Abstract
In recent years, the potential use of silica nanoparticles (SiNPs) among different biomedical fields has grown. A deep understanding of the physicochemical properties of nanoparticles (NPs) and their regulation of specific biological responses is crucial for the successful application of NPs. Exposure to NP physicochemical properties (size, shape, porosity, etc.) could result in deleterious effects on cellular functions, including a pro-inflammatory response mediated via activation of the NLRP3 inflammasome. The aim of this study was to evaluate the potential
Introduction
Nanotechnology has great potential for use in several medical applications such as medical diagnostics, therapy and tissue engineering. As nanotechnology develops, nanoparticles have been used in an increasingly wide range of applications and to obtain different results, owing to their unique physicochemical properties.1,2 Recent studies suggest great potential for silica nanoparticles (SiNPs) as new tools in biotechnology and the biomedical field. NPs have unusual physicochemical properties such as their large surface area, size distribution, crystallinity, surface properties, electronic properties, solubility, and ability to aggregate and shape.3,4 For these reasons, biomedical uses of SiNPs are rapidly increasing and include drug delivery systems, labeling and tissue engineering.4–6 However, owing to the physicochemical properties of SiNPs, which are different from bulk particles, they interact with biological systems differently and require further study. 7 Several investigations have revealed that SiNPs, owing to their foreign nature, can be recognized by the immune system and induce immunomodulatory effects (through stimulation or suppression of immune responses). Interactions between SiNPs and immune-competent cells may be desirable for some applications but unwanted for others.3,4,8
The immune system defends organisms against a wide variety of pathogenic agents and danger signals. The first line of defense is the innate immunity response, which comprises both innate cells and effector molecules involved in host protection. This response is dependent on initial recognition of the pathogen by PRRs, which recognize conserved molecular PAMPs or danger signals, called danger/damage-associated molecular patterns (DAMPs), present in the environment.1,7 It has been reported that innate immune cells such as macrophages, dendritic cells and monocytes internalize SiNPs and produce pro-inflammatory cytokines such as IL-1β and IL-18. 5 This process involves a cytosolic multiprotein complex, called the inflammasome, which plays a major role in innate immunity. One of the best characterized is the NLRP3 inflammasome, which contains the protein NLRP3 (previously termed Nalp3 or cryopyrin). This is a member of the NLR family and consists of a N-terminal PYRIN domain, a central NACHT domain and C terminal leucine-rich repeats (LRRs). The LRR exposes the NACHT domain for homotypic aggregation, triggering a cascade leading to NLRP3/ASC/pro-caspase-1 complex formation. Consequently, triggered by the signaling of PAMPs or DAMPs, the NLRP3 inflammasome regulates caspase-1 activation and the maturation of IL-1β and IL-18.9–13
This is a highly controlled process involving detection of two specific signals. First, a priming signal is required to induce the transcription and translation of pro-IL-1β and pro-IL-18 and the expression of NLRP3. PAMPs, such as LPS, stimulate the activation of TLRs, which results in the generation of a priming signal. Then, a second signal causes inflammasome activation, leading to the formation of the complex and activation of caspase-1, which modifies pro-IL-1β and pro-IL-18, resulting in their maturation. This second signal is triggered by a group of chemically and biologically unrelated PAMPs or DAMPs (endogenous or exogenous), such as ATP, monosodium urate crystals, UVB irradiation and the environmental pollutants silica and asbestos.9,14,15
Although monocytes and neutrophils constitutively express active caspase-1 and need only one signal for IL-1β release,16,17 several studies have shown that SiNPs can act as a second signal to stimulate or potentiate the assembly of the NLRP3 inflammasome.14,15 However, other studies have demonstrated that the effects of SiNPs on cells vary with the physicochemical properties of the SiNPs, such as chemical composition, crystallinity, size, shape and surface area.3,18 For example, it was recently reported that SiNPs are sensed by the NLRP3 inflammasome and induce the release of mature IL-1β, but this response is dependent on the SiNP size. 8 Similarly, silica particle size impacts immune responses, with submicron amorphous silica particles inducing higher inflammatory responses than silica particles >1000 nm in size. 5 However, Sandberg et al. demonstrated that non-crystalline silica particles of nano and sub-micro sizes induced IL-1β release from LPS-primed cells, but the differential response patterns obtained suggest that silica-induced IL-1β responses depend not only on the particle surface area, but also on factors such as particle reactivity or particle uptake. 19 Therefore, owing to these varying results, the interactions and responses of the immune system to the SiNPs must be evaluated before any biomedical application.
Neutrophils and PBMCs are essential players in immunity, providing an effective defense against foreign agents and playing a key role during inflammation by using several mechanisms, including inflammasome activation. These two populations include the major immune cells that act in the defense against different aggressions. In addition, neutrophils are the first cells to be recruited at tissue injury sites.20,21 However, the literature addressing SiNPs activation of the inflammasome in neutrophils and PBMCs is poorly documented, even more so in human primary cells. Thus, the aim of this work was to evaluate the potential
Material and methods
Ethics statement
Following ethical guidelines, all donors gave written informed consent before enrollment in the study. Study protocols were reviewed and approved by the Institutional Research Ethics Boards at the Universidad Cooperativa de Colombia and were conducted according to the principles of the Declaration of Helsinki.
