Abstract
The objective of the present study was to investigate LPS and lipoteichoic acid (LTA)-induced TLRs, associated signaling molecules and inflammatory mediators, as well as to compare their combined effect in porcine alveolar macrophages. Macrophages were incubated for 24 h with various concentrations of LPS, LTA, LPS + LTA or control. Multiple concentrations of LPS elicited marked up-regulation in mRNA for TLR2 and TLR4, CD14, MD2, MyD88, IRAK-4 and TRAF6 compared with the control. LTA had no effect on TLR4 and MD2; only higher doses up-regulated TLR2, CD14, MyD88, IRAK-4 and TRAF6 mRNA. LPS-activated cells released IL1-β, IL12-β, TNF-α, IL-6, IL-8, IFN-γ and IL-10 in a dose-dependent manner, while LTA had no effect on IL-1β, IL-6 and IFN-γ. Higher doses of LTA induced IL-12β, TNF-α, IL-8 and IL-10. Combined stimulation augmented TLR2, CD14 and MyD88 mRNA, and subsequently produced elevated levels of IL-6, TNF-α and IL-8 when compared with LPS and LTA alone. Additionally, phagocytosis of macrophages was significantly increased following low concentration of LPS treatment. Only low levels of NO (nitric oxide) were detected in the LPS group. Overall, compared with LPS, LTA was a relatively weak inducer, and co-stimulation accelerated gene and cytokine production associated with pulmonary innate immune function.
Introduction
TLRs are key receptors in innate immunity and trigger responses following interaction with pathogen-associated molecular patterns (PAMPs).1,2 Well-described PAMPs include LPS and lipoteichoic acid (LTA), representing the major elements of cell envelopes of Gram-negative and Gram-positive bacteria respectively. Both LTA and LPS are antigenic and able to activate monocytes/macrophages to secrete various inflammatory mediators. Although LTA is much less reported than LPS, LTA was regarded as the Gram-positive equivalent to LPS in some aspects.3,4 LTA produces pathophysiological properties and was published to share recognition mechanisms, such as CD14 and TLRs, with LPS. 5 Activated TLRs transmit signals through MyD88-dependent or independent pathways which, in turn, activate intra-cellular signaling cascades and ultimately result in the induction of innate immune inflammatory mediators.1,2,6
Macrophages are involved in the innate immune response through phagocytosis and the production of a variety of compounds, like cytokines and NO. 7 PAMP-stimulated immune cells secret pro-inflammatory cytokines, such as TNF-α, IL1-β and IL-6, initiating both innate and adaptive defense responses, and also release the anti-inflammatory cytokine, IL-10.8–10 NO synthase gene expression is up-regulated in response to PAMPs through activation of NF-κB and mitogen-activated protein kinases. 11 Over-production of these cytokines and NO might cause inflammatory diseases. 12
Infections of the respiratory tract are frequently caused by co-infections of Gram-positive and Gram-negative bacteria. In particular, bacterial pneumonia, often caused by Gram-positive pathogens, is the most frequent source of sepsis. 13 As the primary inflammatory cell found within the airway, alveolar macrophages (AM) 14 play a central role in the constant recognition of potentially pathogenic organisms entering the lung.15,16 Despite the importance of AM cells in host defense, there is limited knowledge of the factors that are crucial for their effectors functions. Recently, we have reported that different cytokines can be produced by porcine AM cells after initial stimulation with LPS.17,18 In addition, some investigations have been published on the effects of LPS and other PAMPs in porcine immune cells,19–22 but AM responses to LTA and co-stimulation with LPS has never been studied in pig. The synergistic interactions between bacterial components and their activation of TLR signaling have been suggested to contribute to the pathophysiology of sepsis.23,24 It has been reported that combinations of TLR ligands synergistically augment the production of various cytokines by macrophages 25 and appears to be crucial for the innate immune defense against a variety of pathogens. 26 We therefore hypothesized that LPS and LTA would induce differential inflammatory mediators from porcine AMs and their combination would have a synergistic effect. It was proposed that LTA is recognized by TLR2, whereas LPS is mediated through TLR4. However, it has been reported that TLR2 mediates signals from other bacterial components, including peptidoglycan (PGN), and lipoproteins and/or lipopeptides. 27 Indeed, highly purified LTA, as well as chemically synthesized LTA analogs, can stimulate TLR2. 28 However, although LTA is regarded as an important mediator of inflammation, the recruitment of TLRs is still unclear because of biologically active contamination of LTA preparations.
In the present study, we first examined the phagocytic ability of AMs with priming of a range of concentrations of LPS for the selected Gram-negative bacteria. Thereafter, we sought to investigate the comparative induction of TLRs, the associated pathway genes and their effector molecules in porcine AMs following stimulation with LPS and LTA in a dose-dependent manner. Furthermore, we evaluated whether combined stimulation of LPS and LTA would result in amplified induction of these genes and inflammatory molecules from porcine AM cells.
Materials and methods
Animals
Three 40-d-old Pietrain piglets (post-weaned) were used in this study. The pigs were clinically healthy and no respiratory diseases based at the time of this investigation on their health history were found. Animals were housed conventionally in the teaching and research station of Frankenforst, University of Bonn, Germany. The experiment was performed according to the institutional guidelines and animal husbandry regulations of Germany. 29
Isolation of alveolar macrophages
Porcine alveolar macrophages from lungs were obtained by bronchoalveolar lavage (BAL) using ice-cold sterile calcium–magnesium-free Dulbecco’s PBS (D-PBS) (pH 7.4; Sigma Aldrich, Taufkirchen bei München, Germany), as described elsewhere.
18
BAL fluids were filtered through sterile gauze and placed on wet ice. The cells were washed twice with D-PBS by centrifugation at 4
Assay of phagocytic activity
The in vitro phagocytic activity was performed with Vybrant phagocytosis assay kit (Molecular Probes, Darmstadt, Germany), according to the manufacturer’s instructions. Using this method, the process of phagocytosis can be quantified by following the internalization of fluorescently-labeled bacterial particles. The protocol takes advantage of the detectability of the intracellular fluorescence emitted by the engulfed bacteria, and Trypan blue was shown to effectively quench the fluorescence of extracellular fluorescein-labelled Escherichia coli.
