Abstract
The present study was conducted to investigate the effects of dietary supplementation of selenium from different sources on the growth performance, nutrient digestibility, and blood immune indices of piglets orally challenged with Salmonella typhimurium (ST). In a 2 × 2 factorial arrangement, 32 piglets (6.43 ± 0.54 kg of body mass) were assigned into four groups with or without dietary inclusion of sodium selenite (SS) or yeast selenium (YS) and with or without ST challenge (5 ml 1 × 109 cfu/ml ST or 5 ml saline) on d 13. In each period, YS increased average daily feed intake and average daily gain but did not reach statistical significance. During the challenged stage, piglets fed YS had higher digestibility of dry matter, crude protein, crude fat, and YS reduced the amount of Escherichia coli in feces. Additionally, YS regulated the composition of T-lymphocyte subset and influenced the production of inflammatory cytokines. In conclusion, in this study selenium-enriched yeast was more effective in enhancing nutrient digestibility, and inhibiting inflammation and oxidative stress by inducing the activity of the lymphocytes, expression of antioxidant enzymes and so on.
Keywords
Introduction
Selenium is one of the micronutrients in animals and has been recognized as an essential dietary nutrient for about 50 years. It can protect against several degenerative diseases, improve the reproductive performance of animals, and strengthen the immunity of animals.1,2 One function of selenium is performed by kinds of selenoproteins acting as redox catalysts (e.g., glutathione peroxidase) at normal internal environment homeostasis. 3 Selenium has additional momentous effects on the immune response, which might happen independent to the enzymatic functions. 1 Also, the apparent digestibility of dry matter in selenium-supplement pigs has been investigated in detail. 4 Traditionally, sodium selenite (SS) is generally supplied in pigs’ diets to improve animal health, but it has the disadvantage of low absorption and high toxicity for animals and residual selenium in excrement can pollute the environment. 5 Selenium of selenium-enriched yeast is biotransformed by yeast cells, which meets the optimal selenium requirement of the animal body without any toxic side effect. 6 Meanwhile, selenium-enriched yeast has proven to be safer, more stable, more absorbent, and less polluting than inorganic selenium. 7
Salmonella typhimurium (ST) is one of the most important bacterial foodborne pathogens that afflicts a large number of humans and animals worldwide. 8 Particularly for intensive livestock and poultry farms, ST represents a non-negligible challenge in causing substantial economic loss. 9
Accordingly, we hypothesized that yeast selenium (YS) supplementation would have better effects on performance, apparent digestibility, and immunity and anti-oxidation function for piglets than SS. This investigation was conducted to determine the effect of selenium-enriched yeast on growth performance, nutrient digestibility, and diarrhea in a piglet model with ST challenge. Also, T-lymphocyte subsets and serum indices were determined to explain the enhancement of immunity and resistance to oxidative stress.
Materials and methods
Animals and diets
The piglets were used in this investigation according to the Animal Care and Use Committee of Wuhan Polytechnic University. Thirty-two healthy weaned crossbred male piglets (Duroc × Large White ×Landrace; weaned at 21 ± 1 d of age; body mass (BM) = 6.43 ± 0.54 kg) were selected from a creditworthy farm, and controlled in the Hubei Key Laboratory of Animal Nutrition and Feed Science (Wuhan Polytechnic University, Wuhan, China). During the 27 d experiment, piglets were housed individually in pens, with free access to feed and water. The pre-starter and starter diets (Table 1) were formulated to meet National Research Council (NRC) nutrient requirements. 10 The two experimental diets consisted of the same amounts of most ingredients, such as cooked corn, expanded corn, expanded soybean and fermented soybean meal and so on, at each period. The only difference between the two diets was the source of selenium in 5% premix, and the content of selenium in both diets was 0.375 mg/kg.
Composition of experimental diets (%, as-fed basis).
