Abstract
The compound m-chlorophenylpiperazine (mCPP), which is known to trigger migraine-like head pain in some subjects, was evaluated for its ability to induce dural plasma protein extravasation (PPE) in guinea pigs. Intravenous mCPP dose-dependently increased PPE. This effect was inhibited by non-selective 5-HT2 receptor antagonists (methysergide, LY53857, LY215840), by a peripherally restricted 5-HT2 receptor antagonist (xylamidine) and by a 5-HT2B selective receptor antagonist (LY202146). These data suggests that peripheral 5-HT2B receptors mediate mCPP-induced PPE. The nitric oxide synthase inhibitor L-NAME and 5-HT1 agonist sumatriptan also blocked mCPP-induced PPE, suggesting a role for nitric oxide (NO) and the trigeminal system, respectively. NO release has been linked to activation of the 5-HT2B receptor on the vascular endothelium. However, LY202146 did not block PPE induced by electrical stimulation of the trigeminal ganglion. These data are consistent with activation of peripheral 5-HT2B receptors initiating PPE and the theory that selective 5-HT2B antagonists might be effective prophylactic therapies for migraine.
Introduction
While selective 5-HT1 receptor agonists like sumatriptan are effective in the acute treatment of migraine (1–3), a number of non-selective 5-HT2 receptor antagonists such as methysergide, pizotifen, cyproheptadine and amitryptiline are used as prophylactic anti-migraine agents (4). Although efficacious, these compounds lack selectivity and have adverse events which limit their medicinal application (5). Kalkman has suggested that the prophylactic activity of these compounds occurrs via blockade of either the 5-HT2C or 5-HT2B receptor (6). His hypothesis centered on the observation that meta-chlorophenylpiperazine (mCPP), an agonist at both the 5-HT2B and 5-HT2C receptors, caused severe migraine-like headaches in patients with a history of migraine (7). In that study, the headache severity correlated significantly with the peak plasma concentrations of mCPP, 2–3 h after administration. Furthermore, the efficacious clinical doses of seven prophylactic therapies correlated with their functional activity at human 5-HT2B and 5-HT2C receptors (P = 0.001 and P = 0.005, respectively) (8). The 5-HT2A receptor is not thought to play a role in migraine, as ketanserin, a 5-HT2A-selective antagonist, did not provide a clear beneficial outcome in a clinical trial of migraine prophylaxis (9).
Emerging evidence suggests that nitric oxide (NO) may also play a key role in migraine (10). A severe and persistent headache is a well-known adverse event associated with administration of the NO donor nitroglycerin for cardiovascular indications. In addition, nitroglycerin induced biphasic headaches of greater severity and longer duration in patients with a past history of migraine. These headaches fulfilled the International Headache Society criteria for classification as a true migraine (11, 12). The role of NO in migraine is further reinforced by a clinical study in which migraine pain, phonophobia and photophobia were significantly reduced by intravenous infusion of the nitric oxide synthase (NOS) inhibitor 546C88, which presumably decreased NO (13).
Fozard has proposed that migraine could result from NO release following activation of either 5-HT2B or 5-HT2C receptors on endothelial cells of the cerebral vasculature (14). The released NO would activate sensory afferents in the meninges, resulting in the release of inflammatory neuropeptides, neurogenic inflammation, vasodilatation and pain. In fact, the NO donor nitroglycerin induced calcitonin gene related peptide (CGRP) release from sensory fibres following direct application onto the meninges (15). Furthermore, released neuropeptides could trigger the formation of additional NO, after which the process would self-perpetuate, with the result being an ongoing inflammatory cascade (16).
Moskowitz has proposed that neurogenic inflammation of the dural membranes in concert with activation of the trigeminal system results in the pain of migraine (16, 17). Specifically, inflammatory neuropeptides such as substance P and CGRP are released onto the dural tissue to cause vasodilatation, plasma protein extravasation (PPE), endothelial cell changes and platelet aggregation. One animal model of migraine uses electrical stimulation of the trigeminal ganglion to induce neurogenic inflammation as indicated by increased PPE in the dura. Commercially available acute migraine treatments, as well as selective 5-HT1F receptor agonists, block the dural PPE induced by trigeminal ganglion stimulation (18–20).