30
Briefly, 150 µl of the adjusted cell suspension (106 cells/ml) was seeded in a 24-well plate and treated with various concentrations of LPS (0.1, 1, 5 and 10 µg/well) for 2 h. The positive and negative controls were prepared by adding 150 µl of adjusted cells and 150 µl of RMPI-1640 media, respectively, to the wells on the microplate. Determinations were performed in five replications for each experimental group (at different LPS dose tested), positive and negative control wells. Following 2 h of incubation, solutions were removed from all microplate wells by vacuum aspiration. After that, fluorescein-labeled E. coli (K-12 core type) bioparticles were added to all the positive, negative control and experimental wells. All plates were incubated for 2 h at 37
Cell culture and stimulation
The collected porcine AM cells were re-suspended in 2 mM
In this study, first, adherent macrophages were treated in triplicate with various concentrations (0, 0.1, 1, 5 and 10 µg/ml) of LPS from E. coli 026:B6 (Sigma-Aldrich) and LTA from Staphylococcus aureus (Sigma-Aldrich). Second, a combination of LPS (at 1 µg/ml) and LTA (at 10 µg/ml) was added to the triplicates wells. Media in triplicate wells were added in the same culture plate as control. All plates were incubated at 37
RNA extraction and cDNA synthesis
Harvested cells were washed in ice-cold PBS, and the total RNA was extracted using Pico-Pure RNA isolation kit following the manufacturer’s manual (Arcturus; Invitrogen). The extracted total RNA was treated with RNase-Free DNase Set (Qiagen, Hilden, Germany) for 15 min at 37
Quantification of mRNA
Product sizes and nucleotide sequence details of primers used for qRT-PCR analysis of mRNA expression of porcine TLRs, associated molecules and two reference genes.

bp: base pair.
Cytokine assays
The concentrations of IL1-β, IL12-β, TNF-α, IL-6, IL-8, IFN-γ and IL-10 in cell culture supernatants were measured using commercially-available specific porcine ELISA kits (R&D System, Minneapolis, MN, USA), following the manufacturer’s instructions. The OD value was detected using ELISA plate reader at 450 nm. Standard and sample dilutions were added in duplicate wells. Concentration was calculated from a standard curve and used as protein level (pg/ml). The concentrations were detected according to the standard using microplate data compliance software SoftMax Pro (Molecular Devices, Sunnyvale, CA, USA).
NO assay
The Griess Reagent assay system (Promega, Mannheim, Germany) was used to measure nitrite formed by the spontaneous oxidation of NO. Briefly, 50 µl cell supernatant was added to each well of the 96-well microplate and incubated with 50 µl of sulfanilamide solution (1% sulfanilamide in 5% phosphoric acid) at room temperature for 5–10 min in darkness. Then, 50 µl of 0.1% N-1-napthylethylenediamine dihydrochloride in water was added to each well and incubated at room temperature for 5–10 min in the darkness. The OD was measured at 530 nm. Concentrations were calculated from a standard sodium nitrite curve. All samples were analyzed in duplicate.
Assay of cell viability
In a separate set of experiments, AMs were cultured as described above and treated with LPS (1 µg/ml), LTA (10 µg/ml) and LPS (1 µg/ml)+LTA (10 µg/ml) or medium (control). Following 24 h incubation, cells were collected, washed twice with D-PBS and re-suspended in RPMI-1640 medium. Cell viability was determined using the principle of Trypan blue dye exclusion. The cells from each well were stained with Trypan blue, and the number of dye-excluding (live) cells and positively-staining (dead) cells were counted using a hemocytomer. The assays were performed in duplicate for each group. The results are presented as the percentage of viable cells and, in general, at least 200 cells per vial were scored for viability.
Statistical analysis
The data were analyzed using the SAS software package v. 9.2 (SAS Institute, Cary, NC, USA). All values are expressed in the figures as means of triplicates data ± SEM. The statistical difference of various concentrations of PAMPs on the mRNA expression, cytokine production and phagocytosis were determined by ANOVA followed by Tukey’s multiple comparison procedure as appropriate. The significance of difference between PAMP-stimulated groups was determined using Student’s t-test. All results were deemed significant for P < 0.05.
Results
Phagocytosis assay
We first examined whether treatment with various concentrations of LPS would affect phagocytosis in AM cells in vitro. As shown in Figure 1, treatment of cultured macrophages with low doses of LPS (0.1 µg/ml) resulted in a statistically significant increase in the rate of phagocytosis compared with untreated and the highest does of LPS-treated cells (22 ± 0.07% and 20 ± 0.04% increase from control and highest dose of LPS respectively). Although LPS at 1 µg/ml (51 ± 3.4%) and 5 µg/ml (48 ± 3.7%) primed macrophages, they were able to phagocytose more bioparticles than the control, but there was no significant difference between them (Figure 1).
