Abstract
Objective
This study evaluated the clinical and pathological effects of the immunosuppressive agent mycophenolate mofetil (MMF) in rats with experimental autoimmune encephalomyelitis (EAE; a model of multiple sclerosis [MS]).
Methods
EAE rats were randomly divided into 4 groups: model alone (
Results
Oral administration of high-dose MMF significantly ameliorated the course of EAE in rats: cumulative clinical scores were lower and weight loss was less than in rats receiving methylprednisolone. The ameliorated disease course was associated with alleviation of histopathological signs of EAE. Treatment increased the blood proportion of CD8+, CD4+CD25+ and CD4+CD45RA+ T cells, with a concomitant reduced proportion of CD4+ T cells and ratio of CD4+ to CD8+ T cells, compared with EAE model alone rats.
Conclusions
MMF may have pharmacological potential in MS treatment and these findings may help in understanding the pathophysiological mechanism of MS.
Keywords
Introduction
Multiple sclerosis (MS) is the most common disabling central nervous system (CNS) disease in young adults. It is characterized by recurrent disease relapse and/or progression attributed to multifocal inflammation, demyelination and axonal pathology, within the brain and/or spinal cord. 1 Experimental autoimmune encephalomyelitis (EAE) is an animal model of MS. 2
Interferon-ß, methylprednisolone, glatiramer acetate, mitoxantrone and natalizumab have been approved for treating MS. 3 Despite their promising efficacy, therapeutic response is rarely optimal. Furthermore, adherence to parenteral forms of these drugs is challenging, which has implications for the effectiveness of treatment. 4 Modest efficacy, adherence and injection-related adverse effects have catalysed a series of investigations to identify an effective oral therapy for MS.
In clinical and animal studies, mycophenolate mofetil (MMF) has been an effective immunosuppressive agent in organ transplantation and in the treatment of some autoimmune diseases.
Most mammalian cells can use both salvage and de novo pathways for purine metabolism; however, antigen-activated B and T cells highly depend on de novo synthesis of purines. 10 MMF selectively inhibits inosine 5′-monophosphate dehydrogenase (IMD) type II, which is responsible for the de novo synthesis of the purine nucleotide guanine within activated lymphocytes and macrophages, without affecting purine salvage pathways. 11 MMF depletes GTP and dGTP, which are required for DNA synthesis by activated B and T cells. 12 Thus, inhibition of IMD by MMF limits the lymphocyte proliferation rate. MMF can inhibit T- and B-cell proliferation but does not affect the initial phases of activation, which include the early induction of cytokine production. 13 The depletion of intracellular GTP also limits the transfer of fucose and mannose to membrane glycoproteins, and inhibits the expression of adhesion molecules on leucocytes, thereby suppressing lymphocyte recruitment to target tissues. 11
Multiple sclerosis is considered to be a T cell-mediated autoimmune disease: this hypothesis is primarily based on the genetic susceptibility of individuals for the disease, the presence of immune cells within active MS plaques and animal models of CNS autoimmunity mediated by T cells.14–16 CD4+ T cells specific for many CNS proteins (including myelin associated glycoprotein, myelin basic protein, myelin oligodendrocyte glycoprotein and proteolipid protein) can be isolated from MS patients and healthy individuals.16–17 These data suggest that in patients with MS, CNS protein-specific CD4+ T cells become activated, cross the blood–brain barrier and induce CNS autoimmunity.15–17 Further support for this hypothesis comes from the many animal models of CD4+ T cell-mediated EAE. 16