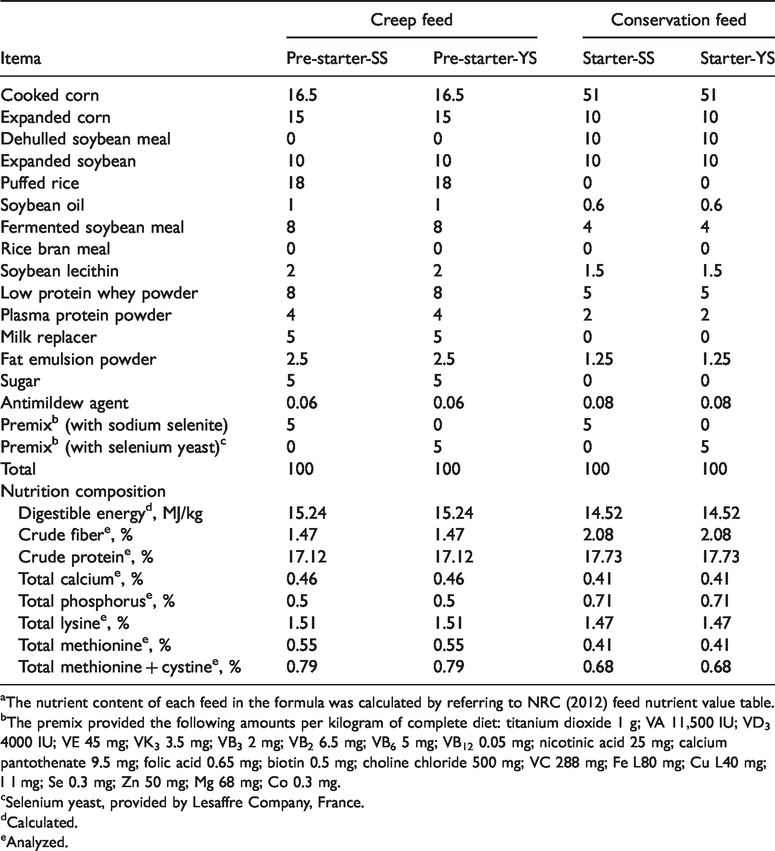
The nutrient content of each feed in the formula was calculated by referring to NRC (2012) feed nutrient value table.
The premix provided the following amounts per kilogram of complete diet: titanium dioxide 1 g; VA 11,500 IU; VD3 4000 IU; VE 45 mg; VK3 3.5 mg; VB3 2 mg; VB2 6.5 mg; VB6 5 mg; VB12 0.05 mg; nicotinic acid 25 mg; calcium pantothenate 9.5 mg; folic acid 0.65 mg; biotin 0.5 mg; choline chloride 500 mg; VC 288 mg; Fe L80 mg; Cu L40 mg; I 1 mg; Se 0.3 mg; Zn 50 mg; Mg 68 mg; Co 0.3 mg.
Selenium yeast, provided by Lesaffre Company, France.
Calculated.
Analyzed.
Bacteria
ST (CICC 21484) was provided from the China Center of Industrial Culture Collection (Beijing, China). To determine the CFUs, the inoculum was diluted with PBS and plated on NB basal medium agar for 24 h at 37°C.
Experimental design
The 2 × 2 factorial experiment was utilized for this study with dietary treatments (SS vs. selenium yeast) and ST challenge (saline vs. ST). Based on similar BM, pigs (n = 32) were randomly divided into four treatments: (i) SS-SC (piglets received a SS diet and injected with 5 ml 1 × 109 cfu/ml ST); (ii) SS-NC (piglets received a SS diet and injected with 5 ml saline); (iii) YS-SC (piglets received a selenium yeast diet and injected with 5 ml 1 × 109 cfu/ml ST); and (iv) YS-NC (piglets received a selenium yeast diet and injected with 5 ml saline). There were eight replicates (n = 8) for each treatment in each sampling time, and each replicate had one pig. After 3 d of acclimatization, the animals received the pre-starter ration during d 1–13 and starter diets during 14–27. On d 13 of the trial, piglets in groups SS-SC and YS-SC were orally challenged with 5 ml of ST inoculum (1 × 109 cfu/ml), while piglets in the groups SS-NC and YS-NC were treated with equivalent amounts of saline. Piglets were weighed on the d 1, d 13, d 20, and d 27 of the trial at 8:00 am. Meanwhile, daily feed intake, diarrhea occurrence, and skin/fur situation of piglets were recorded.
Sample collection
On d 13 (before challenge), d 14, d 20, and d 27 of the trial, two heparinized vacuum tubes of blood were obtained from the precava of each piglet. The blood was centrifuged at 3500 g for 10 min at 4°C and stored at −80°C for further analyses. Following the last blood collection, all piglets were slaughtered, humanely. The fecal samples were collected for measurement of apparent digestibility (on d 10–12, d 17–19, and d 24–26), and microbial composition (ST and Escherichia coli) of feces (on d 13, d 14, d 20, and d 27).
Measurement of nutrient digestibility
For each diet (Table 1), 0.1% TiO2 was mixed individually and supplied in the premix. By measuring the levels of nutrients in feed (NR) and fecal (NF) samples and the concentration of TiO2 in feed (IR) and fecal (IF) samples, apparent digestibilities (DN) were calculated using the equation DN = (1 − IRNF/IFNR) × 100%. 11
Measurement of fecal microbial
The standard culture method (SCM) was used for the present study in determining the content of bacteria in piglets’ fresh fecal sample, which was based on previous studies. 12 The fecal sample was diluted by the sterilized saline. Then, the appropriate diluted sample was inoculated in the bacterial test tablets (Biopeony Beijing Co., Ltd), colonies counted, and the color show test carried out according to the instructions.