Effect of LPS-induced phagocytosis on porcine AMs in vitro. Macrophages were incubated with FITC-labeled E. coli with LPS at the indicated concentrations at 37℃ with 5% CO2. Fluorescence as a result of bacteria adherent to the outside of the cells was quenched with Trypan blue. Cell phagocytic capacity was determined by a fluorescence microplate reader. The amount of fluorescent bacteria that has been engulfed by cells was measured as mean fluorescence intensity. The number of cells that were significantly phagocytic is expressed as a percentage of the whole population. Results represent the mean values for three pigs ± SEM. Five determinations were performed at each concentration of LPS per animal. Values with different letters denote a statistical difference among LPS concentrations (P < 0.05).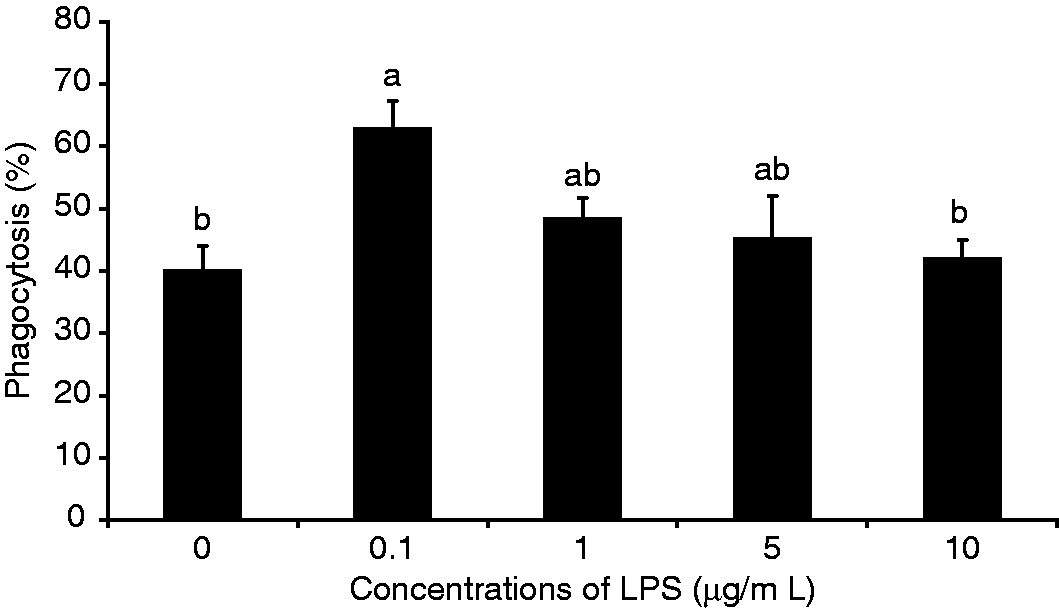
Cell viability
Trypan blue exclusion demonstrated that there were no significant effects in the viability of AMs between samples treated with PAMPs or in the controls after 24 h incubation. As can be seen in Figure 2, the mean viable percentages for the 4 samples were 90.6 ± 4.1, 88.72 ± 5.4, 87.03 ± 5.07 and 87.65 ± 2.7 for control, LPS, LTA and LPS + LTA respectively.
Effects of LPS, LTA and their combined stimulation on AM cell viability. Macrophages were stimulated with LPS at 1 µg/ml, LTA at 10 µg/ml and with LPS (at 1 µg/ml) + LTA (at 10 µg/ml) for 24 h. Cell viability was measured by Trypan blue exclusion. The results are expressed as the mean ± SEM (n = 3) percent cell viability.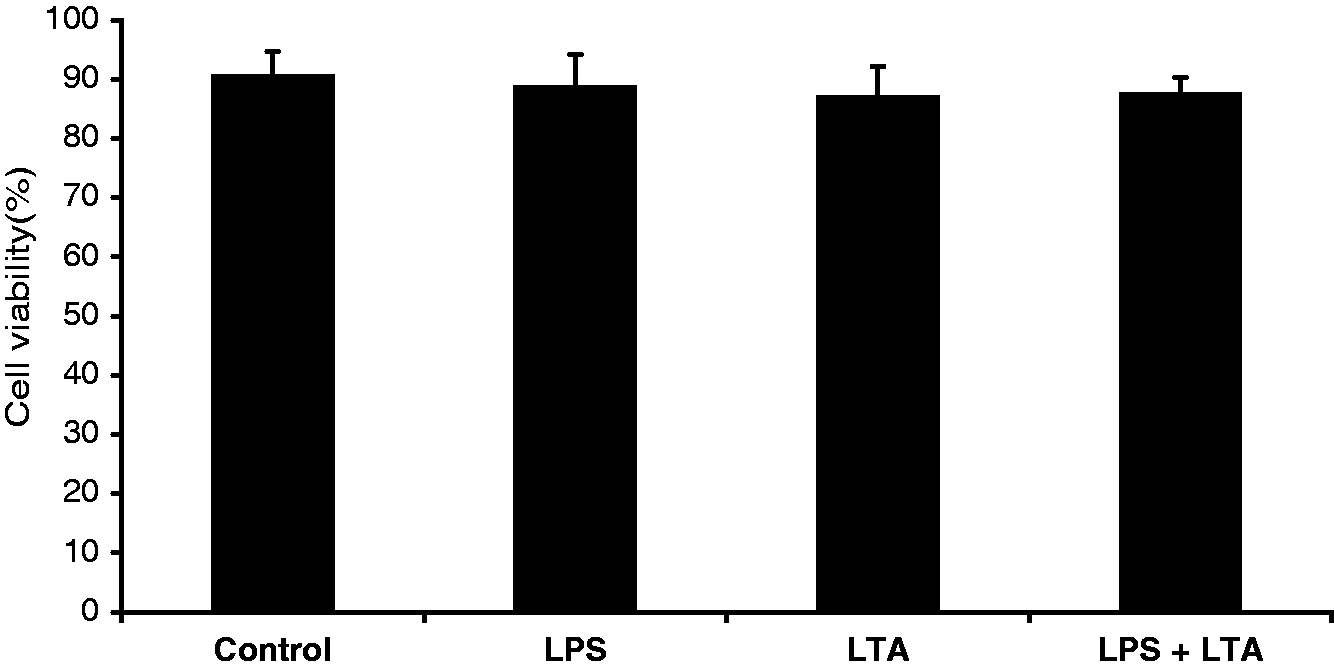
Expression of TLRs and their pathway signaling molecules in a dose-dependent manner
Exposure of cells to LPS resulted in significant up-regulation of TLR4, TLR2, CD14, MD2, IRAK-4 and TRAF6 compared with controls in a dose-dependent way. In contrast, at the concentrations evaluated, LTA had no effect on TLR4 and MD2. Only higher doses of LTA (5–10 µg/ml) caused a significant expression of TLR2, CD14, IRAK-4 and TRAF6 compared with control. In general, it is indicated that LPS was a more potent inducer than LTA, and increasing concentrations of both LPS and LTA induced increased expression of these genes (Figure 3).