Autoreactive CD4+ T cells alone are unlikely to fully account for all of the effector mechanisms that drive the disease process in MS. Regulatory CD8+ T cells may also contribute to limiting disease severity or occurrence in MS patients; early experiments have demonstrated that the defective suppressor function of CD8+ T cells in MS may contribute to the autoimmune process, which may underlie the pathogenesis of MS. 18 A few studies have suggested that CD8+ T cells are pathogenic in EAE: EAE is more severe in mice deficient in (or depleted of) CD8+ T cells, compared with other mice, and disease severity has been correlated inversely with the number of CD8+ T cells.19,20 More recently, longitudinal MRI revealed a significant negative correlation between the proportion of cytokine-producing CD8+ T cells in the peripheral blood of MS patients and the development of lesions. 21 Furthermore, the beneficial effects of treating MS with glatiramer acetate may in part be due to activating regulatory CD8+ T cells. 22 CD8+ T cells that can lyse myelin-specific CD4+ T cells have been detected in MS patients, and vaccination of such patients with irradiated myelin-specific CD4+ T cells induced CD8+ T cells that could specifically kill these CD4+ T cells.16,23,24 Mouse models have also shown potent disease-modifying effects of CD8+ T regulatory cells through the secretion of interleukin 10 and other soluble mediators, regulation of antigen-presenting cell (APC) function, and eliminating activated CD4+ T cells by CD8+ T cells, via recognition of the nonclassical Qa-1 major histocompatibility complex.16,25,26
In the present study, the effect of MMF and methylprednisolone in rats with EAE was investigated. The maximal dose of MMF tolerated by animals was compared with a methylprednisolone maximum dose effect on EAE in rats.13,27,28 The effect on blood proportion of CD4+, CD8+, CD4+CD25+ and CD4+CD45RA+ T cells, and the ratio of CD4+/CD8+ T cells was examined, as well as pathological changes in brain and spinal cord.
Materials and methods
Animals
Fifty female Wistar rats (6–8 weeks old; weight 180–220 g) were obtained from the Animal Experiment Centre of Shandong University (Jinan, China); 15 female guinea pigs (6–8 weeks old; weight 350–400 g) were obtained from the Animal Experiment Centre of Zhongxingwang (Jinan, China). Animals were housed with a standard 12-h light/12-h dark cycle in the animal experiment centre of the Second Hospital of Shandong University, with temperature and relative humidity 22℃ and 65–70%, respectively. After 1 week’s adaptation and feeding with a standard-mixed feed, the animals were used for experiments. The experimental procedures were approved by the Ethics Commission for Experiments on Animals of Shandong University and were conducted in accordance with international standards (the UK Animals [Scientific Procedures] Act, 1986 and associated guidelines, the European Communities Council Directive of 24 November 1986 [86/609/EEC], as previously described). 29
EAE induction
Experimental autoimmune encephalomyelitis was induced, as described previously.
28
Guinea pig spinal-cord homogenate was prepared in normal saline and emulsified in an equal volume of incomplete Freund’s adjuvant (Sigma-Aldrich, St. Louis, MO, USA) containing a suspension of 10 mg/ml of killed and dried
Experimental groups and treatment protocols
Rats with EAE were randomly divided into four groups for treatment: model alone (
To evaluate the EAE model, two rats with EAE were killed after functional evaluation on Day 14 p.i. The rats were deeply anaesthetized with 10% chloral hydrate, then were immediately cardiac perfused with ice-cold phosphate buffer solution (PBS, 0.2 M, pH 7.4) and 4% paraformaldehyde. Next, spinal cords and brains were excised. The remaining rats were killed at Day 29 p.i.; blood samples were taken and spinal cords and brains were excised for pathology and analysis of mononuclear cell phenotype, as described below.
Histology
Rats were anaesthetized by intraperitoneal injection of 10% chloral hydrate to collect blood at Day 29 p.i. for lymphocyte analysis as described below. The rats were then killed and cold PBS (0.2 M, pH 7.4 at 4℃) was perfused through the left ventricle for 3 to 5 min. Brains and spinal cords were removed and fixed in 4% paraformaldehyde, then embedded in paraffin wax. Paraffin wax sections were cut into 5 -µm-thick sections and stained with haematoxylin and eosin to assess inflammation, 33 and with modified trichrome to assess demyelination. 34 Tissue samples were classified by three pathologists who evaluated the inflammatory infiltration and demyelination, and who were blinded to the samples and treatments they received.
