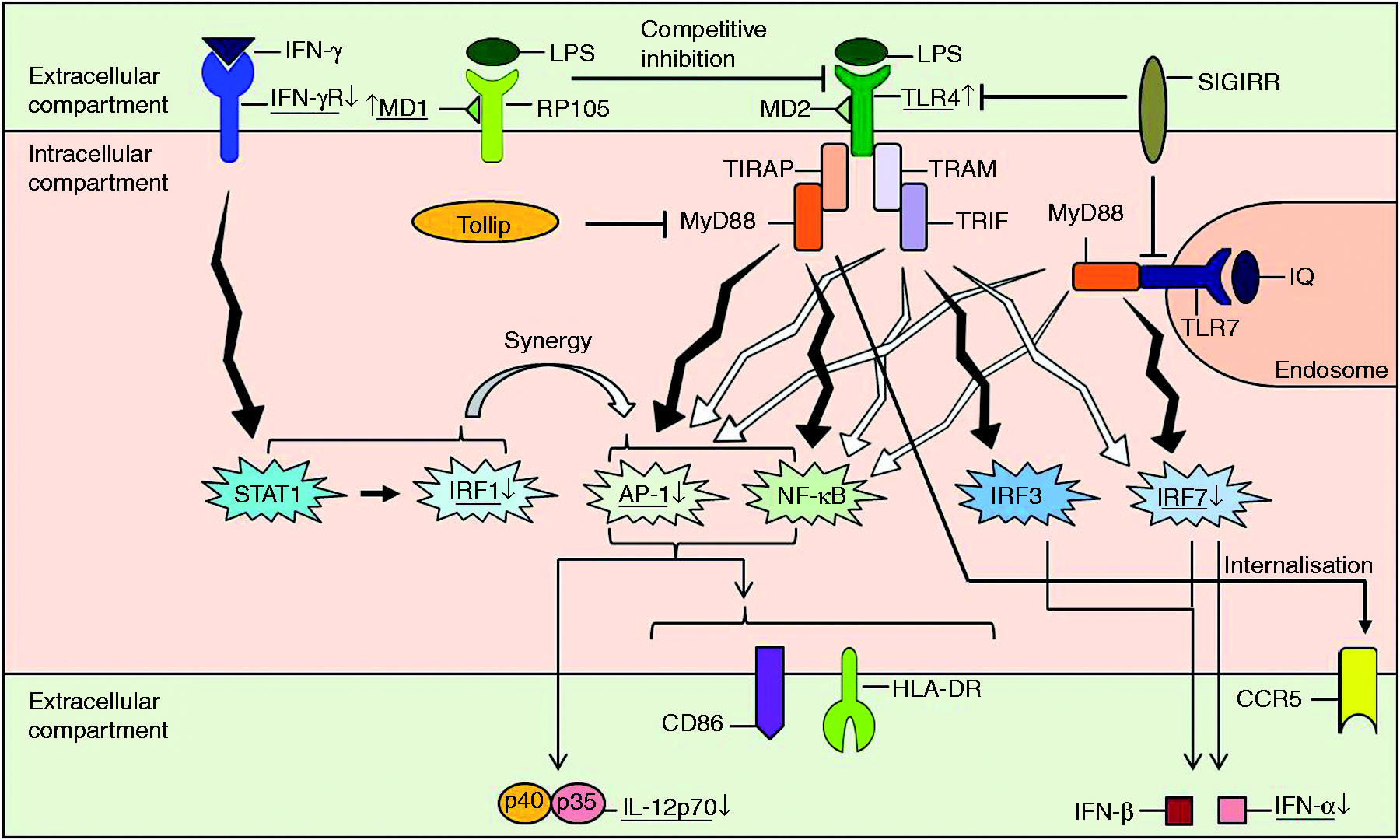
Also in pDC variations in the gene expression profile between patients with RRMS and healthy controls were found. Expression of IRF7, encoding the transcription factor IRF7, which induces IFN-α and IFN-β secretion upon TLR7 stimulation (Figure 2), 26 was reduced in pDC from patients. This may explain the reduced IFN-α secretion in patients with RRMS in steady-state conditions, accounting for possible involvement of IFN-α-producing DC in the pathogenesis of MS. Indeed, two types of pDC have been identified: type 1 pDC are the main source of IFN-α and induce IL-10-producing Treg, while type 2 pDC secrete IL-6 and TNF-α and direct naive T cells toward IL-17-secreting T helper type 17 cells. 27 Schwab et al. have previously reported a reduced ratio of type 1/type 2 pDC in patients with MS, 27 which is in agreement with the reduced steady-state IFN-α secretion observed in this study. In addition, reduced expression of IFNGR1, encoding the ligand-binding subunit of the heterodimeric IFN-γR, 28 in pDC from patients may result in reduced responsiveness to IFN-γ. An interesting function of IFN-γ signaling in pDC is the induction of indoleamine 2,3-dioxygenase (IDO), an inducible tolerogenic enzyme that is involved in the induction of Treg.29,30 Based on these findings, we hypothesize here that decreased IRF7 and IFNGR1 expression in pDC from patients might result in a diminished Treg-inducing capacity of pDC from patients with RRMS. Future studies addressing the effect of molecular changes in DC from patients with RRMS on the function of T cells are, however, needed.
