Abstract
Defective bacterial clearance by macrophages plays an important role in Crohn’s disease (CD). Phenotypes and functions of inflammatory M1 and anti-inflammatory M2 have not been studied in CD. Vitamin D supplementation reduces the severity of CD by unclear mechanisms. We studied macrophage characteristics in CD and controls and the effects of 1,25 vitamin D (1,25D). PBMC were isolated from CD patients and controls. M1 and M2 were generated by culturing of monocytes with GM-CSF and M-CSF, respectively. CD M1 and M2 showed normal phagocytosis and chemotaxis to CCL2 and fMLP. LPS-induced production of TNF-α, IL-12p40 and IL-10 was comparable between groups. Phagocytosis was unaltered with 1,25D; migration only increased marginally. M1 produced more IL-12p40 and TNF-α; IL-10 was greater in M2. 1,25D markedly decreased IL-12p40 by M1 and M2. 1,25D decreased TNF-α in CD M1; IL-10 levels were unaffected. M2 express F13A1, PTGS2, CD163, CXCL10, CD14 and MMP2, whereas TGF-β, CCL1 and CYP27B1 expression was higher in M1. Marker expression was similar between CD and controls. M1 and M2 markers were not differentially modulated by 1,25D. CD macrophages are not functionally or phenotypically different vs. controls. 1,25D markedly decreased pro-inflammatory M1 cytokines but did not modulate polarization to anti-inflammatory M2 phenotype.
Introduction
Inflammatory bowel disease (IBD) is a chronic, relapsing inflammatory disorder of the gastrointestinal tract resulting from a disruption of immune tolerance to the intestinal microflora, leading to mucosal damage in genetically predisposed individuals.1,2 Crohn’s disease (CD) and ulcerative colitis (UC) are the two major types of IBD. 3 The composition of the intestinal microflora in IBD differs from healthy individuals, primarily owing to a lower incidence of dominant commensals (e.g. Firmicutes, Bacteroides, Bifidobacterium).
IBD is estimated to affect over 3.6 million persons in North America and Europe, and the incidence is increasing in Asia and Africa.4,5 Genetic studies have identified > 200 susceptibility loci for IBD, most of which are shared between CD and UC.6–8
The etiology of CD is poorly understood, with increasing evidence suggesting that it results from impaired innate immunity and mucosal barrier dysfunction leading to bacterial persistence and mucosal inflammation.1,2,9 Defective autophagy associated with delayed elimination of intracellular bacteria has been implicated in the chronic intestinal inflammation in CD.10–12 The NOD2 gene is involved in the innate immune response and is highly associated with CD risk. Defects in NOD2 function can affect microbial sensing, 13 Paneth cell function and antimicrobial peptide (AMP) production, 14 as well as intracellular bacterial killing. 12
The intestine encounters more Ags than any other organ and is thus home to the largest compartment of the immune system. Mononuclear phagocytes, including both dendritic cells and macrophages (Mφ), play a central role in discriminating detrimental from harmless Ags. They are major effector cells of innate immunity, responsible for initiating and sustaining protective immune responses mounted towards pathogenic organisms, whilst also ensuring that local and systemic tolerance is generated in response to innocuous Ags. A breakdown of Mφ tolerance against dietary proteins and the resident commensal microbiota is thought to lead to chronic inflammatory disorders such as CD.15 Mφ play an important role in maintaining tissue homeostasis, by clearing apoptotic or senescent cells, and by repairing and remodeling tissue during wound healing. 16
M1/M2 describes the two major and opposing activities of Mφ. M1 activity inhibits cell proliferation and causes tissue damage, whereas M2 activity promotes cell proliferation and tissue repair. 15 The molecules primarily responsible for these ‘fight’ (NO) or ‘fix’ (ornithine) activities both arise from arginine, via enzymatic pathways (iNOS and arginase) that down-regulate each other. 17 M1 and M2 Mφ promote Th1 and Th2 responses, respectively. Products of Th1 and Th2 responses (e.g. IFN-γ, IL-4) also down-regulate M2 and M1 activity, respectively. It is hypothesized that imbalanced M1 or M2 are involved in the pathogenesis of IBD. 17
Multiple factors, including cytokines and microbial products, affect Mφ polarization. 18 Classically activated M1 develop in response to pro-inflammatory factors such as IFN-γ, TNF-α, LPS and GM-CSF. In mice, they differentiate from Ly6Chi monocytes and promote inflammation by producing high levels of TNF-α, IL-1, IL-2, IL-6 and IL-23. M2 are differentiated by various stimuli, including Th2 cytokines (e.g. IL-4 and IL-13), and anti-inflammatory cytokines such as IL-10 and TGF-β. They express scavenger receptors, mannose receptors and arginase; produce large amounts of IL-10; and promote resolution of inflammation and tissue remodeling. 18 Some phenotype markers used to define mouse M1 such as NOS2, as well as the M2 markers Arginase-1 and Ym1, are not differentially expressed by human Mφ.19–21 Studies of the transcription profile of differently polarized Mφ should help to identify human markers.22–24
It has become increasingly apparent that the active form of vitamin D (vD), 1,25-dihydroxyvitamin D (1,25D), is not only important for calcium and bone homeostasis, but is also involved in regulation of the immune response. 25 1,25D exerts a positive effect on innate immunity, enhancing antibacterial defense through stimulation of autophagy and the induction of specific cytokine responses and antibacterial proteins.26,27 The immunomodulatory effects of vD also involve adaptive immunity via inhibition of Th1 and Th17 pathways.28–30
vD Deficiency is suggested to play a role in the pathogenesis of various immune-mediated diseases, including IBD. 29 vD Deficiency is common among patients with IBD, including those recently diagnosed. 31 An inverse association between disease activity and serum vD levels was observed in some studies.31,32 Few studies have been carried out in patients with CD supplemented with vD, with inconsistent results. 33 This may, in part, be explained by the fact that doses of vD supplementation were too low to normalize serum concentrations. In a recent pilot study, the vast majority of patients with CD needed 5000 IU/d to obtain adequate serum concentrations. 34
Recently, it was reported that inhibiting M1 polarization and increasing M2 improved experimental colitis.35,36 Scant information is available on alterations of Mφ populations in human IBD. The mechanisms governing their effect on colitis development are unclear. The aims of this study were to examine M1 and M2 functions in CD vs. Mφ from controls, and determine the effect of 1,25D on polarized Mφ.