Flow cytometry analysis of lymphocytes
The blood proportions of CD4+, CD8+, CD4+CD25+ and CD4+CD45RA+ T cells, and the ratio of CD4+/CD8+ T cells, was assessed by fluorescence-activated cell sorting flow cytometry. Whole-blood samples (100 µl) taken on day 29 p.i. were treated with ethylenediamine tetra-acetic acid (2.25 mg/ml) and analysed within 6 h of sampling. The samples were incubated at room temperature (20–25℃) for 20 min with 10 µl of the appropriate monoclonal antibodies: anti-CD4, anti-CD8, anti-CD25 and anti-CD45RA labelled with fluorescein isothiocyanate, and phycoerythrin; all antibodies were obtained from BD Biosciences, San Jose, CA, USA, and were used according to the manufacturer’s protocol. Erythrocytes were lysed using BD FACS™ Lysing Solution; BD Biosciences, diluted 1 : 10 with deionized water at room temperature (20–25℃), then incubated at room temperature for 10 min. The samples were centrifuged at 1500 r.p.m. for 5 min at room temperature (20–25℃) (Biofuge™ Stratos, ThermoFisher Scientific, Waltham, MA, USA). The supernatants were discarded and the samples were washed with 2 ml PBS (0.2 M, pH 7.4). Then, samples were again centrifuged at 1500 r.p.m. (Biofuge™ Stratos) for 5 min at room temperature (20–25℃). Supernatants were discarded and samples were fixed with 0.5 ml 1% paraformaldehyde in PBS (0.2 M, pH 7.4), then analysed by flow cytometry FACSCalibur™; BD Biosciences) for T-cell populations, using methods described previously. 35 Data analyses were performed using CellQuest™ software, version 6.0 (BD Biosciences). Data were expressed as the proportion of positive cells (compared with cells stained with an irrelevant control antibody) or total number of positive cells.
Statistical analyses
Data were presented as mean ± SD. Differences in maximal disease and weight loss between the different groups were analysed by independent-samples
Results
Control rats were all neurologically asymptomatic, with normal increase in weight. Of the rats that were immunized, 32 of 44 animals showed EAE. The incidence of EAE was 72.1% (Duckers scores: 1:
Treated rats showed clinical recovery from 17–29 days p.i., when rats gradually gained weight. The Duckers scores of the model alone group continuously increased until 21days p.i.; however, the Duckers scores of the animals treated with MMF and methylprednisolone decreased gradually after treatment (Figure 1). The Duckers scores of the high- and low-dose MMF groups were significantly lower than the model alone group on Day 21 p.i. ( Assessment of neurological signs (as assessed by Duckers scores31) in rats with experimental autoimmune encephalomyelitis (EAE), 14 days after treatment with low- (20 mg/kg per day) or high-dose (30 mg/kg/ per day) mycophenolate mofetil (MMF) or methylprednisolone (20 mg/kg per day), and also in untreated EAE model alone rats and healthy control rats. Treatment was started on Day 15 after EAE induction. Data presented as mean ± SD of the groups. *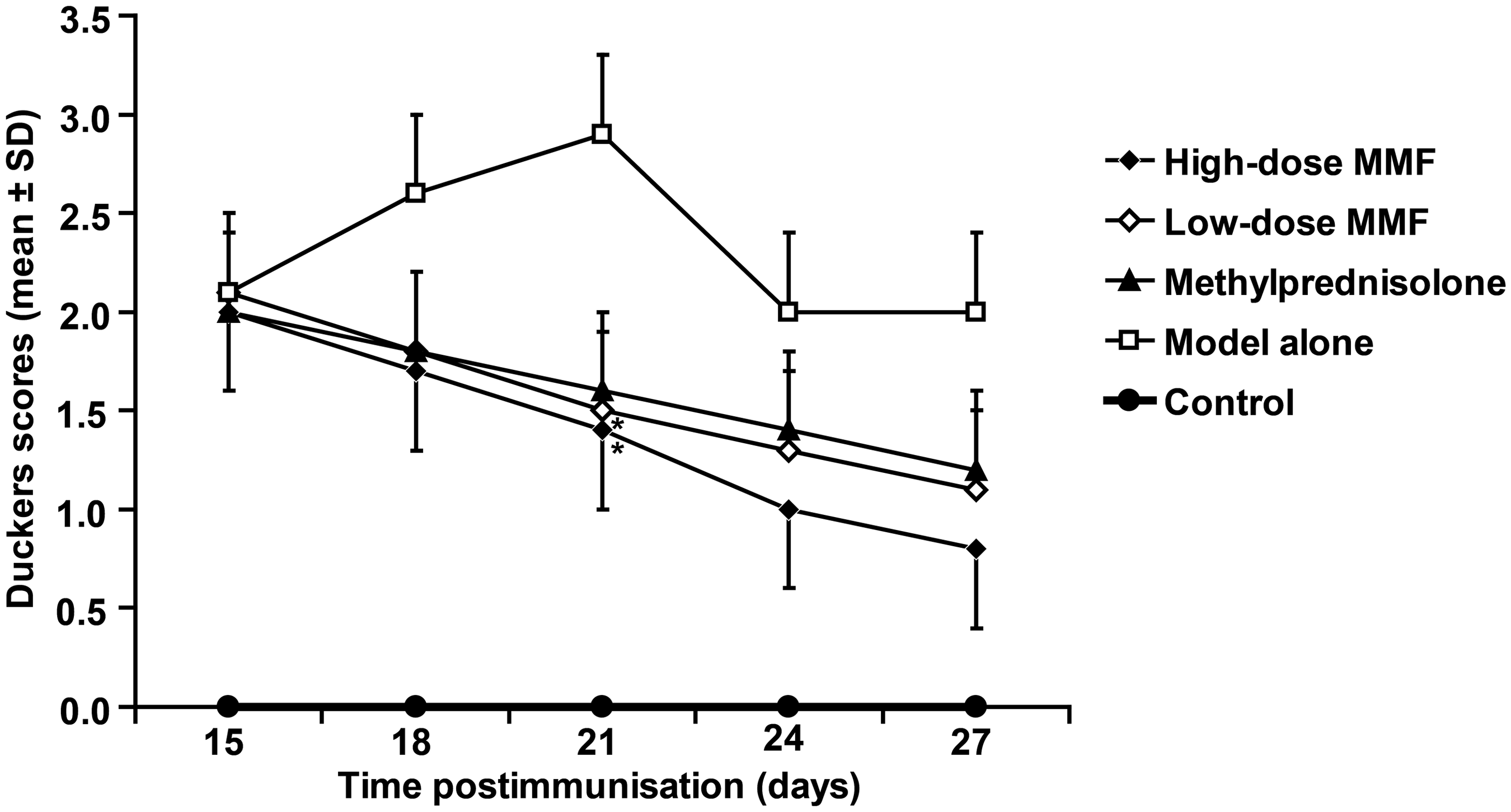
The mean ± SD weight of all rats with EAE before and at 14 days p.i. was 189.80 ± 13.43 g versus 176.60 ± 22.31 g. Low-dose MMF and methylprednisolone treatment resulted in significant weight gain compared with the model alone group during the treatment phase ( Assessment of weight of rats with experimental autoimmune encephalomyelitis (EAE) 14 days after treatment with low- (20 mg/kg per day) or high-dose (30 mg/kg per day) mycophenolate mofetil (MMF) or methylprednisolone (20 mg/kg per day), and also in untreated EAE model alone rats and healthy control rats. Treatment started on Day 15 after EAE induction. Data presented as mean ± SD of the groups. *
The HE staining showed that spinal cord lesions in rats with EAE were distributed almost entirely in the white matter; most were irregular and perivascular with many layers of inflammatory cell infiltration, which had the appearance of “vascular cuffing”. The control group showed no abnormal changes in neurological histopathology, which was consistent with clinical manifestations. Compared with the model alone group, the treated groups (especially the high-dose MMF group) showed alleviated inflammatory infiltration and vascular cuffing. Modified trichrome staining showed that the model alone rats had histopathological changes of myelin-sheath swelling, disintegration and demyelination in the spinal cord. The impairment was more severe than in the brain, especially in the white matter of the lumbosacral segment. The control group showed no abnormal changes in neurological histopathology features. Compared with the model alone group, the treated groups (especially the high-dose MMF group) showed alleviated myelin-sheath swelling, disintegration and demyelination.