Comparative dose-dependent mRNA expression of TLRs and their associated pathway molecules in PAMP-induced porcine AM. Macrophages (2 × 106 cells/ml/well) from 40-d-old piglets were incubated with the indicated concentrations LPS and LTA in triplicate. After stimulation for 24 h, the cells were harvested for quantification of mRNA expression by qRT-PCR. Graph panels are presented as the mean of pigs (n = 3) ± SEM. The x-axis signifies the different concentrations of LPS and LTA (µg/ml). The y-axis indicates the mRNA expression level following stimulation of cells with various doses of LPS and LTA vs controls. Alphabetic characters a, b, c and x, y represent LPS (□) and LTA (▪) respectively. Values with different letters denote a significant expression difference of mRNAs among various concentrations of PAMPs within stimulator group (P < 0.05) analysed by Tukey’s test. Asterisks indicate significant expression differences between LPS and LTA at each concentration (*P < 0.05).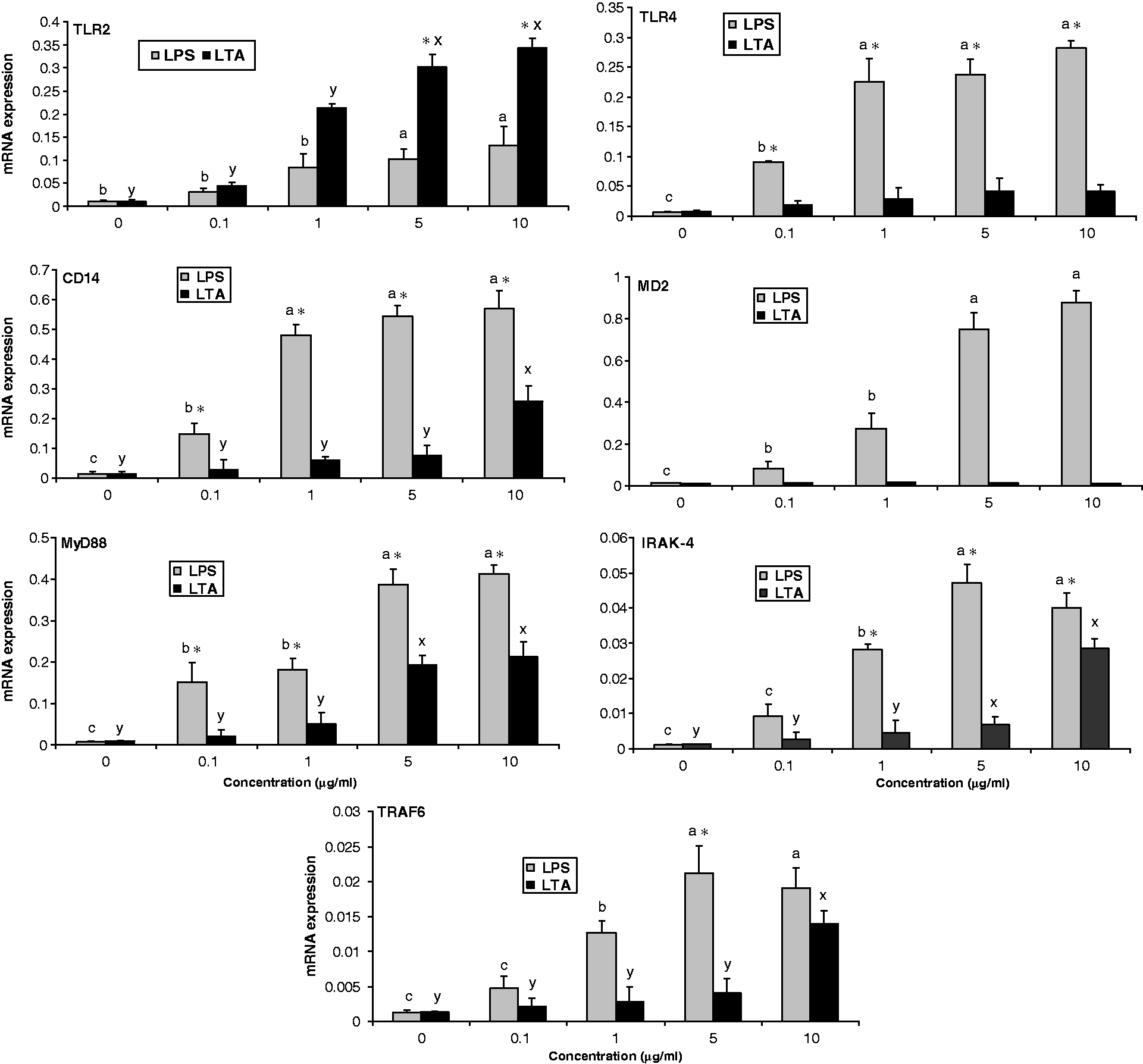
Combined stimulation of LPS and LTA synergistically up-regulated TLR2, CD14 and MyD88 genes
Having shown that various doses of LPS and LTA treatment of macrophages leads to an increase in mRNA expression of TLRs and their pathway molecules, we next assessed whether co-stimulation caused an increase of expression of these genes. Co-stimulation of AMs with LPS and LTA resulted in significant up-regulation of TLR2, CD14 and MyD88 mRNAs levels compared with both stimulator groups alone. LPS at 1 µg/ml and co-stimulation induced significant and similar expression of TLR4 and MD2, but LTA at 10 µg/ml alone had no effect. However there were some synergistic effects of co-stimulation on IRAK-4 and TRAF6 mRNA, but there was no difference compared with LPS and LTA alone (Figure 4).
