Abstract
Here, the potential mechanisms of the protective effects of fish oil against LPS-induced liver injury in a piglet model were investigated by using RNA sequencing. Twenty-four piglets were used in a 2 × 2 factorial design, and the main factors included diet (5% corn oil or 5% fish oil) and immunological challenge (LPS or saline, on d 19). All piglets were slaughtered at 4 h after challenge, and liver samples were collected. Fish oil improved liver morphology and reduced TNF-α, IL-1β and IL-6 productions after LPS challenge. RNA sequencing analysis showed fish oil had significant effect on the expressions of genes involved in immune response during LPS-induced inflammation. Selected gene expression changes were validated using quantitative RT-PCR. Fish oil reduced the expressions of pro-inflammatory genes IL1R1, IL1RAP, CEBPB and CRP, and increased that of anti-inflammatory genes IL-18BP, NFKBIA, IFIT1, IFIT2 and ATF3. Moreover, fish oil restored the expressions of some lipid metabolism-related genes, such as ACAA1, ACACA, ACADS and ACADM, which were only decreased in pigs fed a corn oil diet after LPS challenge. Our RNA sequencing reveals novel gene-nutrient interactions following fish oil supplementation and evoked inflammation, which add to the current understanding of the benefits of n-3 polyunsaturated fatty acids against liver injury.
Introduction
The liver, the main metabolic and detoxification organ, is critical for host defense and tissue repair in severe infection and sepsis. 1 In liver, Kupffer cells are the tissue macrophages and serve as a gatekeeper. However, in sepsis, the activated Kupffer cells produce large amounts of pro-inflammatory cytokines and chemokines, which lead to hepatic dysfunction and acute liver injury. 2
LPS, a vital trigger of sepsis, is found in the outer membrane of Gram-negative bacteria. Recent evidences from animal models indicate that LPS-induced acute liver injury is caused by a variety of factors including inflammation, oxidative stress and lipid metabolism disorders. 3 , 4 LPS can initiate excessive activation of immune cells such as macrophages and neutrophils through recognition by TLR4. 5 MyD88 is a key adaptor molecule for TLR signal. The TLR4/MyD88-mediated pathway activates NF-κB to induce pro-inflammatory cytokine production, such as TNF-α, IL-1β and IL-6. 6 In addition, the signaling downstream of TLR4 is mediated by the adaptor proteins TRIF (Toll–IL-1 receptor (TIR) domain–containing adaptor-inducing IFN-β). LPS-induced activation of the TLR4/TRIF-NLRP3 inflammasome axis is important for the development of liver injury. 7 Therefore, the ability to modulate an excessive liver inflammatory response is crucial to attenuate the progression of the liver injury.
Intake of fish oil (FO) rich in n-3 polyunsaturated fatty acids (PUFAs) is believed to be beneficial against liver injury, because of the benefits of n-3 PUFAs in lipid metabolism modification, oxidative stress and hepatic inflammation.8–10 n-3 PUFAs have anti-inflammatory activity because the NF-κB pathway can be inhibited by eicosahexaenoic acid (EPA (20:5(n-3))) and docosahexaenoic acid (DHA (22:6(n-3))). 11 In addition, recent studies indicate n-3 PUFAs attenuate inflammation associated with inhibition of TLR4 and NOD signaling pathways. 12 , 13 However, the underlying mechanisms remain unclear.
The piglet was evaluated as a good model animal for human nutrition and physiology research. 14 In this study, we established an evoked liver inflammation in pig model by injecting Escherichia coli LPS. We aimed to identify novel n-3 PUFA modulated gene expression using RNA sequencing during evoked inflammation in liver tissues.
Material and methods
Experimental animals and design
The experimental protocol and procedures were approved by the Animal Care and Use Committee of Wuhan Polytechnic University. Twenty-four weaned barrows (8.91 ± 0.74 kg initial body mass (BM), weaned at 28 ± 3 d of age) were arranged in a 2 × 2 factorial of treatments that included diet (5% corn oil (CO) versus 5% FO) and immunological challenge (saline versus LPS). Each treatment group was in six replicated pens. Pigs were placed individually in pens (1.8 × 1.1 m). Feed and water were freely available. Pigs were fed a corn-soybean diet with supplementation of 5% of CO (Xiwang Food Company, Shandong, China) or FO (menhaden fish oil, Fujian Gaolong Company, Fujian, China). The diets (Supplemental Table 1) were formulated to meet or exceed National Research Council requirements. 15 Fatty acid composition of FO or CO is presented in Supplemental Table 2. After 19 d of feeding 5% CO or FO diets, the challenged group received an intraperitoneal injection with E. coli LPS (E. coli serotype 055: B5, Sigma Chemical) at 100 mg/kg BM and the unchallenged group received an equivalent amount of sterile saline. The dose of LPS was chosen according to our previous experiment. 12 , 13
Hepatic fatty acid composition
The fatty acid composition of total fat in the liver tissues was assayed in accordance with Nieto et al. 16
Liver morphology
Tissue slides (5 μm) were deparaffinized and stained with hematoxylin and eosin. Histological analysis was performed in a blinded manner using a light microscope with a computer-assisted morphometric system (BioScan Optimetric, BioScan, Edmonds, WA, USA) by an experienced pathologist.
RNA sequencing and data analysis
There were three biological replicates for each treatment. The total RNA isolation from each sample was carried out as previously described. 12 cDNA libraries construction and RNA sequencing were performed at BGI-Shenzhen Company according to the manufacturer’s specifications. Briefly, mRNA was enriched from total RNA using oligo (dT) magnetic beads. Fragmentation buffer was used to reduce the mRNA into short fragments. The first strand cDNA was synthesized with random primers using the mRNA fragments as templates. Buffer, dNTPs, RNase H and DNA polymerase I were added to synthesize the second strand cDNA. The double-strand cDNA (dscDNA) was purified with a QiaQuick PCR extraction kit and washed with EB buffer for end repair and 5′-end phosphate. Then two blunt end adaptors were ligated to the dscDNA. The ligation product was nick-translated to intact dsDNA followed by PCR amplification. The PCR product was heat denatured, and the single strand DNA was cyclized by splint oligo and DNA ligase to construct cDNA libraries. Finally, RNA sequencing was performed using the sequencing platform of Complete Genomics (BGI-Shenzhen, China). After obtaining the raw reads, the adaptor, high content of unknown bases and low-quality reads were removed before downstream analysis to decrease data noise. After filtering, the clean reads were stored as FASTQ format. 17 The fragments per kb per million map reads were calculated for each gene to normalize the data. The NOISeq method was used to screen differentially expressed genes (DEGs) between the treatment groups. 18 We screened DEGs according to the following default criteria: log2 (fold change) ≥ 1 and diverge probability ≥ 0.8. We applied DAVID 6.8 to perform gene ontology (GO) functional classification and pathway enrichment analysis of DEGs.
Quantitative RT-PCR measurement
Total RNA isolation, quantification, cDNA synthesis and quantitative RT-PCR (qRT-PCR) were carried out as previously described. 12 The housekeeping gene GAPDH was used as an internal normalization control. The primers used for qRT-PCR are listed in Supplemental Table 3. All data were analyzed using the 2−ΔΔCT method. 19
Statistical analyses
All data were analyzed by ANOVA using the general linear model procedure of Statistical Analysis System (SAS Institute). The statistical model included the effects of diet (CO versus FO), challenge (saline versus LPS) and their interactions. Data were presented as means and SEMs. A value of P < 0.05 was considered as significant and 0.05 ≤ P < 0.10 as trends. When a significant interaction or a trend occurred, post hoc testing was performed using Bonferroni's multiple comparison tests.
Results
FO resulted in enrichment of n-3 PUFAs in liver
During the 19-d feeding trial (before LPS or saline injection), there were no differences in average daily feed intake (975 ± 43 g), average daily gain (567 ± 31 g) and feed/gain ratio (1.73 ± 0.05) among the four groups. Fatty acid composition of liver tissues (Table 1) mostly reflected the dietary fatty acid composition. Relative to pigs receiving the CO diet, pigs receiving the FO diet showed higher proportions of palmitoleic acid (16:1(n-7)), lignoceric acid (24:0), nervonic acid (24:1(n-9)), EPA, DHA and total n-3 PUFAs (P < 0.05), and showed lower proportions of oleic acid (18:1(n-9)), linoleic acid (18:2(n-6)), arachidonic acid (20:4(n-6)), total n-6 PUFAs and (n-6):(n-3) ratio in liver (P < 0.05). FO supplementation had no effect on the proportions of palmitic acid (16:0) and stearic acid (18:0) relative to CO. LPS challenge decreased liver EPA and total n-3 PUFA proportion (P < 0.05), but increased lignoceric acid (24:0) proportion (P < 0.05) in pigs fed an FO diet. An LPS challenge × diet interaction was observed for the proportions of EPA, lignoceric acid (24:0) and total n-3 PUFAs (P < 0.05).