SiNPs
Two different monodisperse spherical SiNPs with diameters of 12 nm [nanopowder, spec. surface area 175–225 m2/g (BET Surface Area Analysis of Nanoparticles), 99.8% trace metals basis (Sigma-Aldrich, St. Louis, MO, USA)] and 200 nm (nanoparticles, mesoporous, 200 nm particle size, pore size 4 nm; Sigma-Aldrich) were used to investigate how particle size and concentration affect the immunomodulatory effect of human cells. The 12-nm SiNP has been previously characterized, 22 and its reported physicochemical properties include an amorphous state, with a surface area of 175–225 m2/g, and ζ: −11.6 ± 1.0 mV. Suspensions of SiNPs were freshly prepared before each experiment, from stock solutions previously sonicated (for 5 min, three times), then diluted to 1.0 mg/ml in the indicated medium and stirred for 10 min at room temperature (∼22°C) to achieve dispersion. It is noticeable that unlike other nanoparticles (NPs), the SiNPs show size distributions in cell culture medium (determined by DLS) very similar to their actual particle size as determined by transmission electron microscopy. 22
PBMC and neutrophil isolation
Fresh whole-blood samples were obtained by venipuncture and collected into EDTA-coated tubes. Human PBMCs were isolated using a standard Ficoll/Histopaque-1077 (Histopaque 1077; Sigma Aldrich) gradient and cultured in RPMI medium (RPMI 1640; Gibco, Waltham, MA, USA) supplemented with 10% FBS (Sigma-Aldrich), as previously described. 23 Neutrophils were isolated from blood samples collected into ACD (citric acid, citrate, dextrose) anticoagulant-coated tubes (BD Pharmingen, San Diego, CA, USA). Neutrophils were isolated by dextran sedimentation (Sigma-Aldrich), followed by centrifugation over Ficoll-Hypaque (Sigma-Aldrich), as previously reported.24–26 To avoid cell death or cellular activation during isolation, all buffers and reagents were used at 4°C. Cellular viability was monitored by trypan blue exclusion and found to be consistently ≥ 95%. Cellular purity (≥95%) was verified by Wright stain. Briefly, blood smears were stained with a mixture of eosin and methylene blue, for 3 min. Then, these were rinsed off with water twice, and the slides were dried at room temperature, before examination.
Cell culture and treatment with SiNPs
PBMCs (1.5 × 105 cells/well) or neutrophils (2 × 105 cells/well) were seeded in 96-well plates and treated with RPMI 1640 containing 12 nm or 200 nm SiNPs (10–1–105 ng/ml). Cells unexposed to SiNPs served as a negative control, whereas cells stimulated with 50 ng/ml LPS (Invitrogen, San Diego, CA, USA) were used as a positive control. PBMCs were incubated overnight (16 h), and neutrophils were incubated for 4 h at 37°C and 5% CO2.
To evaluate the effect of priming in the inflammasome activation,
26
cells were pre-treated with 50 pg/ml LPS for 2 h, before exposure to increasing concentrations of the SiNPs for 4 h. Cells were then centrifuged, and the supernatants were collected and immediately frozen at –80°C until cytokine quantification using ELISA. To analyze RNA levels, we followed the same protocol described above, but seeded the cells in a 24-well plate at a density of 1 × 106 cells/well and incubated them for 4 h at 37°C and 5% CO2 to maximize RNA yield for targeted immune outputs. Then, PBMC or neutrophil pellets were used for total RNA extraction. All reagents and materials used in the experiments were endotoxin free, as reported by the chromogenic
Cytokine analysis
To investigate the effect of SiNPs on IL-1β, IL-6 (OptEIA; BD Biosciences, San Jose, CA, USA) and IL-18 (eBiosciences, San Diego, CA, USA) production, cytokine concentrations (pg/ml) in cell culture supernatants were determined using a commercial ELISA assay according to the manufacturer’s instructions. All samples were run in duplicate. Cytokine concentrations were calculated from a standard curve of the corresponding recombinant human cytokine.
Transcriptional analysis of inflammasome components by real-time PCR
Primers.

Cytotoxicity assay
Cell viability was assessed using 3 -(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) reduction. After exposure of cells to SiNPs of each concentration lasting as long as 24 h for PBMC and up to 8 h for neutrophils, supernatant was removed from each well, and the cells were washed with PBS. The cells were then incubated in fresh serum-free medium containing 0.5 mg/ml MTT for 4 h at 37°C. The resulting formazan product was dissolved in 100 µl DMSO. Finally, the absorbance was measured at 570 nm using a microplate reader (Multiskan FC Microplate Photometer; Thermo Scientific). The data were normalized to the absorbance of the untreated control cells. It is important to clarify that as reported in the paper by Guadagnini et al., SiNPs do not influenced the OD readings in MTT assay. 28
Statistical analysis
Data were plotted and analyzed using Prism 5.0 software (Graph Pad Software, La Jolla, CA, USA). All results represent at least three independent experiments. Normality was determined by Shapiro–Wilks test. The data were analyzed using the Kruskal–Wallis test for comparison of all the independent samples (controls and treatment). Dunn’s multiple comparison test was used as a post-test to compare samples within the data set. Values of
Results
SiNPs do not have cytotoxic effects on PBMCs and neutrophils
The cytotoxicity of 12-nm and 200-nm SiNPs on PBMCs and neutrophils was assessed at different concentrations using the MTT reduction method (Figure 1). PBMCs exposed to 12-nm or 200-nm SiNPs for 24 h, at concentrations of 1 × 102, 1 × 103 and 1 × 104 ng/ml, did not show any changes in cell viability. Similarly, 12-nm and 200-nm SiNPs did not have a significant cytotoxic effect on neutrophils at any concentration when cells were exposed for 8 h.