Expression levels of TLRs and their associated signaling molecules by PAMP-stimulated porcine AMs. Macrophages (2 × 106 cells/ml/well) from 40-d-old piglets were incubated in vitro for 24 h with LPS (1 µg/ml), LTA (10 µg/ml) or co-stimulation of LPS (1 µg/ml) and LTA (10 µg/ml). After the designated time course, total RNA was extracted from cells and mRNA expression was quantified by qRT-PCR. Graph panels are illustrated as mean of three animals ± SEM. Asterisks indicate significant expression differences between the control and treated group (*P < 0.05). Symbols Ψ and ¥ indicate a significant expression difference from LPS and LTA respectively (P < 0.05).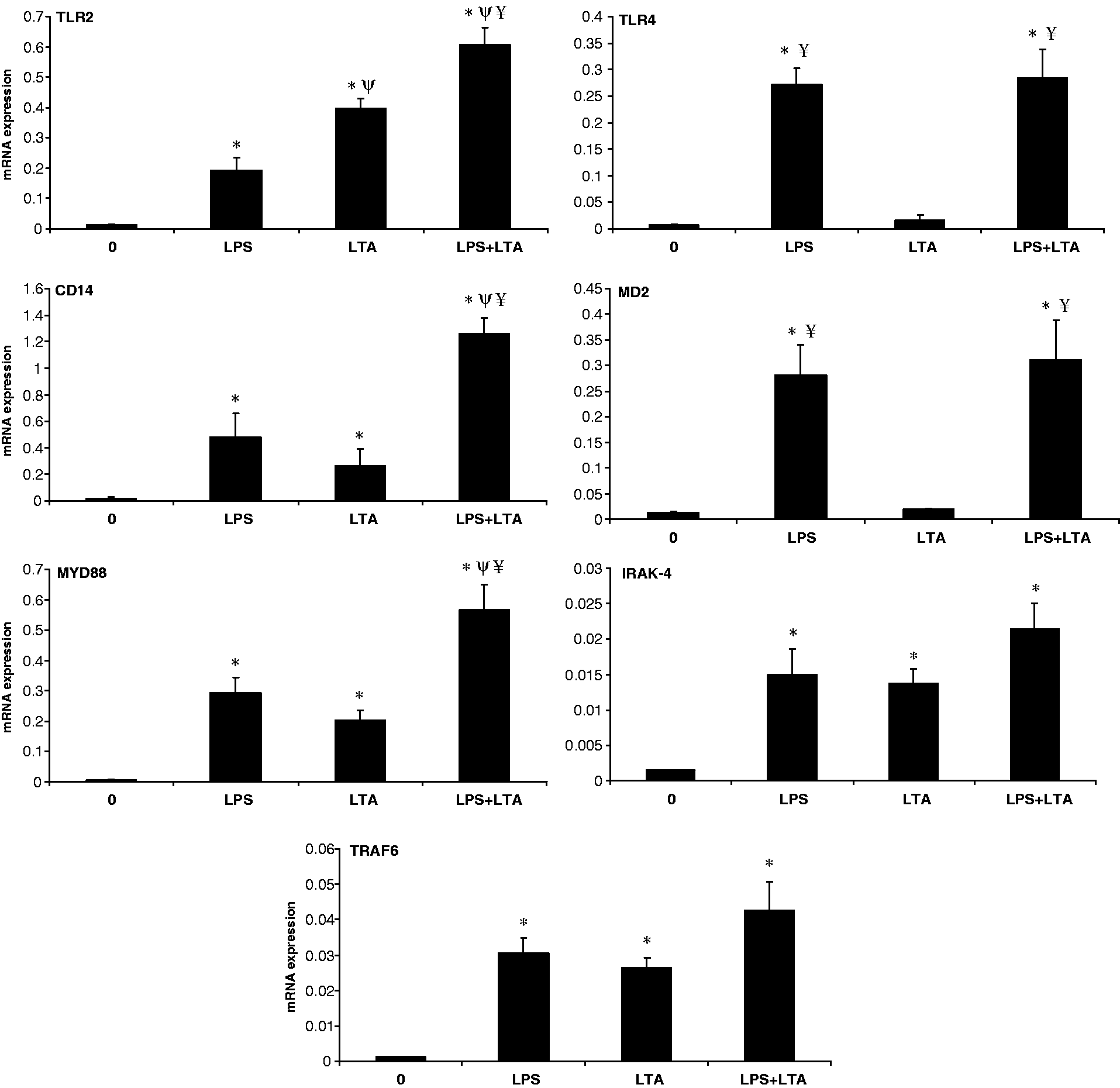
Cytokine production in response to LPS and LTA in a dose-dependent manner
Stimulation of cells with LPS resulted in significant IL-1β, IL-12β, IL-6, TNF-α, IL-8 and IL-10 production compared with the control in a dose-dependent manner. In contrast, LTA stimulation failed to induce IL1-β, IL-6 and IFN-γ production, regardless of the concentrations used. LTA at 5 and 10 µg/ml did stimulate IL12-β, TNF-α and IL-8 production compared with the control. The highest concentration of LTA and LPS resulted in significant production of IL-10 and IFN-γ, respectively, when compared with control (Figure 5).
Comparative cytokine production by PAMP-induced AMs in a dose-dependent manner. Cells were stimulated for 24 h as described in Figure 3. The protein concentrations in the supernatants were measured by ELISA. Graph panels are presented as mean of three pigs ± SEM. The x-axis signifies the different concentrations of LPS and LTA (µg/ml). The y-axis indicates the protein concentrations following stimulation of cells with various doses of LPS and LTA vs controls. Alphabetic characters a, b, c and