Abstract
Preterm infants are highly susceptible to late-onset sepsis (LOS) and necrotizing enterocolitis (NEC), but disease pathogenesis and specific diagnostic markers are lacking. Circulating cell-free DNA (cfDNA) and immune cell-derived proteins are involved in multiple immune diseases in adults but have not been investigated in preterm neonates. We explored the relation of circulating neutrophil-associated proteins and cfDNA to LOS and/or NEC. Using a clinically relevant preterm pig model of spontaneous LOS and NEC development, we investigated neutrophil-associated proteins and cfDNA in plasma, together with cytokines in gut tissues. The changes in cfDNA levels were further studied in preterm pigs and neonatal mice with induced sepsis, and in preterm infants with or without LOS and/or NEC. Fifteen of 114 preterm pigs spontaneously developed both LOS and NEC, and they showed increased intestinal levels of IL-6 and IL-1β and plasma levels of cfDNA, neutrophil-associated proteins, and proteins involved in platelet-neutrophil interaction during systemic inflammation. The abundance of neutrophil-associated proteins highly correlated with cfDNA levels. Further, Staphylococcus epidermidis challenge of neonatal mice and preterm pigs increased plasma cfDNA levels and bacterial accumulation in the spleen. In infants, plasma cfDNA levels were elevated at LOS diagnosis and 1–6 d before NEC. In conclusion, elevated levels of plasma cfDNA and neutrophil proteins are associated with LOS and NEC diagnosis.
Introduction
Fifteen millions infants are born preterm (< 37 wk of gestation) every year and they have high morbidity and mortality, in part owing to infectious complications, such as late-onset sepsis (LOS) and necrotizing enterocolitis (NEC).1,2 The incidence of LOS and NEC in very low birth weight infants (VLBW; < 1500 g) are approximately 21% and 7%, respectively, and the etiology and pathogenesis of these diseases are still incompletely understood.3,4 Many diagnostic biomarkers for LOS and NEC have been suggested but most of them lack adequate sensitivity and precision.2,4,5 For confirmed LOS diagnosis, positive blood cultures are central but they are time-consuming and often associated with false-negative results that increase the risk of septic shock and death shortly after clinical presentation. 2 Similarly, early NEC diagnosis is difficult (e.g. according to Bell scores) and advanced NEC conditions requiring surgery is associated with high mortality. It is therefore important to determine useful markers for early LOS and NEC diagnosis and implement timely treatments without excessive antibiotic usage.
LOS and NEC often occur together and diagnostic markers are frequently developed for combined LOS and NEC, rather than for each disease separately.2,6 LOS may be caused either by systemic entry of environmental bacteria via indwelling catheters (e.g. Gram-positive Staphylococcus epidermidis) 7 or by bacterial translocation across the immature gut, especially when intestinal structure is compromised by NEC lesions. This condition may explain that the bacterial profile in the blood of LOS infants often reflects the gut microbiota composition, especially during NEC.8–10
Neutropenia is frequently observed in a proportion of preterm infants with neonatal sepsis and/or NEC (16.5–60%).11,12 It may result from insufficient number of circulating neutrophils combined with immature neutrophil structure and function. Neutrophils counteracting bacteria or bacterial components can release neutrophil granule proteins and DNA in neutrophil extracellular traps (NETs). 13 This leads to the elevated levels of multiple circulating proteins, including lactoferrin, neutrophil defensins, L-plastin, calprotectin and histones.14–16 Studies in adults show that neutrophil-associated proteins play a key role in bacterial killing owing to their antimicrobial activities, 17 but impaired mechanisms for clearance of these neutrophil-derived components (e.g. DNA and histones) facilitate severe systemic inflammation and aggravate septic conditions.18,19 Cell-free DNA (cfDNA) has been used as a surrogate marker of circulating NETs as cfDNA from other sources such as platelet, mitochondria or bacteria were negligible, especially in pathological conditions.20–22 Elevated circulating cfDNA and neutrophil granule proteins have been suggested as early biomarkers for sepsis in adults,23–25 and elimination of DNA by DNase treatment reduces sepsis severity and organ injury. 18 Recent studies also showed the presence of cfDNA and neutrophil proteins in the intestine during NEC. 26 As yet, no studies have investigated the potential roles of such components in blood during development of the serious neonatal systemic infections associated with LOS and NEC.