Effects of fish oil or corn oil supplementation on selected fatty acids in liver after 4 h LPS challenge in weaned pigs.
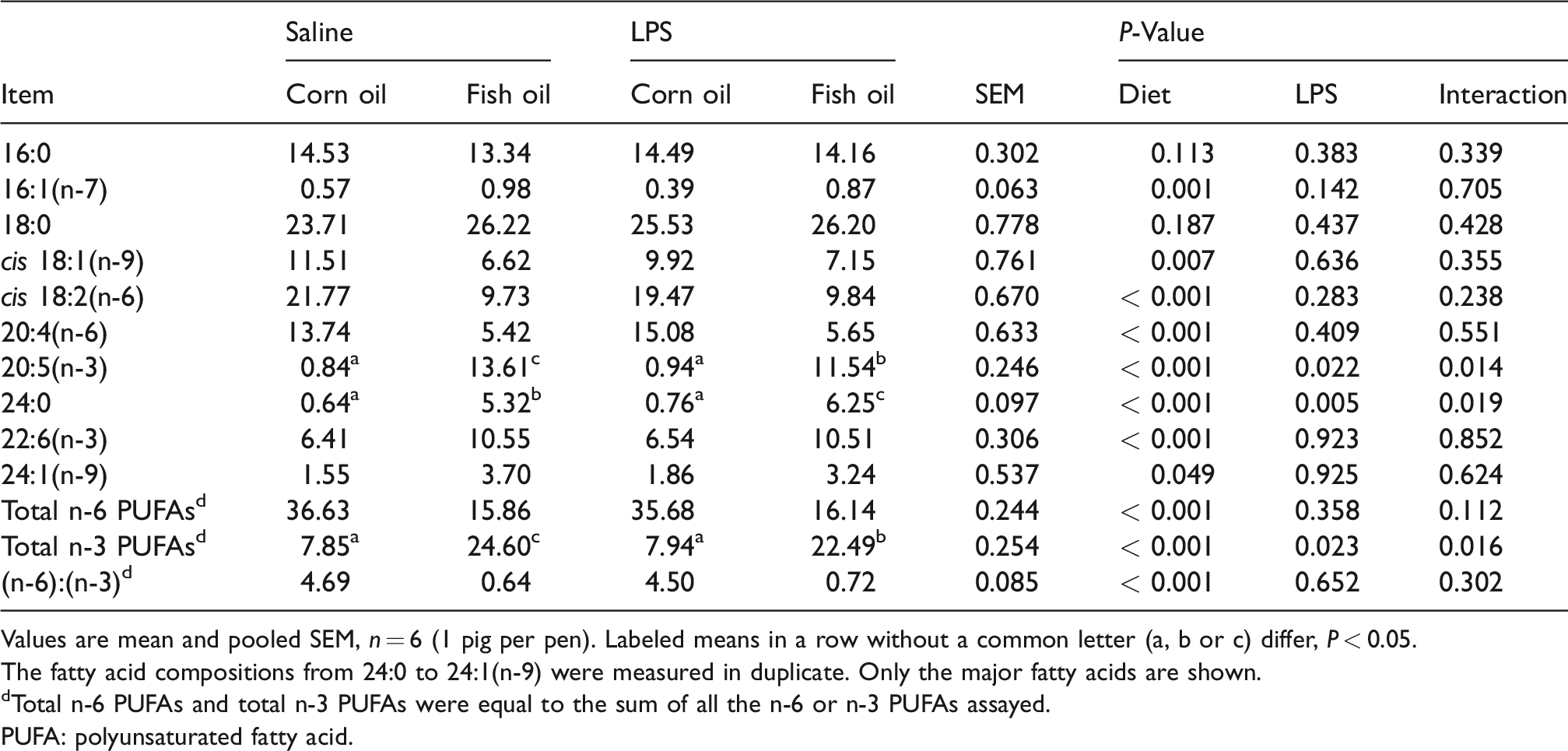
Values are mean and pooled SEM, n = 6 (1 pig per pen). Labeled means in a row without a common letter (a, b or c) differ, P < 0.05.
The fatty acid compositions from 24:0 to 24:1(n-9) were measured in duplicate. Only the major fatty acids are shown.
dTotal n-6 PUFAs and total n-3 PUFAs were equal to the sum of all the n-6 or n-3 PUFAs assayed.
PUFA: polyunsaturated fatty acid.
FO alleviated LPS-induced morphological changes in liver
As shown in Figure 1a and b, no obvious changes of hepatic morphology were observed in the saline-injected pigs fed CO or FO diets. Compared with the saline-treated groups, LPS challenge induced severe hepatocyte karyolysis, cytoplasm vacuolization, inflammatory cell infiltration and disruption of hepatic cell cords in pigs fed CO (Figure 1c). However, FO feeding significantly reduced LPS-induced morphological changes in liver (Figure 1d).
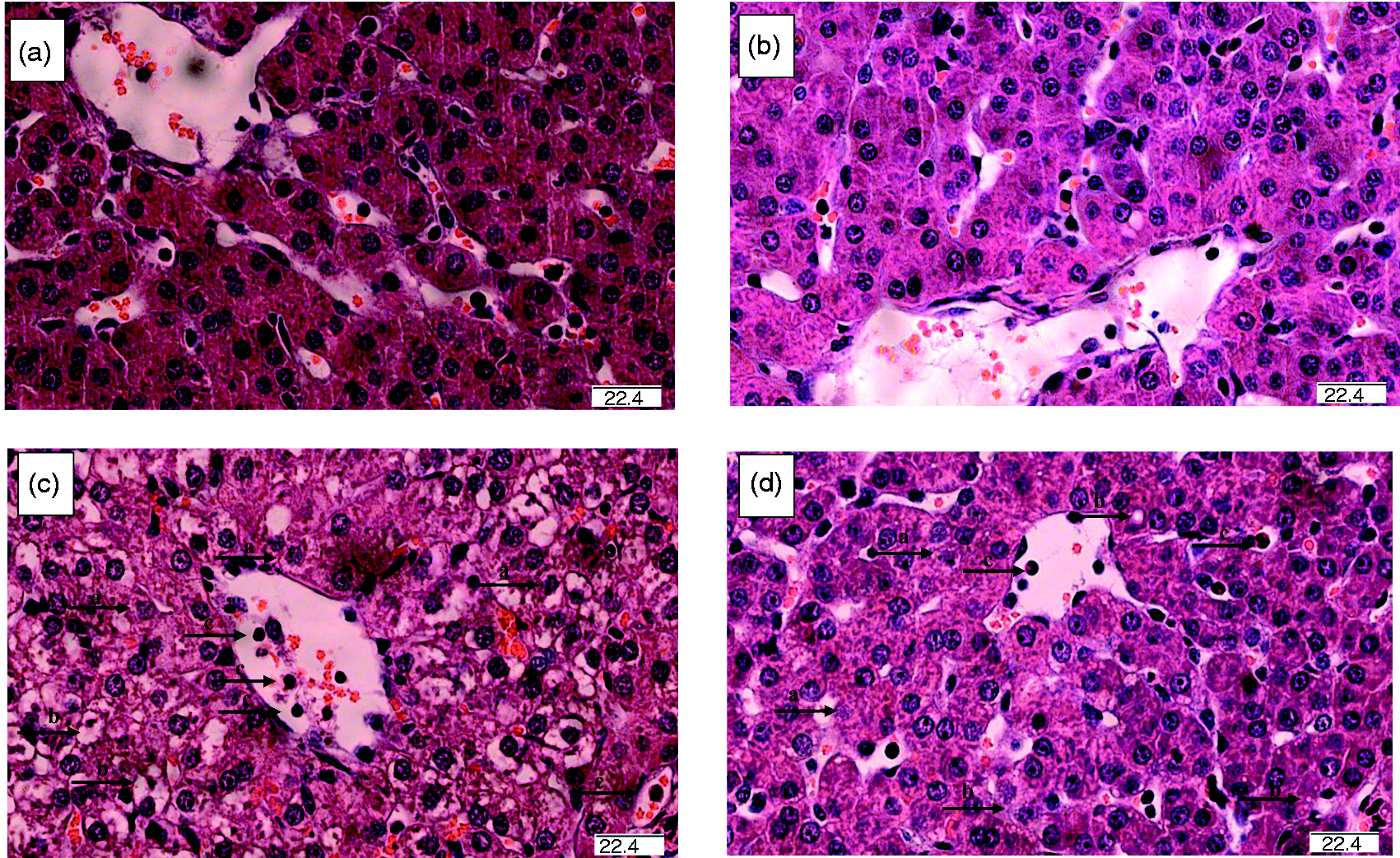
Effects of fish oil or corn oil supplementation on liver morphology after 4 h LPS challenge in weaned pigs. The representative photomicrographs of liver sections stained with hematoxylin and eosin are shown. (a) Piglets fed the corn oil diet and injected with saline. (b) Piglets fed the fish oil diet and injected with saline. No obvious morphologic changes were found. (c) Piglets fed the corn oil diet and challenged with LPS. Significant morphologic changes associated with liver injury, such as hepatocyte karyolysis (a), cytoplasm vacuolization (b), inflammatory cell infiltration (c) and disordered hepatic cell cords arrangement were observed. (d) Piglets fed the fish oil diet and challenged with LPS. Liver injury was significantly attenuated. Original magnifications 400×. Scale bars = 22.4 μm.