x, y represent LPS (□) and LTA (▪) respectively. Values with different letters denote a significant production difference of cytokines among various concentrations of PAMPs within the stimulator group (P < 0.05), analysed by Tukey’s test. Asterisks indicate significant expression differences between LPS and LTA at each concentration (*P < 0.05).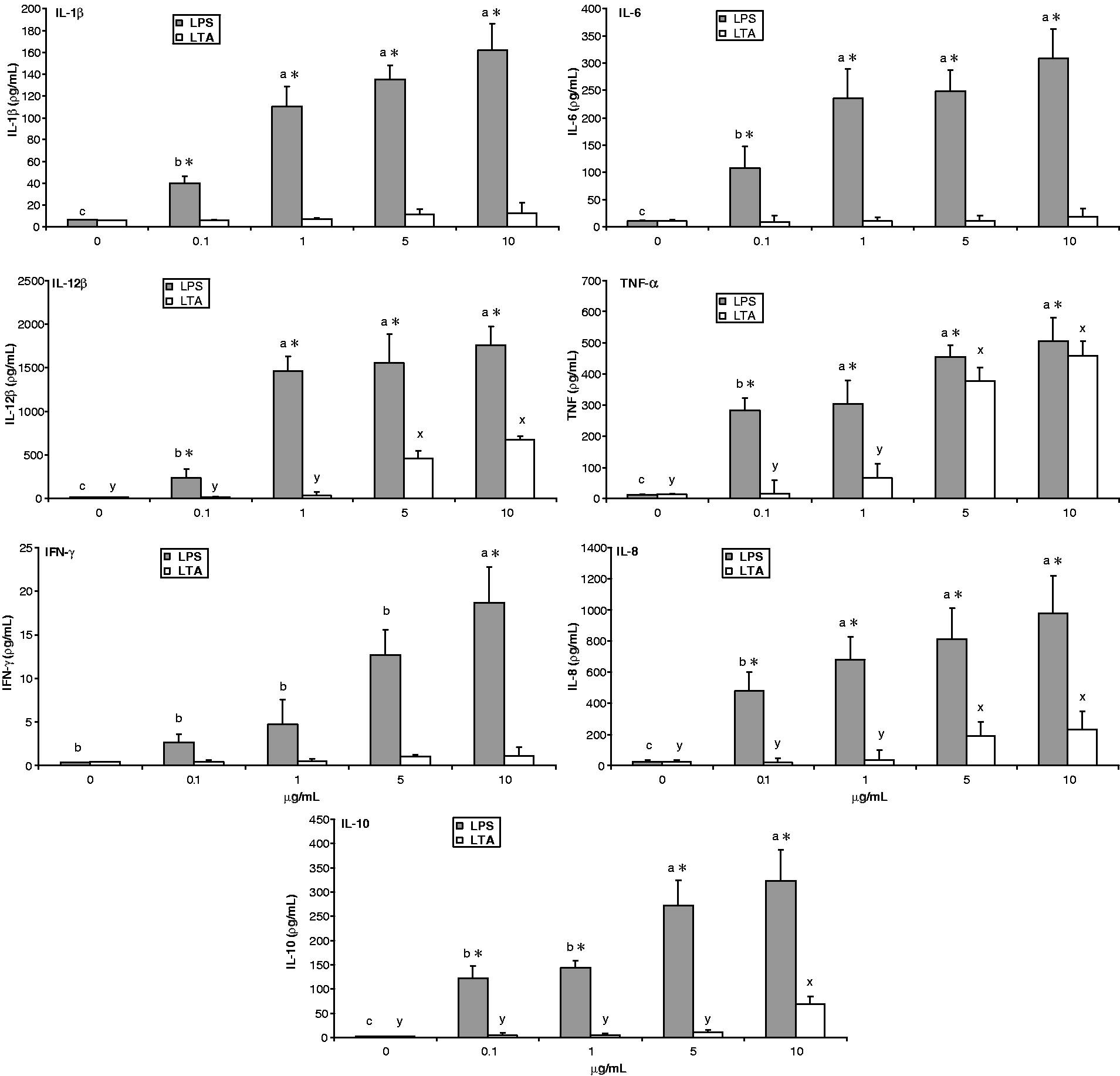
Co-stimulation of LPS and LTA increases cytokine production synergistically
Given the results that LPS and LTA-stimulated cells lead to different patterns of cytokine production in a dose-dependent fashion, we assessed whether combined stimulation of PAMPs would result in amplified induction of cytokines. Stimulation of AMs with the combination of LPS at 1 µg/ml and LTA at 10 µg/ml resulted in significant production of TNF-α and IL-8 compared with LPS and LTA alone. In addition, co-stimulation resulted in significantly higher production of IL1-β, IL12-β and IL-10 compared with control or LTA alone. LTA at 10 µg/ml had no effect but with LPS at 1 µg/ml had synergistic effect in IL-6 and IL-1β production. Co-stimulation with PAMPs failed to induce IFN-γ (Figure 6).
Synergistic effect of LTA with LPS on induction of cytokines in AMs. Cells were stimulated for 24 h, as described in Figure 4. Cytokine concentrations from culture supernatants were measured by ELISA. Graph panels are illustrated as mean of three animals ± SEM. Significant differences (P < 0.05) between the control and treated groups are indicated with an asterisk (*). Symbols Ψ and ¥ indicate a significant production difference from LPS and LTA respectively (P < 0.05).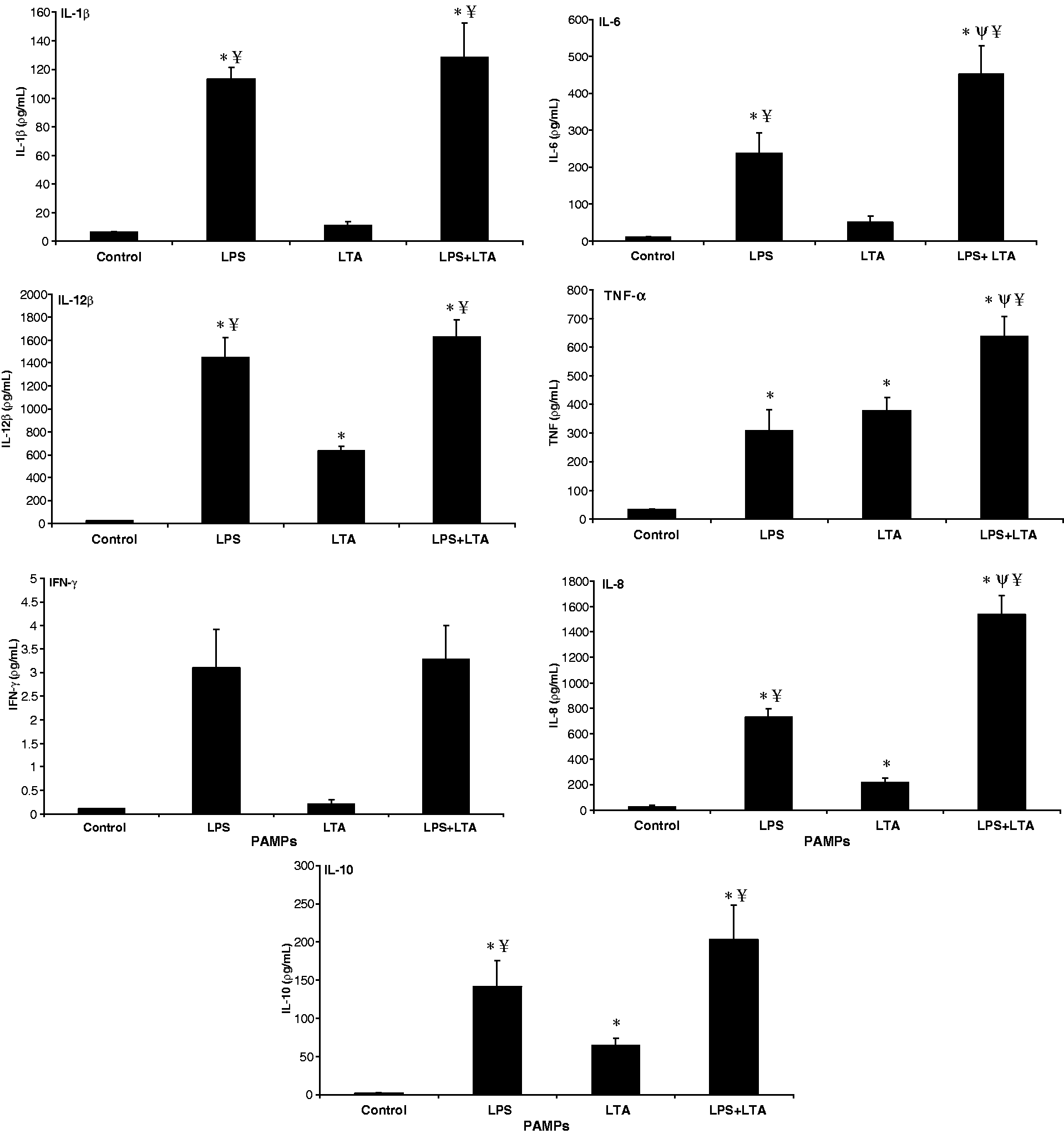
NO production
There was no significant difference in NO production between the treated samples and controls (data not shown).