Material and methods
Patients and isolation of monocytes
Patients were recruited at the Centre of Excellence in IBD at the McGill University Health Center, Montreal, QC, Canada. Healthy controls consisted of age- and sex-matched individuals. Informed consent was obtained as per the protocol approved by the Research Ethics Board of the McGill University Health Centre (study number 14-499BMD). This research study involved human subjects and was conducted in accordance with the World Medical Association’s Declaration of Helsinki. Patients taking vD supplementation were excluded from study. Overall, 44 patients with CD and 33 controls were included.
PBMC were isolated from heparinized venous blood using density-gradient centrifugation with Ficoll-Paque PLUS (GE Healthcare, Baie d’Urfe, QC, Canada) and plated at a concentration of 3 × 106 cells/ml. Monocytes were isolated using CD14 microbeads, MS columns and the VarioMACS Separation System (Miltenyi Biotec, Auburn, CA, USA).
Differentiation of monocytes into macrophages and stimulation
Monocytes were isolated using CD14 microbeads and plated at a concentration of 5 × 105 cells/ml. Inflammatory M1-type Mφ were generated by culturing monocytes in the presence of GM-CSF, whereas M-CSF (800 U/ml; Peprotech, Rocky Hill, NJ, USA) was used to produce anti-inflammatory M2. After 6 d, M1 and M2 Mφ were pre-incubated for 20 h with 12.5 nM 1,25D. Mφ were then used or were activated with LPS for an additional 24 h. Supernatants were collected and stored at −80°C until cytokine levels were determined by ELISA (R&D Systems, Minneapolis, MN, USA).
Phagocytosis
Differentiated Mφ were cultured with different concentrations of 1,25D. Phagocytosis was measured using the Phagocytosis Assay kit (Cayman Chemical Co., Ann Arbor, MI, USA). In brief, FITC-latex beads were added to macrophages and cultured at 37°C for 30 min and then analyzed by flow cytometry (FACSCalibur; BD Biosciences, San Jose, CA, USA). Alternatively, phagocytosis was determined by reading the fluorescence intensity on a plate reader (Infinite M200, Tecan Group, Männersdorf, Switzerland).
RNA extraction and real-time PCR
Total cellular RNA was isolated from differentiated Mφ using RNeasy plus mini kit (Qiagen, Valencia, CA USA). cDNA was generated using Transcriptor First Strand cDNA (Roche Diagnostics, Laval, QC, Canada). Real-time RT-PCR was performed by a StepOnePLus RT-PCR (Life Technologies, Burlington, ON, Canada) using PerfeCTa SYBR green Fast Mix (Quanta Biosciences, Beverly, MA, USA). Primers were ordered from Integrated DNA Technologies (Coralville, IA, USA).
The relative expression of each gene was calculated by the ΔCt method, where ΔCt is the value obtained by subtracting the Ct value of GADPH mRNA from the ΔCt value of the target gene. The amount of target relative to the GADPH mRNA was expressed as 2–(ΔCt).
Modified Boyden chamber chemotaxis
Macrophages were plated on culture inserts (8-µm pore; Corning, Corning, NY). fMLP 250 nM or CCL2 25 ng/ml was added to the lower chamber of a 24-well plate. The plate was then incubated (4 h, 37°C). The assay was terminated by detaching the filter and wiping to remove non-migrated cells from the filter top. Migrated cells were then fixed with 1% formalin and stained with hematoxylin. Migrated cells were then counted under a microscope.
Statistical analysis
Data were analyzed using GraphPad Prism, version 5.03 (GraphPad Software, La Jolla, CA, USA). All data are presented as median. The statistical significance of the differences induced by 1,25D was determined using the Wilcoxon matched-pairs signed rank test. The difference between groups was determined using the Mann–Whitney test. Difference between paired samples was assessed using the Wilcoxon signed-rank test. A P-value ≤ 0.05 was considered statistically significant.