SiNPs do not have cytotoxic effects in PBMCs and neutrophils. (a) PBMCs treated with 12-nm and 200-nm SiNPs at 102–104 ng/ml for 24 h and (b) neutrophils treated with 12-nm and 200-nm SiNPs at 102–104 ng/ml for 8 h. C+: positive control (DMSO).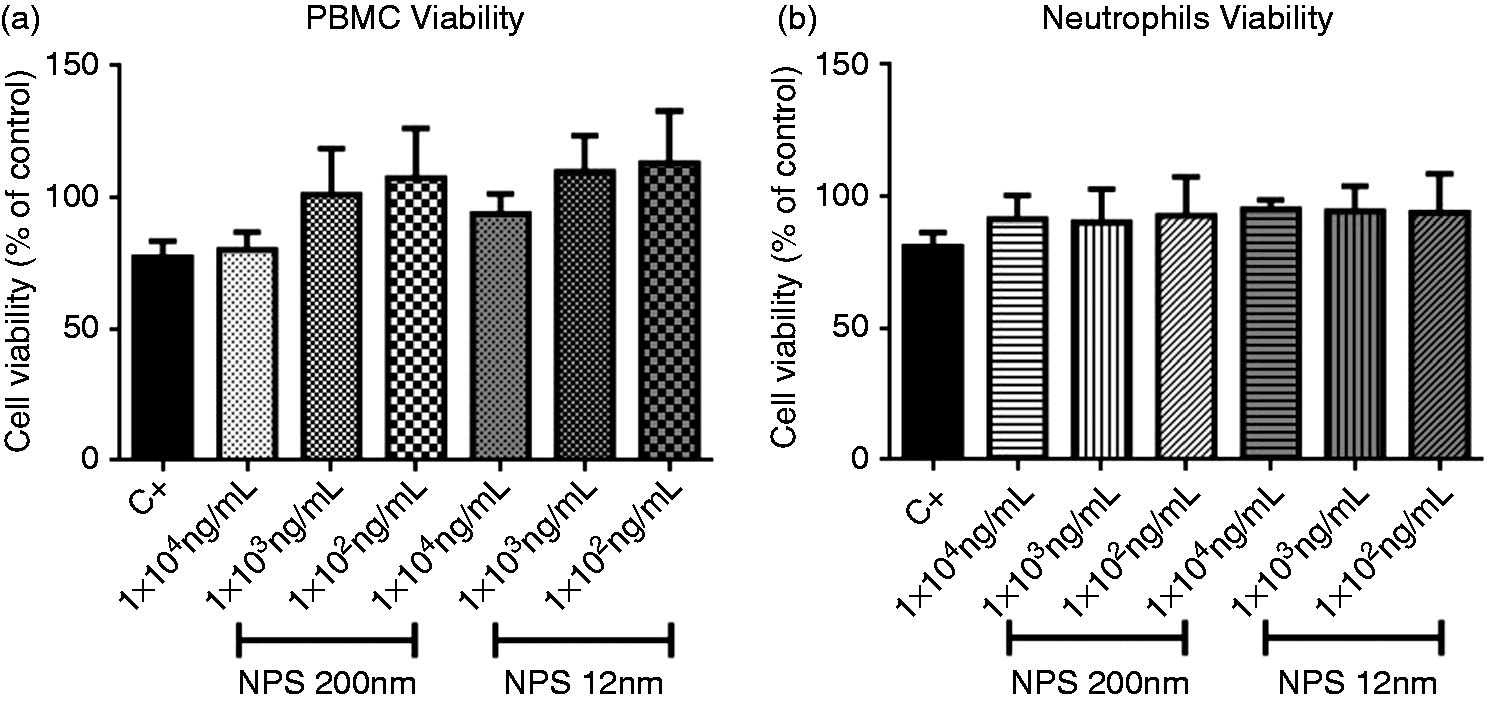
SiNPs induce IL-1β and IL-6 release in PBMCs
The release of two pro-inflammatory cytokines after treatment with 12-nm and 200-nm SiNPs was assessed in the culture supernatants of PBMCs. The results indicate that 200-nm SiNPs induced the release of IL-6 at SiNP concentrations > 1 × 104 ng/ml (Figure 2); 12-nm SiNPs induced detectable levels of IL-1β at SiNP concentrations > 1 × 101 ng/ml compared with negative control, but only resulted in significant differences at concentrations > 1 × 104 ng/ml (Figure 2). The 12-nm NPs also induced the release of IL-6 at concentrations > 1 × 102 ng/ml (Figure 2). Neutrophil stimulation with 12-nm and 200-nm SiNPs did not result in the significant production of IL-1β, as compared with the cells without stimulation (Figure 3). IL-6 