Measurement of antioxidase and inflammatory cytokines of blood samples
The contents of catalase (CAT), superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), malondialdehyde (MDA), IgA, IgG, and IgM were tested using the commercial assay kits according to the manufacturers’ instructions (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). The concentrations of IL-1, IL-2, and IL-6 were measured with the commercial porcine ELISA kit (Shanghai Enzyme-linked Biotechnology Co., Ltd. (Shanghai, China)). White blood cells (WBCs) were analyzed by Bayer ADVIA2120i according to the instructions. Servicebio Bio-Technology Co., Ltd. (Wuhan, China) was entrusted with measuring CD3+ cell, CD4+ cell, CD8+ cell, contents of total protein (tp), urea, NO, C-reactive protein (CRP) in whole blood.
Statistical analysis
All data were analyzed as a 2 × 2 factorial design by ANOVA using the general linear model (GLM) procedures of SPSS version 17.0 (SPSS Inc., Chicago, IL, USA). The statistical model consisted of the main effects of dietary treatment (SS or selenium yeast) and challenge (saline or ST), and their interactions. Post hoc testing was conducted using Duncan’s multiple comparison tests. The data were considered significant at P < 0.05, and 0.05 < P < 0.1 was considered as trend. All data are presented as mean ± SEM.
Results
Growth performance
Before ST challenge, YS supplementation enhanced average daily feed intake (ADFI) significantly (P < 0.05; Table 2). In other groups, no differences were observed in ADFI after ST challenge (P > 0.05). During d 14–20, piglets infected with ST had a lower average daily gain (ADG) than that of the piglets treated with saline (P < 0.05). There was no significant difference in feed conversion rate (FCR) among all treatment groups throughout the trial.
Effects of YS supplementation on growth performance of weaned piglets during the whole test.
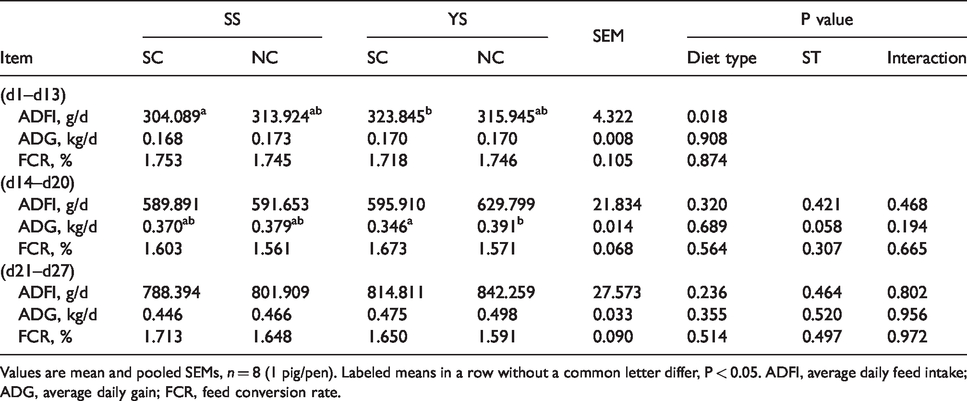
Values are mean and pooled SEMs, n = 8 (1 pig/pen). Labeled means in a row without a common letter differ, P < 0.05. ADFI, average daily feed intake; ADG, average daily gain; FCR, feed conversion rate.
Nutrient digestibility
As shown in Table 3, at the non-challenged stage, diet treatments did not significantly affect the digestibility of crude fat. Piglets supplied YS had higher digestibility of dry matter and crude protein. It is worth mentioning that the YS had contributed significantly to the development of dry matter and crude protein digestibility, as indicated by statistics showing before ST (P < 0.05). Form d 14 to 27, the digestibility of dry matter, crude protein and crude fat in YS group were remarkably higher than that in SS group (P < 0.001). There was an interaction between diet and ST on the digestibility of crude fat (P < 0.001).
Effects of YS supplementation on nutrient digestibility of weaned piglets during the whole test.
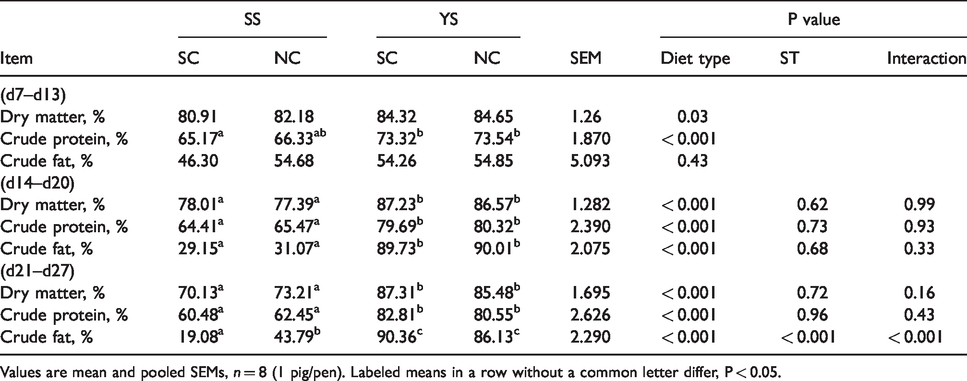
Values are mean and pooled SEMs, n = 8 (1 pig/pen). Labeled means in a row without a common letter differ, P < 0.05.
