Abstract
In 2004, a novel mechanism of cellular death, called ‘NETosis’, was described in neutrophils. This mechanism, different from necrosis and apoptosis, is characterized by the release of chromatin webs admixed with microbicidal granular proteins and peptides (NETs). NETs trap and kill a variety of microorganisms. Diverse microorganisms, including Mycobacterium tuberculosis, are NET inducers in vitro. The aim of this study was to examine whether M. tuberculosis can also induce NETs in vivo and if the NETs are bactericidal to the microorganism. Guinea pigs were intradermally inoculated with M. tuberculosis H37Rv, and the production of NETs was investigated at several time points thereafter. NETs were detected as early as 30 min post-inoculation and were clearly evident by 4 h post-inoculation. NETs produced in vivo contained DNA, myeloperoxidase, elastase, histones, ROS and acid-fast bacilli. Viable and heat-killed M. tuberculosis, as well as Mycobacterium bovis BCG were efficient NET inducers, as were unilamellar liposomes prepared with lipids from M. tuberculosis. In vitro, guinea pig neutrophils also produced NETs in response to M. tuberculosis. However, neither the in vivo nor the in vitro-produced NETs were able to kill M. tuberculosis. Nevertheless, in vivo, neutrophils might propitiate recruitment and activation of more efficient microbicidal cells.
Keywords
Introduction
Tuberculosis is a chronic inflammatory disease caused by Mycobacterium tuberculosis. The disease has a worldwide distribution and high rates of morbidity and mortality. 1 Many efforts have been made to unravel the immunologic mechanisms that confer protection against the disease, and the coordinated role of T cells and macrophages has been recognized as a fundamental mechanism for tuberculosis control. 2 It is only recently that the participation of polymorphonuclear (PMN) cells in the immunology of tuberculosis has been pondered, but their role is still highly debated. 3
As macrophages and dendritic cells, PMN cells are essential components of innate immunity and actively participate in the capture and killing of microorganisms. These cells are the best endowed with immediate microbicidal properties because they are completely mature cells. They rapidly respond to infection by surrounding invaders to engulf and kill them through both oxygen-dependent and oxygen-independent mechanisms, including reactive oxygen species (ROS), lysosomal enzymes and antibiotic peptide pathways.4–8
In 2004, a new microbicidal strategy of neutrophils was described; 9 it involved the release of neutrophil extracellular traps (NETs) through which these cells also ensnare and kill microorganisms. Neutrophils stimulated in vitro with LPS, phorbol 12-myristate 13-acetate (PMA) or IL-8 release NETs made of chromatin (DNA and histones) intermingled with granular (lysosomal) proteins but lacking cytoskeletal (actin) components.9–11 Additionally, diverse bacteria such as Staphylococcus spp., Shigella spp. and Salmonella can induce NETs, in which they are trapped and inactivated. 9 In addition to bacteria, other microorganisms, including protozoa,12–14 fungi10,15–17 and viruses18–20 can also induce the production of NETs with the properties mentioned above. NETs inactivate microorganisms through antimicrobial agents intermixed with chromatin. The antimicrobial agents so far described include granular proteins such as elastase, myeloperoxidase (MPO), cathepsin G, lactoferrin, proteinase 3, calprotectin,10,21–23 histones and the antibiotic peptides LL37, BPI and lipocalin-2. 24 Some microorganisms, notably Streptococcus pneumoniae and Staphylococcus aureus, resist the microbicidal effect of NETs because they secrete DNases that disarrange and inactivate the NETs.25,26 NETs produced in vivo may be beneficial because of their antimicrobial properties. 27 However, the continued in vivo production and accumulation of NETs, and their persistent deposition in host tissues may be detrimental for the host as has been reported for autoimmune and other inflammatory diseases.28–31 Two recent reviews on the meaning of NETs in health and disease, are those by Brinkmann and Zychlinsky, 32 and Jorch and Kubes. 33
Thus, NETs are produced in response to diverse chemical agents, auto-Ags and microorganisms. In a previous study, we have reported on the production of NETs by human neutrophils stimulated in vitro with M. tuberculosis and Mycobacterium canetti. 11 In that study, both mycobacteria induced the release of NETs in vitro, but these structures were unable to destroy the microorganisms. It was envisaged, however, that in vivo NETs could have an effect on M. tuberculosis, thus directing the outcome of the disease.
In the present article, we show that neutrophils participate in the very early response of guinea pigs to in vivo intradermal infection with M. tuberculosis, and that these cells respond to infection with the production of NETs. We also show that neutrophils, at a time when NET expression is at peak (6 h), are unable to kill M. tuberculosis.
Guinea pig was used as the experimental model because this animal is highly susceptible to tuberculosis, and the intradermal route was chosen because the skin is an appropriate organ for the study of the in situ host–parasite interactions occurring during infection as, for instance, in cutaneous tuberculosis, one of the several extra-pulmonary forms of the disease in the human being.
Materials and methods
Ethics statement and safety measures
Animal procedures were approved by the Committee for the Ethical Principles in Research of the National School of Biological Sciences, National Polytechnic Institute (Approval: CEI-ENCB-013/2013). All of the experiments with mycobacteria were carried out in a class 2 biosafety cabinet within a BSL3 laboratory.
Culture and labelling of mycobacteria
M. tuberculosis H37Rv (MTB), Mycobacterium bovis BCG (vaccine Japan BCG) and Mycobacterium smegmatis (TMCC 1515) were grown in Middlebrook 7H9 broth supplemented with 10% OADC (Becton Dickinson, Sparks, MD, USA). To avoid bacterial clumps, mycobacteria were grown under constant agitation by using a magnetic stirrer and the integrity of mycobacteria was evaluated by means of the Ziehl–Neelsen stain. Mycobacteria were used either intact (>90% viability in all cases) or heat-inactivated (121℃/10 min). The number of bacteria used in each experiment was calculated from the number of CFUs grown on Middlebrook 7H10 agar (Becton Dickinson). Ten µl of serially diluted bacterial suspensions were seeded on 7H10 agar plates and the plates were incubated at 37℃ for 2–3 wk; the number of colonies developed was counted under an inverted microscope.
In some experiments, M. tuberculosis was pre-labelled with Iris Fuchsia, a lipophilic fluorescent (588 nm) dye (Cyane Technologies, Turin, Italy). One microliter of Iris Fuchsia (at 1 mg per ml DMSO) was diluted 1:1000 in PBS, and 1 ml of the solution was incubated with 1010 bacteria for 20 min in the dark. Stained bacteria were washed three times with PBS (2700 g for 5 min), and then suspended in DMEM supplemented with 10% FCS (DMEM-FCS) to a concentration of 1 × 1010 bacilli per ml. Staining with Iris Fuchsia did not affect the viability of M. tuberculosis in a significant manner as deduced from the count of CFUs on 7H10 agar medium (> 80% of bacteria remained viable).
Characterization of mycobacteria-induced NETs in guinea pigs
Albino adult Hartley guinea pigs were obtained from the local Institutional Animal House, and they were handled according to the regulations of the Mexican Official Norm for the Production, Care and Use of Laboratory Animals, 1999 (NOM-062-ZOO-1999; 22 August, 2001). Over the 2 yr of the project, at least 14 animals were employed; thus, the results shown are representative of several experiments, many of them repeat experiments. Guinea pigs were intradermally inoculated with 107 bacteria in 10 µl PBS using a 0.3-cc syringe with a 27-G needle. Lesser amounts of bacteria, e.g. 106, gave less conspicuous infiltrates, inadequate for the experiments intended. The shaved flanks of guinea pigs were inoculated at single sites at sequential time points to cover several periods of infection, namely 0.5, 1, 2, 4, 6 and 8 h. The injection sites were outlined with permanent ink to mark the areas. Guinea pigs were anaesthetized with 6.5 mg/kg of intramuscular Ketalar (ketamin chlorhydrate; Pfizer, Mexico City, Mexico) and then killed by CO2 inhalation in a confined container. The injection sites were carefully removed and divided in two. One half was preserved in 4% formalin for histopathological analysis, and the other half was embedded in Tissue-Tek OCT Compound (Sakura Finetek, Torrance, CA, USA) for cryosectioning.
In some experiments, guinea pigs were inoculated with liposomes made of lipids extracted from M. tuberculosis. Whole lipids were extracted according to Folch et al. 34 from the biomass of M. tuberculosis grown in synthetic PBY medium. With these lipids, unilamellar liposomes were prepared according to a protocol by Chatterjee and Banerjee. 35 Briefly, 4 mg whole mycobacterial lipids dissolved in 1.0 ml chloroform were placed into a 20 × 100 mm screw-cap tube and the solvent was removed by rotary evaporation at 60℃ to produce a thin lipid film. Next, 5 ml 0.01 M PBS, pH 7.4, was added, and the tube was vigorously vortexed to produce multilamellar vesicles (MLVs). MLVs were then sonicated at 60℃ for 15 min to produce small unilamellar liposomes. Ten µl of the liposome suspension were intradermally inoculated into the shaved back of a guinea pig and, 4 h later, the animal was killed and skin sampled for sectioning.