FO reduced LPS-induced TNF-α, IL-1β and IL-6 productions in liver
qRT-PCR results (Table 2) showed that LPS challenge upregulated the expression levels of the hepatic pro-inflammatory cytokines TNF-α, IL-1β and IL-6 (P < 0.001). However, the responses of these variables to LPS treatment were lower in pigs fed an FO diet (P < 0.01), indicating FO supplementation alleviated LPS-induced liver inflammation.
Effects of fish oil and corn oil supplementation on mRNA expression of TNF-α, IL-1β and IL-6 in liver after LPS challenge.
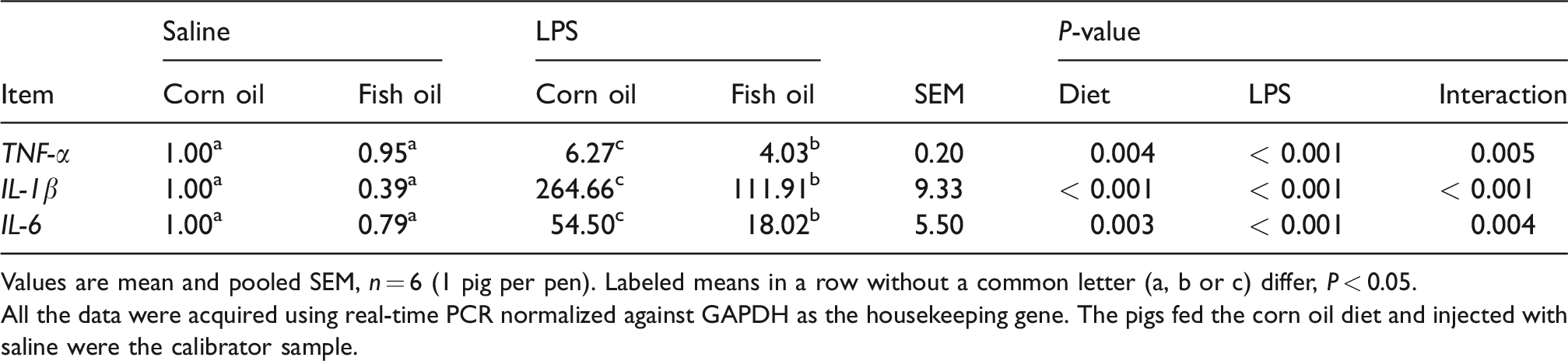
Values are mean and pooled SEM, n = 6 (1 pig per pen). Labeled means in a row without a common letter (a, b or c) differ, P < 0.05.
All the data were acquired using real-time PCR normalized against GAPDH as the housekeeping gene. The pigs fed the corn oil diet and injected with saline were the calibrator sample.
LPS elicited significant changes in liver gene expression in both FO- and CO-treated pigs
The volcano plot of DEGs in each pairwise is shown in Figure 2. As expected, there were substantially more DEGs following LPS challenge in both CO-fed and FO-fed pigs. Following pairwise comparisons of the saline versus LPS treatment in each diet group, we detected 1584 DEGs (622 up-regulated and 962 downregulated genes) in CO-fed pigs and 1420 DEGs (740 up-regulated and 680 down-regulated genes) in FO-fed pigs, respectively (Figure 3a). Details of all genes significantly altered in each group are presented in Supplemental Table 4. Furthermore, we performed a Venn analysis and the results are shown in Figure 3b. There were 616 DEGs and 452 DEGs that specifically regulated in the CO diet group and the FO diet group, respectively. Moreover, a total of 968 DEGs were overlapped in both diet groups, in which 48 DEGs were also found in CO-LPS versus FO-LPS.
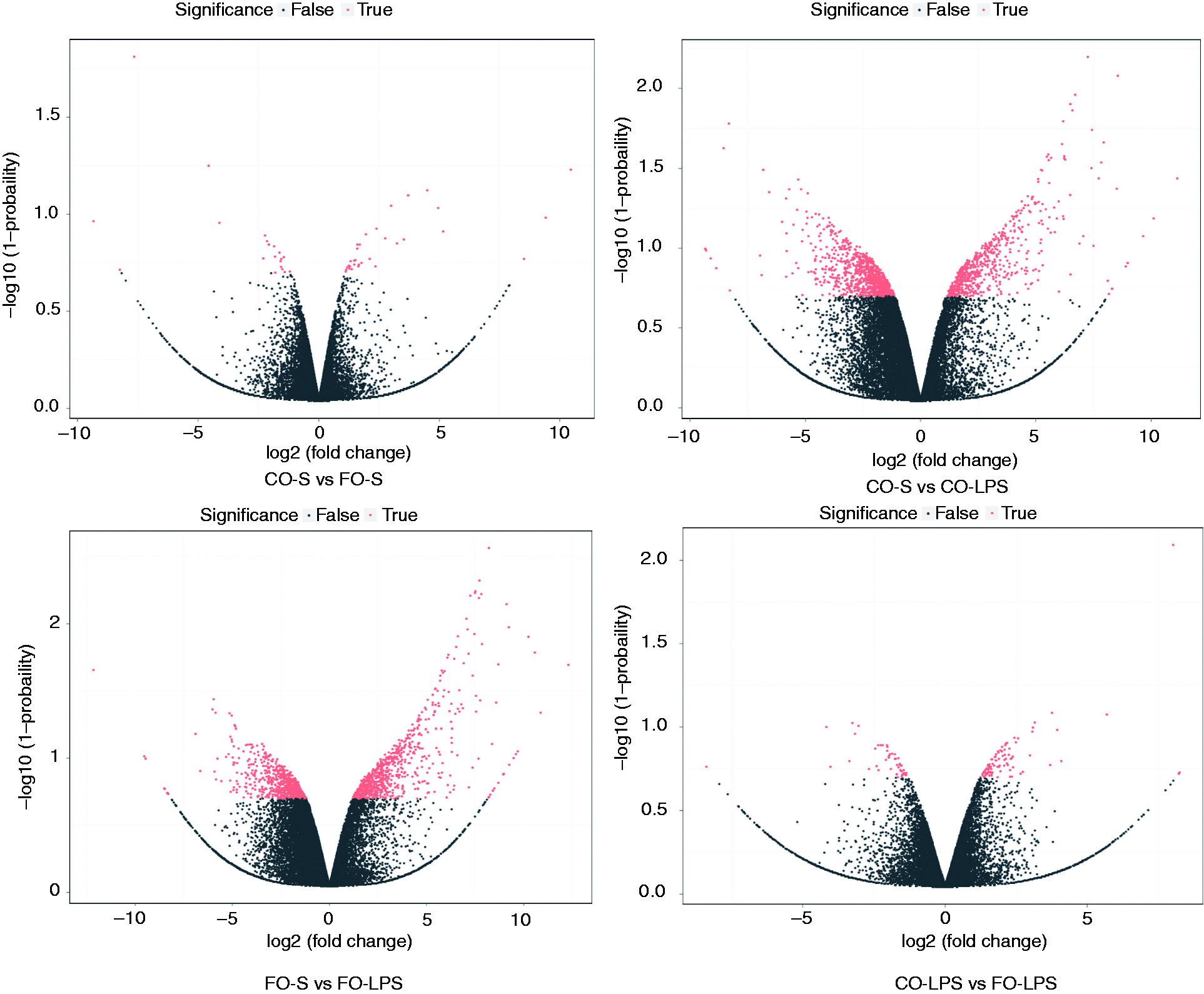
Volcano graph of all expressed genes in each pairwise. x-axis and y-axis present threshold value in log transform. Each dot is a differentially expressed gene (DEG). Dots in red mean significant DEGs which passed screening threshold and black dots are non-significant DEGs. The threshold was defined as: log2 (fold change) ≥1 and diverge probability ≥ 0.8.