Results
Patients recruited for the study
Characteristics of the patients with CD and controls.
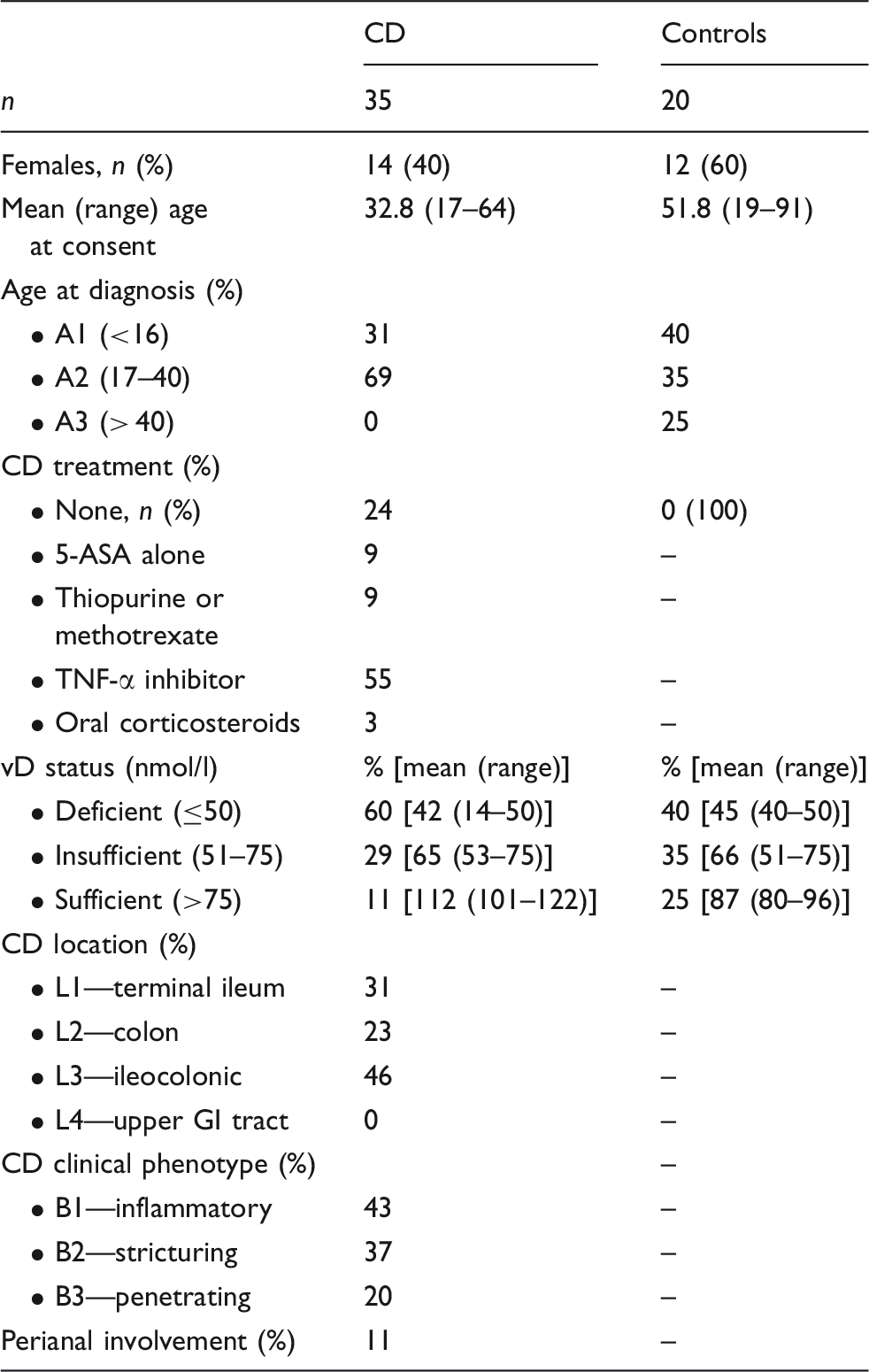
Crohn’s disease age, location and clinical phenotype as per Levine et al. 57 5-ASA: 5-aminosalicylic acid; GI, gastrointestinal.
Effect of 1,25D on phagocytosis and chemotaxis of macrophages
Phagocytic activity of M2 was greater than M1. However, phagocytic activity did not differ between Mφ from patients with CD and controls. 1,25D had no effect on phagocytic activity by any macrophage population (Figure 1). The proportion of untreated and 1,25D-treated M2 that had phagocytosed latex beads was comparably high. Chemotaxis of M1 and M2 generated in CD are shown in Figure 2. M2 migrated in slightly higher numbers toward CCL2 compared with M1. No difference was seen in response to fMLP. 1,25D only marginally increased of both types of Mφ toward fMLP and that of M2 towards CCL2 (data not shown).