Compared with the control group, the model alone and treatment groups showed increased proportions of CD4+ T cells and decreased proportions of CD8+ T cells (Figure 3). Additionally, the proportions of CD4+CD25+ and CD4+CD45RA+ T cells were lower in the model alone and treatment groups compared with controls (Figure 3). The ratio of CD4+/CD8+ T cells was higher in the model alone and treatment groups compared with controls (Figure 4). Compared with the model alone group, treatment groups showed lower proportions of CD4+ T cells and higher proportions of CD8+ T cells (Figure 3). The ratio of CD4+/CD8+ was lower, and the proportion of CD4+CD25+ T cells and CD4+CD45RA+ T cells was higher, in the treatment groups, especially in the high-dose MMF group; in adition, there was no significant difference between low-dose MMF and methylprednisolone (Figures 3 and 4).
Proportions of CD4+, CD8+, CD4+CD25+ and CD4+CD45RA+ T cells in blood samples from rats with experimental autoimmune encephalomyelitis (EAE), 14 days after treatment with low- (20 mg/kg per day) or high-dose (30 mg/kg per day) mycophenolate mofetil (MMF) or methylprednisolone (20 mg/kg per day) and also in untreated EAE model alone rats and healthy control rats. Treatment was started on Day 15 after induction of EAE. * Ratio of CD4+/CD8+ T cells in blood samples from rats with experimental autoimmune encephalomyelitis (EAE), 14 days after treatment with low- (20 mg/kg per day) or high-dose (30 mg/kg per day) mycophenolate mofetil (MMF) or methylprednisolone (20 mg/kg per day) and also in untreated EAE model alone rats and healthy control rats. Treatment was started on Day 15 after induction of EAE. *

Discussion
The effects of the immunosuppressive agent MMF and methylprednisolone on EAE in a rat model of MS were investigated in this study. A monophasic remission of disease with MMF or methylprednisolone, after the onset of clinical signs in this rat model of MS, was observed. Treatment with MMF at Day 15 p.i. had a similar effect to methylprednisolone, in that it prevented further deterioration of clinical paralysis and weight loss, which are both associated with EAE. HE staining of spinal cords showed ameliorated inflammatory infiltration and “vascular cuffing” of treatment groups, especially with high-dose MMF. Modified trichrome staining showed alleviated myelin-sheath swelling, disintegration and demyelination in spinal cords after treatment. High-dose MMF was more effective than methylprednisolone in reversing established EAE, with no significant difference being observed between low-dose MMF and methylprednisolone.
Treatment of MS consists of direct symptom management, brief corticosteroid administration for acute exacerbations and the regular use of disease-modifying drugs (DMDs). DMDs, including interferon β-1a and -1b, glatiramer acetate, natalizumab and mitoxantrone are licensed worldwide to reduce relapse frequency in patients with MS. Despite their promising efficacy, many patients show poor response. 36 Furthermore, the principal DMDs are injected or are given by intravenous infusion, which can be associated with reduced convenience and compliance as well as with injection- or infusion-related adverse effects. 36 Therefore, much attention has been paid by the MS community to clinical programmes that employ oral drugs, including MMF, fingolimod and other selective immunomodulatory or immunosuppressive agents, which may serve to overcome some of the limitations of previous therapies, and may help to improve adherence. Michel et al. conducted a multicentre retrospective study, in which the annualized relapse rate and the Expanded Disability Status Scale score were assessed: their results suggested that MMF can improve or stabilize MS patients and can be used as an alternative form of therapy. 37 However, despite intensive research, the mechanism of action of MMF in MS is still not precisely known. In the present study, levels of CD4+ T cells in blood samples taken from rats with EAE were increased, compared with levels in blood samples from control group rats. This result is consistent with other research which showed that CD4+ and CD8+ T cells play important roles in disease pathogenesis. 38 Compared with the model alone group, treatment with MMF or methylprednisolone decreased the proportion of CD4+ T cells in the blood. MMF may therefore affect immune suppression by decreasing the proportion of CD4+ T cells. Downmodulating inflammation with MMF may diminish disease symptoms in EAE during an inflammatory episode.