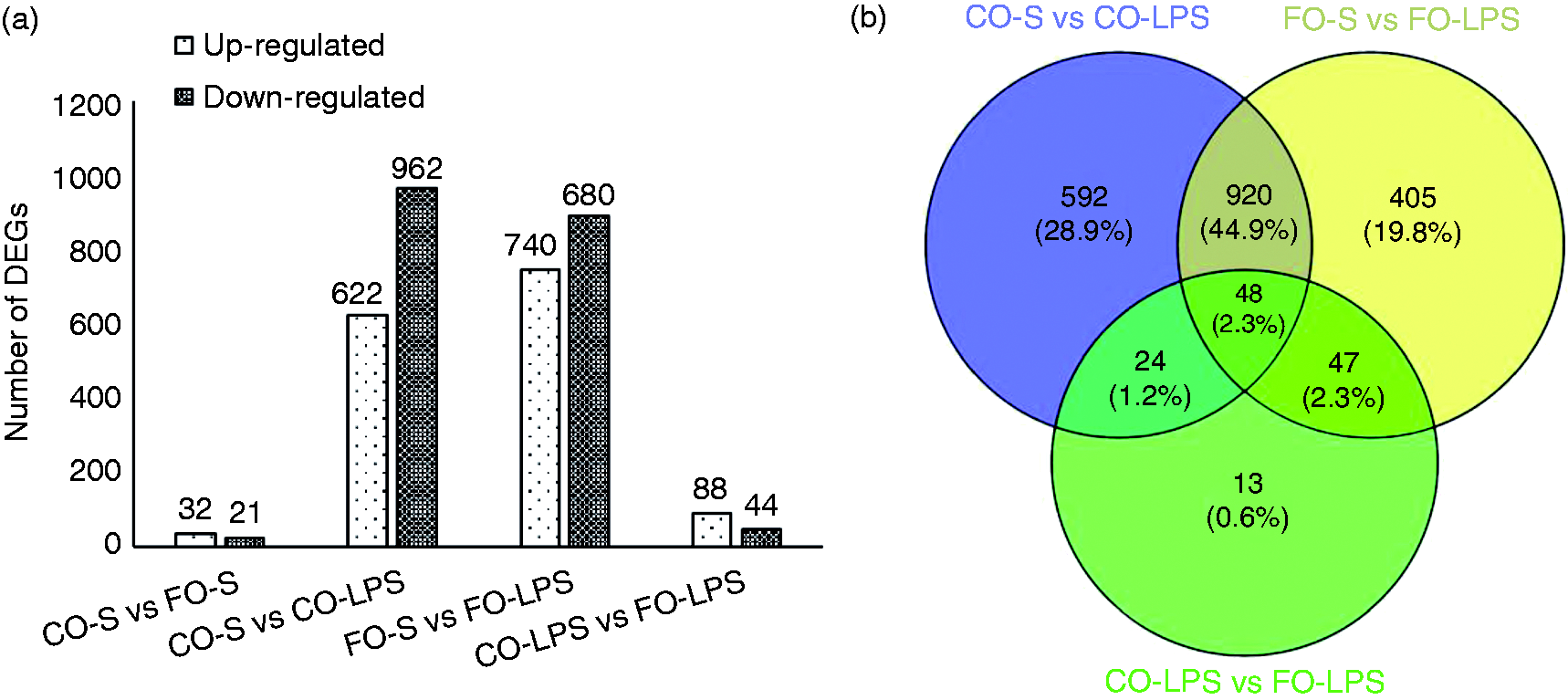
Effect of diet on LPS-induced alterations in hepatic gene expression. Statistics of differentially expressed genes. (a) x-axis represents pairwise, and y-axis represents number of screened differentially expressed genes (DEGs). (b) Venn chart of overlapped DEGs between (among) CO-S versus CO-LPS, FO-S versus FO-LPS and CO-LPS versus FO-LPS pairwise.
We detected only 53 DEGs between the FO and CO diet groups without LPS challenge. However, there were 132 DEGs (88 up-regulated and 44 down-regulated genes) between the FO and CO diet groups with LPS treatment. We selected four hepatic metabolism-related genes with abundant expression in liver for further verification by qRT-PCR (Figure 4), including 3-hydroxy-3-methylglutaryl-CoA synthase 2 (HMGCS2), apolipoprotein A4 (APOA4), fatty acid desaturase 1 (FADS1) and thyroid hormone responsive (THRSP). For pigs injected with saline, FO supplementation significantly increased mRNA expression of APOA4 (P < 0.05), whereas it decreased that of THRSP (P < 0.05) and FADS1 (P < 0.01) compared with CO, which was consistent with the results of the RNA sequencing. However, FO supplementation had no significant effect on the mRNA expression of HMGCS2 (P > 0.05).
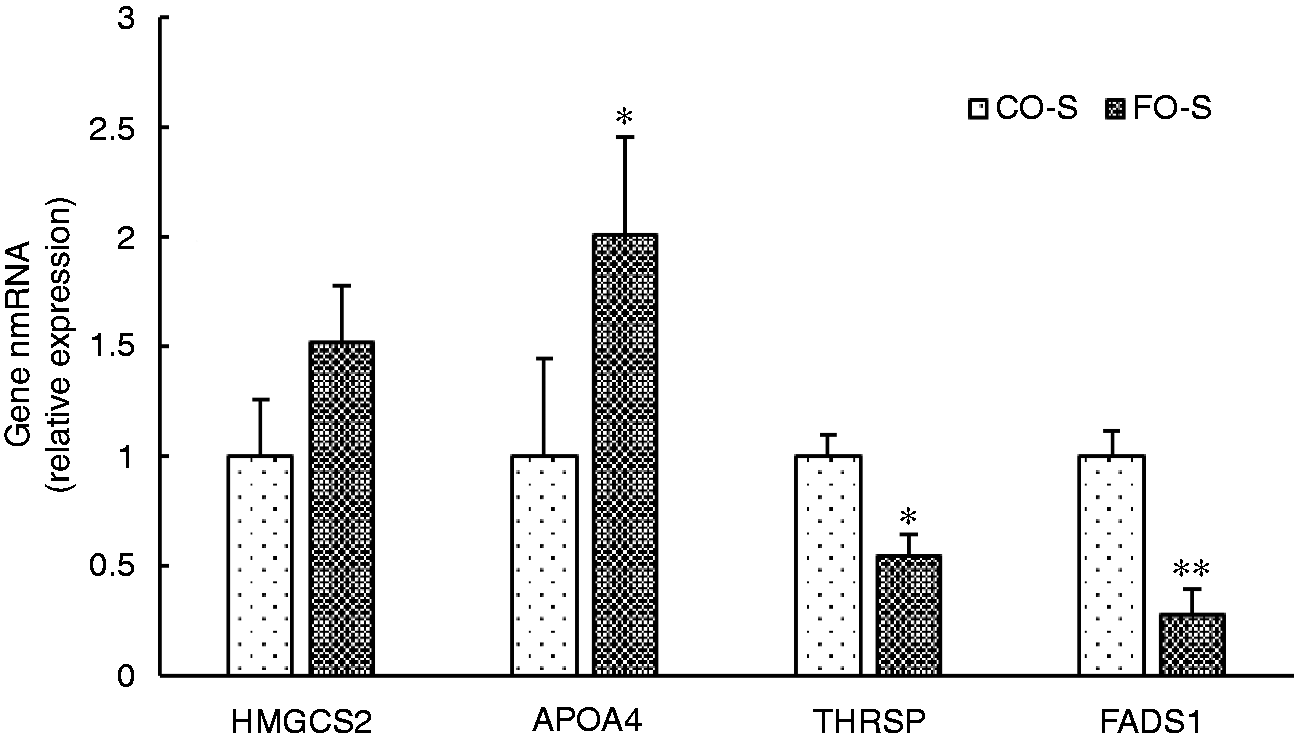
The mRNA expressions of liver HMGCS2, APOA4, FADS1 and THRSP in pigs fed corn oil or fish oil diet with saline treatment. GAPDH was the housekeeping gene, and the pigs fed the corn oil diet and injected with saline were the calibrator sample. *P < 0.05, **P < 0.01.