Skin sampling
Paraffin-embedded samples
Skin samples were collected and fixed in 10% neutral formalin for 8 d. Then they were rinsed in water and subjected to a standard process for paraffin sectioning (http://www.openwetware.org/wiki/Paraffin embedding and sectioning). Three-µm-thick paraffin sections were prepared in a Leica RM2125RT microtome (Leica Biosystems, Nussloch, Germany) and the sections were kept at 25–26℃ until used for hematoxylin and eosin, acid-fast bacilli and Toluidine blue staining.
Tissue-Tek-embedded samples
Some skin samples were collected, rinsed in PBS (pH 7.4), properly trimmed, oriented tangentially in a cryo-mold to display all the skin layers, embedded in tissue freezing medium (Tissue-Tek OCT Compound) and snap-frozen in a dry ice–acetone blend. The frozen blocks were then cut in a cryostat (Lab-Tek Division, Miles Laboratories, Naperville, IL, USA) at −20℃. Four-µm-thick sections were prepared, fixed with 1% paraformaldehyde in PBS for 10 min, rinsed twice with PBS, air-dried and kept under vacuum until used.
Staining
Acid-fast bacilli staining
Paraformaldehyde-fixed cryostat tissue sections were hydrated in PBS for 10 min, and then stained with the Merck (Ziehl-Neelsen) acid-fast bacilli staining kit 109215 (Merck, Darmstadt, Germany).
DNA staining
For DNA staining, Hoechst 33342 fluorescent dye (Thermo Fisher Scientific, Rockford, IL, USA) was added at a concentration of 0.1 µg/ml in PBS for 10 min at 25℃ in the dark. This fluorescent dye stains the DNA from intact and disrupted cell nuclei and emits blue fluorescence with excitation at 350 nm and emission at 461 nm. NETs were identified by their finely reticular fluorescent structure at low magnifications and fibrillar fluorescent structure at higher magnifications. In experiments where Iris Fuchsia-stained bacteria were inoculated, they appeared as red fluorescent rods. For examination under the microscope (Nikon Eclipse E800; Nikon, Tokyo, Japan), the stained sections were protected with anti-fade Vectashield (Vector Laboratories, Burlingame, CA, USA).
MPO, elastase and histone staining
For MPO, elastase and histone staining, adjacent cryostat skin sections were (a) fixed with 1% paraformaldehyde in PBS, (b) rinsed in PBS, (c) blocked with 1% BSA in PBS-Tween for 2 h, (d) incubated 24 h at 4℃ with goat anti-histone (H3) Ab (Ab11946; Abcam, Cambridge, MA, USA), rabbit polyclonal to anti-MPO Ab (Ab9535; ABCAM) or rabbit anti-elastase polyclonal Ab (Ab21595; Abcam), all diluted 1:3000 in PBS–0.4% Tween 20. As secondary Abs, donkey anti-rabbit IgG (Alexa Fluor 647, Ab150075; Abcam) and donkey anti-goat IgG (Alexa Fluor 488, Ab150129; Abcam) were used diluted 1:5000 in PBS-T for 4 h. Finally, sections were mounted in Vectashield Mounting Medium with DAPI (H-1200; Vector Laboratories). Images were taken and analyzed with an Axioscop 2 mot plus confocal fluorescence microscope (Carl Zeiss, Mexico City, Mexico).
Nitro blue tetrazolium staining
To assess the in vivo reduction of nitro blue tetrazolium (NBT; indicative of the presence and activity of superoxide anion), the bacteria, suspended in 0.1% NBT in 0.15 M NaCl, were injected into the guinea pig skin at a dose of 107 microorganisms in 10 µl of inoculum. Sampling was performed at 6 h thereafter. At this time, (a) skin fragments were excised, (b) embedded in Tissue-Tek, (c) frozen on a blend of dry ice in acetone, (d) sectioned at 4 µm, (e) fixed in 1% paraformaldehyde, (f) stained for DNA, (g) rinsed in distilled water and (h) mounted with Vectashield for microscope examination. Except for DNA (which requires staining with Hoechst reagent), no counter stain was necessary because the reduced NBT produces a blue formazan deposit at the reaction site.
Semi-quantitative assessment of NETs
A semi-quantitative assessment of NETs (in pixel density per area) was performed using ImageJ 1.47v software (http://imagej.nih.gov/ij). Statistical analysis of the data was done with the Kruskall–Wallis test, as suggested by ImageJ.
Determination of the viability of M. tuberculosis trapped in NETs in vivo
Ten million M. tuberculosis cells in 10 µl PBS were intradermally inoculated in the skin of guinea pigs. After 6 h of infection (when NET expression was at the highest) guinea pigs were sacrificed; control sites were produced by injecting the mycobacteria immediately before sacrifice. The inoculated sites were excised with a 5-mm Ø puncher tool. The skin samples were collected in microtubes containing 1.0 mg dispase (D4818; Sigma-Adrich, Toluca, Mexico) in 1 ml PBS, and left at 25℃ for 18 h. Next, the skin samples were rinsed three times with saline solution containing 0.1% Tween-80 (SS-T80), transferred to a fresh tube with 200 µl SS-T80 and grounded with a 3-cc glass homogenizer. The suspension was adjusted to 1.0 ml with SS-T80 and filtered through a 70-µm cell sieve (Becton Dickinson); the filtrate was diluted 1:10, 1:100, 1:1000 and 1:10,000 in SST80. Ten microliters of each dilution were dropped on Middlebrook 7H10 agar plates (Becton Dickinson), and the plates were incubated at 35℃ for 2–3 wk. Mycobacterial colonies were counted thereafter.
Mycobacteria-induced NETs in vitro
Neutrophils were isolated from heparinized blood taken from deeply anesthetized guinea pigs by centrifugation on Polymorphprep (Axis-Shield PoC, Oslo, Norway) according to the manufacturer’s instructions. The cells (> 95% neutrophils) were seeded at the density of 1 × 106 cells per ml/well in 24-well microplates (containing one 18-mm Ø glass-coverslip per well). The cells were kept unstimulated, or they were stimulated with 25 nM PMA or M. tuberculosis at a MOI of 20:1 for 6 h at 37℃/5% CO2. The cells were then (a) fixed with 1.0 ml of 4% paraformaldehyde in PBS for 10 min, (b) gently rinsed with PBS, (c) stained with the Hoechst reagent 1:1000 (in PBS) for 10 min, (d) rinsed with distilled water, (e) air dried, (f) protected with Vectashield and (g) mounted on glass slides for UV examination (blue filter). Some cell preparations were stained for 10 min with the hot Ziehl–Neelsen stain, briefly discolored with acid alcohol (1% HCl in 70% ethanol), rinsed with distilled water and stained with the Hoechst reagent as above.
In vitro assessment of the microbicidal activity of NETs on M. tuberculosis
To determine the microbicidal activity of ‘netotic’ neutrophils on M. tuberculosis, guinea pig neutrophils were isolated from heparinized blood as described above. The viability of cells was > 98% as assessed by Trypan Blue dye exclusion. Neutrophils were cultured in RPMI (Gibco, Invitrogen, Carlsbad, CA, USA) supplemented with 2% FCS (Sigma-Aldrich, St. Louis, MO, USA) and allowed to adhere for 30 min to 24-well flat bottom plates at 2 × 105 cells per well. The cells were infected with M. tuberculosis H37Rv at MOIs of 0.1:1, 1:1 and 10:1 for 30, 90, 180 and 360 min. After infection, cultures were lysed with Triton-X 100 (0.25% in PBS) for 3 min. Neutrophil lysates were serially diluted and plated on 7H10 medium supplemented with OADC at the indicated time points. As a control, M. tuberculosis was incubated with Triton-X 100 alone. Surviving colonies were counted after 2–4 wk of culture at 37℃.
Results
M. tuberculosis induces the formation of NETs in the skin of guinea pigs
The normal histology of the skin of a guinea pig is shown in Figure 1. The skin sections were stained with the toluidine blue stain (Figure 1A) and Hoechst reagent (Figure 1B). Whereas the toluidine blue stain shows the integral structure of the skin, including the epidermis, dermis with annexes (hair follicles and sebaceous glands) and hypodermis, the Hoechst stain shows the normal distribution of the cells and their intact nuclear structure. To determine the initial intradermal location of mycobacteria, we removed the M. tuberculosis-inoculated skin site immediately after inoculation, before recruitment of inflammatory cells. Figure 1(C) depicts a panoramic image comprising the injected site (rectangle), in which no inflammatory cells are observed (Hoechst stain, 10 × 10×); Figure 1(D) shows the same tissue section stained for acid-fast bacilli (Ziehl-Neelsen stain, 10 × 10×). While at this magnification, acid-fast bacilli appear as an amorphous spot in red fuchsia (rectangle), close to the root of a hair follicle, bacilli are clearly distinguished at a higher magnification (100 × 10×) (Figure 1D, inset).