We hypothesized that levels of circulating neutrophil-associated components would be elevated in preterm neonates with LOS and/or NEC. First, we used a model of preterm pigs, which spontaneously develop NEC associated with variable LOS symptoms (like in infants), to characterize the levels of circulating neutrophil-associated proteins and cfDNA. Second, we investigated the possible sepsis-induced elevation of cfDNA in preterm pigs and neonatal mice following systemic challenge with S. epidermidis, a bacterial species commonly found in LOS infants. Finally, we performed a retrospective study in preterm infants to investigate circulating cfDNA levels shortly before and at diagnosis of LOS and/or NEC.
Materials and methods
Spontaneous preterm pig model of LOS and NEC
Preterm pigs and infants show many similarities of immaturity, including respiratory, metabolic and cardiovascular deficiencies, together with high susceptibility to NEC and sepsis after preterm birth and enteral feeding. 27 We performed a preterm pig study with similar feeding and parenteral nutrition regimes to that in preterm infants, in order to analyze multiple gut and circulating parameters. Detailed experimental procedures are described in Supplementary material 1. Briefly, preterm pigs (n = 114, eight litters) were delivered by caesarean section at day 106 (90% of gestation) and fed donor human milk (Hvidovre Hospital, Hvidovre, Denmark) with various types of milk fortifiers for 8–9 d. At euthanasia, plasma was collected for proteomics and cfDNA analyses and the small intestine was macroscopically scored for evaluation of NEC-like lesions. 28 Distal intestinal tissues were snap-frozen and stored at −80℃ for cytokine analysis. The intact left femur was aseptically collected for bacterial culture of bone marrow. A pig with severe peritonitis, or multiple organ injuries (except small intestine), or bacteria detected in the bone marrow (> 500 CFU/g) with organ injury (except small intestine) was diagnosed as sepsis. All piglet procedures were approved by the Danish National Committee of Animal Experimentation.
Preterm pigs and neonatal mice with sepsis induced by S. epidermidis
To investigate the association of cfDNA with only LOS (no NEC), we challenged newborn preterm pigs and mice with S. epidermidis, a species often found in patients with LOS. Twenty-one caesarean-delivered preterm pigs (90% gestation) were fed minimal amount of enteral nutrition to avoid NEC development. At postnatal d 4 (PND4), pigs was systemically injected with S. epidermidis at 109 CFU/kg (SE pigs, n = 11) or saline (SAL pigs, n = 10) and plasma was collected 12 h post-infection. Mouse experiments were approved by the Animal Ethical Committee of Gothenburg. Sepsis was induced in C57BL/6 wild-type mice (Charles River Laboratories, Sulzfeld, Germany) by an i.p. injection of 7 × 107 S. epidermidis (SE) at PND9 or PND14 (n = 7 or 5, respectively). Controls were injected with saline (SAL, n = 5 and 4 for PND9 and 14, respectively). Plasma and spleen tissue were collected at euthanasia of 24 h post-infection.
Blood cell counts and functions and intestinal cytokines in the piglet model
Blood leukocytes at euthanasia were characterized by an automatic cell counter (Advia 2120i Hematology System, Siemens, Germany). Whole blood was collected from three litters (37 pigs, five NEC cases) at PND5 prior to any NEC symptoms and analyzed for neutrophil phagocytosis using pHrodo Red Escherichia coli Bioparticles Phagocytosis Kit for Flow Cytometry (Life Technologies, Nærum, Denmark), as described in Supplementary material 2. Distal small intestinal tissues were homogenized and analyzed for IL-6 and IL-1β by ELISA (R&D Systems, Abingdon, UK) as previously reported. 28 Plasma CRP levels were analyzed by ELISA according to the manufacturer’s instruction (R&D Systems).
Circulating cfDNA levels and bacteria in organs
Circulating cfDNA was analyzed in infants, piglets and mice using the Quant-iT PicoGreen dsDNA Assay Kit (Life Technologies) as previously described. 24 Blood and homogenized bone marrow of preterm pigs were cultured on blood agar for enumeration of bacterial colonies, which were identified by a MALDI-TOF mass spectrometer. 10 The spleens of mice were homogenized, spot-plated in serial dilutions on tryptic soy agar, and incubated overnight at 37℃ to enumerate the S. epidermidis.