Microbial counting of feces
Before ST challenge, the concentration of ST in YS-NC group was higher than the SS-NC group (P < 0.05; Table 4), and E. coli in YS-SC group was higher than SS-SC (P < 0.05). One d post ST challenge, fecal ST level in SS-NC group was higher than that in YS-NC group (P < 0.05). Fecal E. coli was lower in SS-NC group when compared with the YS-NC (P < 0.05). Seven d after challenge, the level of ST in the YS-SC group was higher than that of YS-NC group (P < 0.05). Moreover, the concentration of E. coli in the SS-NC group was higher than the YS-NS group (P < 0.05). Compared with the SS treatment, there was a significant difference in E. coli between SS-SC and YS-SC groups at d 14 post challenge (P < 0.05), and SS-SC group had a higher E. coli level than that of SS-NC group (P < 0.05). There was an interactive effect between diet and ST on the level of E. coli.
Microflora (ST and E. coli) counting of feces at d14, d16, d22, and d28.
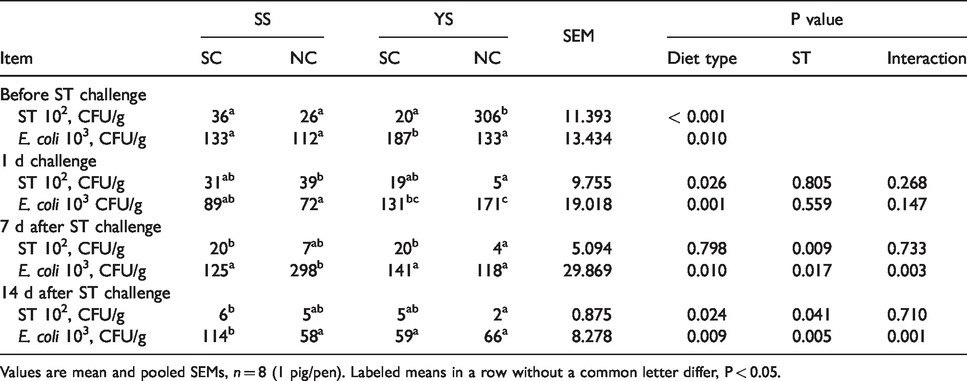
Values are mean and pooled SEMs, n = 8 (1 pig/pen). Labeled means in a row without a common letter differ, P < 0.05.
T-Lymphocyte subset
Before ST challenge, piglets that had been fed YS had a higher CD4+ T cells concentration compared with piglets supplied SS (P = 0.001; Table 5). No significant difference was found in the CD3+, CD8+, and CD4+/CD8+ ratio (P > 0.05). One d after ST challenge, CD4+ cells and CD3+ cells in YS-SC group were higher than that in SS-SC group (26.75% and 35.64%, respectively). CD8+ cells in YS-SC group declined significantly by 21.98% compared with SS-SC group (P > 0.05), while the immune cells of CD3+ in YS-SC advanced. There was a significant interaction between SS challenge and diet on the CD3+ cells (P < 0.05). Seven d after ST challenge, compared with piglets supplied with SS, piglets fed with YS had a higher CD4+ cells and CD4+/CD8+ ratio (P < 0.05). Fourteen d after ST challenge, the piglets fed YS diet had a higher number of CD3+ cells, and a lower one of CD8+ cells compared with piglets fed SS diet (P < 0.05). There was a significant increase of CD4+/CD8+ ratio in piglets infected by ST at 14 d post-challenge.
Effects of YS supplementation on CD4+, CD3+, CD8+ and CD4+/CD8+ ratio of whole blood in weaned pigs.