Effect of 1,25D on phagocytic activity of M1 and M2 macrophages generated from circulating monocytes in patients with CD (n = 44) and healthy controls (Con; n = 33). Macrophages were incubated with vehicle or vD (12.5 nM 1,25D) for 20 h. Phagocytosis of FITC-labeled latex beads was measured as described in the ‘Materials and methods’. Data represent % of beads phagocytosed. ***P < 0.001. Macrophage chemotaxis in CD. M1 and M2 macrophages were generated from peripheral blood monocytes in 44 patients with CD as described in the ‘Materials and methods’. Chemotaxis was measured by the modified Boyden method in response to CCL2 and fMLP, as described in the ‘Materials and methods’. Data are presented as the number of migrated cells counted under a microscope in response to each chemokine relative to unstimulated conditions for the same patient.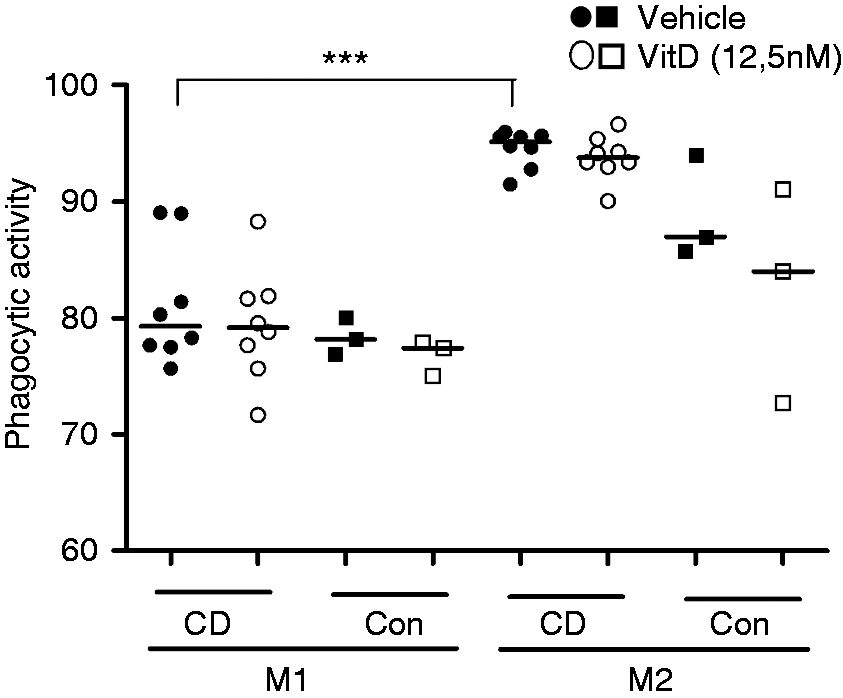
1,25D inhibited inflammatory cytokines production by M1 and M2 macrophages
We next determined the effect of 1,25D on the production of anti- and pro-inflammatory mediators in Mφ from patients with CD and healthy controls. We measured the production of TNF-α and IL-12/IL-23p40, two pro-inflammatory cytokines that play a central role in CD pathogenesis. M1 macrophages from patients with CD produced slightly more TNF-α than M2 macrophages (P = 0.069). LPS-induced TNF-α by CD and control Mφ was comparable. 1,25D decreased TNF-α production by M1 from CD patients (P < 0.005) (Figure 3).
Effect of vD on cytokine production by M1 and M2 macrophages generated from peripheral blood monocytes in patients with CD (n = 44) and controls (n = 33). Polarized macrophages were incubated with vehicle or 12.5 nM 1,25D for 20 h. Cytokine levels were determined by ELISA after stimulation with 100 ng/ml LPS for 24 h. *P < 0.05, **P < 0.005, ***P < 0.001 vs. corresponding samples cultured without 1,25D; #P < 0.05 vs. control samples without 1,25D.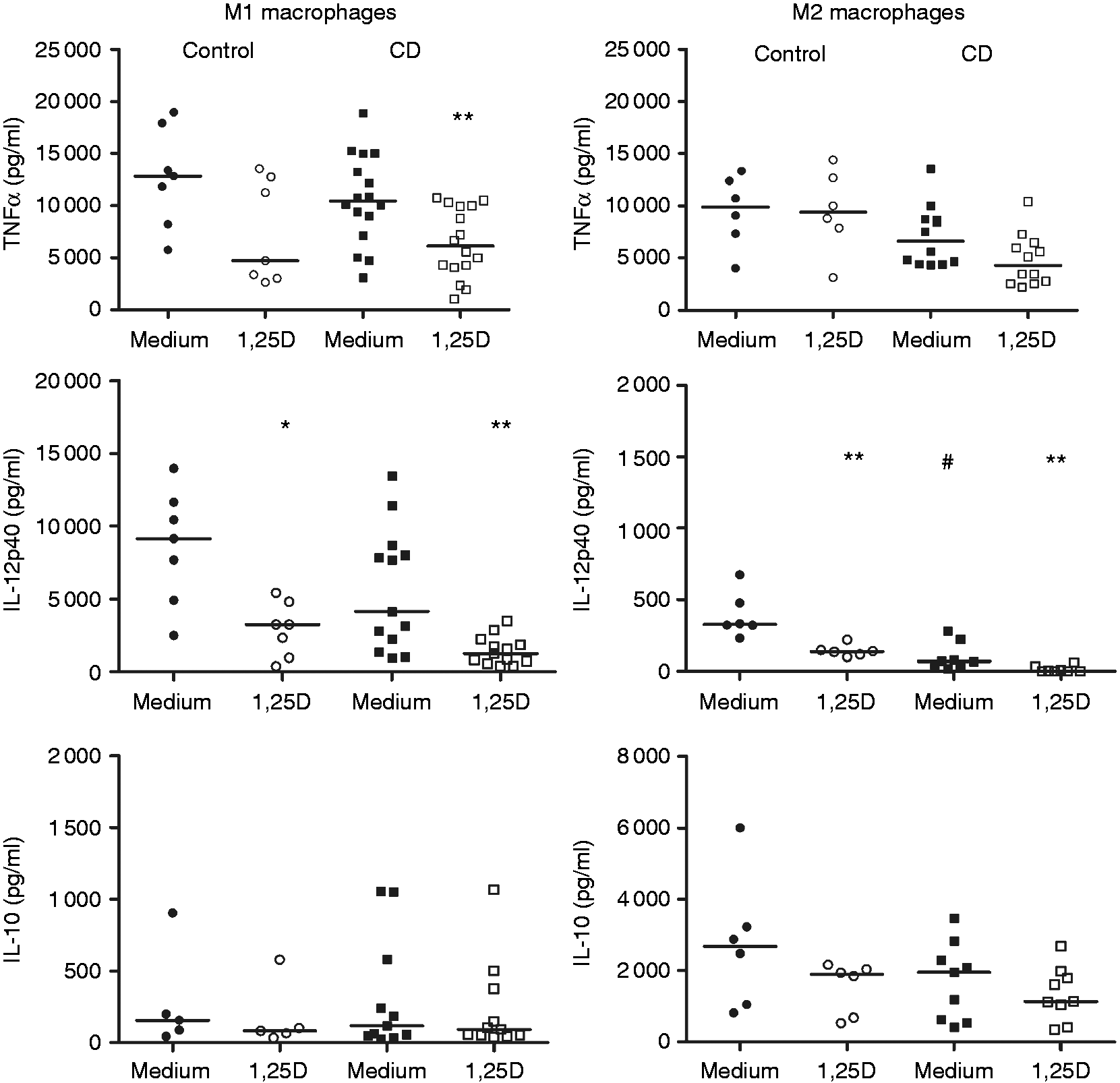