was only detected in neutrophils stimulated with 200 nm NPs at 1 × 104 ng/ml. Levels of IL-18 from PBMCs and neutrophils stimulated with 12-nm and 200-nm SiNP were not detected (data not shown).
SiNPs induce IL-1β and IL-6 release in PBMCs. PBMCs were treated with SiNPs overnight, and supernatant levels of IL-1β and IL-6 were assessed by ELISA. Data represent the medians ± IR of at least three independent experiments. Kruskal–Wallis analysis shows significance. * SiNP induction of IL-1β and IL-6 release in neutrophils. Neutrophils were treated with SiNPs for 6 h, and supernatant levels of IL-1β and IL-6 were assessed by ELISA. Data represent medians ± IR of three independent experiments. Kruskal–Wallis analysis shows significance. *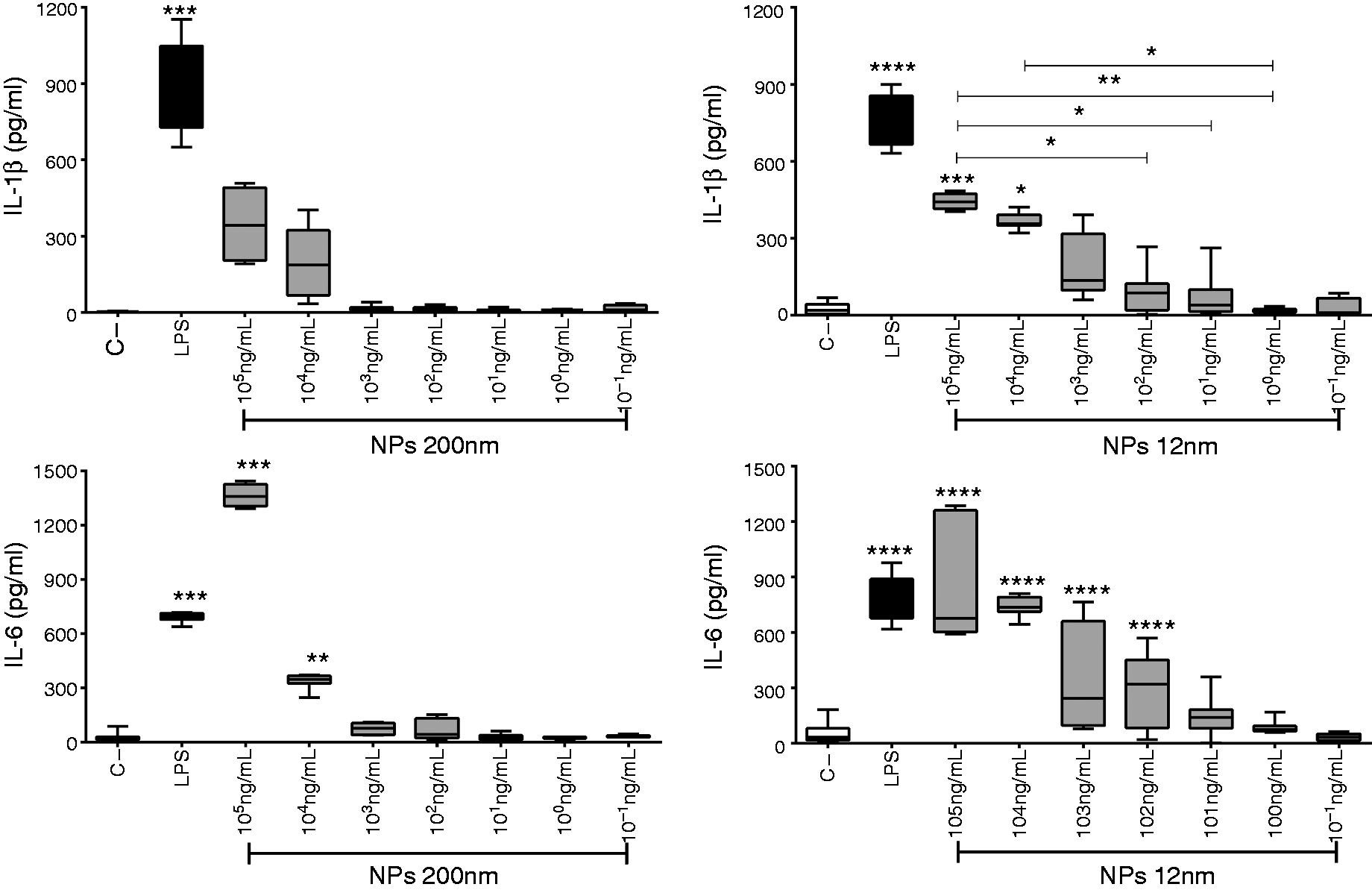
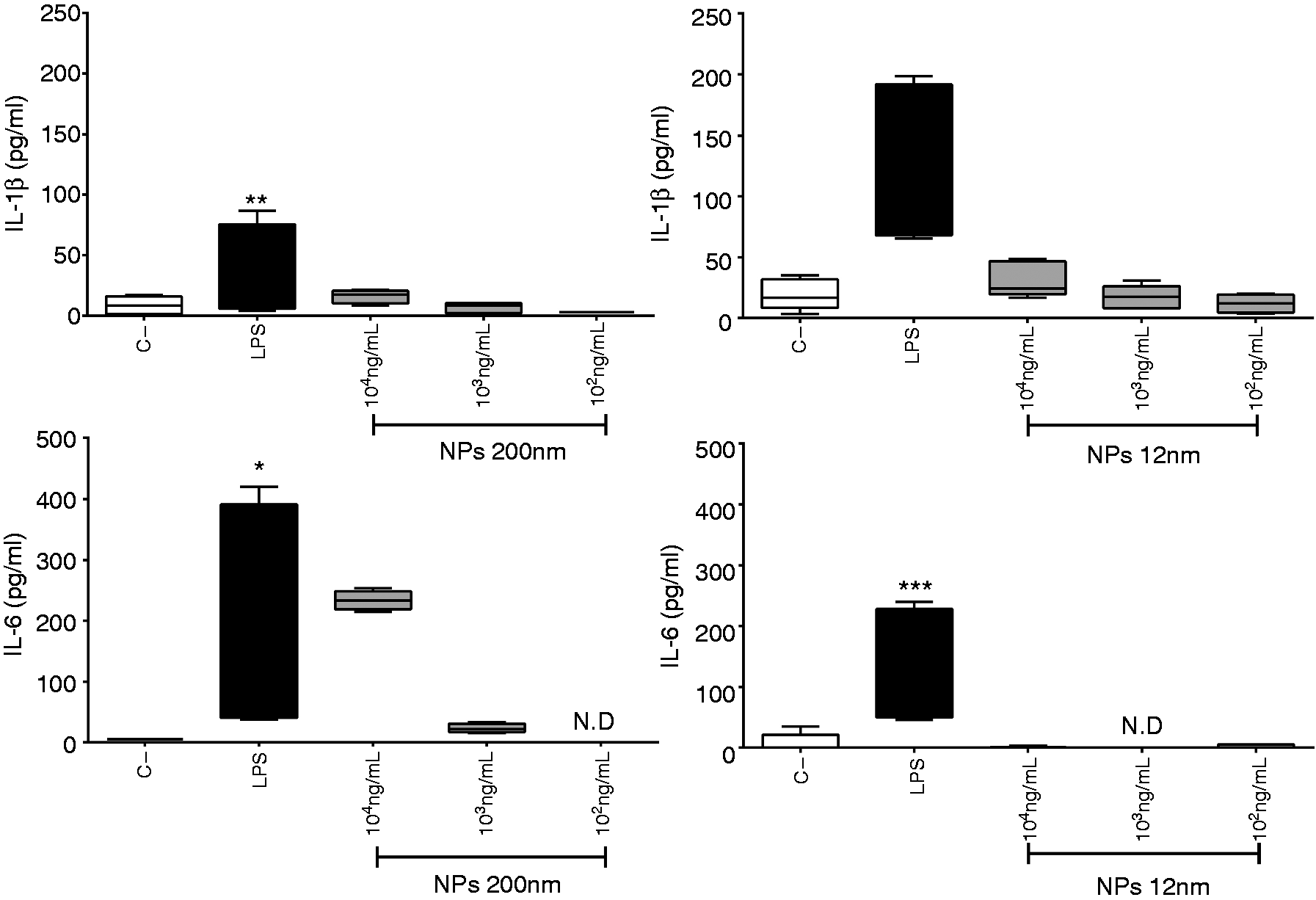
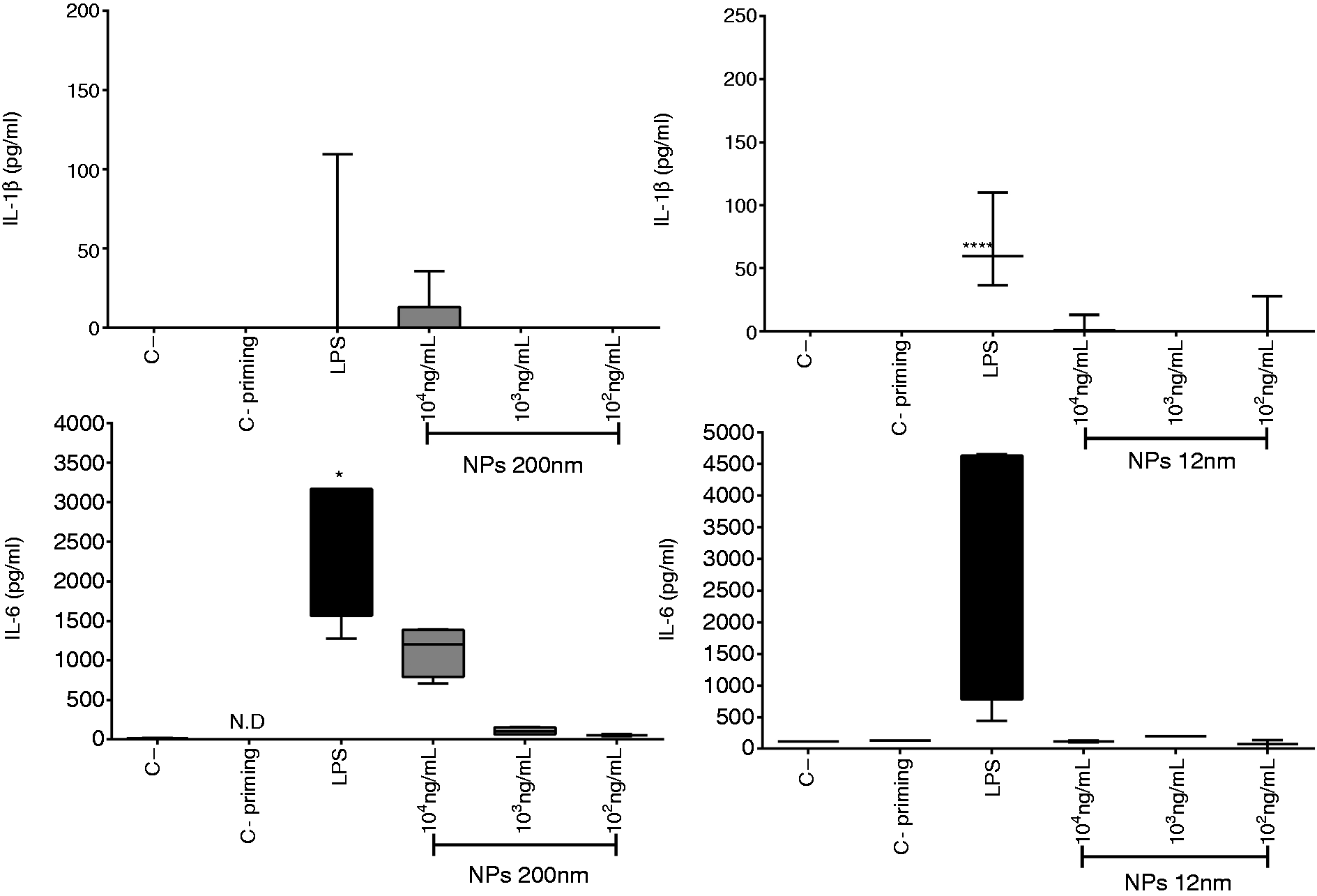
SiNPs induce IL-1β and IL-6 release in primed PBMCs