Functional analysis highlighted FO effect on genes involved in immune response
According to the Venn analysis (Figure 3b), the differentially regulated genes between FO and CO supplementation were selected for further GO and KEGG analysis, including the specific DEGs from CO-S versus CO-LPS pairwise (616 genes), FO-S versus FO-LPS pairwise (452 genes), and co-DEGs of the three sets (CO-S versus CO-LPS, FO-S versus FO-LPS and CO-LPS versus FO-LPS, 48 genes). The genes associated with the enriched GO terms and KEGG pathways are listed in Supplemental Tables 5 and 6, respectively. As shown in Figure 5a, the specific DEGs from CO-S versus CO-LPS were predominately associated with extracellular exosome, mitochondrion, cytosol, oxidation-reduction process, mitochondrial inner membrane, oxidoreductase activity, peroxisome, mitochondrial matrix, fatty-acyl-CoA binding and electron carrier activity. However, the majority of the specific DEGs from FO-S versus FO-LPS were involved in nucleus, cytoplasm, immune response, innate immune response, positive regulation of I-κB kinase/NF-κB signaling, positive regulation of NF-κB transcription factor activity, regulation of apoptotic process and response to LPS (Figure 5b). The co-DEGs of the three sets were enriched in extracellular space, defense response to virus, immune response, inflammatory response, chemokine-mediated signaling pathway, chemokine activity, regulation of cell proliferation and response to lipopolysaccharide (Figure 5c).
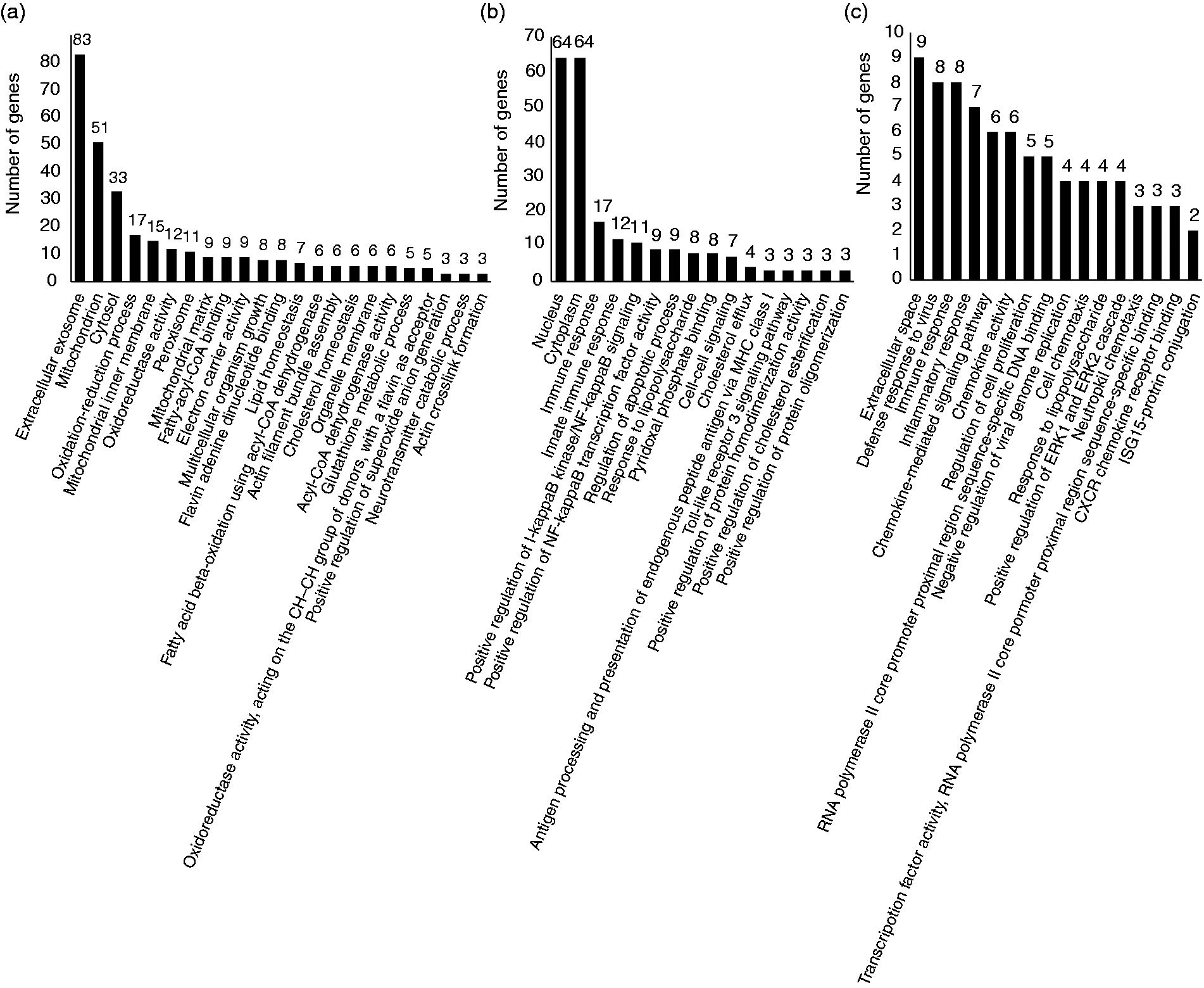
Gene ontology (GO) functional analysis of the specific differentially expressed genes (DEGs) from CO-S versus CO-LPS pairwise (a), FO-S versus FO-LPS pairwise (b), and CO-LPS versus FO-LPS pairwise (c). X-axis represents number of DEGs. Y-axis represents GO terms. We displayed the enriched GO terms with P-value < 0.01 according to DAVID (6.8).
Furthermore, KEGG pathway analysis demonstrated the specific DEGs from CO-S versus CO-LPS were mainly involved in hepatic metabolism, such as metabolic pathways, carbon metabolism, valine, leucine and isoleucine degradation, butanoate metabolism and fatty acid metabolism (Figure 6a). However, the specific DEGs from FO-S versus FO-LPS (Figure 6b) and the co-DEGs of the three sets (Figure 6c) were significantly enriched in TLR signaling pathway, NF-kB signaling pathway, TNF-α signaling pathway, and cytokine-cytokine receptor interaction, which were associated with LPS-induced inflammatory response. These data supported that FO supplement had an effect on the expression of immune-related genes in liver during evoked inflammation.