Plasma proteomics for preterm pigs with spontaneous development of LOS and NEC
Thirteen pigs diagnosed with NEC and sepsis (LOS + NEC) and 15 pigs without any intestinal lesions (CON) were randomly selected for plasma proteomics with detailed procedure described in the Supplementary material 3. Briefly, plasma samples were denatured, reduced, alkylated, filtered by YM-10 kDa spinfilter (Millipore) and digested overnight prior to analysis by UPLC-MS using a nano-UPLC system coupled online to a Q-Exactive mass HF spectrometer (Thermo Scientific). 29 Peptide signaling intensities from full MS scans were imported and further processed in MaxQuant 1.5.2.6 software and Perseus software (http://www.coxdocs.org/doku.php?id=:perseus:start). 30 Protein identification was performed by data searching against Uniprot Sus Scrofa protein reference proteome databases and an in-house optimized PeptideAtlas database for Sus scrofa. 31
Pilot case–control study in preterm infants
This study was a retrospective case–control study with very preterm infants (< 32 wk of gestation) recruited in an ongoing sample salvage study (SERVIS) at the neonatal intensive care unit of the Royal Victoria Infirmary, Newcastle upon Tyne, UK. Ethical approval was obtained from County Durham and Tees Valley Research Ethics Committee and parenteral consents were signed for all recruited infants. Twenty-seven infants diagnosed as LOS and/or NEC (13 LOS, 13 NEC and one combined), as previously described, 32 with salvaged blood available on the day of diagnosis and/or up to 6 d prior to diagnosis, were identified as ‘cases’. Each case was matched with one control infant (no LOS or NEC) by age at blood sampling and gestational age. Standard feeding, antibiotic and antifungal guidelines were followed. 33 Serum samples stored at –80℃ were used for analysis of cfDNA.
Statistics and bioinformatics
Statistics were performed in JMP 11.0 (JMP, SAS Institute, Cary, NC, USA) for all data except proteomic data with FDR control analyzed in Perseus software. For comparing infant groups before the disease onset or at disease diagnosis, cfDNA data were fitted into linear mixed models with disease as a fixed factor and matched gestational age as a random factor. Data from the pig study (cfDNA, cytokine, CRP and phagocytosis) were fitted into linear models with disease and sow as fixed factors. Data from study of sepsis-induced preterm piglets were fitted into linear models with treatment as a fixed factor. Data from mice study were fitted into linear mixed models with treatment and age as fixed factors and experiment as random factor. ROC curve analysis in all 114 pigs (LOS + NEC vs. CON) was performed to evaluate the diagnostic usefulness of cfDNA. Proteomic data were initially tested by a two-sided t-test (NEC + LOS vs. CON pigs) followed by permutation-based false-positive control to correct for multiple testing (FDR <0.2). Abundances of significant proteins with FDR control were further verified by fitting data into linear models with disease and pig study time as fixed factors. The MS data were deposited to the ProteomeXchange Consortium via PRIDE partner repository with data set identifier PXD004082. Significant proteins were also analyzed in the open-source pathway database Reactome (www.reactome.org) to elucidate pathways involved in the disease pathogenesis. All differential proteins identified by proteomics were further analyzed for hierarchical clustering to determine if levels of neutrophil-associated proteins had similar regulatory trends. Further, circulating cfDNA levels and abundance of neutrophil-associated proteins identified by proteomics were analyzed for pair-wise Pearson correlations. Values are means ± SEM. P < 0.05 was regarded as statistically significant.
Results
Clinical evaluation, gut cytokines and circulating parameters in LOS + NEC preterm pigs
NEC is often associated with systemic inflammation and multiple organ injuries. Using the spontaneous model of combined LOS and NEC in preterm pigs, we carefully evaluated gut lesions and other organ injuries coupled with culture of blood and bone marrow. Fifteen of 114 pigs from eight litters (13%) developed NEC in the small intestine, a rate similar to the NEC rate of 2–15% in preterm infants. 34 Twelve of 15 NEC pigs had organ culture data available, and 11/12 of these had positive bacterial cultures in bone marrow, severe peritonitis or multiple organ injuries (atelectasis, renal petechia or hemorrhage, severe hepatic steatosis, gastric hemorrhage or hemorrhage in lymph nodes). Three remaining NEC pigs also had severe peritonitis or multiple organ injuries. These pigs were categorized as NEC and sepsis (LOS + NEC), and they had no sign of thrombocytopenia (mean 165–225 × 109 cells/l).