M1 produced much more IL-12/IL-23p40 than M2 macrophages (P < 0.0005) when stimulated with LPS (Figure 3). This is consistent with the known association between IL-12/IL-23p40 and M1 macrophages. M2 from patients with CD released less IL-12/IL-23p40 than those from controls (P < 0.005). Pre-incubation with 1,25D greatly decreased IL-12/IL-23p40 production by M1 macrophages from CD and controls (P < 0.005 and P < 0.05, respectively). IL-12/IL-23p40 released by M2 macrophages was also decreased that by 1,25D (P < 0.005).
IL-10 is an anti-inflammatory cytokine whose production is associated with M2 Mφ.
As expected, IL-10 was produced in higher amounts by M2 than by M1 (P < 0.005). Mφ from patients with CD and controls produced similar amounts of IL-10. Pre-incubation with 1,25D had no effect on IL-10 levels.
Effect of 1,25D on macrophage polarization markers
We next examined if 1,25D alters markers associated with M1 and M2. We found that CD80 and CCR7 cannot discriminate human M1 and M2, at least at the mRNA level. Using a recently published array,37,38 we validated a set of genes that was preferentially expressed by M1 or M2 Mφ. In CD, F13A1, CD163, PTGS2 and CD14 were preferentially expressed by M2 (Figure 4). M1 were associated with higher levels of CYP27B1 and CCL1 mRNA. Other genes, such as MMP2, CXCL10, FN1 and TGFB, were also found to be expressed with some specificity to one of the Mφ populations (Figure 4a). As cells were not always in sufficient numbers to generate both M1 and M2 Mφ from the same patient, we analyzed the expression of the markers relative to GADPH as a housekeeping gene. Although there was a loss in the power to discriminate M1 and M2 by relying on transcript abundance, several markers can still be used. Marker expression in controls and patients with CD was generally similar. PTGS2 expression tended to be higher in patients with CD than in controls, whereas M2 from controls seemed to express more CCL1 (Figure 4b).