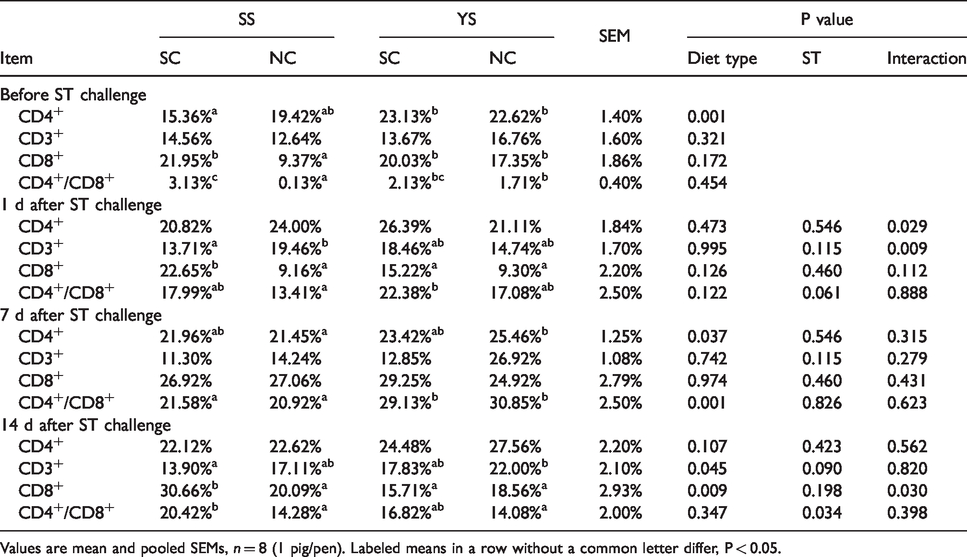
Values are mean and pooled SEMs, n = 8 (1 pig/pen). Labeled means in a row without a common letter differ, P < 0.05.
Serum biochemical indexes
Before ST challenge, no difference was seen in IgA, IgG, IgM, CAT, GSH-PX, and WBCs between different selenium sources (P > 0.05; Tables 6 and 7), while the piglets supplied with SS had higher contents of urea, SOD, MDA, compared with pigs fed YS (P < 0.05). Compared with SS group, piglets fed YS had higher levels of NO, CRP, IL-1, IL-6 (P < 0.01), and IL-2 (P < 0.05).
Effects of YS supplementation on antioxidase of serum in weaned pigs.
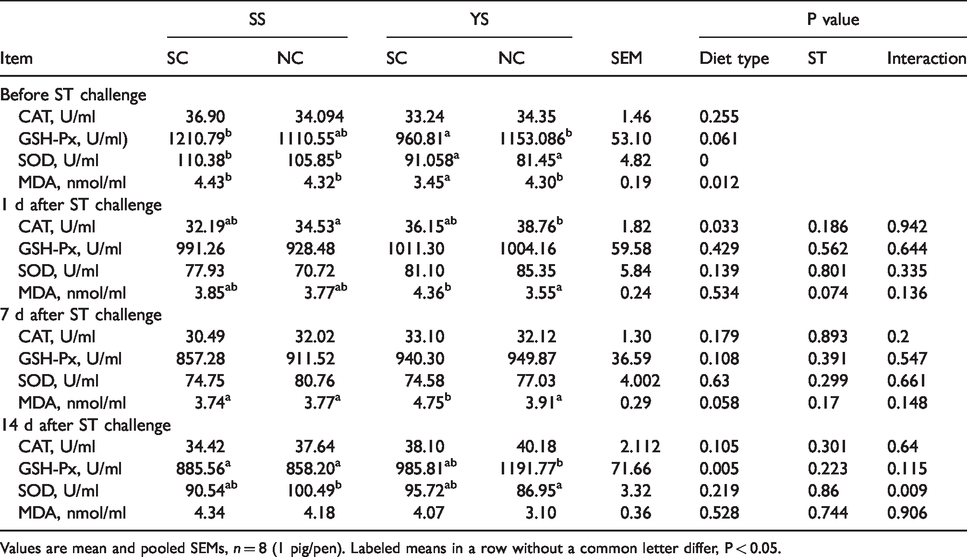
Values are mean and pooled SEMs, n = 8 (1 pig/pen). Labeled means in a row without a common letter differ, P < 0.05.
Effects of YS supplementation on inflammatory cytokines and other performance of serum in weaned pigs.