Levels of inflammatory cytokines IL-6 and IL-1β were higher in intestinal tissues of LOS + NEC vs. CON pigs (P < 0.001; Figure 1a, b), reflecting inflammatory responses associated with NEC lesions. Plasma CRP concentrations in LOS + NEC pigs were 2.5-fold higher than in CON pigs (P < 0.05; Figure 1c). On day 5 before any clinical signs of LOS or NEC, neutrophil phagocytic rate (percentage of neutrophils that exert phagocytic capacity) in LOS + NEC pigs tended to be lower than in CON pigs (P = 0.09; Figure 1d).
Intestinal cytokines, plasma CRP and ex vivo blood neutrophil phagocytosis in LOS + NEC vs. CON pigs. (a–c) intestinal IL-6 and IL-1β, and plasma CRP and (d) neutrophil phagocytic rate. Values are means ± SEM. *P < 0.05 and ***P < 0.001.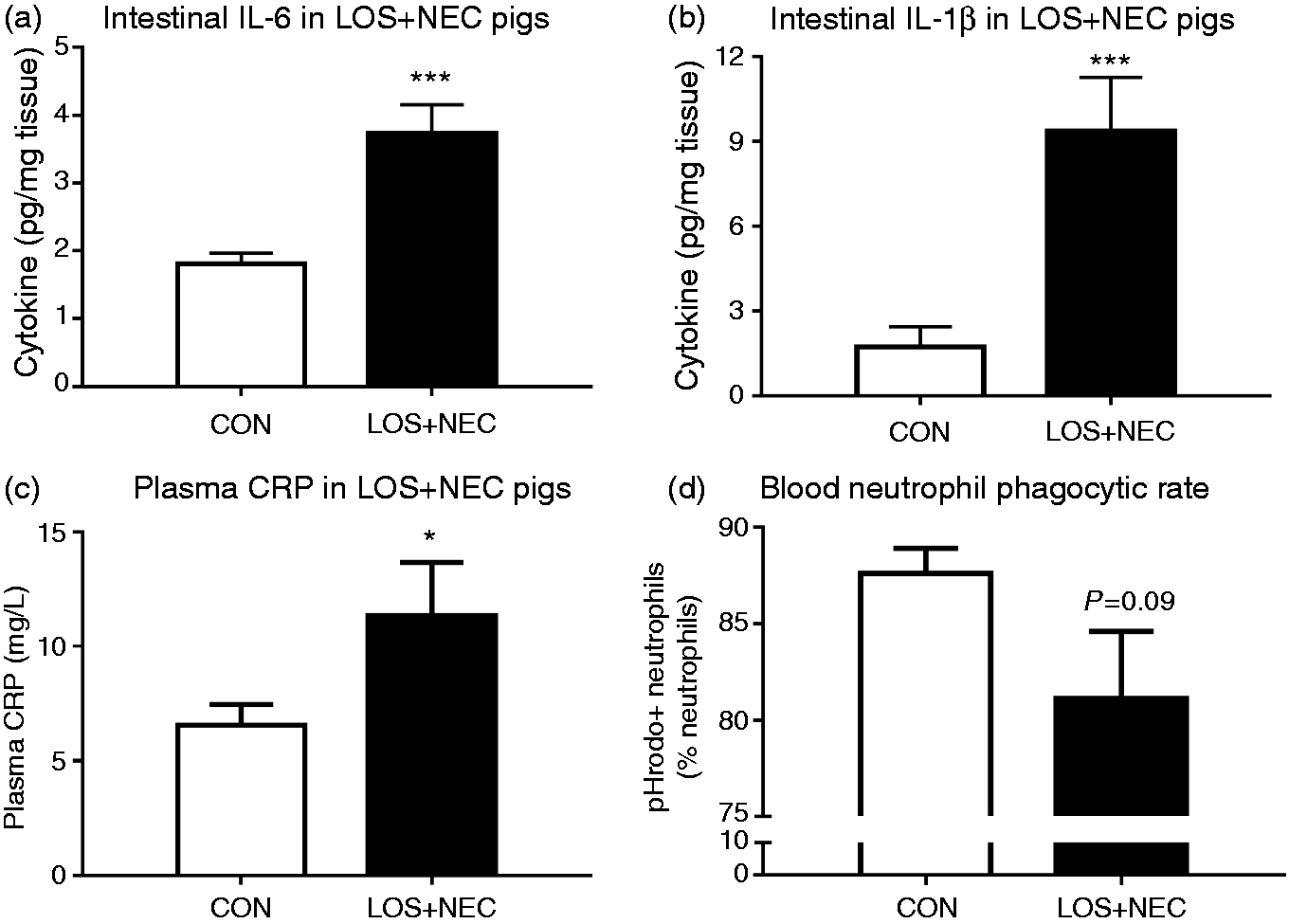
LOS + NEC pigs had 1.5-fold higher circulating cfDNA levels than CON pigs (P < 0.001; Figure 2a). Receiver operating characteristics (ROC) analysis for cfDNA across all 114 pigs showed that the area under the curve (AUC) was 0.83 ± 0.07, indicating a good diagnostic accuracy, together with the best sensitivity and specificity coordinates of 100% and 57%, respectively (Figure 2b), corresponding to the cut-off values of 100.3 ng/ml cfDNA. These values were greater than that of plasma CRP (AUC of 0.69 ± 0.08 with the best sensitivity and specificity coordinates of 73% and 62%, respectively).