Verification of M1 and M2 markers for macrophages generated from peripheral blood monocytes in patients with CD (n = 44) and controls (Con; n = 33). (a) M2/M1 marker ratio in CD. (b) Marker transcript abundance relative to GADPH in CD and controls. *P < 0.05, **P < 0.005, ***P < 0.001.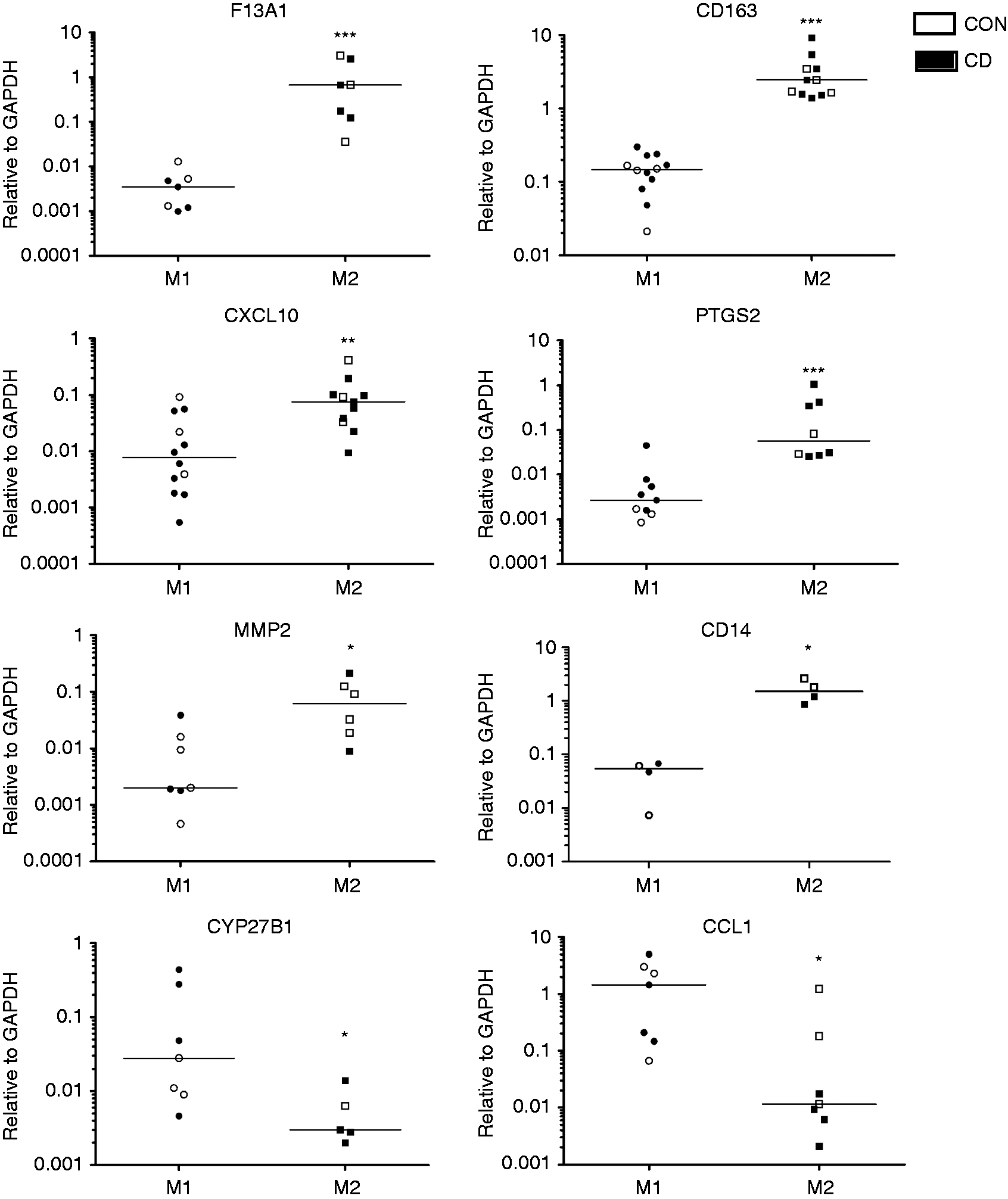
We then evaluated the effect of 1,25D on Mφ polarization markers in CD and controls. Only one of the M1 markers was modulated by 1,25D. CCL1 expression was increased twofold after incubation with 1,25D. However, this difference did not achieve statistical significance (P = 0.06; Figure 5), owing to the limited sample size available for analysis.
