Abstract
We performed a meta-analysis to evaluate potential associations between vitamin D receptor (VDR) genetic variants and tuberculosis (TB). Systematic literature research was conducted in PubMed, Web of Science, and Embase. We calculated odds ratios (ORs) and 95% confidence intervals (CIs) to estimate strength of associations in all possible genetic models, and P values ≤ 0.05 were considered to be statistically significant. In total, 42 studies were enrolled for analyses. Pooled overall analyses suggested that VDR rs1544410 (dominant model: P = 0.02; allele model: P = 0.03) and rs731236 (dominant model: P = 0.04; recessive model: P = 0.02; allele model: P = 0.01) variants were significantly associated with TB. Further subgroup analyses by ethnicity revealed that rs1544410 (dominant and allele models) and rs731236 (dominant, recessive, and allele models) variants were both significantly associated with TB in South Asians. When we stratified data by type of disease, positive results were detected for rs7975232 variant in EPTB (dominant, recessive, over-dominant, and allele models) subgroup, and for rs2228570 variant in PTB (dominant, recessive, and allele models) and EPTB (dominant, recessive, over-dominant, and allele models) subgroups. Our meta-analysis supported that rs7975232, rs1544410, rs2228570, and rs731236 variants might serve as genetic biomarkers of certain types of TB.
Keywords
Introduction
Tuberculosis (TB) is a commonly seen chronic infectious disorder which includes pulmonary tuberculosis (PTB) and extrapulmonary tuberculosis (EPTB). 1 In spite of rapid advancements achieved in early diagnosis and pharmacological therapy over the past few decades, TB remains a serious public health problem. According to a recent investigation, over 30% of the general population is infected with Mycobacterium tuberculosis (MTB), and around 5–10% of these infected individuals will eventually develop active TB. 2 The course of MTB infection depends on a complex interaction of pathogen, host, and environmental factors, and the fact that only a small portion of infected individuals finally develop active TB suggests that host genetic background may play a crucial role in its development.3,4
Recently, it became evident that the vitamin D metabolic pathway might be involved in the pathogenesis of TB. First, previous epidemical investigations showed that vitamin D deficiency was much more prevalent in patients with TB, and the serum level of vitamin D was reversely correlated with disease severity.5–7 Second, several experimental studies demonstrated that vitamin D could activate macrophages and promote elimination of MTB.8–10 It is well acknowledged that vitamin D exerts its biological functions by binding with vitamin D receptor (VDR). Therefore, it is possible that VDR variants, which may result in diminished function of vitamin D, might also be involved in the development of TB.
To date, numerous studies already investigated potential associations between VDR variants and TB. But the results of these studies were not consistent.11–52 Thus, we performed the present meta-analysis to obtain a more conclusive result.
Materials and methods
Literature search and inclusion criteria
This meta-analysis followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guideline. 53 Potentially relevant literature published before January 2019 was retrieved from PubMed, Web of Science, and Embase using the following searching strategy: (vitamin D receptor OR VDR) AND (polymorphism OR variant OR mutation OR genotype OR allele) AND (tuberculosis OR TB). We also checked the references of enrolled articles to identify other potentially related studies.
To test the research hypothesis of this meta-analysis, included studies must meet all the following criteria: (1) case-control study on associations between VDR variants and TB; (2) provide genotypic/allelic frequency of investigated VDR variants in cases and controls; (3) full text in English available. Studies were excluded if one of the following criteria was fulfilled: (1) not relevant to VDR variants and TB; (2) case reports or case series; (3) abstracts, reviews, comments, letters, and conference presentations. For repeated reports, we only included the study with the largest sample size for analyses.
Data extraction and quality assessment
We extracted following data from included studies: (1) the name of the first author; (2) publication time; (3) country and ethnicity; (4) sample size; and (5) genotypic/allelic distribution of VDR variants in cases and controls. The P value of the Hardy–Weinberg equilibrium (HWE) was also calculated. When necessary, we wrote to the corresponding authors for extra information. We used the Newcastle–Ottawa scale (NOS) to assess the quality of eligible studies. 54 This scale has a score range of 0–9, and studies with a score of more than 7 were thought to be of high quality. Data extraction and quality assessment were performed by two independent reviewers. Any disagreement between two reviewers was solved by discussion until a consensus was reached.