In addition to promoting inflammation, the CD4+ T-cell lineage can also prevent and downmodulate inflammation via specific CD4+ T regulatory cells (Tregs) and other regulatory feedback mechanisms. 39 CD4+CD25+ Tregs, named nTregs, are a subtype of CD4+ T cells developed in the thymus that can express high levels of CD25 (interleukin-2Rα). 40 The nTregs constitute 5–10% of peripheral CD4+ T cells in humans and mice. 41 CD4+CD25+ Tregs can suppress CD4+ and CD8+ T-cell proliferation and cytokine production. 42 nTregs can also suppress B cells, natural killer cells, monocytes/macrophages and dendritic cells, thus affecting both innate and adaptive immunity. 42 A decreased population of CD4+CD25+ Tregs in acute-stage MS has been found, as well as restored function and proportion of CD4+CD25+ Tregs in monkeys with remitting EAE. 43 Sustained reduction of the frequency of CD4+CD25+ Tregs was found in MS patients. 44 Furthermore, the effector function of CD4+CD25+ regulatory T cells from peripheral blood was found to be lower in patients with MS than in healthy donors. 45
A subset of naïve CD4+CD45RA+ regulatory T cells has been detected in peripheral blood and lymphoid organs, and is thought to represent precursor cells of antigen-experienced CD45RO+ Tregs, analogous to the development of conventional memory T cells. 46 The CD4+CD45RA+ population is reduced after antigen stimulation, so their numbers are decreased in immune stress conditions such as autoimmune diseases, 39 which confirms the hypothesis that CD4+CD45RA+ T cells play a key role in EAE. Progressive MS shows a selective loss of circulating CD4+CD45RA+ Tregs;. CD4+CD45RA+ T cells can travel to the secondary lymphoid organs, where they encounter APCs. After contact with APCs loaded with an antigen, CD4+CD45RA+ T cells become activated and start to proliferate. They express high levels of FOXP3 and possess an equivalently strong suppressive capacity, compared with their memory counterparts.47–49
Together, these data suggest that both CD4+ and CD8+ T cells are involved in MS and EAE pathogenesis. The present study found lower proportions of CD4+CD25+, CD4+CD45RA+ Tregs and CD8+ T cells in blood samples from rats with EAE than in samples from control rats. These results are consistent with previous findings. 49 Compared with the model alone group, MMF and methylprednisolone treatment increased the blood proportion of CD4+CD25+, CD4+CD45RA+ Tregs and CD8+ T cells. MMF might, therefore, effect immune suppression by increasing the proportion of CD4+CD25+, CD4+CD45RA+ Tregs and CD8+ T cells.
In the present study, MMF was more effective than methylprednisolone in preventing established EAE, and it may therefore be a viable therapy for MS. The results of this study may help to understand the pathophysiological mechanisms of MS and the possible therapeutic role of MMF in this disease. The therapies available for MS have increased tremendously over the last few years. In addition to the licensed corticosteroids and DMDs, there are now more drugs close to registration, the most promising of which are MMF, cladribine, BG-12, teriflunomide, laquinimod (ABR-215062) and fingolimod (FTY720). 50 Although a final cure for MS is still missing, MS is likely to be manageable for most patients with these treatments.
In conclusion, MMF treatment was found to be associated with reduced infiltration of lymphocytes in the brain and spinal cord of rats with EAE. Examination of the mechanism of action of MMF in EAE, through the analysis of subsets of T lymphocytes and ratio of CD4+/CD8+, showed that MMF may effect immune suppression by decreasing the proportion of CD4+ T cells and the ratio of CD4+/CD8+ T cells, and by increasing the proportion of CD8+, CD4+CD25+ and CD4+CD45RA+ T cells.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research was supported by Shandong Province Natural Science Foundation [grant number ZR2010HL039].
Acknowledgements
We gratefully acknowledge the assistance of Chengjun Zhou, Tingguo Zhang and Bingxin Guan (Department of Pathology, Second Hospital of Shandong University, China) in observing the sections and evaluating the inflammatory infiltration and demyelination.