Although pDC are generally shown to be negative for TLR4, 7 pDC from patients with RRMS showed increased expression of TLR4 compared with pDC from healthy controls. Nevertheless, pDC from patients and controls displayed a 10 times lower expression of TLR4 than cDC (Supplementary Figure 1). Others, demonstrating increased TLR4 expression in PBMC from patients with MS compared with PBMC from healthy controls, suggested that this may represent a state of innate immune activation. 31 Similarly, the expression level of LY86 in pDC is six times lower than in cDC but is significantly increased in pDC from patients compared with healthy controls. LY86 encodes MD1, which together with RP105 forms a competitive inhibitor for LPS binding to the TLR4/MD2 complex (Figure 2). 32 Up-regulated gene expression of the inhibitory molecule MD1 in pDC from patients could be a mechanism to counteract the increased state of innate immune activation. Whereas we were able to demonstrate TLR4 expression by pDC on a protein level using flow cytometry (Supplementary Figure 2), we next investigated whether altered molecular expression profiles affect the TLR responsiveness of pDC following LPS and IFN-γ stimulation; we found a significantly up-regulated expression of HLA-DR on pDC in both patients and controls. However, no difference could be detected between patients and controls regarding the fold change in the surface expression of HLA-DR upon LPS and IFN-γ stimulation (data not shown). No differences in the expression levels of other surface markers, CCR5 and CD86, on pDC could be detected following TLR4 ligation (data not shown).
Here, we included a clearly defined homogeneous study population of 16 untreated patients with RRMS. In doing so, we have ascertained that our findings were not confounded by treatment of the patients or clinical course. However, as we previously demonstrated possible involvement of DC not only in RRMS, but also in chronically-progressive MS (CPMS), 9 it would be interesting to also investigate molecular changes in DC from patients with CPMS. Furthermore, by evaluating TLR responsiveness in patients receiving first- or second-line treatment in comparison with untreated patients, better insights into the treatments' mechanisms of action could be obtained, potentially identifying responders and non-responders to immunomodulatory treatment in the nearby future. In this perspective, ustekinumab, an anti-IL-12p40 Ab treatment effective for psoriatic arthritis and Crohn's disease, fails to show efficacy in MS.33,34 Based on our observations, it is possible that the lack of treatment effectiveness of ustekinumab in MS might be due to impaired IL-12p70 secretion in steady-state conditions. In this regard, interesting findings can be obtained when comparing TLR responsiveness and relative gene expression of molecules that are involved in TLR signaling between responders and non-responders to specific immunomodulatory treatments.
In conclusion, we demonstrated that an impaired steady-state release of IL-12p70 and IFN-α secretion by DC in RRMS patients could be mediated by changes in the gene expression level of molecules involved in the TLR-signaling pathways in circulating DC subsets. Ultimately, lessons learned from molecular changes may provide better knowledge with regard to the disease pathogenesis of MS, as well as new insights for patient stratification and the development of new drugs.
Footnotes
Acknowledgements
We would like to thank all patients with MS and healthy controls for participating in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant no. G.0168.09 of the Fund for Scientific Research (FWO) – Flanders, Belgium (FWO-Vlaanderen). Further support was provided through the Special Research Fund (BOF), a BOF-GOA grant (ID PS 28313), Medical Legacy Fund (UZA), the Methusalem Funding Program, the Belgian Hercules Foundation and the Belgian Charcot Foundation, the “Belgische Stichting Roeping”.