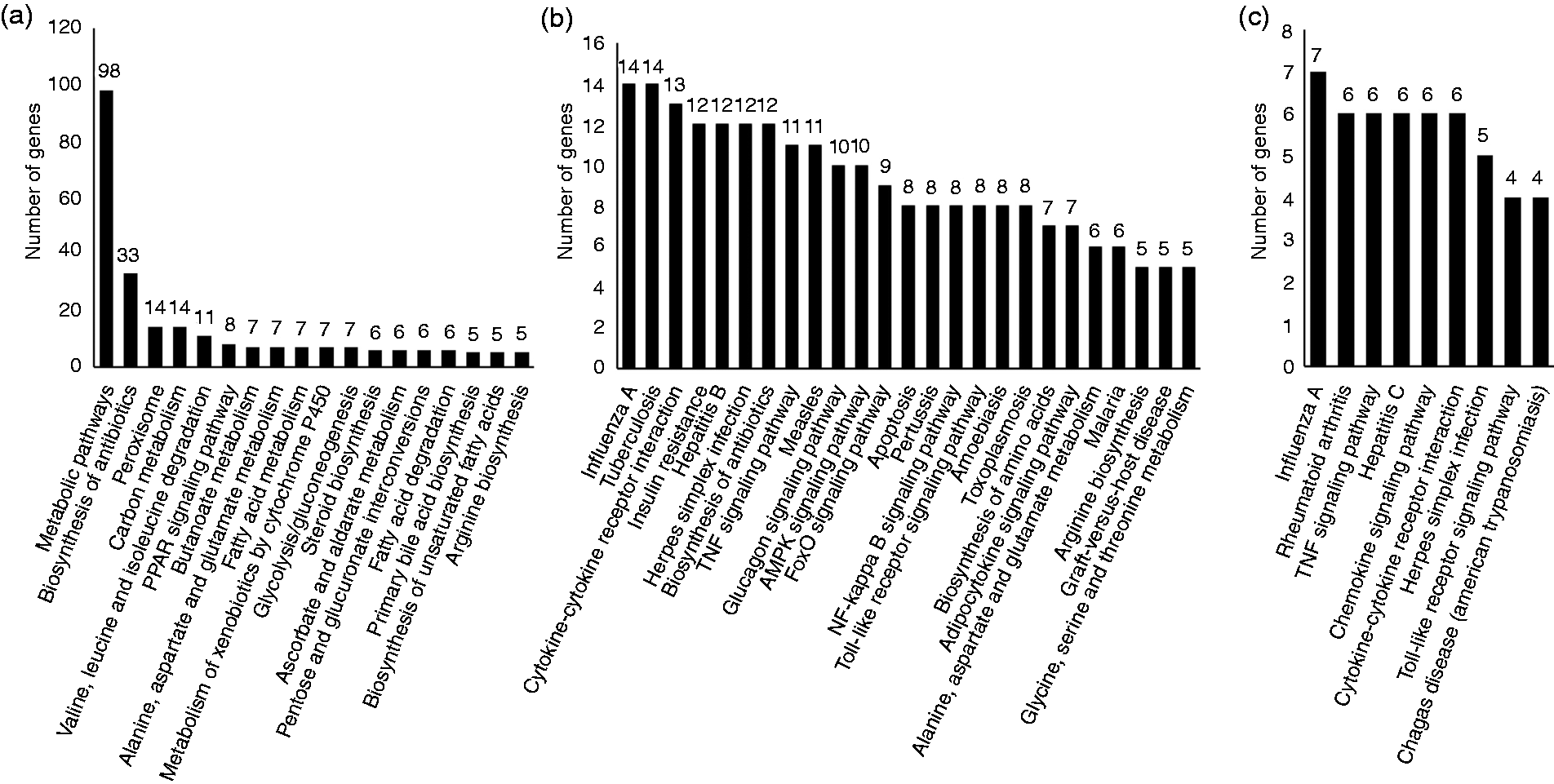
KEGG pathway analysis of the specific differentially expressed genes (DEGs) from CO-S versus CO-LPS pairwise (a), FO-S versus FO-LPS pairwise (b) and CO-LPS versus FO-LPS pairwise (c). x-axis represents number of DEGs. y-axis represents pathway terms. We displayed the enriched pathway terms with P-value < 0.01 according to DAVID (6.8).
Validation of RNA sequencing data by qRT-PCR
Based on our RNA sequencing data and previous literature, some important immune and lipid metabolism-related genes are listed and summarized in Table 3. To verify the RNA sequencing data, we selected 15 genes for further qRT-PCR (Table 4). LPS challenge increased mRNA abundance of liver C-X-C motif chemokine ligand 10 (CXCL10), C-X-C motif chemokine ligand 11 (CXCL11), IL-1 receptor type 1 (IL1R1), IL-1 receptor accessory protein (IL1RAP), IL-18 binding protein (IL-18BP), NF-kB inhibitor alpha (NFKBIA), CCAAT/enhancer binding protein (C/EBP) beta (CEBPB), C-reactive protein (CRP), IFN-induced protein with tetratricopeptide repeats 1 (IFIT1), IFN-induced protein with tetratricopeptide repeats 2 (IFIT2) and activating transcription factor 3 (ATF3) (P < 0.05). A diet × LPS interaction was observed for CXCL10, IL1R1, IL1RAP, IL-18BP, NFKBIA, CEBPB, CRP and ATF3 (P < 0.05) and a trend for interaction observed for IFIT1 and IFIT2 (P < 0.10). The responses of the variables (CXCL10, CXCL11, IL-18BP, NFKBIA, IFIT1, IFIT2 and ATF3) to LPS treatment were higher in pigs fed FO than pigs fed CO, whereas the responses of the variables (IL1R1, IL1RAP, CEBPB and CRP) to LPS treatment were lower in pigs fed an FO diet. For pigs injected with saline, diet had no effect on these variables.
Genes with evidence for differential regulation after LPS challenge in corn oil and fish oil diet.
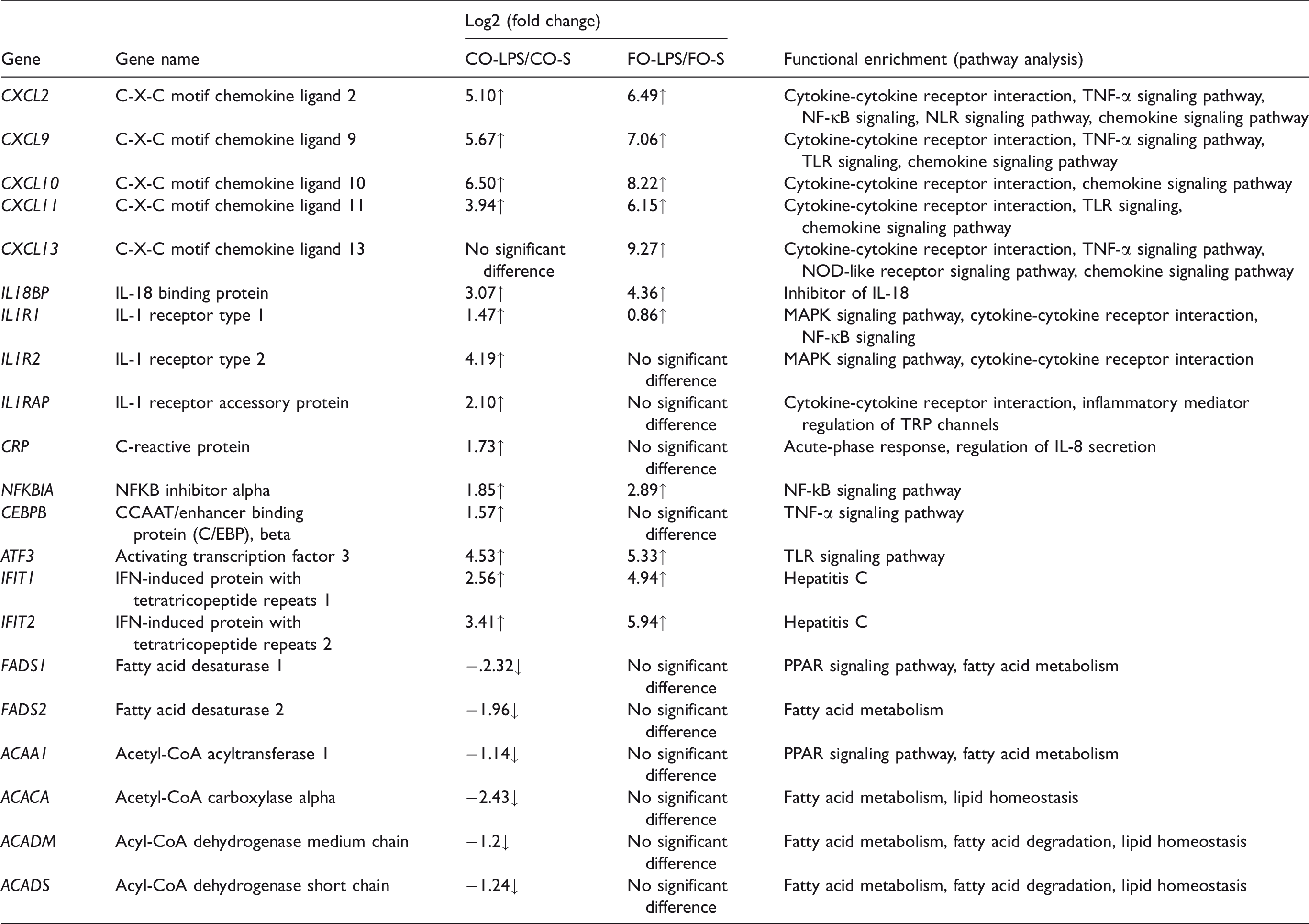
Effects of fish oil and corn oil supplementation on mRNA expression of 15 selected genes in liver after LPS challenge in weaned pigs.