The activation of the NLRP3 inflammasome involves two specific signals; during the first (priming-signal), PAMPs such as LPS stimulate multiple signaling receptors and induce the expression of pro-IL-1β, pro-IL-18 and NLRP3.26,29 To generate a priming signal, PBMC and neutrophil cultures were primed with LPS (50 pg/ml, 2 h) before exposure to the SiNPs for 4 h. When PBMCs were primed and then exposed to 200-nm SiNPs (1 × 104 ng/ml), there was release of IL-1β at higher levels than when the PBMCs were not primed (Figure 4). However, the release of IL-1β was the same when the cells were exposed to 12-nm SiNPs with and without priming. IL-6 release was significantly elevated when PBMCs were exposed to 12-nm SiNPs at a concentration of 1 × 104 ng/ml (Figure 4). Levels of IL-18 from LPS-primed PBMCs stimulated with 12 nm and 200 nm SiNPs were not detected (data not shown). IL-6 production in LPS-primed neutrophils was detected after stimulation with 1 × 104 ng/ml 200-nm SiNPs (Figure 5) but not when exposed to any concentration of 12-nm NPs (data not shown). Levels of IL-1β or IL-18 from LPS-primed neutrophils stimulated with 12-nm or 200-nm SiNPs were not detected (data not shown).