Circulating cfDNA and neutrophil-associated proteins in LOS + NEC vs. CON pigs. (a) Circulating cfDNA. (b) ROC curve analysis of circulating cfDNA in diagnosing LOS + NEC in 114 preterm pigs. The AUC was 0.83. The gray line at 45o angle tangent to ROC curve marks the best cut-off point of sensitivity and specificity coordinates showing a diagnostic ability of 100% true-positive rate (correctly classified LOS + NEC) but 43% controls with false-positive results. (c) Fold change of five neutrophil-associated proteins (gray) and four proteins associated with neutrophil-platelet interaction during systemic inflammation (black) as identified by proteomics. Values in (a) are means ± SEM. *P < 0.05, **P < 0.01 and ***P < 0.001.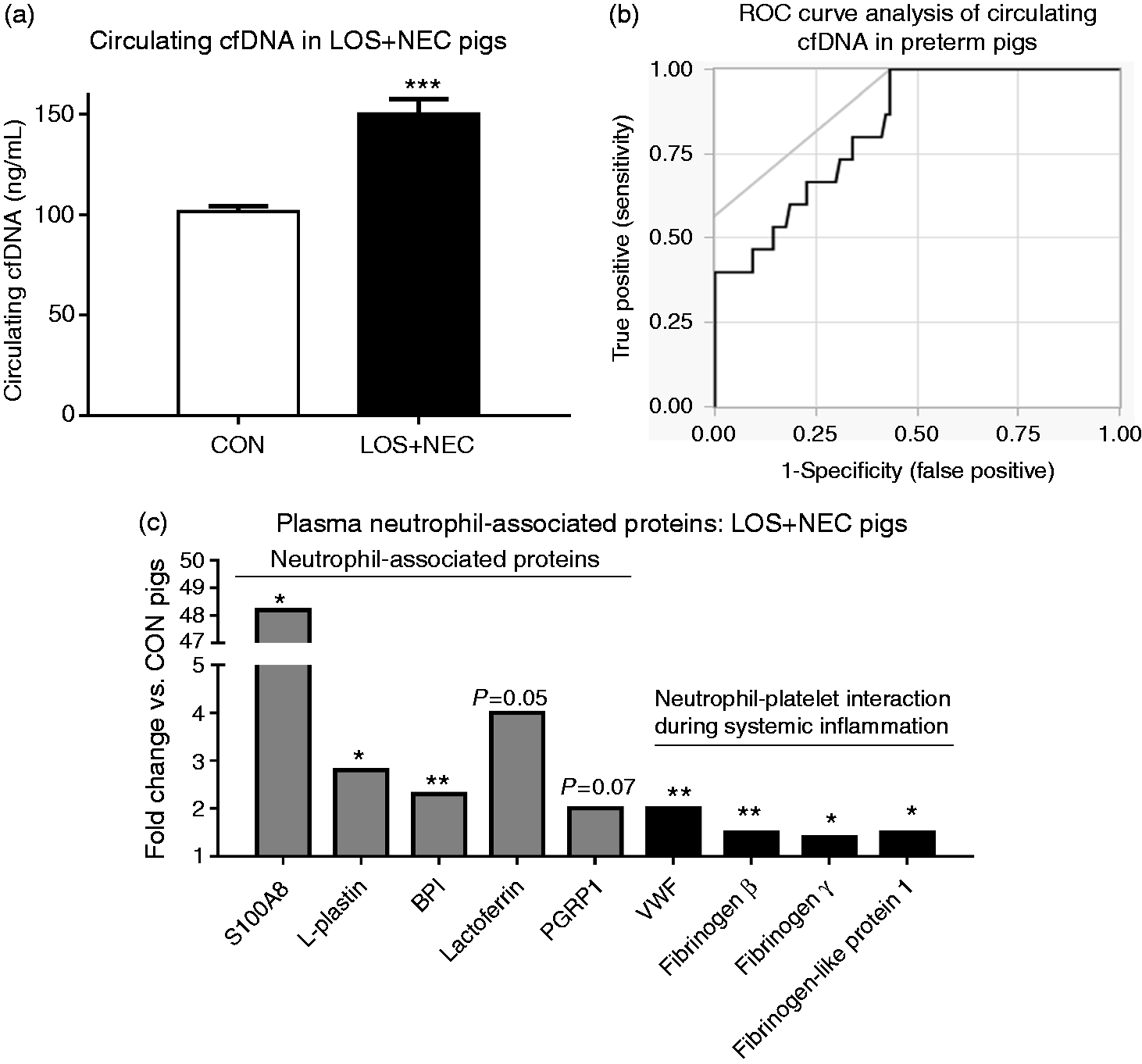
Plasma proteomics in preterm pigs with LOS + NEC
To further explore circulating neutrophil-associated components which may potentially be released during LOS and NEC, we performed a proteomic analysis using plasma samples randomly selected from 15 CON pigs without any organ lesions or positive blood cultures, and 13 LOS + NEC pigs. CON pigs had either undetected bacteria in organs (12/15) or minimal bacterial load present in the bone marrow (one pig with level > 500 CFU/g without additional organ injury, 103 ± 103 CFU/g vs. 2.5 × 105 ± 1.4 × 105 CFU/mg in LOS + NEC pigs; P < 0.01).
List of plasma proteins identified and quantified by proteomics with statistically significant changes of the mean of abundance between LOS + NEC vs. CON pigs.