Statistical analyses
We used Review Manager Version 5.3.3 (The Cochrane Collaboration, Software Update) to conduct statistical analyses. We calculated odds ratios (ORs) and 95% confidence intervals (CIs) to estimate strength of associations in all possible genetic models, and P values ≤0.05 were considered to be statistically significant. Q test and I2 statistic were employed to assess between-study heterogeneities. If the P value of Q test was less than 0.1 or I2 was greater than 50%, random-effect models (REMs) were used to pool the data. Otherwise, fixed-effect models (FEMs) were applied for synthetic analyses. Subgroup analyses by ethnicity of participants and type of disease were performed. Stabilities of synthetic results were evaluated with sensitivity analyses, and publication biases were evaluated with funnel plots.
Results
Characteristics of included studies
We found 370 potentially relevant articles. Among these articles, 42 eligible studies were finally included for pooled analyses (see Figure 1). The NOS score of eligible articles ranged from 7 to 8, which indicated that all included studies were of high quality. Baseline characteristics of included studies are summarized in Table 1.11–52
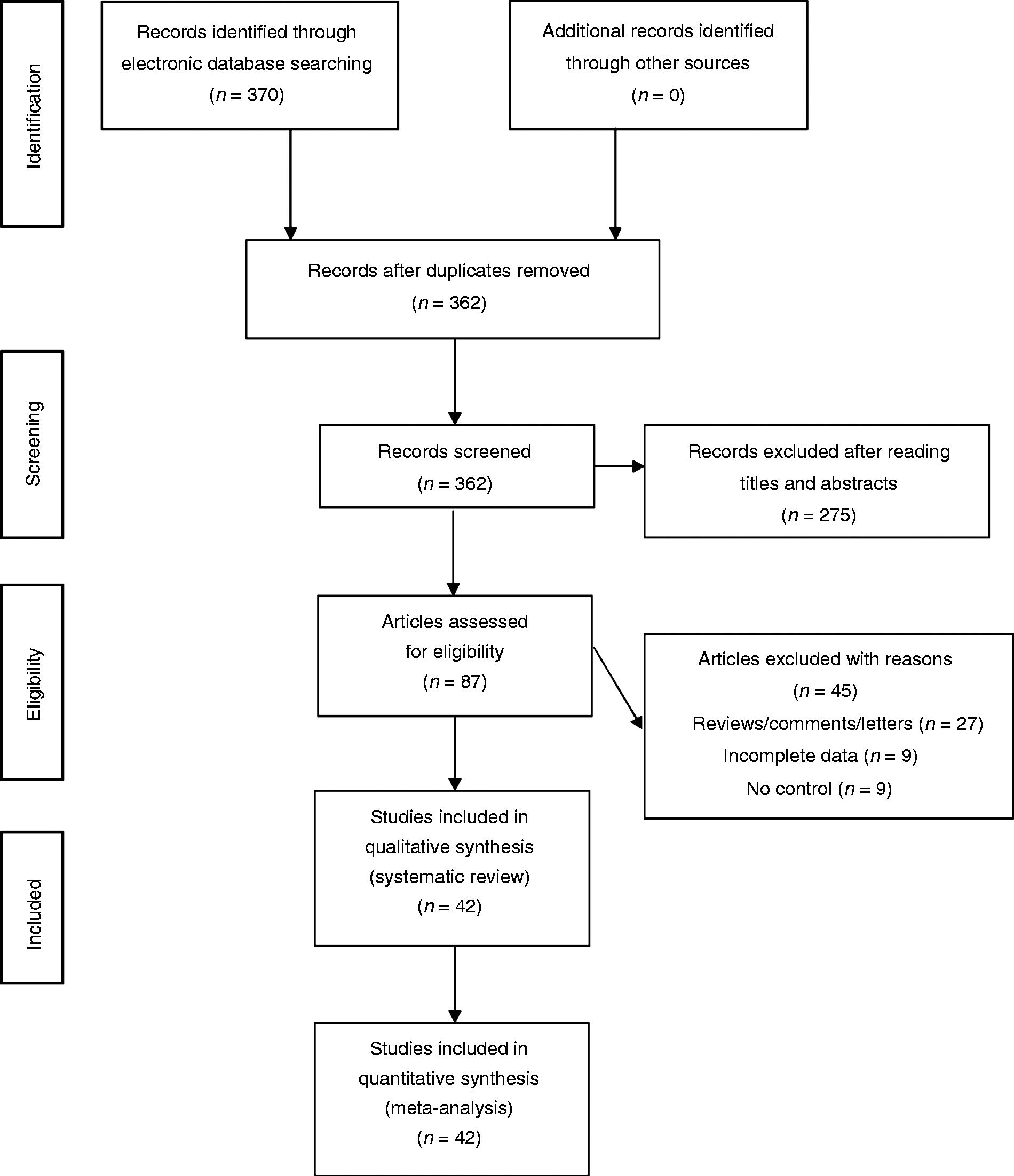
PRISMA diagram for the selection of studies of the present meta-analysis.
The characteristics of included studies.
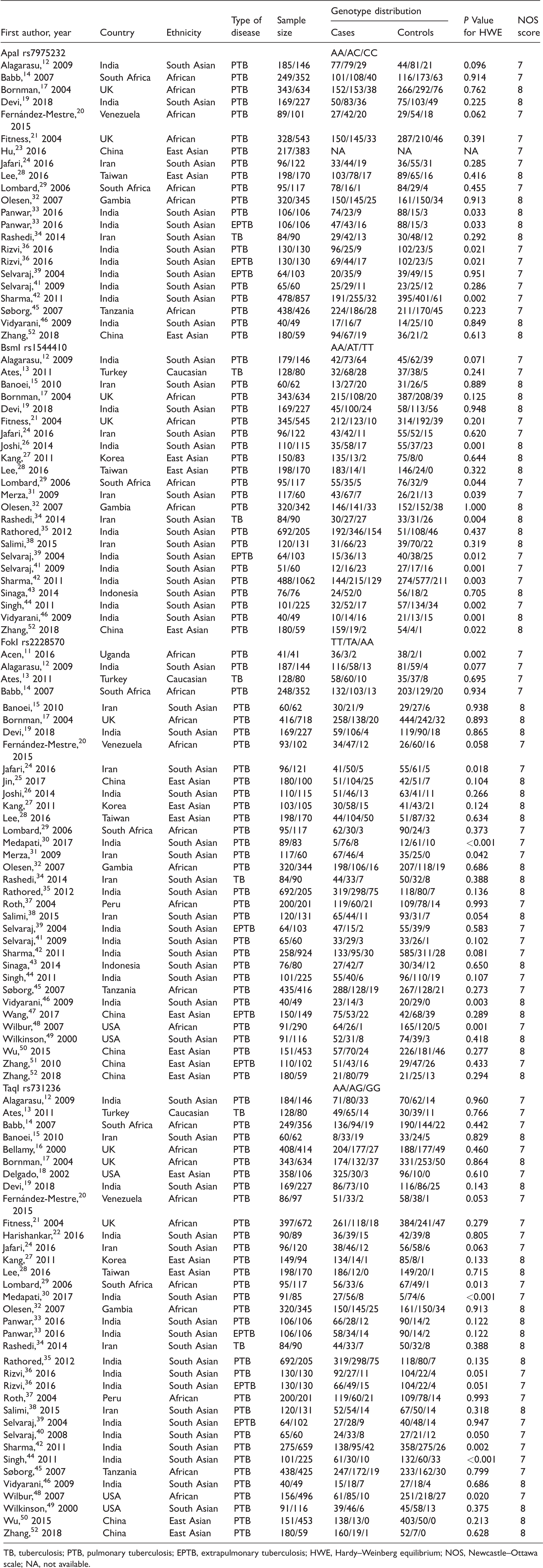
TB, tuberculosis; PTB, pulmonary tuberculosis; EPTB, extrapulmonary tuberculosis; HWE, Hardy–Weinberg equilibrium; NOS, Newcastle–Ottawa scale; NA, not available.