SiNPs induce IL-1β and IL-6 release in primed PBMCs. PBMC cultures were primed with LPS (50 pg/ml, 2 h) and then treated with SiNPs (4 h). Supernatant levels of IL-1β and IL-6 were assessed by ELISA. Data represent medians ± IR of three independent experiments. Kruskal–Wallis analysis shows significance. * SiNPs induce IL-6 release in primed neutrophils. Neutrophils were primed with LPS (50 pg/ml, 2 h) and then treated with SiNPs (4 h). Supernatant levels of IL-6 were assessed by ELISA. Data represent medians ± IR of three independent experiments. Kruskal–Wallis analysis shows significance. *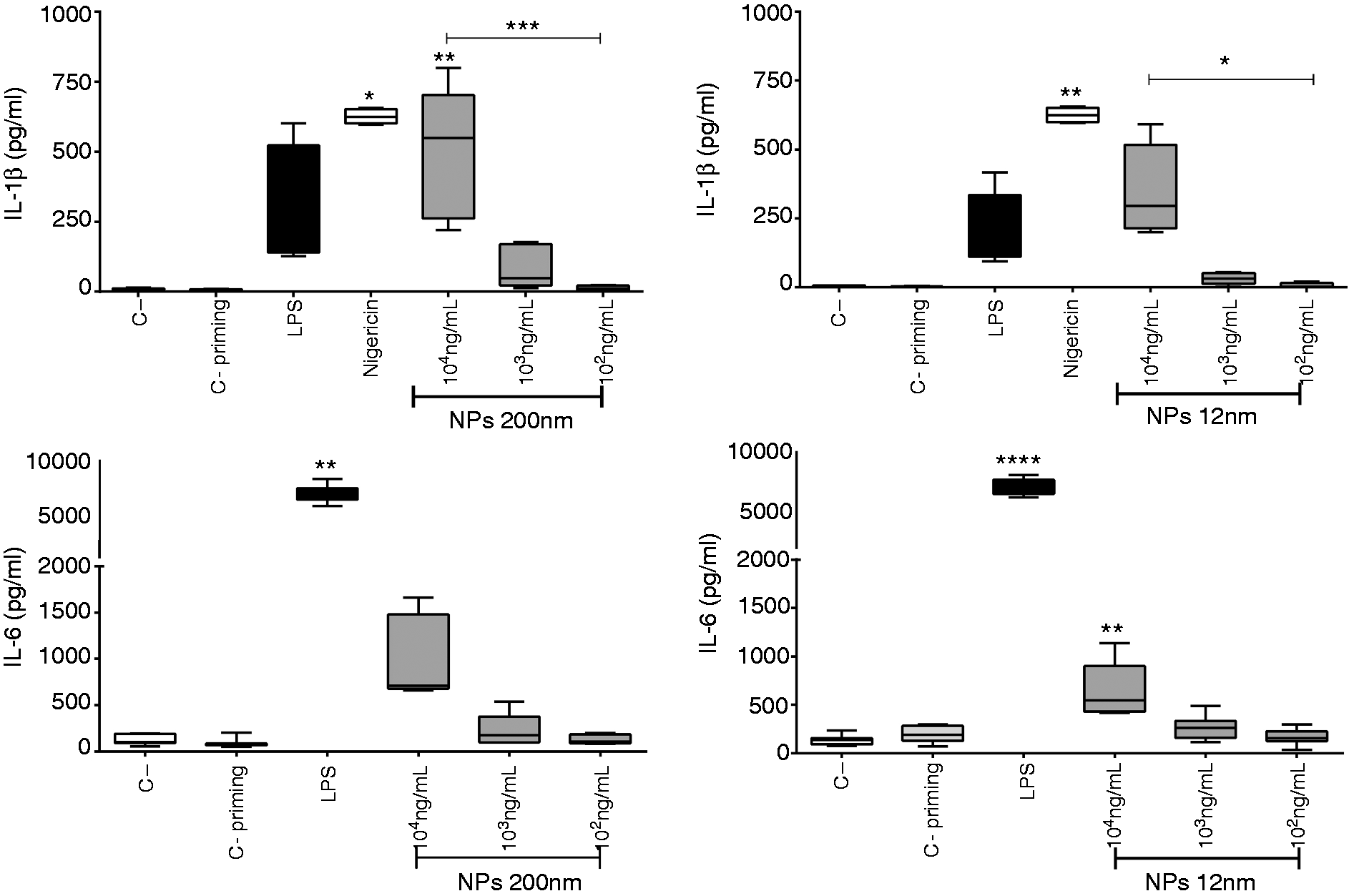
SiNPs induce the expression of NLRP3 inflammasome components and pro-inflammatory cytokines
Quantification of mRNA expression in PBMCs revealed that both 12-nm and 200-nm SiNPs trend toward increasing the mRNA of IL-6 and IL-1β at two concentrations tested (1 × 104 and 1 × 103 ng/ml) in PBMCs. Likewise, the 200-nm SiNPs administered at 1 × 104 ng/ml seems to increase the expression of TLR2, TLR4, ASC and NLRP3 (Figure 6). The 12-nm SiNPs administered at 1 × 103 ng/ml seems to decrease expression of TLR4 and trend toward to increased expression of NLPR3 (Figure 6).
SiNPs induce the expression of NLRP3 inflammasome components and pro-inflammatory cytokines in PBMCs. Expression of TLR2, TLR4, NLRP3, ASC, caspase-1, IL-1β, IL-18 and IL-6 genes in PBMCs stimulated for 4 h with 12 nm and 200 nm SiNPs (1 × 104 and 1 × 103 ng/ml) analyzed by quantitative PCR. C+: positive control (LPS 50 ng/ml); CPRI: priming control (LPS 50 pg/ml).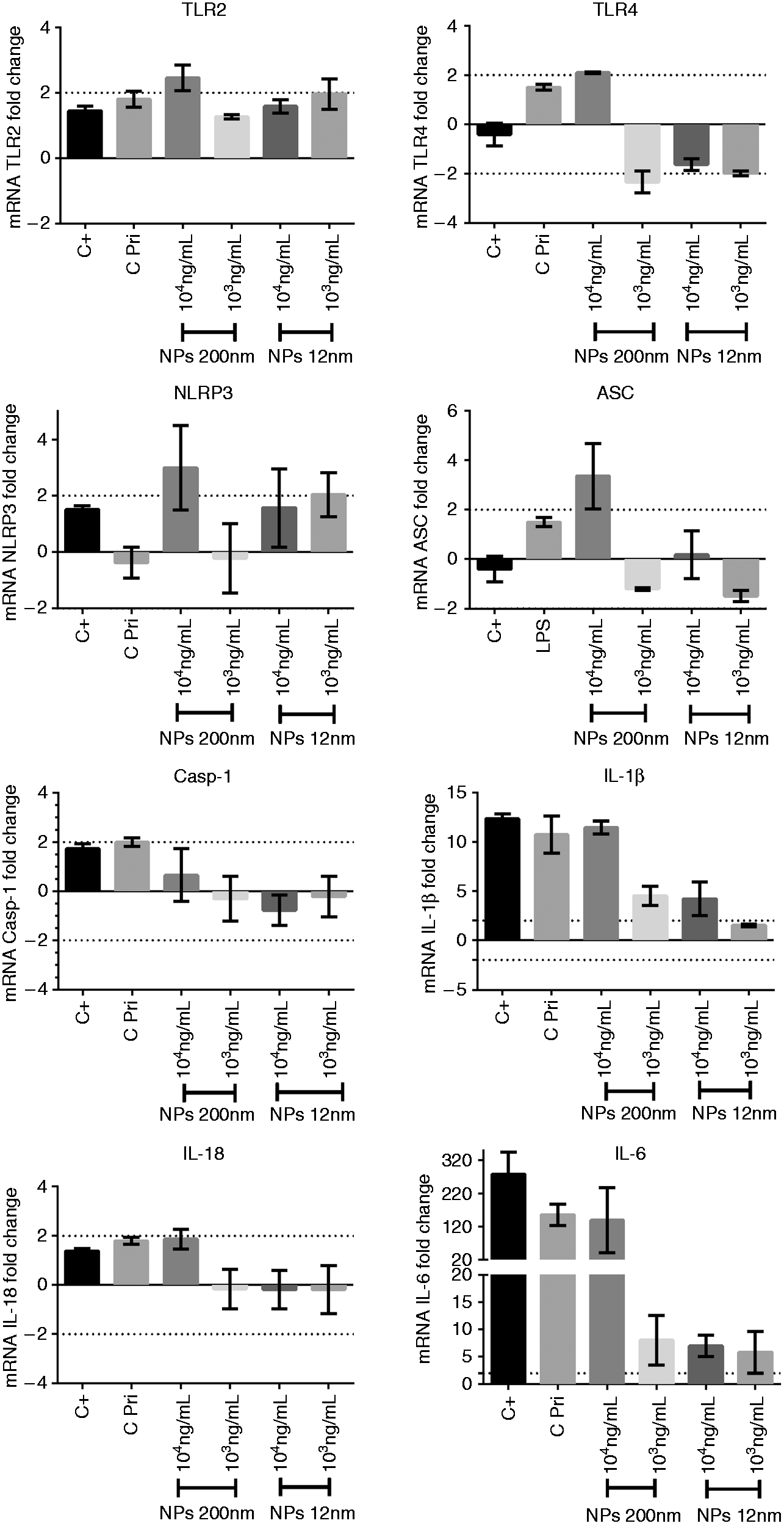