Although quite useful for certain studies, direct electrical stimulation of the trigeminal ganglion does not allow researchers to study the endogenous factors that eventually lead to the increased firing of the trigeminal nerve. To address this limitation and investigate the role of specific 5-HT2 receptors in the initiation of migraine, we utilized intravenous infusion of mCPP to induce dural PPE. We also evaluated blockade of the mCPP-induced dural protein extravasation by antagonists with varying affinities for the 5-HT2 family of receptors. Finally, to determine the role of NO and trigeminal nerve activation in the extravasation induced by mCPP, the effect of the NOS inhibitor Nω-nitro-L-arginine methyl ester (L-NAME) and the 5-HT1 receptor agonist sumatriptan on mCPP-induced extravasation was determined.
Materials and methods
Chemicals
1-(3-chlorophenyl) piperazine dihydrochloride (mCPP) and methysergide maleate were obtained from Research Biochemicals Inc. Natick, MA, USA L-NAME was purchased from Sigma (St Louis, MO, USA). Sumatriptan succinate (Imitrex®; Cerenex Pharmaceuticals Shawnee Mission, KS, USA) was purchased commercially. Xylamidine tosylate was a gift from The Wellcome Research Laboratories. LY53857 (ergoline - 8 - carboxylic acid, 6 - methyl - 1 - (1-methylethyl)-,2-hydroxy-1-methylpropyl ester), LY215840 (ergoline-8-carboxamide, N-(2-hydroxycyclopentyl)-6-methyl-1-(1-methylethyl)-,[8β(1S,2R)], LY202146 (1H-Pyrido[3,4-b]indole,1-[2-bromo-3,4-dimethoxyphenyl) methyl]-2,3,4,9-tetrahydro-6-methyl-,monohydrochloride) and LY310898 (4 - [[3 - [2 - [4 - (4 - fluorobenzoyl) -1- piperidinyl]ethyl]-2,3 -dihydro-2 - oxo - 1H - benzimidazol -1- yl]methyl]-, monohydrochloride) were synthesized in the Lilly Research Laboratories.
Radioligand binding
Drug competition for binding was performed as described by Wainscott et al. (21) using homogenates of cells expressing the cloned human 5-HT2A, 5-HT2B or 5-HT2C receptors. The 5-HT2B receptor was labelled with 3H-5-HT. The 5-HT2A and 5-HT2C receptors were labelled with 125I-DOI. The Kd of 3H-5-HT for the cloned human 5-HT2B receptor was 7.76 ± 1.06. The Kd of 125I-DOI for the cloned human 5-HT2A and cloned human 5-HT2C receptor was 0.672 ± 0.078 n
mCPP-induced extravasation
Male guinea pigs from Charles River Laboratories (Wilmington, MA, USA) (250–350 g) were anaesthetized with sodium pentobarbital (45 mg/kg i.p.). The femoral vein was exposed and mCPP (0.1–100 µg/kg) or saline (1 ml/kg) was injected intravenously. Exactly 2 min later, Evans Blue (50 mg/kg), a fluorescent dye with high affinity for endogenous proteins, was also injected intravenously. Fifteen minutes later, the animals were killed by perfusion exsanguination performed by injecting saline (40 ml, 1 ml/s) into the left ventricle of the heart after the right atrium had been cut. The descending aorta and the vena cava were clamped at the level of the diaphragm to enhance rostral perfusion. The top of the skull was removed to facilitate the collection of the dural membrane. The membrane was removed from one hemisphere, rinsed with deionized water, and spread flat on a microscope slide. Once dried, the tissues were coverslipped with a 70% glycerol/water solution.
PPE, as indicated by the intensity of Evans Blue fluorescence in each of the dural tissue samples, was measured using a fluorescence microscope (Zeiss, Germany) equipped with a grating monochromator and a spectrophotometer. An excitation wavelength of approximately 535 nm was utilized and the emission intensity at 600 nm was determined. Fluorescence measurements were normalized with respect to the fluorescence measured in tissues from saline-treated animals.
In some experiments, animals were dosed intravenously with a test compound or vehicle 2 min prior to the mCPP injection.
Trigeminal ganglion stimulation-induced extravasation
Male guinea pigs from Charles River Laboratories (250–350 g) were anaesthetized with sodium pentobarbital (45 mg/kg i.p.) and placed in a stereotaxic frame (David Kopf Instruments Tujunga, CA, USA) with the incisor bar set at −4.0 mm as described previously (19). Following a midline sagittal scalp incision, two pairs of bilateral holes were drilled through the skull (4 mm posteriorly and 3.2 and 5.2 mm laterally – all coordinates referenced to bregma). Pairs of stainless steel stimulating electrodes, electrically insulated except at the tips (Rhodes Medical Systems, Inc. Tujunga, CA, USA) were lowered through the holes in both hemispheres to a depth of 10.5 mm from dura.