Histological structure of a guinea pig skin section and the intradermal deposition of injected M. tuberculosis. (A) Normal skin of a guinea pig stained with toluidine blue; epidermis (e), dermis (d) with annexes (a) (hair follicles and glands) and hypodermis (h) are shown. (B) Normal skin of a guinea pig stained with the Hoechst stain for DNA; nuclei of all the cells in the tissue, including those of hair follicles, dermal glands and fibroblasts, appear fluorescent blue. (C) A skin section stained for DNA taken immediately after inoculation of M. tuberculosis. (D) The same skin section stained for M. tuberculosis with the Ziehl–Neelsen stain and no counterstaining. Rectangles mark the deposit of bacilli at the injection site, and inset in (D) is a 100 × 10 × magnification of bacilli in the rectangle; Apart from the presence of bacilli (red stain), no cell infiltration is observed. All main images at 10 × 10×.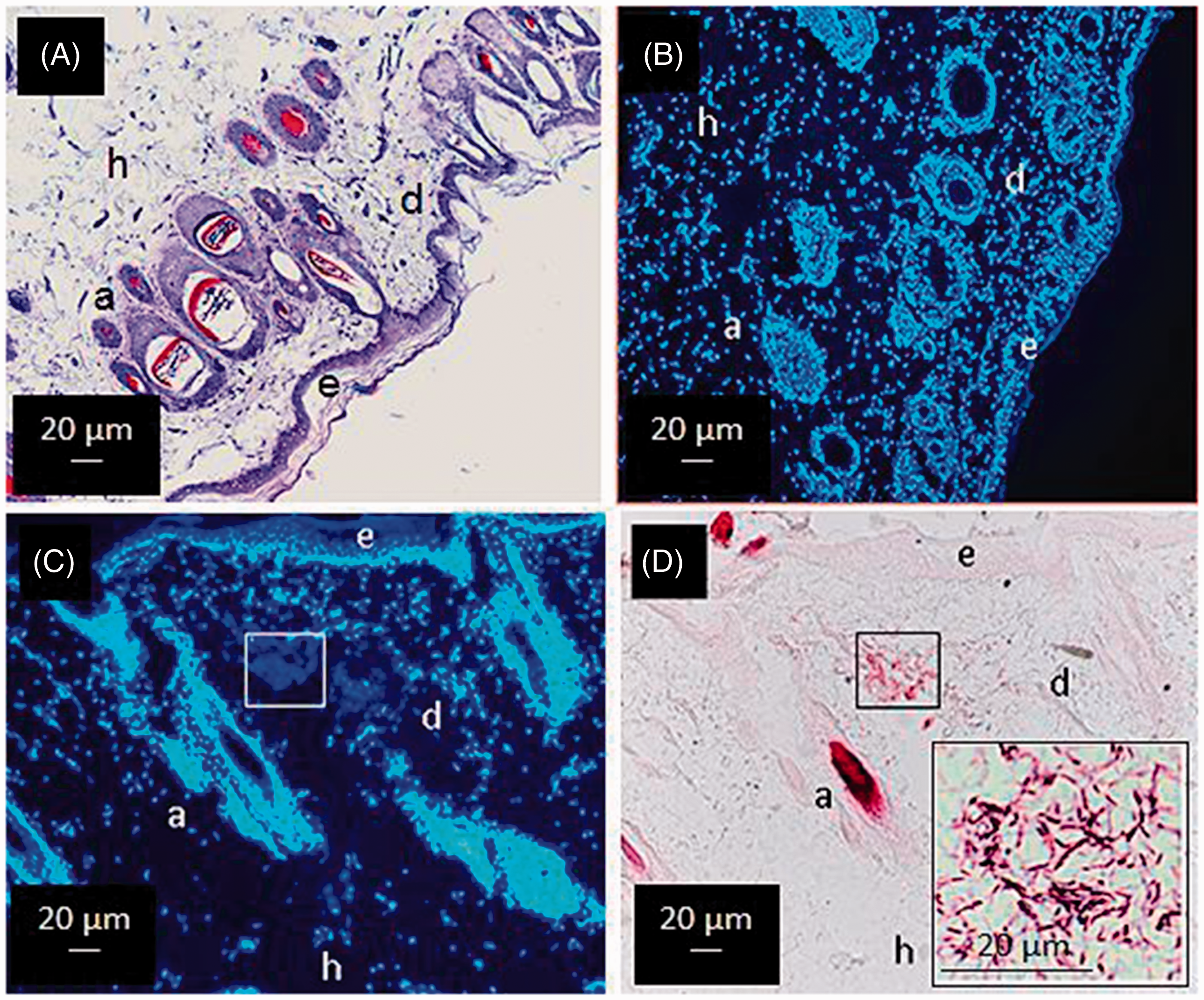
Neutrophils accumulate early at the infection site
Accumulation of cells at the injection site starts shortly after inoculation; a noticeable cell accumulation was evident by 2 h after infection in most cases, and > 95% of these cells were polymorphs (Figure 2A, a skin lesion stained with toluidine blue; Figure 2B an adjacent section stained with the Hoechst reagent).
Intradermal accumulation of inflammatory cells following inoculation of M. tuberculosis. Panel A: a prominent, localised cell infiltrate at the site of inoculation of M. tuberculosis in the dermal skin of a guinea pig 2 h after the injection (toluidine blue stain, 10 × 10×) (arrow). (B) An adjacent section stained for DNA (Hoechst stain) showing the nuclear staining of the infiltrating cells (arrow). (C) An adjacent section stained with hematoxylin and eosin (and Ziehl–Neelsen), from which the predominantly PMN nature of the infiltrate (dotted circle) was established; the inset shows a higher magnification (100 × 10×) of the infiltrate showing polymorphs containing bacilli (red). (D) A high magnification of the infiltrate stained for DNA showing the majoritarian PMN structure of the cells. Several cells seem to have initiated the process of NETosis: chromatin de-compaction and chromatin release (short arrows).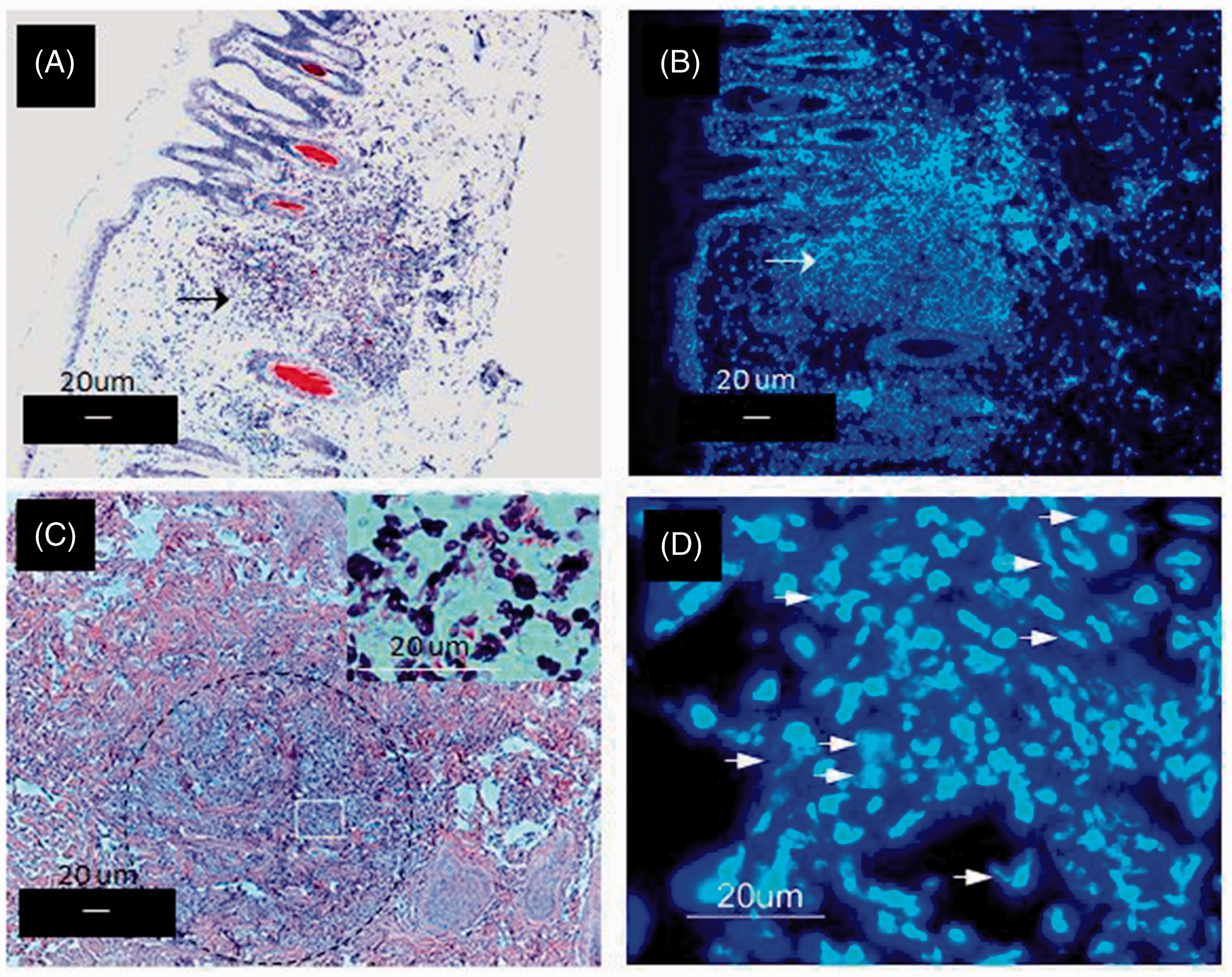
The cells of the early infiltrate intermingled with the collagen fibres, noticeably altering the dermal histology (Figure 2C), and most of the neutrophils contained bacilli (Figure 2C, inset). At this stage of infection, most cells were not producing NETs, although some of them did as deduced from the Hoechst staining (Figure 2D).