Effect of 1,25D on M1 and M2 polarization marker expression in macrophages from patients with CD (▪) and controls (□). Macrophages were derived from peripheral blood monocytes in patients deficient in vD. Changes in M1 markers induced by 12.5 nM 1,25D for 20 h are shown in the upper panel. Changes in M2 markers are similarly illustrated in the lower panel.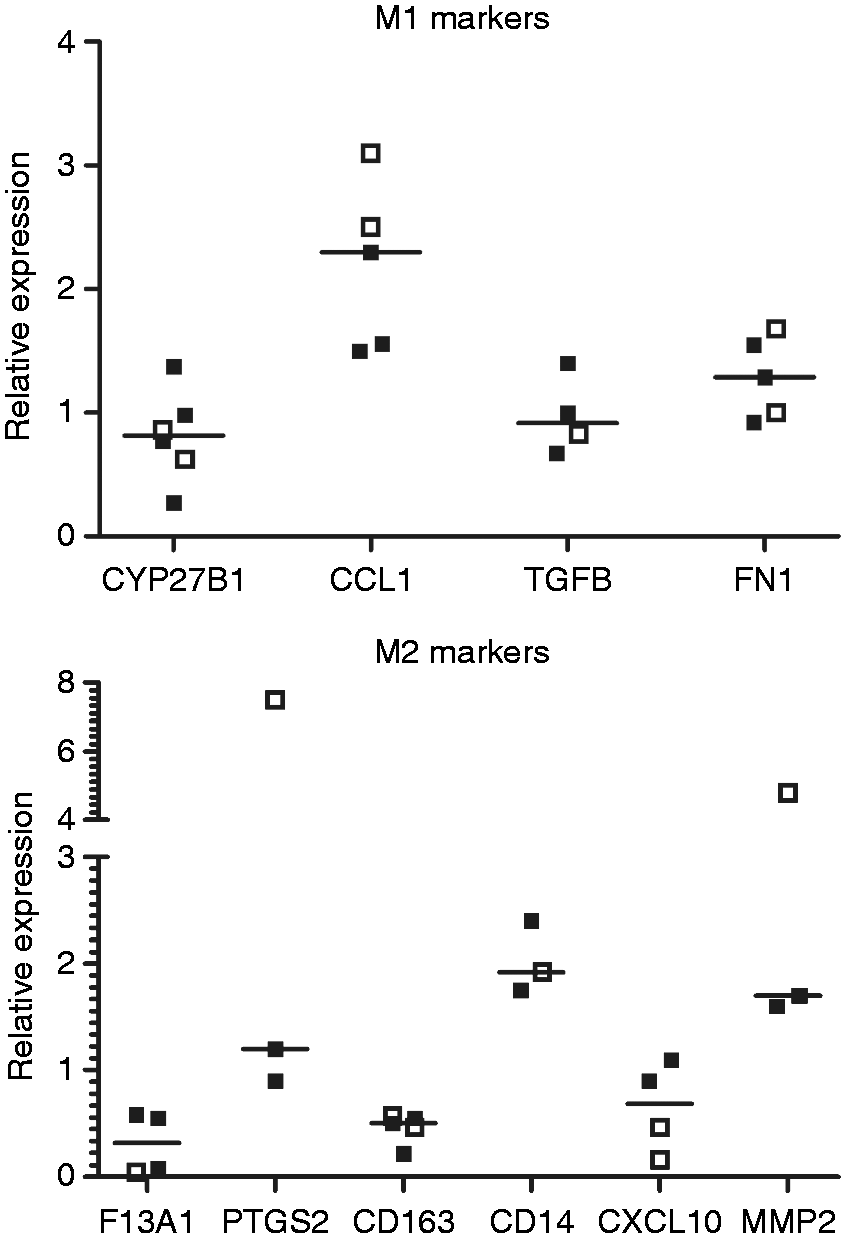
None of the M2 markers was decreased by 1,25D. Expression of CD14 and MMP2 by M2 was enhanced following 1,25D treatment, but the differences were not significantly different, likely owing to the small number of samples analyzed.
Discussion
Macrophages are one of the most abundant leukocytes in the intestinal mucosa, where they play an essential role in maintaining homeostasis. 15 However, they are also implicated in the pathogenesis of disorders such as IBD, offering potential targets for novel therapies. Depending on the signals received, Mφ can be pro- or anti-inflammatory, immunogenic or tolerogenic, and can repair or destroy tissue. Mφ are classified as pro-inflammatory M1 or anti-inflammatory M2.17,18 The role of M1 and M2 in IBD has not been extensively investigated. M1 contribute to experimental dextran sulfate sodium (DSS) colitis, whereas M2 macrophages attenuate colitis severity. 36 Inhibiting M1 polarization was reported to ameliorate colitis.36,39 Using iNOS and TNF-α as M1 markers and CD163 and stabilin-1 as M2 markers, Lissner et al. 40 provided evidence that Mφ subtypes in the intestinal lamina propria are shifted towards pro-inflammatory M1 in CD. Using a co-culture model, M1 reduced transepithelial resistance as a marker for epithelial barrier integrity. 40 The mechanisms for paracellular leakage included intracellular re-localization of tight junction proteins and epithelial cell apoptosis. Using specific cytokine blockade, it was found that M1 exerted their deleterious effect mainly through TNF-α. 40 Using double-labelling immunohistochemistry in situ, Barros et al. 41 also reported that M1 were higher in Th1- associated diseases such as CD. In this study, we examined if M1 and M2 Mφ from patients with CD differed in phenotype and function from those of control patients, and whether vD can modulate their function in vitro.
Our data reveal that Mφ from control and patients with CD display comparably high phagocytic ability. Chemotactic response of CD and control Mφ was also similar. Our results are in line with a recent study that showed that monocyte-derived Mφ isolated from patients with CD and healthy controls display a similar ability to kill phagocytosed bacteria and produce equivalent cytokine levels in response to Escherichia coli. 42 Similarly, phenotypic and functional studies on peripheral blood monocytes from patients with CD in clinical remission were not found to be impaired compared with healthy controls. 43 These results, taken together, suggest that CD, rather than associated with an underlying immunodeficiency, may be related to defective innate immune mechanisms in the inflamed intestinal mucosa rather than in peripheral blood.