The femoral vein was exposed and test compound was injected intravenously. Approximately 8 min later, Evans Blue (50 mg/kg) was also injected intravenously. Exactly 10 min post-injection of the test compound, the left trigeminal ganglion was stimulated for 3 min at a current of 1.0 mA (5 Hz, 4 ms pulse duration) with a Model 273 potentiostat/galvanostat (EG&G Princeton Applied Research Princeton, NJ, USA).
Fifteen minutes following the stimulation, the animals were euthanized by exsanguination with saline as described above. Tissue harvest, processing, and fluorescence measurements were identical to the procedure described above, except dural tissue from both hemispheres was collected.
The extravasation induced by electrical stimulation of the trigeminal ganglion was an ipsilateral effect (i.e. primarily only on the side of the dura in which the trigeminal ganglion was stimulated). This allowed the contralateral (unstimulated) dura to be used as a control. The ratio of the amount of extravasation in the ipsilateral dura compared with the contralateral dura was calculated.
Statistical analysis
The results are expressed as mean values with standard errors of the mean (± SEM). All statistical evaluations were made with respect to vehicle-treated controls utilizing
Results
mCPP-induced extravasation
Intravenous mCPP (0.3, 1 and 100 µg/kg) dose-dependently increased PPE in the dura of guinea pigs (Fig. 1). The increased extravasation observed following administration of 1 µg/kg and 100 µg/kg mCPP was similar in magnitude. Based on these data, 1 µg/kg of mCPP was chosen as the dose for all subsequent experiments.

Plasma protein extravasation in dura of guinea pigs following intravenous treatment with m-chlorophenylpiperazine (mCPP) or saline. The normalized extravasation is the average fluorescence intensity of the dura from mCPP- or saline-treated animals divided by the average fluorescence intensity of the dura from a separate group of saline-treated control animals. Data are represented as mean ± SEM (n = 3). Statistical analysis was performed by comparison with saline control group using
Blockade of the mCPP-induced dural extravasation by 5-HT2 receptor antagonists with varying affinities for the three receptor subtypes (Table 1) and by sumatriptan was evaluated. The dural extravasation induced by mCPP (1 µg/kg, i.v.) was totally blocked by pretreatment with methysergide (10 µg/kg, i.v.), a non-selective 5-HT2 receptor antagonist (Fig. 2). Two additional non-selective 5-HT2 receptor antagonists, LY53857 (22) and LY215840 (23), also significantly inhibited the extravasation following i.v. infusions (10 µg/kg) (Fig. 2). LY202146 (0.1 µg/kg, i.v.), a potent and somewhat selective 5-HT2B receptor antagonist, also blocked the mCPP-induced extravasation. However, LY310898, an antagonist with high affinity for 5-HT2A and 5-HT2C, but not 5-HT2B receptors, had no effect on dural extravasation at doses as high as 100 µg/kg (Fig. 2). Thus, 5-HT2B receptor activation appears to mediate mCPP-induced dural extravasation.