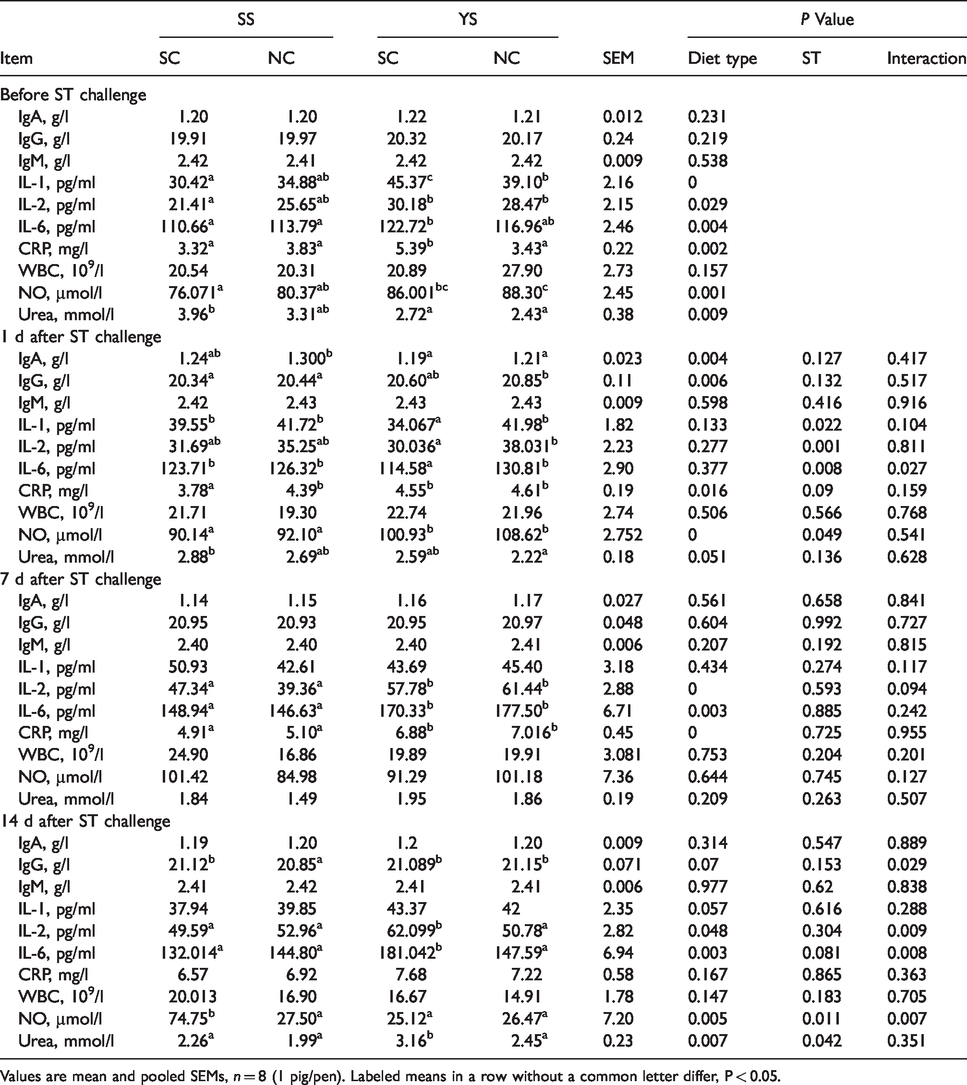
Values are mean and pooled SEMs, n = 8 (1 pig/pen). Labeled means in a row without a common letter differ, P < 0.05.
One d after ST challenge, piglets fed with YS had higher IgG content (P < 0.01), CAT activity (P < 0.05) and a lower IgA level (P < 0.01), and tended to have a lower proportion of urea compared to piglets fed with SS (P = 0.051). Concentrations of NO and CRP in YS group were remarkably higher than that in SS group (P < 0.01). Except for IL-6 concentration, there was no interaction between ST treatment and diet observed concerning the selected immune factors. Compared with pigs treated with ST, animals injected with saline had higher levels of IL-1, IL-2, and IL-6 (P < 0.05) compared with piglets treated with ST.
Seven d after ST challenge, no different changes in some indices were found in piglets fed between YS and SS diets, except that CRP, IL-2, and IL-6 levels in YS-SC group were higher than that in SS-SC group (P < 0.01). The ST treated piglets fed YS diet tended to have a higher MDA content than piglets of SS-NS group (P = 0.058). ST treatment and diet did not affect the indices shown in Tables 6 and 7.
Fourteen d post ST challenge, YS diet up-regulated the contents of urea, GSH-Px, IL-2, and IL-6 (P < 0.05), and down-regulated the NO level compared with piglets fed with SS (P < 0.01). Pigs fed YS tended to have a higher IgG content than that in pigs fed SS (P = 0.07). As shown in the present data, there was an interaction between ST challenge and diet observed in the abundance of IgG, SOD, NO, IL-2, and IL-6 (P < 0.05).