Tissue-resident M2 Mφ contribute to mucosal tolerance by secreting IL-10. 44 The lamina propria of the inflamed intestine in patients with IBD, especially CD, is massively infiltrated by monocytes and proinflammatory M1 Mφ. 40 Infiltrating intestinal macrophages are distinct in phenotype and function from their resident counterparts. They strongly express CD14, TREM-1, an amplifier of pro-inflammatory responses, and the human myeloid IgA Fc receptor CD89. 44 In addition, they exhibit activated NF-κB and secrete pro-inflammatory cytokines such as TNF-α. These Mφ are thought to contribute to IBD pathogenesis by disrupting the epithelial barrier. 40 However, Smith et al. 45 reported that in CD Mφ, an abnormal proportion of cytokines are routed to lysosomes and degraded rather than being released through the normal secretory pathway, leading to impaired bacterial clearance. We did not observe significant differences in cytokine secretion between Mφ from patients with CD and controls. TNF-α and IL-10 production by Mφ from patients with CD was comparable with that of controls. LPS elicited a lesser amount of IL-12/IL-23p40 in CD M2 than controls. IL-12/IL-23p40 production by M1 had large inter-individual variations in both groups.
Studies on phenotypic characterization of M1 and M2 Mφ in human diseases are limited. We observed that M1 were associated with higher levels of CYP27B1 and CCL1 mRNA levels. This is consistent with a previous study in which global gene expression profiles of Mφ derived from human monocytes by GM-CSF or M-CSF were compared with their mouse counterparts. 22
Once formed by CYP27B1, 1,25D carries out its diverse biological functions in the intestine, bone and kidney by signaling through the vD receptor. 1,25D is involved in many biological processes, such as apoptosis, cell proliferation and immune function.46,47 As a modulator of innate and adaptive immunity, vD can affect carcinogenesis via changes in systemic inflammation, by down-regulating the TLR signals, cytokines, adipokines and chemokines responsible for Mφ infiltration of adipose tissue. 48 vD modulates the composition of the gut microbiome and its deficiency provokes impaired epithelial integrity, as well as increased inflammation.49–51 1,25D stimulates innate immunity by inducing NOD2 expression and by stimulating genes encoding AMPs, such as defensins and cathelicidin.52,53
Low plasma levels of 25(OH)D are associated with an increased risk of CD, as well as colorectal cancer. 54 In a recent publication, 55 we reported that vD-deficient mice displayed more severe DSS colitis than vD-supplemented mice, with lower survival rates. Increased histological inflammation score and increased IL-6 were also observed in the mucosa of vD-deficient mice. Moreover, vD supplementation decreased the number of inflammation-associated colorectal tumors in mice, independent of the NOD2 gene. 55
CCL1 is a chemokine that attracts Th2 and T regulatory cells. 56 F13A1, CD163, PTGS2 and CD14 were preferentially expressed by M2 Mφ. F13A1 encodes the coagulation factor XIII A subunit that was previously associated with M2 Mφ, 22 whereas the Mφ scavenger receptor CD163 is a classic M2 marker. Inflammatory cytokines such as IL12p40 and TNF-α have been associated with M1, whereas production of IL-10 has been linked with M2. We investigated the potential of vD to modulate Mφ function in patients with CD who were not taking supplements and who were mostly deficient (Table 1). The resulting data show that for both control and CD Mφ, 1,25D decreased inflammatory cytokines while maintaining IL-10 production. Previous reports showed that 1,25D often stimulates IL-10 production, thereby suppressing inflammation. We recently reported that 1,25D pre-treatment decreased the production of TLR2-, TLR4- and TLR7/8-induced cytokines. 52 IL-12/IL-23p40 levels were reduced by 45–56%, whereas those of IL-23 were reduced by 64–70%. IL-10 was less affected, being decreased by up to 25%. These data are in keeping with the effects of 1,25D viewed herein. The different cell types studied, with dissimilar culture conditions and stimuli used can explain the inconsistent IL-10 response to vD in vitro.
Treatment with 1,25D did not change expression of M1 markers except for CCL1, which increased slightly. Expression of the two major M2 markers was not significantly altered by 1,25D for either group. There was a modest increase in PTGS2 and MMP2 following 1,25D incubation. CD14, a known vD receptor target, was upregulated. Several confounding factors may account for the lack of polarization of M1 to M2 in response to 1,25D. First and foremost is the limited number of patients with cells that were available after completing the other experiments. Other explanations include the process used to generate Mφ, the culture conditions and concentration of 1,25D employed.
In summary, 1,25D treatment did not significantly affect Mφ functions such as phagocytosis and chemotaxis, but exerted an inhibitory effect on pro-inflammatory cytokine production. Certain limitations exist in interpreting the results generated in this research. The cells studied were cultured and treated with agents to generate Mφ. These manipulations may yield cells that do not necessarily reflect the real physiological or pathological status in vivo. Moreover, it would be of interest to study intestinal rather than peripheral Mφ. However, the cell isolation process may alter Mφ function. Nevertheless, we believe the data presented here will serve as a starting point for future studies into Mφ biology in CD, ideally focusing on intestinal-derived cells.
Footnotes
Acknowledgements
Ernest Seidman is a Tier 1 Canada Research Chair in immune-mediated gastrointestinal disorders and holds the Bruce Kaufman Chair in IBD at McGill University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by a grant from the Dairy Farmers of Canada.