Incipient NETs are observed as early as 30 min post-infection
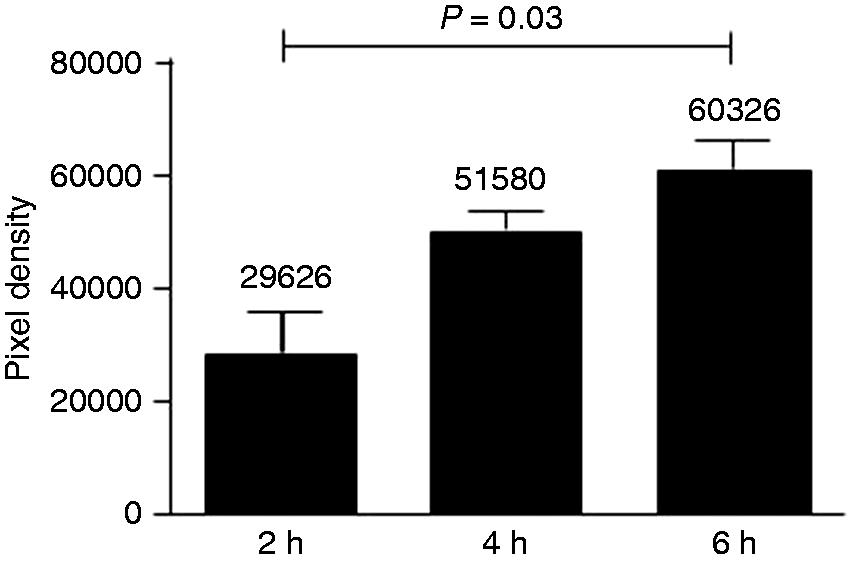
NETs appeared distinctively as early as 30 min after inoculation and these NETs contained mycobacteria (Figure 3). As time elapsed, the size of NETs increased in proportion to the number of neutrophils involved and they were always associated to the presence of bacilli. NETs reached their maximal expression between 4 and 8 h post-inoculation, with 6 h being an adequate time for their observation. NETs were identified by their finely reticular structure, showing a cloudy image at lower magnification (10 × 10×) and a fibrillar structure at higher magnification (60–100 × 10×) (Figure 4). A semi-quantitative assessment of the NETs observed in Figure 4 is illustrated in Figure 5. The analysis was performed with ImageJ and the data, in pixel density, were analyzed by the Kruskall–Wallis test. Bars are the mean values and SDs from three measurements on the same image. A significant difference (P < 0.042) was observed between the results at 2 h and 6 h.
NETs are produced as early as 30 min after inoculation of M. tuberculosis, and they do contain bacilli. Early detection of NETs (30 min) in the dermis of a guinea pig inoculated with M. tuberculosis pre-stained with red fluorescent Iris Fuchsia. (A) A small, NET-forming cell cluster (rectangle) (Hoechst stain, 10 × 10×). (B) The DNA-stained cell cluster observed at a higher magnification (40 × 10×). (C) The cell cluster observed under ultraviolet light; M. tuberculosis appears fluorescent red (100 × 10×). (D) A merged image of red fluorescent bacteria and the NET-forming cell cluster (100 × 10×). Kinetics of MTB-induced NET production in the skin of a guinea pig. NET production in the skin of a guinea pig intradermally inoculated with M. tuberculosis for (A) 2, (B) 4 and (C) 6 h (10 × 10×). A detailed image of the fibrillar structure of NETs is shown in (D) (Hoechst stain, 100 × 10×). Semi-quantitative assessment of DNA (NETs) released from the cells recruited at the MTB-injection site. A semi-quantitative analysis of NETs appearing from 2 to 6 h after inoculation of M. tuberculosis revealed a progressive increase in the area of DNA released, and this increment was statistically significant (ImageJ pixel density, P < 0.042) (n = 3 measurements).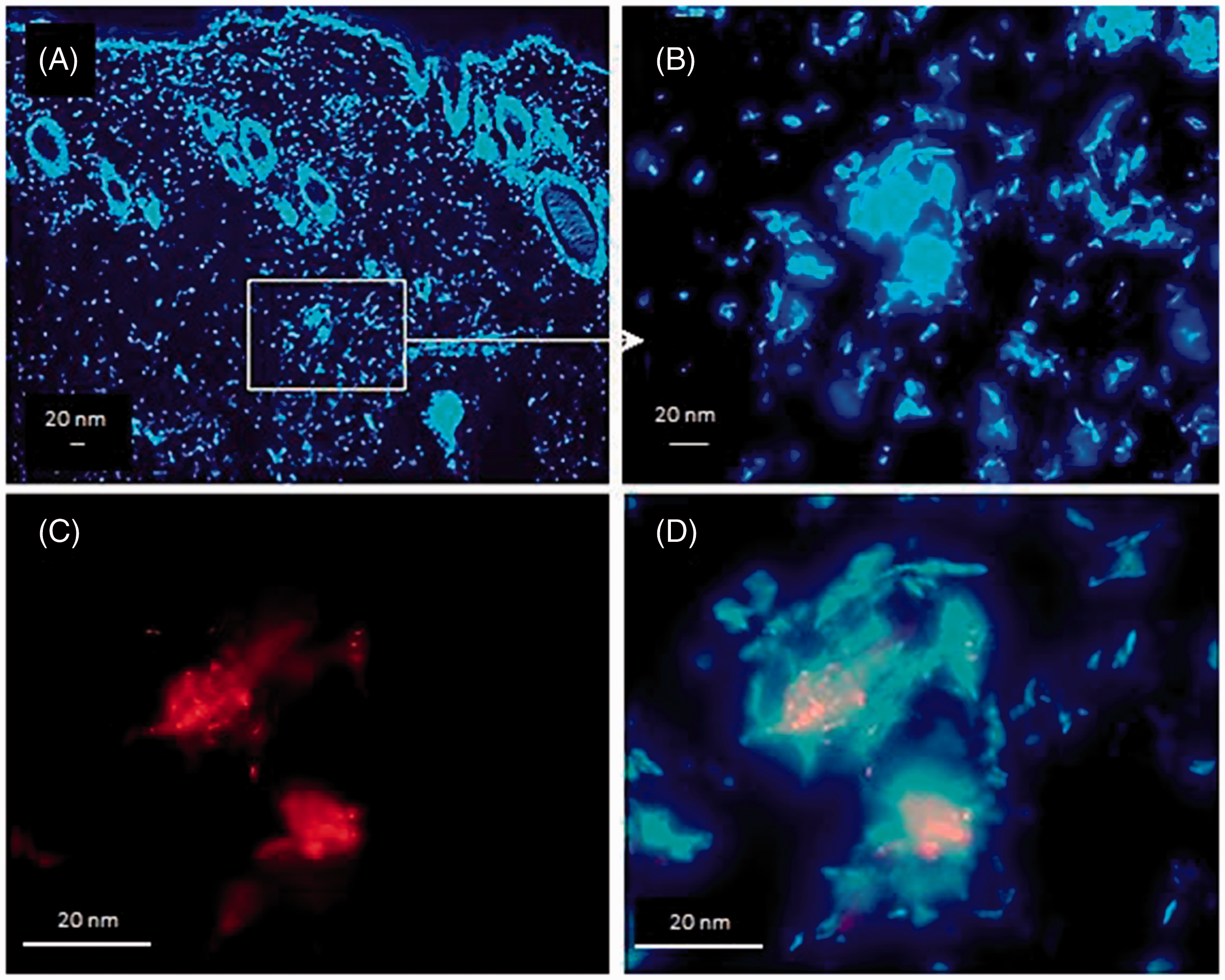
NETs contain chromatin admixed with elastase, histones and MPO
NETs were made of DNA, as deduced from their staining with the Hoechst reagent, and contained elastase, histone and MPO, just as was originally described by Brinkmann et al. in their in vitro studies (Figure 6).