Pharmacological profile of serotonergic ligands at the cloned human 5-HT2A, 5-HT2B, or 5-HT2C receptors
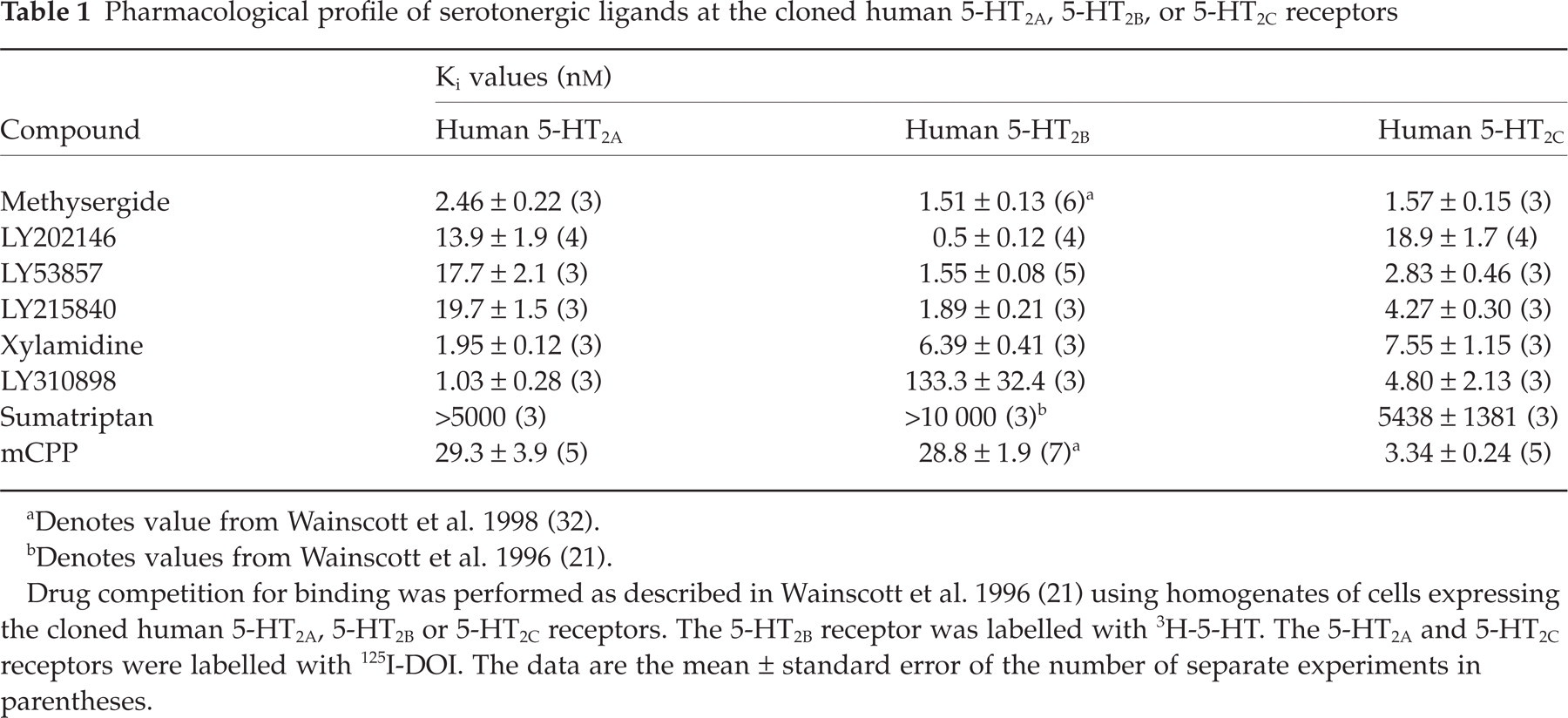
aDenotes value from Wainscott et al. 1998 (32).
bDenotes values from Wainscott et al. 1996 (21).
Drug competition for binding was performed as described in Wainscott et al. 1996 (21) using homogenates of cells expressing the cloned human 5-HT2A, 5-HT2B or 5-HT2C receptors. The 5-HT2B receptor was labelled with 3H-5-HT. The 5-HT2A and 5-HT2C receptors were labelled with 125I-DOI. The data are the mean ± standard error of the number of separate experiments in parentheses.

Plasma protein extravasation (PPE) in dura of guinea pigs due to m-chlorophenylpiperazine (mCPP) (1 µg/kg i.v.) following i.v. pretreatment with either saline, a 5-HT2 antagonist or sumatriptan. The dose of compound, in µg/kg, used for these studies is indicated in parentheses behind the compound name or number on the graph. The normalized extravasation is the average fluorescence intensity of the dura from animals treated with either saline, a 5-HT2 antagonist or sumatriptan prior to mCPP divided by the average fluorescence intensity of the dura from control animals given saline for both treatments. A normalized extravasation ratio of 1 would indicate a complete blockade of mCPP-induced dural extravasation. Xylamidine is a peripherally restricted non-selective 5-HT2 antagonist. Sumatriptan is a potent 5-HT1 receptor agonist, with no affinity for 5-HT2 receptors, thought to block PPE through the inhibition of inflammatory neuropeptide release (substance P, calcitonin gene related peptide) from trigeminal sensory afferents in the dura. Data are expressed as mean ± SEM (n = 3). Statistical analysis was performed by comparison with saline/mCPP group using
To determine if the 5-HT2B receptor was located peripherally, xylamidine (10 µg/kg, i.v.), a 5-HT2 receptor antagonist that does not cross the blood–brain barrier (24), was studied and found to inhibit significantly the mCPP-induced dural extravasation (Fig. 2). To confirm that mCPP was activating the trigeminal pathway, the 5-HT1 receptor agonist sumatriptan (10 µg/kg, i.v.) was also shown to block significantly the dural extravastion induced by mCPP (Fig. 2).