Discussion and conclusion
In the present study, dietary supplementation with YS resulted in a great increase of ADFI before the challenge. YS showed significant improvement in ADG, FCR and feed intake in chicken and cattle, which is consistent with the present findings. 13 YS acts as the anti-stimulation nutrient to attenuate the growth inhibition under infection and stress conditions and increases the immune response of weaning pigs.14,15 In addition, results of this study indicated that no significant effect of YS on the growth performance occurred in the following 2-wk period. The previous study on weaned piglets showed that the level of selenium meeting the requirement did not affect the performance of piglets. 16 Therefore, the difference was probably due to the selenium supplementation (0.375 mg/kg) in the two diets which was adequate for maintaining healthy growth of weaned pigs. We also measured the selenium content in SS and YS diets. The results were 0.445 ± 0.0396 mg/kg (pre-starter-SS, Table 1), 0.487 ± 0.0346 mg/kg (pre-starter-YS), 0.365 ± 0.0502 mg/kg (starter-SS), and 0.398 ± 0.0272 mg/kg (starter-YS). There were no significant differences between the two group diets in the same phases. Meanwhile, only SC group showed a trend to deduce ADG. Accordingly, it had been reported earlier that the ADG of weaned pigs was decreased in response to ST injection. 17 Also, it was reported that pigs challenged with ST (3 × 109 cfu) produced a physiological febrile and reduced ADG. 18 Based on this result and the previous study, it was possible that YS supplementation partially alleviated growth inhibition in the ST infection. Additionally, the model of ST challenge in weaned piglets was established successfully.
In investigations with weaned piglets, it was usual to evaluate the effectiveness of nutrient digestion by measuring the digestibility.19,20 Based on the results, titanium dioxide was selected as the exogenous indicator to evaluate the apparent digestibility of nutrients. 21 In this study, YS supplementation increased the digestibility of dry matter and crude protein before SC, and this was consistent with earlier findings. 22 Likewise, the appropriately improved apparent digestibility of nutrients in the total tract was found in diets supplemented with YS for lactating dairy cows and sheep.22,23 The results may be associated with the improvement of rumen microbial activity and the change of fermentation mode caused by YS addition. 23 The present results indicated that YS supplementation in weaned piglets diet could significantly improve the digestibility of nutrients in an experimental challenge model of ST. This might be because cells of selenium-enriched yeasts are rich in protein, B vitamins, fat, carbohydrates, enzymes, and some coordination factors.6,24 Therefore, it was likely that the selenium yeast product resulted in superior effects on improving nutrient digestibility to SS.
In the present study, we observed that dietary supplementation of YS improved ST and E. coli in feces compared with control piglets before ST challenge. However, several experiments have shown that YS supplementation reduced the fecal microflora in finishing pigs.25,26 It was reported that piglets which were fed the yeast diet had limited effects on microbial counts in fresh feces. 27 The different results might be explained by the experimental animals, proper doses of YS, and types of YS. Then, we found YS supplementation resulted in a decrease of ST compared with piglets fed SS after 1 d of ST challenge, which was consistent with previous reports. 26 Expectedly, ST up-regulated this bacterium in excrement microflora during the late stage of challenge, indicating that the treated pigs may be successful in infection. Nowadays, overpopulation of E. coli might cause diarrhea and the inhibition of animal growth performance has been accepted.28,29 A numerically significant decrease was found in E. coli of the fresh feces on d 21 of piglets fed YS diet, and a marked reduction of E. coli in ST-injected pigs fed selenium-enriched yeast at the last day before slaughter.
In the current study, we evaluated the effect of YS in the diet on cytokine production after stimulation. We simulated the pathological state of weaned piglets by infecting with ST. 18 ST invades the host gut by attacking specific members of the microbiota selectivity in the gastrointestinal tract. 30 ST is known as common Gram-negative intracellular bacterium which triggers inflammasome assembly via the cytosolic receptors (NLRP and NLRP4), thereby recruiting a series of cytokines and inflammatory medium.31,32 Thus, it was considered that the anti-oxidant should be able to contribute some effects of protection on cells and tissues against stimulation. The cellular immune response plays a vital role in the organism to response the intracellular pathogens by inhibiting pathogen replication and accelerating the clearance of infected cells. 33 T-Lymphocyte subset is commonly measured as indication for the immune status in pigs. 34 It was demonstrated that feeding YS could improve the immune-boosting of body, which was consistent with the data of our study. 35 We found that YS supplementation enhanced the numbers of CD4+ T-lymphocytes during d 1 to 7. Earlier, it had been reported that selenium-enriched yeast capsules improved immunity (i. e., the CD4+ T-lymphocytes count) of HIV-positive children, and it was indicated that the moderate nutritional doses of YS to the aged had an effectiveness of promoting a T-lymphocyte response for stimulation.36,37 However, selenium supplementation from this study had no significant effect on changes of the CD3+ and CD4+ lymphocyte levels when it was administered before infection on d 14, which had been present in previous research. 