9
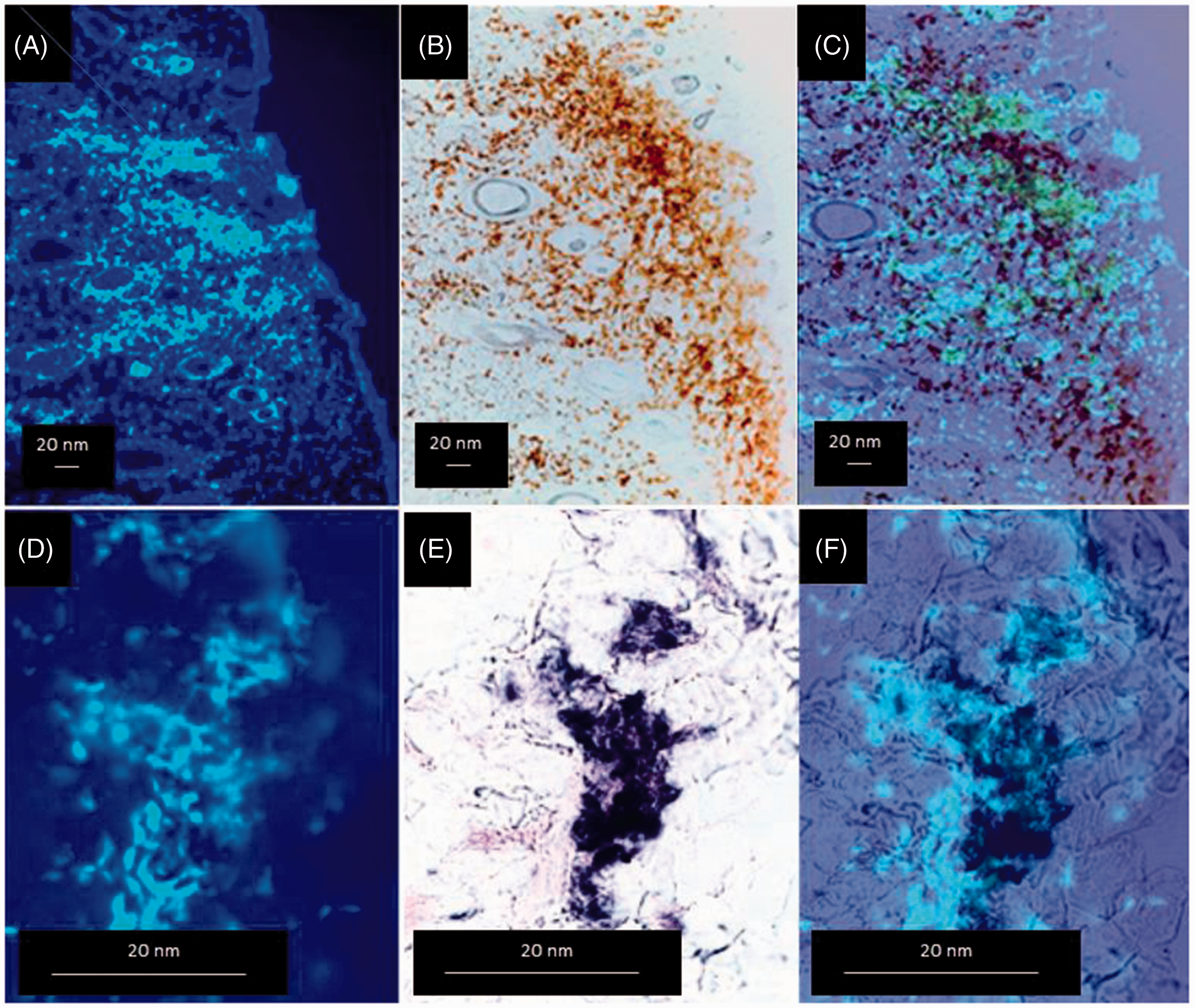
M. tuberculosis NETs contained active MPO and ROS. This was deduced from the presence of oxidized ortho-dianisidine (brown) and the presence of reduced NBT (purple) in the NETs. MPO activity and reduced NBT appeared intermingled with the DNA in the NETs (Figure 7), suggesting that NETs are conceivably microbicidal.
Characterization of MTB-induced NETs in the skin of a guinea pig. Adjacent skin sections of a guinea pig inoculated with M. tuberculosis, stained for DNA, MPO and histone (upper set), or DNA, elastase (ELAS) and histone (lower set). Notice co-localization in all cases (confocal microscopy) (n = 3). MPO and ROS activity in the MTB-induced NETs in the skin of a guinea pig. DNA released from neutrophils contain (A–C) active peroxidase and (D–F) ROS. Because of the colored insoluble products of MPO and ROS, these preparations did not require counterstaining. Together with the presence of elastase, these findings suggest that NETs are microbicidal. (A–C) 10 × 10×; (D–F) 100 × 10× (n = 3).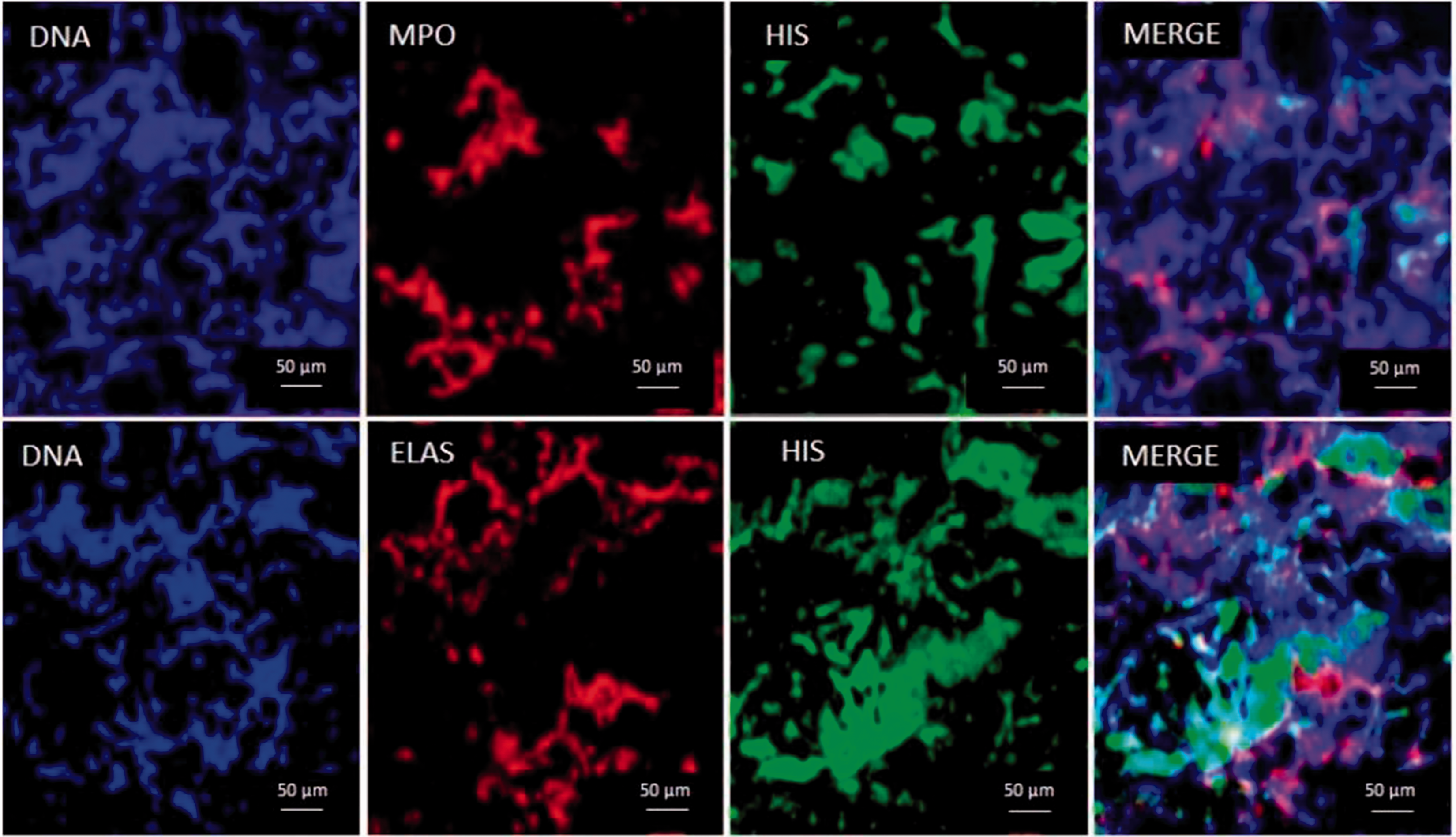
In vivo NET induction does not require the presence of live mycobacteria, and mycobacteria, other than M. tuberculosis, are also NET inducers
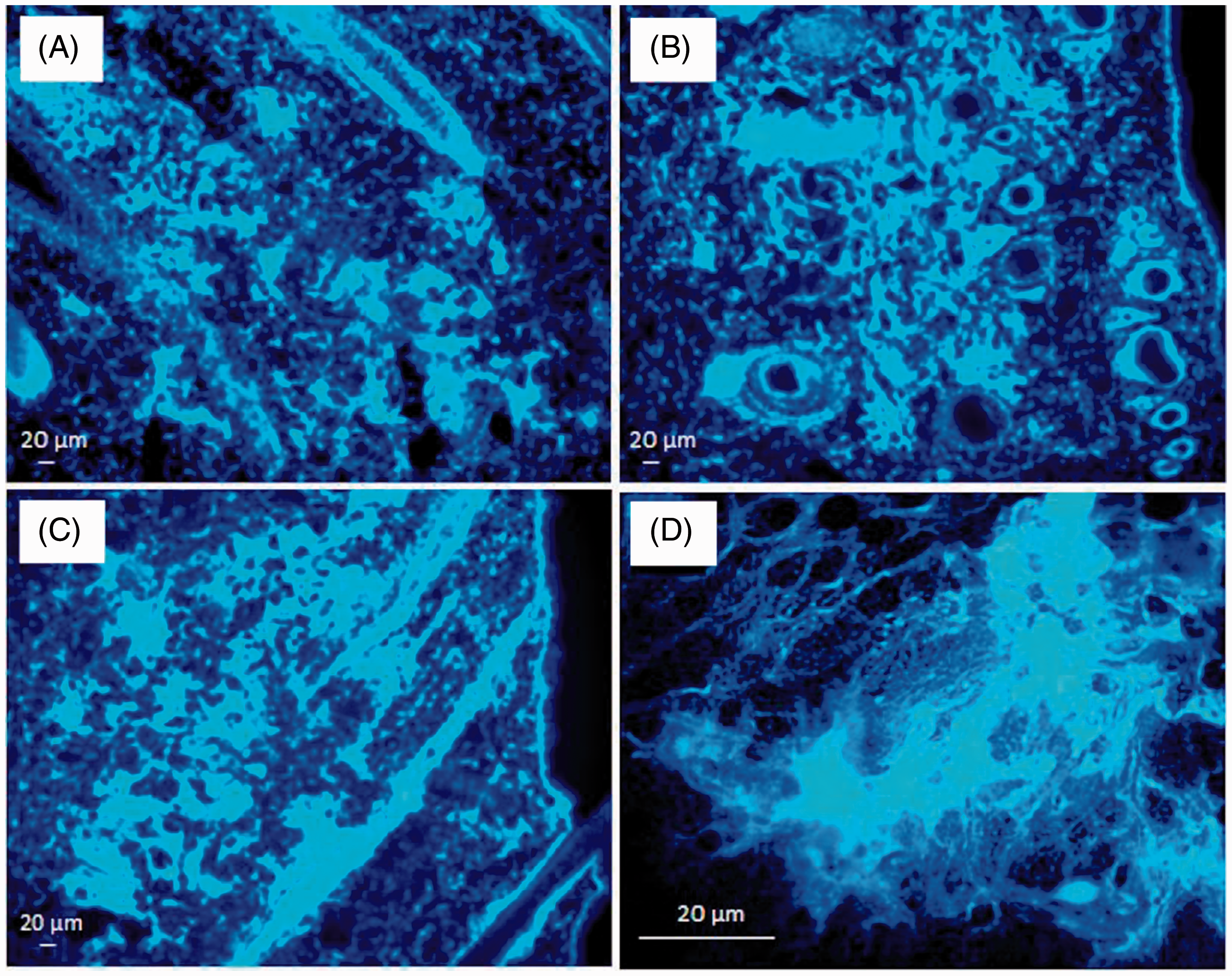
The theoretical requirement of bacterial viability for NET induction was tested by inoculating equivalent amounts of viable and heat-killed M. tuberculosis in separate skin sites of the same guinea pig. Both preparations induced the production of NETs with similar characteristics (Figure 8A, B). In addition to M. tuberculosis, M. bovis BCG and M. smegmatis were also efficient NET inducers in guinea pigs (Figure 8C, D).