Discussion
AMs are the primary airway cells responsible for innate immune defense, including phagocytosis of respiratory pathogens. LPS triggers inflammatory responses by inducing inflammatory cytokine production and thus influences the rate of phagocytosis of macrophages in an autocrine manner. 33 In this study, we first focused on the role of a various concentrations of LPS in regulating phagocytosis of fluorescein-labeled E. coli K-12 bioparticles by AMs. We found that macrophages exhibited a significant enhancement in their ability to phagocytise bacteria when exposed to as little as 0.1 µg/ml LPS. In addition, we observed that macrophages exhibited a slight increase in phagocytosis with the other concentrations of LPS used. It is well known that the production of cytokines by LPS-stimulated immune cells is time- and dose-dependent.18,18,34 Recently, we reported that TNF-α, a key pro-inflammatory factor, can be released by porcine AMs at 1 h after LPS stimulation. 18 A recent study reported that LPS-mediated induction of TNF-α inhibited the phagocytic ability of macrophages. 33 Hence, from the findings of the present study, we may speculate that the lack of significant enhancement of phagocytosis after stimulation with higher LPS concentrations can be attributed to LPS-mediated early induction of TNF-α. The finding of our study is also consistent with the observations that phagocytic activities of macrophages are augmented following LPS treatment.35,36 Exposure of AMs to LPS induced early release of TNF-α and IL-8. 17 Thus, the LPS-caused induction of inflammatory cytokine production provides another possible mechanism for the stimulation of phagocytic activities by this endotoxin. Phagocytic action of macrophages requires energy. 37 Thus, LPS may promote mitochondrial ATP synthesis that enhances the phagocytic activities of macrophages.
In this study, one time point, i.e. 24 h, was observed following LPS and LTA treatment according to previously published work. 38 Moreover, we are aware that all the results reported here correspond to this unique time point, i.e. 24 h after stimulation. It has been reported that time points earlier than 24 h are more relevant to decipher the onset of the response to stimulus as shown in kinetics studies in cows 39 , pigs 40 , mice 41 and humans. 42 Moreover, kinetics studies have revealed that many genes return to their basal expression level by 48 h after stimulation, suggesting that homeostasis is restored at that time.39,40 Therefore, in this study, we were interested in investigating the stimulation of AMs at the time when cytokines released into the medium are measured efficiently. It should be noted here that there was no difference in the viability of AMs at 24 h between cells incubated with LPS and LTA or controls. Although this assay does not measure apoptosis directly, it indicates that cell death was not a significant issue when using LPS, LTA or LPS + LTA with these macrophages. Notably, the reference genes used in this study, PPIA and B2M, are stably expressed in porcine AM cells. 31 These two reference genes did not show any significant variation for TLRs or downstream gene expression at the different concentrations of PAMP.
Taking into account that AM cells play a crucial role in the inflammatory process in the pulmonary system, we investigated the dose–response effects of PAMP on the expression of TLRs and downstream signaling genes from porcine AMs. The findings of the present study have shown that AMs responded to the presence of both LPS and LTA by up-regulating CD14 and MyD88 in a dose-dependent manner. We also demonstrated that LTA was unable to stimulate TLR4 and MD2, whereas LPS induced an increased expression of TLR4, TLR2 and MD2, regardless of the concentrations used. In accordance with our results, it has been shown that LTA activates immune cells via TLR2, whereas TLR4 and MD2 are not involved. 43 The identification of the responsible TLR in the recognition of bacterial LTA is controversial. It is noteworthy that commercial preparation of LTA might have contamination with lipopeptide or lipoproteins. Hence, it appears likely that up-regulation of TLR2 was due to impurities of LTA in our study. Many studies involving LTA, especially those using commercially-available preparations, are confounded by contaminating product.44,45 From the present study, we could only state that both LPS and LTA represent immuno-stimulatory compounds; however, the LPS receptor is TLR4/MD2, and that of LTA is still unclear. Moreover, the effects of LPS and LTA on the up-regulation of IRAK4 and TRAF6 gene expression on cells may play a role in further augmentation of innate immune responses.