Finally, to investigate whether NO release was necessary for the production of dural protein extravasation following mCPP infusion in guinea pigs, animals were preteated with L-NAME, a non-specific NOS inhibitor. L-NAME (100 µg/kg, i.v.), at a dose that by itself had no effect on PPE in the dura, significantly blocked the dural extravasation due to mCPP (Fig. 3).

Plasma protein extravasation (PPE) in dura of guinea pigs due to either m-chlorophenylpiperazine (mCPP) (1 µg/kg i.v.) or saline (1 ml/kg i.v.) following intravenous pretreatment with either saline (1 ml/kg, i.v.) or NG-nitro-L-arginine methyl ester (L-NAME) (100 µg/kg), a general nitric oxide synthase inhibitor. These data suggest a role for nitric oxide in the generation of dural protein extravasation following mCPP infusion. In addition, these data show that L-NAME by itself has no effect on PPE at a dose that significantly blocked mCPP-induced PPE. The normalized extravasation is the average fluorescence intensity of the dura from animals treated with either saline or L-NAME prior to mCPP divided by the average fluorescence intensity of the dura from control animals given saline for both treatments. A normalized extravasation ratio of 1 would indicate a complete blockade of mCPP-induced dural extravasation. Data are expressed as mean ± SEM (n = 3). Statistical analysis was performed by comparison with either the saline/saline or saline/mCPP group using
Trigeminal ganglion stimulation-induced extravasation
To determine if 5-HT2B receptor antagonists could directly inhibit the release of substance P (SP) and CGRP like sumatriptan, LY202146 was evaluated in the trigeminal stimulation-induced extravasation model. LY202146 (10 µg/kg, i.v.) did not inhibit the dural PPE resulting from electrical stimulation of the trigeminal ganglion (Fig. 4), despite the dose being 100 times higher than necessary to significantly inhibit mCPP-induced extravasation (Fig. 2).

Electrical stimulation-induced dural protein extravasation in guinea pigs following intravenous treatment with either saline or the selective 5-HT2B antagonist LY202146 (10 µg/kg). The extravasation ratio is the average fluorescence intensity of the dura from the stimulated side divided by the average fluorescence intensity of the dura from the unstimulated side. A ratio of 1 would indicate a completely effective treatment. Data are expressed as mean ± SEM (n = 3).
Discussion
These data demonstrate for the first time that a pharmacological agent that produces migraine or migraine-like symptoms in humans can also induce PPE in the dural membranes of laboratory animals. mCPP (7) has been reported to cause severe migraine or migraine-like symptoms in subjects with a past history of migraine attacks, and it increased dural protein extravasation in guinea pigs. Further, methysergide, an efficacious prophylactic migraine treatment, inhibited dural protein extravasation induced by mCPP in guinea pigs. The ability of these compounds to produce or inhibit clinical migraine and modify neurogenic inflammation in animals as measured by dural protein extravasation provides additional support for the neurogenic inflammation theory of migraine and for the use of dural extravasation as an animal model of migraine.