Alive and dead MTB, and mycobacteria other than MTB, are NET inducers in the skin of the guinea pig. This image shows skin sections of guinea pigs inoculated for 6 h with (A) alive or (B) heat-killed MTB, or with alive (C) M. bovis BCG or (D) M. smegmatis. NETs are produced in all cases, with similar characteristics (Hoechst stain, 10 × 10×) (n = 3). In vitro, guinea pig neutrophils also release NETs when stimulated with (E) PMA or (F) Latex particles (Hoechst stain, 100 × 10×) (n = 3).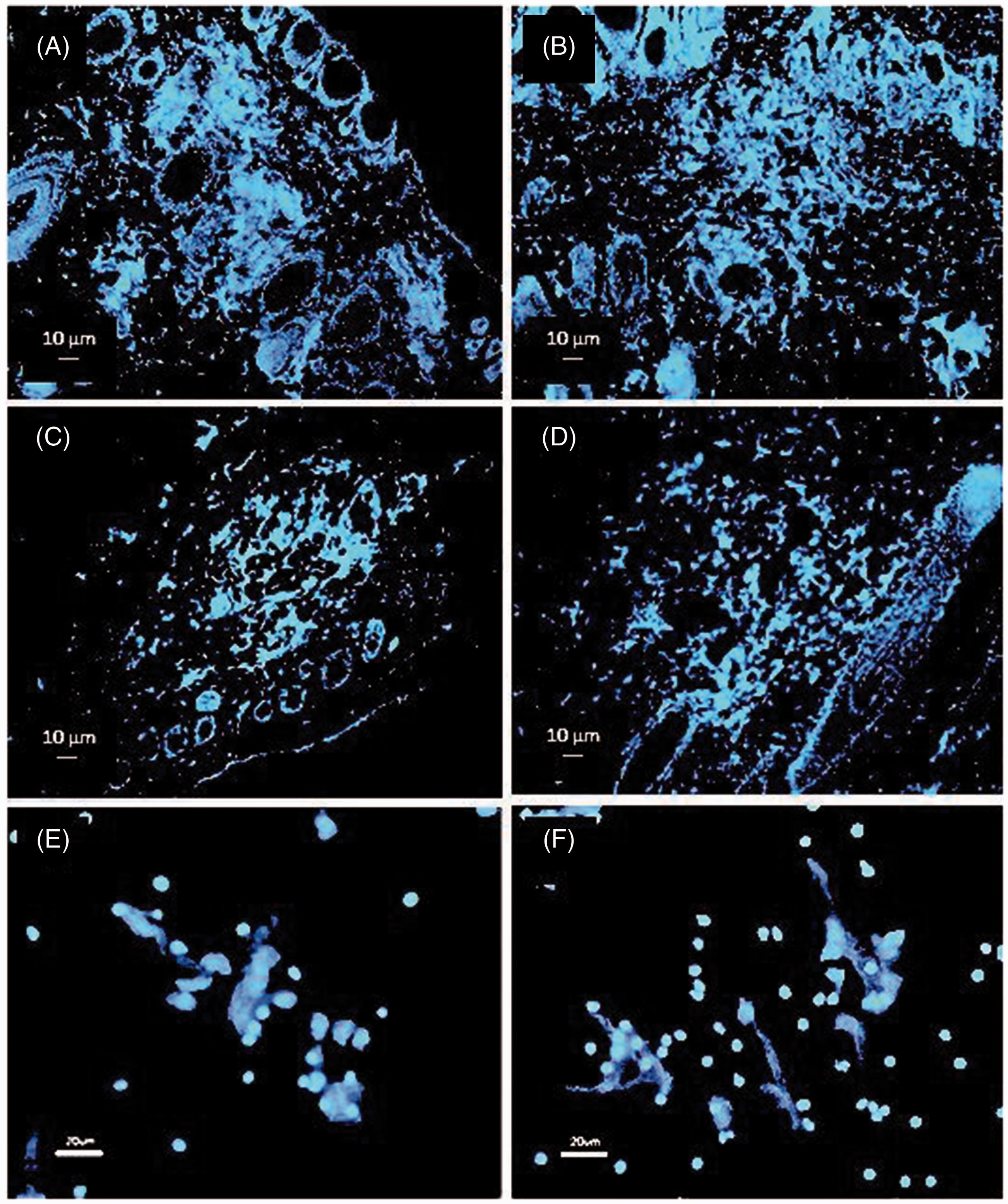
Liposomes with whole M. tuberculosis lipids also induce the release of NETs in vivo
To determine if the induction of NETs required the presence of whole mycobacteria, we prepared unilamellar liposomes with lipids from M. tuberculosis (Figure 9). Intradermal injection of mycobacterial liposomes induced the production of NETs; however, these NETs appeared more defined (more ‘hairy’) than the ones induced by M. tuberculosis itself (compare with Figure 4). Liposome-induced NETs had the same kinetics than NETs induced by the intact bacilli; they were noticeable by 30 min after inoculation and were clearly evident 4 h later. Although we did not characterize the lipids contained in the liposomes, it is very likely that they correspond to the most superficial, solvent-extractable, lipids of the mycobacteria, namely trehalose dimycolate, sulfolipid, lipomannan, lipoarabinomannan, phthiocerol dimycocerosate and others.