Overall and subgroup analyses
Pooled overall analyses suggested that VDR rs1544410 (dominant model: P = 0.02, OR = 0.79, 95% CI 0.65–0.96, I2 = 71%, REM; allele model: P = 0.03, OR = 0.86, 95% CI 0.75–0.99, I2 = 70%, REM) and rs731236 (dominant model: P = 0.04, OR = 0.85, 95% CI 0.73–1.00, I2 = 74%, REM; recessive model: P = 0.02, OR = 1.38, 95% CI 1.05–1.82, I2 = 69%, REM; allele model: P = 0.01, OR = 0.84, 95% CI 0.74–0.97, I2 = 79%, REM) variants were both significantly associated with TB.
Further subgroup analyses by ethnicity revealed that rs1544410 (dominant and allele models) and rs731236 (dominant, recessive, and allele models) variants were both significantly associated with TB in South Asians. When we stratified data by type of disease, positive results were detected for rs7975232 variant in EPTB (dominant, recessive, over-dominant, and allele models) subgroup, and for rs2228570 variant in PTB (dominant, recessive and allele models) and EPTB (dominant, recessive, over-dominant, and allele models) subgroups. No any other positive findings were observed in overall and subgroup analyses (see Table 2 and Supplemental Figure 1).
Results of overall and subgroup analyses.
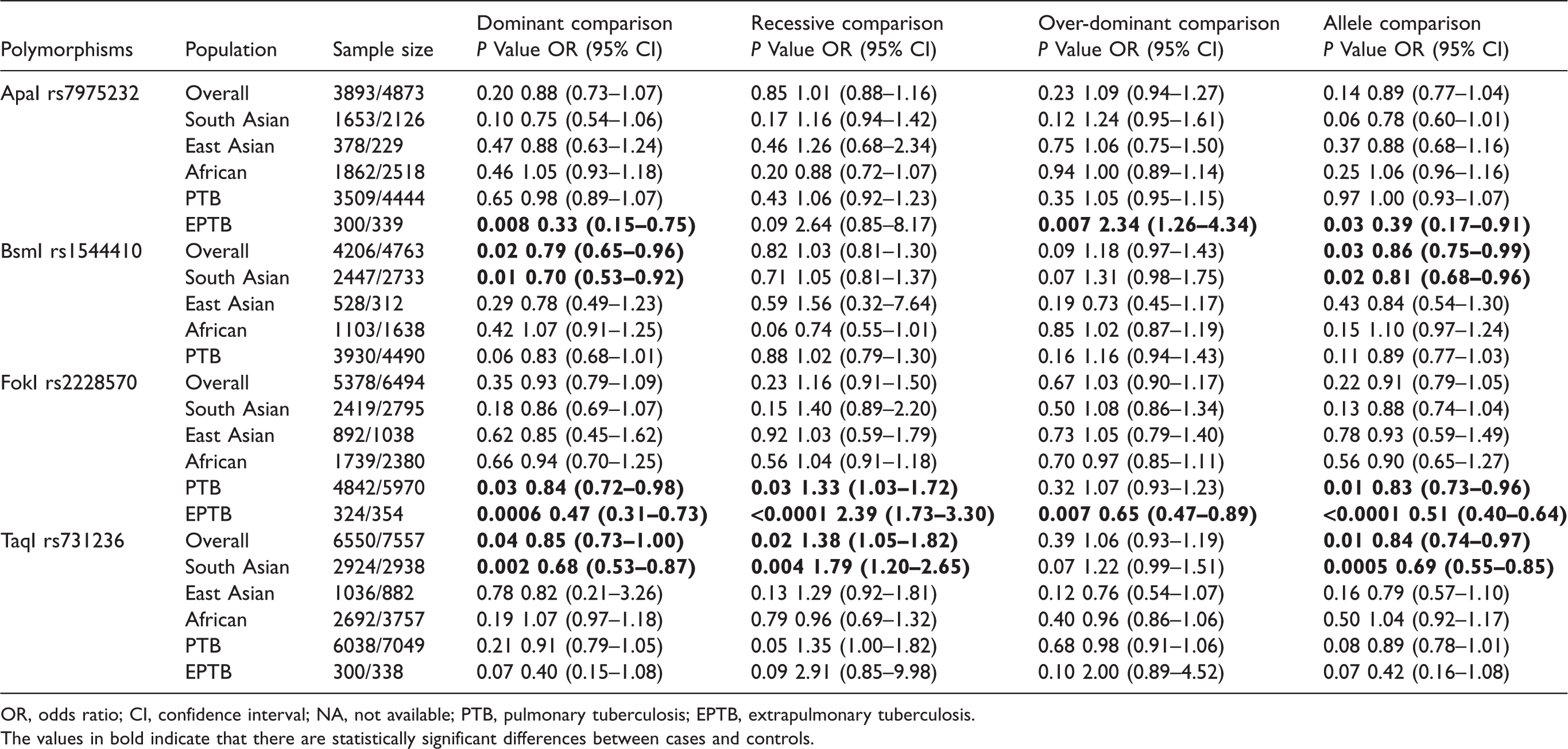
OR, odds ratio; CI, confidence interval; NA, not available; PTB, pulmonary tuberculosis; EPTB, extrapulmonary tuberculosis.
The values in bold indicate that there are statistically significant differences between cases and controls.
Sensitivity analyses
We performed sensitivity analyses to test stabilities of pooled results by excluding studies that violated HWE. No any altered results were observed in overall and subgroup comparisons, which indicated that our findings were statistically stable.
Publication biases
We used funnel plots to assess publication biases. We did not find obvious asymmetry of funnel plots in any comparisons, which suggested that our findings were unlikely to be impacted by severe publication biases.
Discussion
To the best of our knowledge, this is so far the most comprehensive meta-analysis on roles of VDR variants in TB, and our pooled analyses suggested that VDR rs7975232, rs1544410, rs2228570, and rs731236 variants were all significantly associated with certain types of TB.