As shown in Figure 7, the 200 nm SiNPs at 1 × 104 ng/ml increased the expression of ASC, NLR3, IL-1β and IL-18 in neutrophils, and also increased the expression of caspase 1 and at IL-6 1 × 103 ng/ml. The 200-nm SiNPs also tended to increase the mRNA level of TLR2 at two concentrations tested, NLRP3 at (1 × 103 ng/ml) and caspase 1 (1 × 104 ng/ml). Although 12-nm SiNPs increased the expression of NLRP3, TLR2 and caspase 1 at 1 × 103 ng/ml, and also increase the expression of IL-1β at 1 × 104 ng/ml. Likewise, the 12-nm SiNPs seems to increase de mRNA levels of TLR2, ASC and caspase 1 (1 × 104 ng/ml), and IL-18 (at 1 × 103 ng/ml) (Figure 7).
SiNP induce the expression of the inflammasome NLRP3 components and pro-inflammatory cytokines in neutrophils. Expression of TLR2, TLR4, NLRP3, ASC, caspase-1, IL-1β, IL-18 and IL-6 genes in neutrophils stimulated for 4 h with 12 and 200 nm SiNPs (1 × 104 y 1 × 103 ng/mL) was analyzed by quantitative PCR. C+: positive control (LPS 50 ng/ml); CPRI: priming control (LPS 50 pg/ml).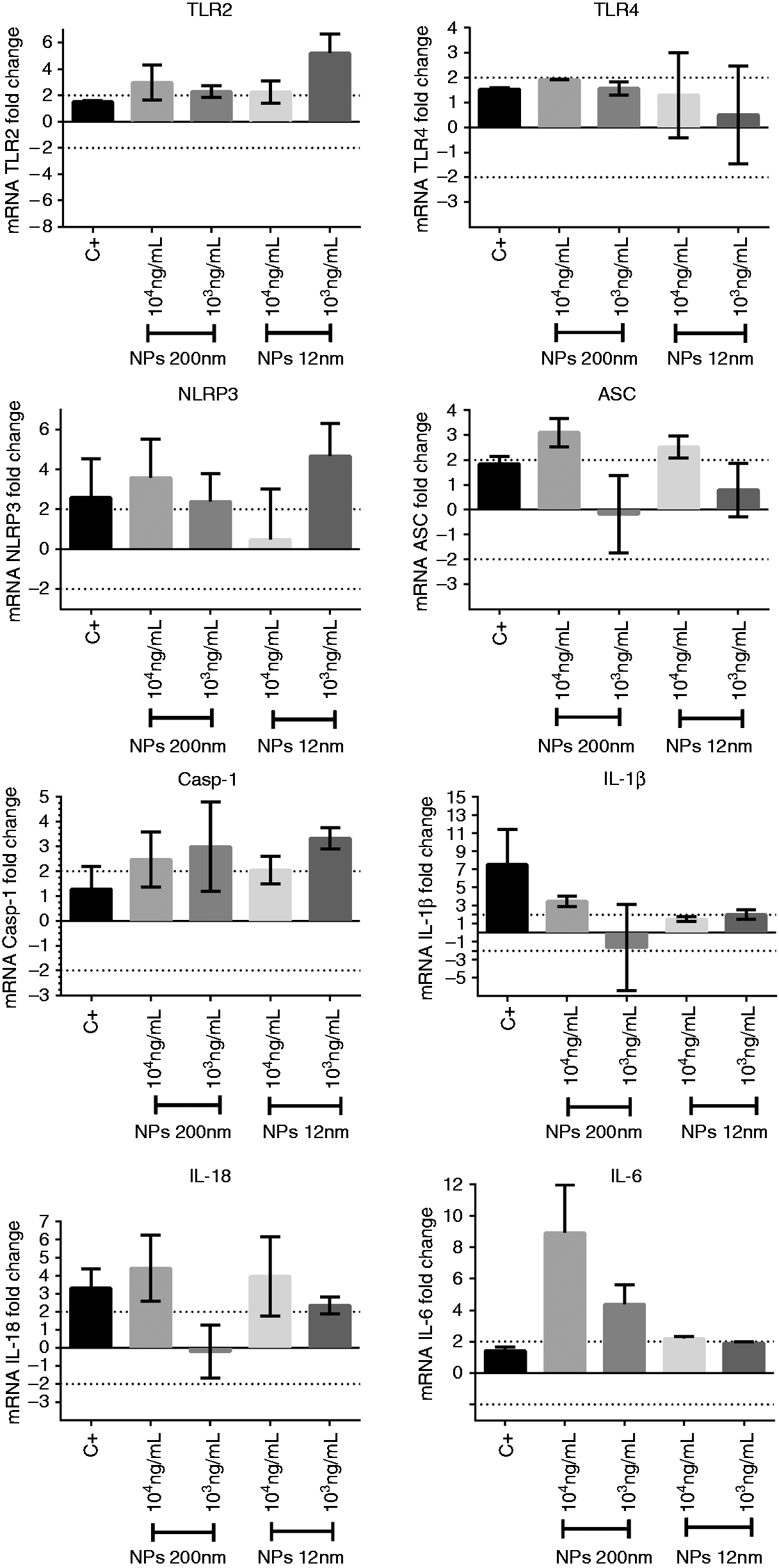
Discussion
SiNPs have been used in nanobiotechnology for various applications, such as controlled drug/gene delivery matrices, biosensors, diagnostic imaging and tissue engineering.3,30,31 However, SiNPs can induce immune responses affecting their safe use for human applications.