NET production induced by liposomes containing lipids of MTB. NET-like structures in the dermis of a guinea pig at 6 h post-inoculation with liposomes prepared with solvent-extractable lipids from M. tuberculosis. (A) Liposomes (100 × 10×); (B–D) images of dermal NET-like structures stained with the Hoechst reagent, 100 × 10×) (n = 3).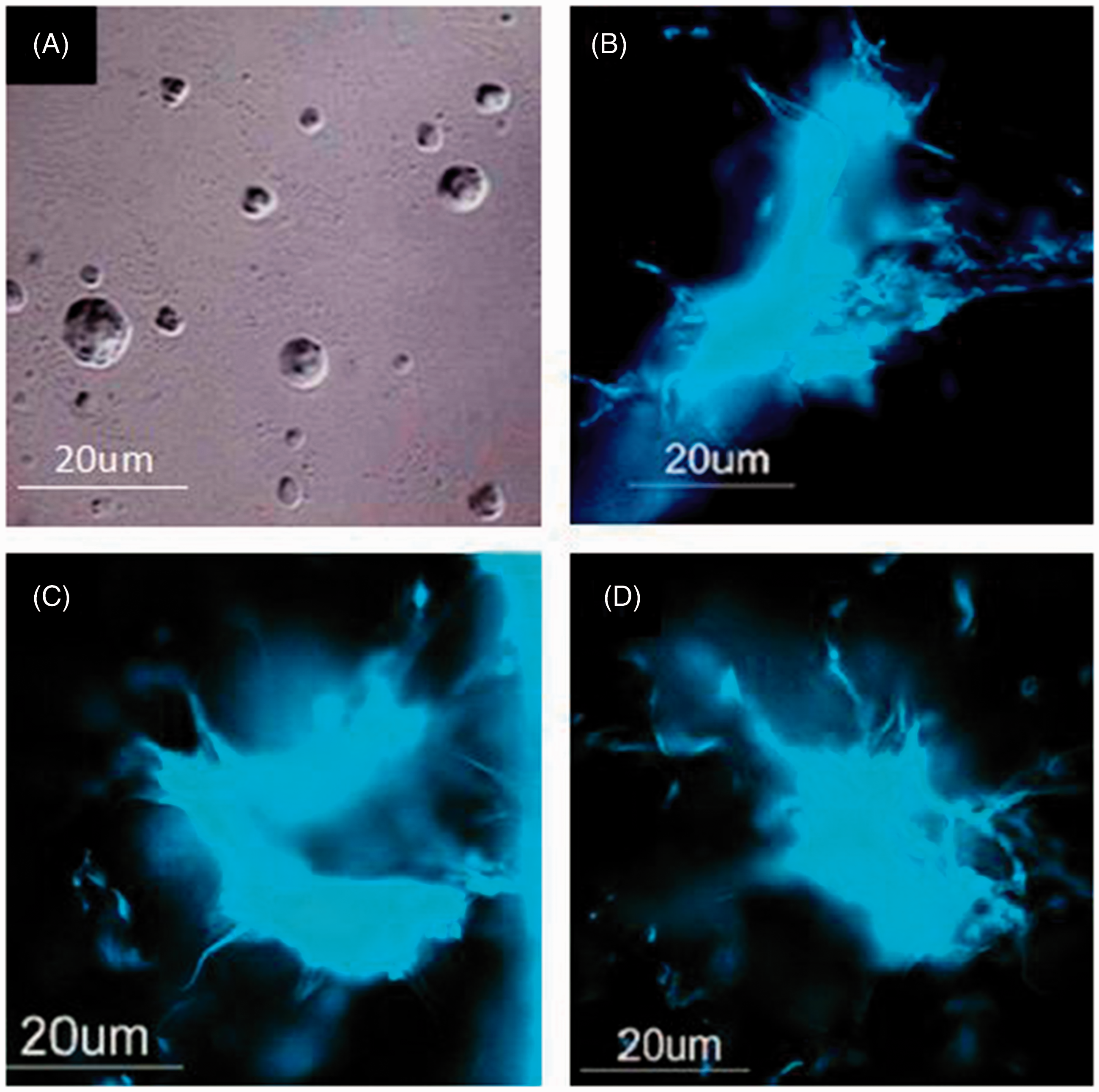
In vivo-induced NETs have no effect on the viability of trapped M. tuberculosis
Dispase-treated skin homogenates prepared from inoculated sites 6 h after inoculation with M. tuberculosis (when NET expression was at the highest) allowed the recovery of only a fraction of the inoculated bacteria. However, comparing the number of CFUs from the 6 h-sampled skin with that from the 0 h sample, no decrease in the number of recovered bacteria was observed (Figure 10A).
In vivo and in vitro induced NETs do not kill MTB. (A) The inflammatory infiltrates produced in vivo in response to the inoculation of M. tuberculosis do not kill the bacteria; the numbers of bacteria recovered from the injected skin 6 h after inoculation (when NET production is at maximum) are similar to the ones recovered immediately after inoculation (0 min). (B) Neutrophils incubated in vitro for 6 h in the presence of M. tuberculosis (MOI 20:1) undergo NETosis; in the image shown (100 × 10×), bacilli (dark rods) appear intermixed with extracellular DNA. (C) However, neutrophils are unable to kill the microorganism within the period examined (0.5–6 h), and (D) this is also true when M. tuberculosis-infected neutrophils are incubated for 4, 6 or 8 h in the presence of PMA (a strong NET and ROI inducer). Bars and vertical lines are the mean and standard error values of three experiments performed (one-way ANOVA, P ≥ 0.05).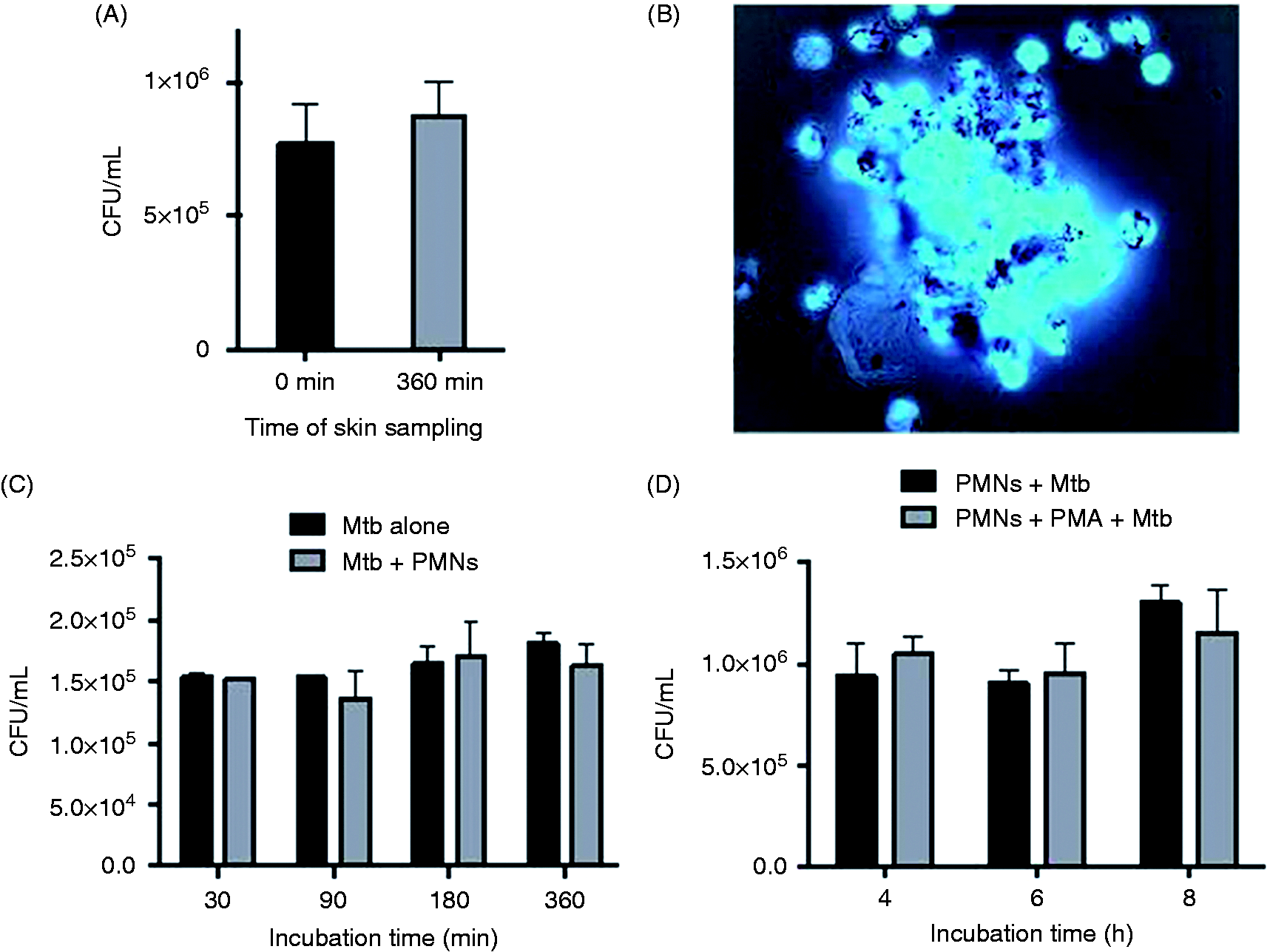
In vitro cultivated guinea pig polymorphnuclear cells do not kill M. tuberculosis
Figure 10(B) shows an image of guinea pig neutrophils infected in vitro with M. tuberculosis; bacilli appear intermixed with extracellular DNA (NETs). However, neutrophils did not kill the microorganism, at least within the period of 30–360 min examined (Figure 10C) and this was also true for M. tuberculosis-infected neutrophils simultaneously stimulated with PMA for 4, 6 and 8 h (Figure 10D).