There are several points that need to be addressed about this meta-analysis. First, although the investigated VDR variants were intensively analyzed with regard to their potential associations with TB, the functional significances of these variants were still undetermined,55,56 and thus future investigations still need to explore the underlying molecular mechanisms of our positive findings. Second, the pathogenic mechanism of TB is highly complex, and therefore it is unlikely that a single genetic variant could significantly contribute to their development. So to better illustrate potential associations of certain genetic variants with TB, we strongly recommend further studies to perform haplotype analyses and explore potential gene-gene interactions.
As with all meta-analysis, this study certainly has some limitations. First, our results were based on unadjusted analyses, and we have to admit that lack of further adjusted analyses for potential confounding factors might impact the reliability of our findings. 57 Second, associations between VDR variants and TB might also be modified by gene–gene and gene–environmental interactions. However, most eligible studies ignore these potential interactions, which impeded us to perform relevant analyses accordingly.58,59 Third, only retrospective case-control studies were included in this meta-analysis, and thus direct causal relation between investigated variants and TB could not be established. 60 On account of above mentioned limitations, our findings should be cautiously interpreted.
In conclusion, our meta-analysis suggested that VDR rs7975232, rs1544410, rs2228570, and rs731236 variants might serve as genetic biomarkers of certain types of TB. However, further well-designed studies are still warranted to confirm our findings. Moreover, future investigations also need to explore potential roles of other VDR variants in the development of TB.
Supplemental Material
Supplemental material for Associations between vitamin D receptor genetic variants and tuberculosis: a meta-analysis
Supplemental Material for Associations between vitamin D receptor genetic variants and tuberculosis: a meta-analysis by Xun Xu and Minghao Shen in Innate Immunity
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