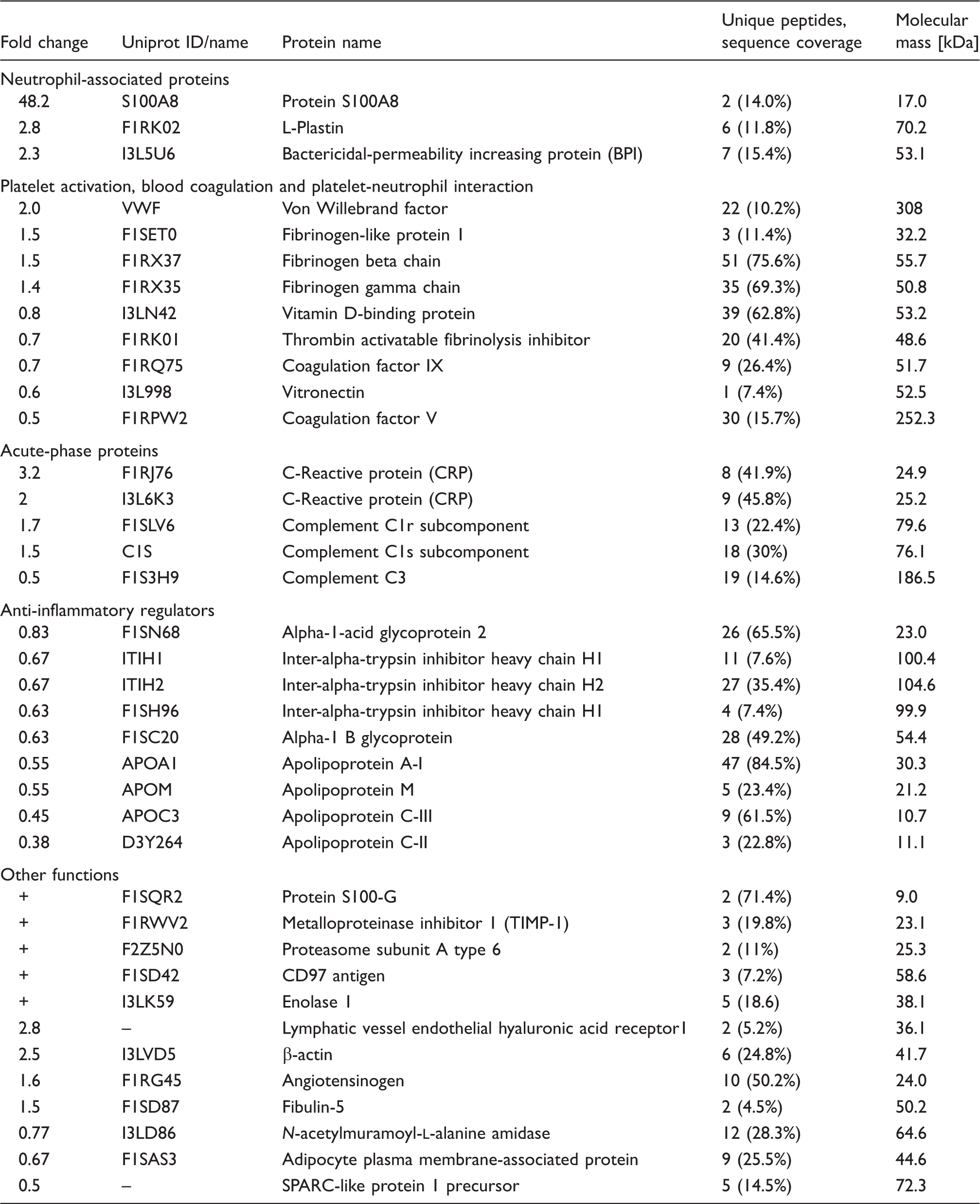
Three proteins, including lactoferrin, S100A8 and peptidoglycan-recognition protein 1 (PGRP1), which are mainly abundant in neutrophils and present in NET structures,14–16 were absent in 80–87% of the CON pigs. Statistics with linear models showed a significant increase in S100A8 levels (48.2 fold, P < 0.05) and a strong tendency to elevated lactoferrin and PGRP1 levels [four- and twofold (P = 0.05–0.06), respectively] in LOS + NEC vs. CON pigs. The fold changes for five neutrophil-associated proteins with significant or strong tendency to significant elevation in the plasma of LOS + NEC are shown in Figure 2c. These five proteins have repeatedly been reported as proteins specifically derived from neutrophils, with bactericidal-permeability increasing protein (BPI), S100A8 and lactoferrin being mainly derived from neutrophils in the blood.14–16,35,36 Fibrinogen chains and von Willebrand factor (vWF) are additional up-regulated proteins in LOS + NEC pigs (Figure 2c), which have been reported to be involved in the interaction between activated neutrophils and platelets during systemic inflammation.19,37,38
Functional analysis by the Reactome database showed key regulated pathways during LOS/NEC. The top 20 pathways (lowest FDR) are shown in Supplementary Table S1 with 8/20 pathways related to platelet activation, clot formation, immunity and inflammatory response. Hierarchical clustering analysis of differential proteins