Discussion
Accumulation of neutrophils in tuberculosis has been repeatedly observed in both human and experimental tuberculosis.3,36 Although early neutrophil infiltration observed in tuberculosis has been attributed to the response of these cells to chemokines produced by local (or passing by) lymphoid cells primed by M. tuberculosis at the sites of infection, 37 some have suggested that neutrophils are the first cells to make contact with bacilli and that they are attracted by sensing the presence of these microorganisms through PRRs, chemotactic factors or soluble PAMPs.38,39 In addition, neutrophils have been shown to rapidly migrate via the lymphatic system after M. bovis BCG intradermal vaccination and shuttle live bacilli to the draining lymph nodes. 40
The role of neutrophils in the immunity to tuberculosis remains a widely debated subject; there are reports that recognize a protective role of these cells,24,41,42 other reports that do not 43 and still others that consider neutrophils to be pathogenic participants in the disease. 44 Most studies on NET production recognize this phenomenon as dependent on ROS,10,45–48 independent of the cytoskeleton, different from necrosis and apoptosis, and lethal for both the cells (NETosis) and the microorganisms ensnared in the NETs.32,49 However, the function of NETs is widely divergent, and reports range from those that indicate that they have microbicidal activity to those that indicate that NETs trap microorganisms but do not kill (all of) them,9,11,50 a matter that might be explained by the different study models.
Here, we provide evidence that NETs are induced in vivo by M. tuberculosis in guinea pigs, and that M. bovis BCG and M. smegmatis also triggered their production. As already mentioned, the guinea pig was used because this animal is highly susceptible to tuberculosis, and the intradermal route was chosen because the skin is an appropriate organ for the study of the in situ host–parasite interactions that occur during the infection. Moreover, we must keep in mind that cutaneous tuberculosis is one of the several extra-pulmonary forms of the disease and a great deal of enlightening information on the pathology of tuberculosis has, indeed, emerged from the study of the skin lesions both in the guinea pig and the rabbit. 51
We found that NETs contain active MPO and ROS, namely superoxide anion (O2–), which causes the in situ reduction of NBT. Superoxide anion dismutates, spontaneously or by the effect of the superoxide dismutase, to produce H2O2. MPO acting on H2O2 in the presence of halides produces very toxic halogenating intermediaries lethal to most microorganisms. In vitro, the MPO–H2O2–Cl– system has been found to have microbicidal activity on M. tuberculosis.51–53 The presence of active MPO and ROS in our study suggested that NETs might have microbicidal activity on the microorganism; however, we were unable to demonstrate any microbicidal activity of the in vivo-induced NETs on M. tuberculosis. Resistance of this microorganism to the microbicidal activity of NETs could be explained by the multiple detoxifying mechanisms possessed by this bacterium, as emphasized by Mendoza-Aguilar et al. 53 It is also probably that M. tuberculosis uses the extracellular DNA to produce biofilms which can be later used as a nourishment. 54
Very early (2 h) following its intradermal injection in guinea pigs, M. tuberculosis caused a noticeable accumulation of neutrophils that released NETs. The size of these NETs increased in time and reached a maximum 6–8 h after inoculation. Thereafter, the NET-containing infiltrates became disorganized and merged with the surrounding tissue. The production of NETs was, indeed, stimulated by M. tuberculosis, because tubercle bacilli were always found trapped in the NETs. These results suggest that recognition of M. tuberculosis microbial-associated molecular patterns and tissue-derived danger or damage-associated molecular patterns (DAMPs) by neutrophil PRRs signaled for chromatin re-organization, dissolution of nuclear and lysosomal membranes, intermixing of granular proteins with the now-loose chromatin and the discharge of this material in the form of NETs.
The early production of NETs in vivo (30 min) compared with the delayed release of NETs in vitro (>3 h) suggest that several inflammatory mediators participate in the process, namely DAMPs, PAMPs, complement, platelet-activating factor, IL-8, GM-CSF, IFN-α and other mediators, are all NET-inducers, 55 which are produced following the infection.
Finally, it has been proposed that NETs, in trapping the mycobacteria limit their dissemination to other tissues via blood and lymphatic vessels. It has been found that the dermal distribution of NETs follows the distribution pattern of fibronectin since this protein has interaction sites with DNA; this would explain why in vivo NETs appear wavy, whereas in vitro they appear as long filaments. 56 By trapping viable M. tuberculosis, NETs may facilitate the participation of macrophages in the killing of the microorganism. 57
The molecular basis of NET production remains poorly understood. Rac-1, Rac-2 and the Raf–MEK–ERK pathway seem to be involved in NET formation through activation of NADPH oxidase and up-regulation of anti-apoptotic proteins.30,58,59 After neutrophils have captured and processed microorganisms, they undergo NETosis, necrosis or apoptosis, at the near-end stage of the phagocytic process. Yipp et al. 60 observed that neutrophils can release NETs while still alive (while crawling and trapping microorganisms); however, this finding does not contradict the opinion that NETs are formed while neutrophils are in the process of dying. Chemicals that traverse the cell membrane (such as PMA), 9 molecules that passively diffuse through the membrane (such as Glc oxidase) 10 and Ab-coated inert particles (such as latex-IgG) will also cause NETosis (Figure 8C, D).
We found that the viability of mycobacteria was not a prerequisite for NET induction. Heat-killed M. tuberculosis induced NET production in the same manner as alive bacilli. This suggests that mycobacterial components rather than mycobacteria metabolism are involved in NET induction. The lipid coat of M. tuberculosis (the outmost structure of the bacterium) might participate in NET induction, as it has been observed that M. tuberculosis-derived lipids interact in vitro with neutrophils and increase both their oxidative response61–63 and capacity to secrete chemokines. 64 Here we showed that the intradermal injection of unilamellar liposomes prepared with whole lipids extracted from M. tuberculosis also leads to NET production in vivo; thus, lipids could be the mycobacterial NET-inducer moiety first recognized by neutrophils. In this study, no ‘empty’ control liposomes were included, because liposomes were prepared only with the lipids extracted from the mycobacteria, and no extra lipids were added. We found that NETs were always present in association with neutrophil infiltrates at the M. tuberculosis infection site of normal guinea pigs.
Conclusions
The present research demonstrates that M. tuberculosis induces NET production in vivo, that M. tuberculosis does not need to be alive to be a NET inducer, that other mycobacteria such as M. bovis BCG and M. smegmatis, as well as M. tuberculosis-derived liposomes, are also NET inducers. NETs are possibly bactericidal as they contain elastase, active peroxidase and ROS. They are, however, unable to kill M. tuberculosis owing, perhaps, to its complex cell wall structure, and the many detoxifying systems it possesses. In vivo killing of M. tuberculosis will probably require the participation of the adaptive cell-mediated immunity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Secretaría de Investigación y Posgrado, Instituto Politécnico Nacional (SIP 20130695) and Consejo Nacional de Ciencia y Tecnología (CONACYT). GFR is a fellow-holder of CONACyT; IEG, MMA and ORE, hold fellowships from SNI (CONACyT), COFAA (IPN), and EDI (IPN); PAP is a fellow holder of COFAA (IPN) and EDD (IPN).