38 The reason why CD4+ lymphocyte cells would respond differently between the two periods was unclear. Maybe, the effects of YS were more evident in pre-feeding pigs which were just weaned, and then the piglets were accustomed to the daily diet after 7 d. The percentages of CD4+ lymphocyte and CD4+/CD8+ ratio of YS group was determined to have also improved during d 15–21 after a single infection in this study. Moreover, piglets fed YS developed the higher CD3+ lymphocyte, which was in agreement with a previous study in chicken, and an unexpected decrease of the CD8+ cell number. 39 This motivated us to investigate the reason. In general, there were significant effects of YS on reducing stress and enhancing cellular immunity when the body was under stress or infection, as shown in this study. To further explore the intuitionistic anti-inflammatory effects of YS in piglets, we investigated various inflammatory indices that responded to weaned pigs infected with ST, such as immune globulins (IgA, IgG, and IgM), antioxidant enzymes, cytokines (e.g. IL-1, IL-2, IL-6) and WBCs. In the present study, supplementation of YS remarkably improved all levels of IL-1, IL-2, IL-6, as well as the NO and CRP production compared with SS weaned pigs before infection. There were several reports showing that IL-l would stimulate macrophages and T lymphocytes to respond to inflammation, IL-2 can promote and maintain culturing of T cells over a long time, and IL-6 is responsible for elevating the differentiation and maturation of T lymphocytes to modulate immunity.40–42 This was possibly indicated by our finding that YS supplementation had a better impact in coping with residual effects of weaning stress than that of SS. It was reported that MDA, one of the critical products of membrane lipid peroxidation, also can aggravate the damage of membranes, which represents an indirect index of the degree of the impaired membrane system. 43 NO plays a key regulatory role in the inflammatory cascade, especially in the development of inflammatory responses and signaling. 44 CRP, a part of the body’s non-specific immune mechanisms, is an acute-phase protein synthesized by hepatocytes in case of inflammatory stimuli such as microbial invasion or tissue damage.45,46 Increase of NO, CRP, and decrease of MDA corresponded to changes of ILs in YS piglets. Urea is mainly affected by renal function, dietary protein intake, and catabolism, generally acting as biochemical evaluation of renal function. 47 Therefore, preliminary results showed that the decrease of urea in YS piglets might indicate better renal function, which was similar to earlier findings. 48 GSH-Px, an important peroxidase, is widely distributed in the body, and its viability and concentration can reflect the body’s selenium level. 49 In this study, we found that weaned piglets fed with YS had higher concentrations of GSH-Px in serum than pigs fed with selenite. Approximately, it was reported that Se-enriched yeast was more effective than SS in enhancing the blood GSH-Px activity in lambs. 50 Having investigated of the activity of GSH-Px in the serum of growing-finishing pigs, researchers indicated that pigs fed SS were more active of GSH-Px than selenium yeast, which contradict our results. 51 The effects of different Se sources on GSH-Px activity may be the difference between ruminants and non-ruminants, or Se of YS gets more deposited in tissues and leads to less GSH-Px in serum. SOD, the primary substance for scavenging free radicals, is an important antioxidant enzyme in the organism. 52 Experiments have shown that high levels of erythrocyte SOD activity are assumed to be due to increased levels of O2− or intense oxidative stimulation. 53 Consistent with this, we found that the concentration of SOD in serum increased in piglets fed SS, indicating that YS was more effective in alleviating oxidative stress than SS in weaned piglets.
One day after ST challenge, piglets fed YS increased the production of IgG, an immune globulin known to play an important role in host defense against several pathogens. The results showed a trend that the concentration of urea of YS treatment was also less than piglets fed SS, in agreement with the improved renal function in weaned piglets. CAT is considered as decomposition enzyme of H2O2 to give H2O and O2 in vitro and catalyze the oxidation of H donors. 54 Fortunately, we found that piglets fed YS had higher activity of CAT compared with SS pigs. All indicated that dietary YS supplementation might be more effective in eliminating H2O2 in the body. Uniformly, we found that NO and CRP in the serum of YS pigs were higher than those in pigs fed SS. Therefore, YS was associated with improved function of the inflammatory responses. It had been reported that data showed a downwards shift of IL-1 and IL-6 contents after 24 h infection of ST, which was consistent with our findings and indicated that the challenge might be successful at an inflammatory level. 55 On d 7 post-infection, the levels of IL-2 and IL-6 in piglets fed YS were higher than those in SS pigs, which might again indicate the function of YS in inflammation. During this period, groups YS and SS did not show discriminating effects on other serum indices. We found that the YS diet improved the levels of IgG, GSH-Px, IL-2, and IL-6, compared with SS treatment. So, based on the discussion above, these results might provide evidence that YS was more effective in enhancing immunity and alleviating oxidative stress. However, some of the effects of YS could not be explained by the present study, which motivates us to continue with further studies to fully evaluate the effects of YS in pigs.
In summary, dietary supplementation of selenium-enriched yeast exerted effects in piglets after ST infection. Particularly, YS seemed to be more effective in enhancing growth performance and nutrient digestibility, compared with SS. Also, YS supplementation improved kidney and immune functions, and alleviated oxidative stress according to serum biochemical indices (e.g., urea IL, immune globulins, GSH-Px, SOD) in weaned piglets.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