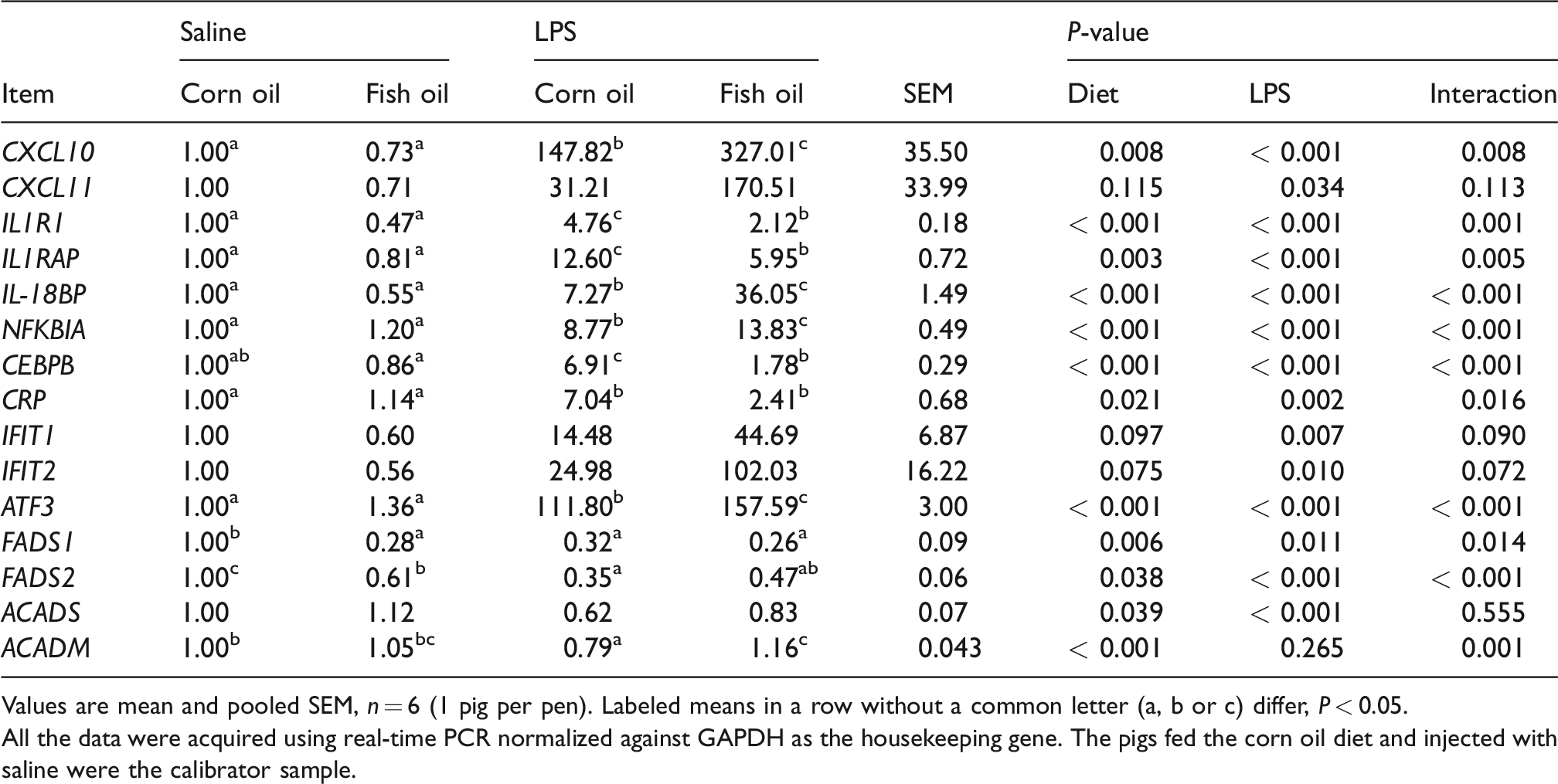
Values are mean and pooled SEM, n = 6 (1 pig per pen). Labeled means in a row without a common letter (a, b or c) differ, P < 0.05.
All the data were acquired using real-time PCR normalized against GAPDH as the housekeeping gene. The pigs fed the corn oil diet and injected with saline were the calibrator sample.
LPS challenge reduced mRNA abundance of liver FADS1, FADS2, acyl-CoA dehydrogenase short chain (ACADS) and acyl-CoA dehydrogenase medium chain (ACADM) (P < 0.05) in pigs fed a CO diet. However, LPS challenge had no effect on these genes in pigs fed an FO diet. A diet × LPS interaction was observed for FADS1, FADS2 and ACADM (P < 0.05). For pigs injected with saline, there are lower expressions of FADS1 and FADS2 (P < 0.05) in pigs fed an FO diet than pigs fed a CO diet.
Discussion
Accumulating literatures indicate that n-3 PUFAs (EPA and DHA) can dampen the inflammatory responses and exert protective effects on many inflammatory diseases. Production of pro-inflammatory cytokines by macrophages is dependent on activation of the NF-κB and JNK pathways. 20 EPA and DHA can prevent NF-κB from translocating to the nucleus to induce transcription of pro-inflammatory cytokines. 11 In addition, EPA and DHA bind to G protein-coupled receptor 120, and inhibit NF-κB and JNK pathways. 20 Our previous studies demonstrated that 5% FO supplementation alleviated LPS-induced inflammation in intestine and muscle. 12 , 13 This study was focused on the protective effects of n-3 PUFA-rich FO against LPS-induced acute liver injury. Thus, we firstly investigated the effects of FO on liver fatty acid composition to confirm that FO feeding can increase liver EPA, DHA and total n-3 PUFA proportions. Studies have demonstrated that LPS caused acute liver histological injury and over-release of pro-inflammatory cytokines (TNF-α, IL-1β and IL-6) within 3-6 h LPS injection. 21 , 22 In this study, severe liver injury was observed in LPS-challenged pigs fed CO through histological observations, such as hepatocyte karyolysis, cytoplasm vacuolization, inflammatory cell infiltration and disruption of hepatic cell cords. However, only mild liver injury was observed in the LPS-challenged pigs fed FO. In addition, compared with CO, 5% FO supplementation reduced the mRNA expression of TNF-α, IL-1β and IL-6 in liver. These results supported that FO attenuated LPS-induced liver inflammatory response.
To our knowledge, this study is the first to study the gene expression profiles of liver tissue in a trial of FO supplementation during LPS-induced inflammation in a piglet model. We found that FO had a limited effect on liver tissue gene expression without LPS treatment but had significant effects during evoked inflammation. We detected only 53 DEGs between pigs fed a CO diet and an FO diet with saline treatment but 132 DEGs with LPS treatment, which highlighted the difficulty in detecting gene changes due to nutritional interventions in the resting state without disease or other stimulation. n-3 PUFAs have been reported to modulate the expression of genes related to lipid and lipoprotein metabolism in liver. 23 According to the RNA sequencing data and qRT-PCR results, APOA4, THRSP and FADS1 were the genes with evidence of n-3 PUFA regulation in liver tissue before administration of LPS. APOA4 is a major component of high-density lipoprotein cholesterol and chylomicrons, and is involved in lipid and Glc metabolism and anti-inflammatory response. 24 , 25 In the current study, 5% FO supplementation significantly increased the mRNA expression of liver APOA4, which might be beneficial against hepatic gluconeogenesis and inflammation. Fatty acid desaturase genes FADS1 (Δ5-desaturase) and FADS2 (Δ6-desaturase) participate in biosynthesis of n-6 and n-3 PUFAs. 26 Reardon et al. 27 and Ralston et al. 28 reported that FADS1 and FADS2 gene expression are reduced by DHA and EPA. In this study, we observed that EPA, DHA and total n-3 PUFAs were enriched in liver through 5% FO supplementation, which might lead to the reduction of hepatic FADS1 and FADS2 expressions. THRSP, mainly expressed in liver and adipocytes, plays a key role in the de novo lipogenesis by regulating lipogenic-related genes. 29 THRSP is reported to promote the synthesis of medium chain fatty acids in mammary epithelial cells. 30 Vahmani et al. 31 reported that feeding FO downregulated hepatic expression of FADS2 and THRSP in lactating dairy cows. n-3 PUFAs can enhance hepatic fatty acid oxidation and suppress de novo fatty and triacylglycerol synthesis. 31 In the current study, we found that the mRNA expression of liver THRSP was reduced in pigs fed an FO diet, which might be related to the inhibitory effect of n-3 PUFAs on the de novo lipogenesis.