These data also provide insight into the role of serotonin, the 5-HT2 family of receptors and NO in dural extravasation and possibly the initiation of migraine attacks. Compounds such as reserpine and fenfluramine that cause the release of serotonin from platelets and other stores also induced migraine-like headaches in migraineurs (25–27). Previous studies have found a correlation between functional activity of compounds at the 5-HT2B and 5-HT2C receptor subtypes and their active doses for prophylactic clinical treatment of migraine attacks (8). To evaluate the role of the 5-HT2B and 5-HT2C receptors in dural extravasation, we determined the ability of 5-HT2 receptor antagonists of varying potency and selectivity for the receptor subtypes to inhibit mCPP-induced dural protein extravasation. The non-selective 5-HT2 receptor antagonists methysergide, LY53857, LY215840 and the peripherally restricted antagonist xylamidine all significantly attenuated the dural protein extravasation. The fact that xylamidine was able to decrease the mCPP-induced dural extravasation suggested that the effect was induced via activation of a peripheral 5-HT2 receptor. The selective 5-HT2B receptor antagonist LY202146 also inhibited mCPP-induced dural protein extravasation. In contrast, LY310898, which had high affinity for the 5-HT2A and 5-HT2C receptors, but 20-fold lower affinity for the 5-HT2B receptor, did not block the protein extravasation at doses three to five orders of magnitude higher than efficacious doses for the other 5-HT2 antagonists. Although binding affinities were determined at cloned human receptors, comparison of the effects of LY202146 and LY310898 provides convincing evidence that 5-HT2B receptors, rather than 5-HT2A or 5-HT2C receptors, are involved in mCPP-induced dural protein extravasation in guinea pigs. The 5-HT2B receptor has been detected in the vascular endothelium, along with other tissues, using pharmacological techniques (28), immunohistochemical methods (29) and mRNA analysis (28). Taken together, these findings suggest that the 5-HT2B receptor involved in the initiation of neurogenic inflammation, and possibly migraine, is located outside of the blood–brain barrier.
It has been suggested that activation of the 5-HT2B receptor in the vascular endothelial layer plays a role in the endothelium-dependent vasorelaxation of cerebral vessels, probably via NO release (8). Our data suggest the generation of NO was necessary for the production of dural protein extravasation following mCPP infusion in guinea pigs, since the general NOS inhibitor L-NAME significantly blocked mCPP-induced dural protein extravasation, despite having no effects on dural protein extravasation by itself. These data are consistent with the proposal that activation of 5-HT2B receptors results in NO release, which is required for the subsequent generation of neurogenic inflammation. In fact, Moskowitz and co-workers have shown that nitroglycerin induces CGRP release from sensory fibres following direct application onto the dural tissue (15). In humans, the peak headache intensity occurred 5.5 h after nitroglycerin infusion, even though NO is an unstable free radical with a half-life of approximately 3 s in biological tissues (30). Thus, other mediators or mechanisms could be involved in the slow cascade of events initiated by nitroglycerin that eventually leads to a migraine attack.
5-HT1 receptor agonists such as sumatriptan are efficacious for the acute treatment of migraine (1–3). Sumatriptan is known to inhibit activity in trigeminal fibres, as illustrated by its ability to attenuate CGRP levels in animal plasma (31) and block neurogenic PPE in the dura following electrical stimulation of the trigeminal ganglion (18, 19). The fact that intravenous pretreatment with sumatriptan significantly blocked the mCPP-induced dural extravasation in guinea pigs suggests that mCPP was activating the trigeminal pathway (Fig. 2). These data suggest that the increase in PPE following mCPP infusion probably occurs via formation of NO, activation of the trigeminal system and the release of inflammatory neuropeptides such as CGRP and SP.
The inflammatory neuropeptides released from the trigeminal sensory afferents following activation could induce the formation of additional NO (16), in addition to causing dural protein extravasation and neurogenic inflammation, resulting in an inflammatory cascade and potentially a migraine attack of long duration. To determine if the secondary NO formation was mediated via the 5-HT2B receptor, we evaluated the ability of a relatively high dose of the 5-HT2B antagonist LY202146 to block PPE induced by electrical stimulation of the trigeminal ganglion, instead of mCPP administration. LY202146 was unable to block the protein extravasation following electrical stimulation of the trigeminal ganglion, despite being able to block mCPP-induced dural protein extravasation at considerably lower doses. These observations suggest that the 5-HT2B receptor could be involved in the initiation of an attack, but not directly associated with the release of peptides from the trigeminal sensory afferents. These data would be consistent with the clinical data illustrating that the efficacious prophylactic agents pizotifen and methysergide could not terminate an ongoing migraine attack (8).
In conclusion, these data suggest that activation of peripheral 5-HT2B receptors initiate the release of NO, which subsequently causes the release of inflammatory neuropeptides from trigeminal sensory afferents, resulting in neurogenic inflammation and dural PPE. Several of the efficacious prophylactic agents for migraine are 5-HT2B receptor antagonists, although burdened with lack of receptor selectivity and undesirable side-effects such as weight gain. The data contained in this study suggest that a potent and selective 5-HT2B receptor antagonist could be a safe and efficacious prophylactic therapy for migraine.
Footnotes
Acknowledgements
The authors thank Dr Marlene L. Cohen for her excellent editorial comments during the preparation of this manuscript.