The measurement of cytokine production is the easiest assessment for cell responsiveness used for different in vitro models. 46 Our study shows differences in cytokine release in vitro in response to LPS compared with LTA, regardless of the concentrations used. Overall, LTA appears to be a less potent cytokine-inducer than LPS. In this study, the lowest concentration of LPS was relatively more potent to induce cytokines IL-12β, TNF-α and IL-8 compared with the highest concentration of LTA. LTA resulted in IL-10 secretion, but to a lesser extent than LPS, and was influenced by their doses. However, the production of inflammatory cytokines is reported to be affected by the concentration of PAMP in swine. 19 By contrast, the lack of detectable IL-1β, IL6 and IFN-γ in response to LTA may be related to the relative insensitivity to cell type in pigs. This finding may speculate the low sensitivity of lung and pulmonary cells to Gram-positive bacterial stimuli via the MyD88-dependent pathway. We detected IFN-γ expression by LPS-induced AM cells, which is somewhat consistent with previously published work. 47 There is contradictory data that IFN-γ was not evident in LPS-induced porcine AMs. 48 This might indicate, in the present study, that AMs contaminated with PMN cells and lymphocytes may lead to the alteration of cytokine secretion. Differences in our study between LPS- and LTA-activated cytokines in vitro concur with previous findings. Our finding is similar to what is observed in humans and mice with lower concentrations of LPS being required to stimulate pro-inflammatory cytokine production compared with LTA and PGN.49,50 Similarly, in a range of human and murine macrophage cells, LTA was consistently less potent than LPS in causing cytokine and or chemokine release.51,52 LPS is a more potent inducer of TNF-α, IL1, IL-6 and IL-8 than other PAMPs from equine and feline whole blood.53,54 Furthermore, IL-1, IL-6 and IL-18 levels have been reported as significantly higher in Gram-negative bacterial infections compared with Gram-positive bacterial infections. 55
Next, we determined the potential synergistic production of inflammatory cytokines provoked by combination of LPS and LTA. Simultaneous stimulation of TLR2 and TLR4 results in amplified cytokine signaling during polymicrobial infections in humans and leads to poor outcomes and an increased risk of septic shock and mortality.56,57 In the present study, co-stimulation of LTA with LPS led to the synergistic activation of TLR2, CD14 and MyD88 genes. Additionally, the combined effects cause significant release of IL-6, TNF-α and IL-8 than either LPS or LTA alone at 24 h post-stimulation. Although co-stimulation resulted in greater IL-6 production, there was no significant production when stimulated with LTA alone. Collectively, our data suggest that during pulmonary infections, an exacerbated inflammatory response may occur inducing the enhanced release of pro-inflammatory cytokines and chemokines, which may lead to septic shock and death. The mechanism underlying the synergistic up-regulation of TLR-associated genes and inflammatory cytokines by PAMP combinations is of interest. Co-stimulation with TLR ligands accelerated gene expression and synergistically induced pro-inflammatory cytokines that are primarily associated with cellular immune function. 58 Previous studies of cytokine protein production by ELISA and mRNA levels of TLR-associated molecules by qRT-PCR confirmed the synergistic activation of certain PAMPs,59,60 and are consistent with our study. Notably, pure LTA appears to be a less potent cytokine inducer than LPS and the ability of LTA to induce higher concentrations of cytokines has been attributed to its synergistic interactions with other bacterial components, such as muramyl dipeptide 61 , PGN or host macromolecules, such as glycosphingolipids. 62 Commercial preparations of LTA, contaminated with LPS and other components, are able to trigger cytokine production in immune cells.44,45 Our findings support the hypothesis of Yang et al. 61 that bacterial cell surface components in various combinations might exert more powerful activities on host cells than the expected levels based on the activities of individual components. The pathophysiology of an infectious disease may be caused not only by the pathogen involved, but also by excessive inflammatory responses, which are sometimes more damaging to the host than the virulent activity of the pathogen that elicited them. Thus, the understanding of the response of AMs to co-stimulation will provide important insights into some effects of Gram-negative and Gram-positive bacteria in the pulmonary infection.
NO is a very important molecule involved in a wide range of physiologic and pathologic processes in mammalian systems, and its production by macrophages is fundamental for immune defense. 63 In the present study, at the tested concentrations, LPS and LTA failed to stimulate NO production from porcine AMs, as assessed by the supernatant–Griess reaction. Our results confirmed previous findings of a failure to induce NO production from porcine AMs, Kupffer cells and peripheral blood mononuclear cells upon stimulation with LPS, LTA and IFN-γ.53,64–66 However, NO production was induced in LPS-activated rat and bovine AMs. 65 But, the fundamental differences among species in the abilities of macrophages to produce NO are not clear.
The current study has several limitations. First, we studied a relatively small number of animals that may be not reflecting the real scenario of a large-sized animal population. Second, we took a limited concentration range of LPS and LTA—a single concentration for combined stimulation with one time point for inflammatory response of cells. Hence, using a wide range of PAMP concentrations, particularly when evaluating the synergistic effects of PAMPs, may have yielded different results. The form of LPS and LTA used in this study also may influence the immuno-stimulatory effect on cells. In this respect, it should be noted that our study does not provide direct evidence for a mechanism underlying the effect of PAMPs on AM cells.
Conclusion
To our knowledge, the present study is the first to provide insight into the in vitro effects of LPS, LTA and their co-stimulation in innate immune responses of AM cells in pigs. Additionally, the present study showed that LPS-activated macrophages led to increased phagocytic activity. Taken together, LTA is a potent mediator of innate immune response, but, compared with LPS, it was a relatively weak inducer in immuno-stimulating response and induced less cytokine secretion. In addition, our findings suggest that the mimic of Gram-negative bacteria synergizes with the mimic of Gram-positive bacteria in macrophages inducing pro-inflammatory cytokines. These data could be helpful in understanding the molecular mechanisms involved in the pathogenesis of infections caused by Gram-negative and Gram-positive bacteria, and their co-infections, and may be potentially useful in developing novel intervention strategies. However, further experiments are clearly awaited to clarify the mechanisms underlying the enhanced responsiveness of AM cells upon co-stimulation of Gram-negative and Gram-positive bacterial components.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Acknowledgements
We are indebted to Mr Ludger Buschen at the research station ‘Frankenforst’ at the University of Bonn for providing the pigs. We are also thankful to Ms Birgit Koch-Fabritius for her technical assistance during the experiments.
Conflict of interest
The authors declare that they have no competing financial or other interest in relation to this work.