In the present study, both the FO and CO diet groups exhibited significant changes in liver gene expression following LPS challenge, although we found that the liver inflammatory response was attenuated by 5% FO supplementation. According to the Venn analysis (Figure 3b), a total of 968 genes reached statistical significance for expression change after LPS treatment in both diet groups, indicating that LPS had a similar broad effect on hepatic gene expression in pigs fed an FO diet and a CO diet. However, through the GO and KEGG analysis of the specific DEGs in each diet group after LPS treatment, we found that FO mainly regulates immune-related genes in liver during evoked inflammation. We confirmed the expressions’ changes of 15 immune-related genes by qRT-PCR. Relative to CO, FO reduced the LPS induction of IL1R1, IL1RAP, CEBPB and CRP, but increased the expressions of IL-18BP, NFKBIA, IFIT1, IFIT2 and ATF3. IL-1R1 and IL-1RAP are two membrane-bound receptors of the cytokine IL-1. As the ligand-recognition receptor, IL-1R1 binds IL-1β directly leading to activation of NF-kB and induction of a range of downstream proteins related to inflammatory response. 32 Although IL-1RAP cannot bind IL-1β directly, it is essential for the formation of an activated membrane receptor complex, which recruit intracellular adaptor proteins and kinases, including MyD88, IL-1 receptor-associated kinase 1 (IRAK1) and IRAK4. 33 CEBPB (also named NF for IL-6, NF-IL6) is a transcription factor activated by LPS or IL-17, and itself regulates numerous genes involved in inflammation, including IL-6 and IL-23R. 34 , 35 CRP is a major acute-phase reactant and marker of inflammation, which is produced by the liver. 36 Some researchers have reported an association between lower CRP levels and increased dietary intake of EPA and DHA. 37 , 38 IL-18 is necessary for the development of Cutibacterium acnes/LPS-induced liver injuries. 39 Blockade of IL-18 can protect against the liver damages. 40 Kupffer cells secrete IL-18 in a manner dependent on TRIF and the NLRP3 inflammasome after stimulation with LPS. 2 IL-18BP, a specific antagonist of IL-18, binds to IL-18 and functions as a competitive inhibitor of receptor binding of IL-18. 41 In our study, FO increased the expression of IL-18BP after LPS challenge, which might prevent IL-18 activation and protect against the liver injury. NF-kB is a critical transcriptional regulator in inflammation and is normally inhibited by IKBα (encoded by NFKBIA gene). The IKBα conjugates with the NF-κB, resulting in its cytoplasmic sequestration and inhibition of transcriptional activation. 42 IFIT1 and IFIT2 play an important role in restricting virus infection. 43 Although IFIT1 and IFIT2 are both strongly induced by LPS, they have opposite functions in LPS-induced inflammation. 44 , 45 IFIT1 was defined as a novel hepatoprotective factor in LPS-induced hepatitis and this protection was associated with the negative regulation of the JNK pathways. 44 However, IFIT2 was found to be an effector protein of amplification of LPS-induced cytokine production and endotoxin shock. 45 Nardo et al. 46 reported that ATF3 was a key transcriptional repressor of innate immune response genes, which was induced by TLR activation and acted in a negative feedback system to limit TLR-driven inflammatory responses in macrophages. Previous studies have reported that ATF3 protects against LPS-induced inflammation in various cells. 47 , 48 Therefore, in the current study, we found that n-3 PUFAs negatively regulated the expression of IL1R1, IL1RAP, CEBPB and CRP, whereas positively regulated that of IL-18BP, NFKBIA, IFIT1, which was beneficial against LPS-induced inflammation. In addition, our RNA sequencing data showed the mRNA expressions of liver CXCL2, CXCL9, CXCL10, CXCL11 and CXCL13 were higher in pigs fed an FO diet than pigs fed a CO diet after LPS challenge. The expressions of two selected chemokines (CXCL10 and CXCL11) were examined by qRT-PCR. The results were consistent with the RNA sequencing data, suggesting n-3 PUFA had no inhibitory effect on these chemokines’ production.
We observed some specific DEGs (ACAA1, ACACA, ACADS and ACADM) from CO-S versus CO-LPS were involved in hepatic fatty acids metabolism according to the RNA sequencing data. The expression changes of ACADS and ACADM were confirmed by qRT-PCR. ACAA1 is a key enzyme in peroxisomal fatty acid β-oxidation. 49 ACACA positively regulates fatty acid synthesis in the liver. 50 ACADS and ACADM play vital roles in free fatty acid β-oxidation and regulate energy homeostasis. 51 , 52 These gene expressions were decreased in pigs fed a CO diet after LPS challenge, indicating lipid metabolism disorders might be induced by LPS in liver. In contrast, LPS challenge had no effect on these genes in pigs fed an FO diet, indicating the protective effect of n-3 PUFA on lipid metabolism.
In conclusion, dietary treatment of FO attenuates LPS-induced liver inflammation in weaned pigs. Supplementation with FO versus CO had only modest effect on liver tissue gene expression without LPS challenge. In contrast, the transcriptomic response to evoked endotoxemia was modified by FO supplementation, with several genes demonstrating significant gene-nutrient interactions, such as IL1R1, IL1RAP, CEBPB, CRP, IL-18BP, NFKBIA, CXCL10, CXCL11 and others. In this study, we highlighted liver genes induced or repressed in inflammation that were modulated by n-3 PUFA and might represent candidates for nutrient targeting of inflammatory stress in pigs.
Supplemental Material
Supplemental Tables1-3 - Supplemental material for Dietary fish oil supplementation alters liver gene expressions to protect against LPS-induced liver injury in weanling piglets
Supplemental material, Supplemental Tables1-3 for Dietary fish oil supplementation alters liver gene expressions to protect against LPS-induced liver injury in weanling piglets by Jing Zhang, Xin Xu, Huiling Zhu, Yang Wang, Yongqing Hou and Yulan Liu in Innate Immunity
Supplemental Material
Supplemental Table4 - Supplemental material for Dietary fish oil supplementation alters liver gene expressions to protect against LPS-induced liver injury in weanling piglets
Supplemental material, Supplemental Table4 for Dietary fish oil supplementation alters liver gene expressions to protect against LPS-induced liver injury in weanling piglets by Jing Zhang, Xin Xu, Huiling Zhu, Yang Wang, Yongqing Hou and Yulan Liu in Innate Immunity
Supplemental Material
Supplemental Table5 - Supplemental material for Dietary fish oil supplementation alters liver gene expressions to protect against LPS-induced liver injury in weanling piglets
Supplemental material, Supplemental Table5 for Dietary fish oil supplementation alters liver gene expressions to protect against LPS-induced liver injury in weanling piglets by Jing Zhang, Xin Xu, Huiling Zhu, Yang Wang, Yongqing Hou and Yulan Liu in Innate Immunity
Supplemental Material
Supplemental Table6 - Supplemental material for Dietary fish oil supplementation alters liver gene expressions to protect against LPS-induced liver injury in weanling piglets
Supplemental material, Supplemental Table6 for Dietary fish oil supplementation alters liver gene expressions to protect against LPS-induced liver injury in weanling piglets by Jing Zhang, Xin Xu, Huiling Zhu, Yang Wang, Yongqing Hou and Yulan Liu in Innate Immunity
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Project of the Hubei Provincial Department of Education (T201508), National Natural Science Foundation of China (31772615), and the State’s Key Project of Research and Development Plan (2016YFD0501210).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