In these studies, we ensured that all experiments focused on measuring cytokine levels were conducted in non-cytotoxic conditions (Figure 1). It has been previously reported that cytotoxic effects of SiNPs appear at concentrations > 1 × 104 ng/ml in different cellular models.28–30 Similar to previous reports, 22 our results provide evidence that both 12-nm and 200-nm SiNPs induce the production of IL-1β and IL-6 by PBMCs. However, SiNPs do not induce detectable IL-18 production (as observed in response to microbial stimulation), likely owing to low levels of this cytokine that cannot be detected by the ELISA test used.35,36 The induced cytokine levels could have an impact on individuals treated with SiNPs, resulting in initiation of the innate immune response and also regulation of adaptive immunity.37,38
Neutrophils are among the first immune cells to arrive at the site of inflammation, where they initiate a rapid, non-specific, phagocytic response and the release of soluble mediators.
39
However, our results indicate that 200-nm SiNPs induce IL-6 but not IL-1β or IL-18 production in our
Similar to others reports, 5 our results indicate that cytokine production after exposure to SiNPs is dose-dependent, but contrary to reports from other groups,22,46 this response was not dependent on the size of the NPs. These contradictory findings suggest that various nanomaterials induce biological effects in response to differences in particle characteristics, but this response is also dependent on the cellular model evaluated, as indicated by our results.
Maturation of IL-1β and IL-18 is strongly regulated by the NLRP3 inflammasome. Thus, even if pro-IL-1β/18 protein is present in the cell, it cannot be released until NLRP3 inflammasomes are formed. After the correct assembly of NLRP3 inflammasomes, caspase-1 is activated and cleaves the pro-form of IL-1β/18 to generate mature IL-1β/18.12,13 Therefore, we evaluated the expression of NLRP3 inflammasome components after the cells were stimulated with SiNPs. Our results provide evidence that SiNPs induce expression of the inflammasome components (NLRP3, ASC, Casp-1), as well as IL-1β, IL-6 and IL-18 in PBMCs and neutrophils in a dose-dependent manner.
The mechanism by which PBMCs and neutrophils recognize SiNPs and then activate the NLRP3 inflammasome with the consequent release of IL-1β and IL-18 is not fully understood. A recent report indicated that scavenger receptor B1 is a silica receptor associated with canonical inflammasome activation associated with caspase-1-mediated inflammatory responses in mouse macrophages and human peripheral blood monocytes. 47 . Lately, different authors have reported that cathepsin B leakage after lysosomal rupture and the presence of cytoplasmic ROS play a crucial role in the activation of the NLRP3 inflammasome. However, it is unclear if these phenomena act separately or in a coordinated manner or how endosomal rupture occurs.5,15 Some studies have reported that SiNPs activate NLRP3 inflammasomes after the generation of ROS, leading to IL-1β release.14,48
Some reports suggest that ROS trigger destabilization of the lysosomal membrane by lipid oxidation. Kojima et al. demonstrated that ATP released from the P2X7 receptor in response to the stimulation of KUP5 cells with SiNPs induces ROS production via cell-membrane NADPH oxidase. 46 The ROS activation of inflammasomes leads to caspase-1-dependent processing of IL-1β.
However, some studies suggest that phagocyte membrane binding with particles may directly regulate NLRP3 inflammasome activity accompanied by K+efflux. The tension generated during particle binding changes the surface curvature and triggers ion channel opening. Recent papers reviewed a list of NLRP3 activators and identified K+ efflux as an event common among them all; however, mechanisms of ion channel opening related to NLRP3 activation remain unclear. 9
Several studies regarding immunological studies with NPs have been conducted using macrophage cell lines, but not in PBMCs or neutrophils; it was recently reported that macrophages differ in their mechanisms of inflammasome activation compared to other cells. The signals involved in inflammasome activation can be characterized as primers or activators of IL-1β production. The priming signals include PAMPs such as LPS and trigger the synthesis of inactive pro-IL-1β in macrophages via activation of TLRs, whereas elements such as K+ efflux, ROS or lysosomal rupture are considered upstream of inflammasome activation. While macrophages require two signals to produce mature IL-1β, monocytes and neutrophils constitutively express active caspase-1 and need only one signal for IL-1β release.16,17 However, our study shows that in PBMCs, priming with LPS and then stimulating with SiNPs results in greater production of IL-1β than in non-primed cells.
In conclusion, our results demonstrate that SiNPs induce the production of pro-inflammatory cytokines in a dose-dependent manner and that this stimulation takes place independently of NP size. Although our data suggest that the production and release of IL-1β occurs through the formation of the NLRP3 inflammasome, both 200 nm and 12 nm SiNPs are slightly inflammatory when these were used at low concentrations; this could be very useful for biomedical applications and devices where immune response is not desired. However, further studies are needed.
Footnotes
Acknowledgements
The authors thank the donor individuals who participated in this study and the collaboration of the personnel from Grupo Inmunovirología.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Universidad Cooperativa de Colombia and Univesidad de Antioquia UdeA. The funders had no role in the study design, data collection and analysis, decision to publish or preparation of the manuscript.