revealed that the neutrophil-associated proteins (marked in red) and proteins involved in neutrophil-platelet interaction during systemic inflammation (marked in blue) clustered well together with small distance among proteins in each group (red and green rectangles) as well as among proteins in both the groups (blue rectangle, Figure 3a). Multi-pairwise correlation analysis between cfDNA levels and abundance of neutrophil-associated proteins (36 pairs) demonstrated 14 significant correlations (P < 0.05) with strong Pearson r values (Supplementary Table S2). Among these, cfDNA levels showed strong correlations with the abundance of lactoferrin, L-plastin, BPI and vWF (r = 0.781, 0.658, 0.478 and 0.485, respectively; Figure 3b–e). These results strongly suggest that cfDNA and neutrophil-associated proteins may be released together during systemic inflammation and NEC in preterm pigs.
Clustering and correlation analyses of neutrophil-associated proteins and cfDNA analyzed in the plasma of LOS + NEC vs. CON pigs. (a) Hierarchical clustering analysis of differential proteins identified by plasma proteomics. (b) Correlation between cfDNA levels and abundance of neutrophil-associated proteins. Red, green and blue rectangles indicate clusters of only neutrophil-associated proteins (red font), only proteins involved in neutrophil-platelet interaction during systemic inflammation (blue font), or both protein groups, respectively. FGB: fibrinogen β; FGG: fibrinogen γ; LCP1: L-plastin; LTF: lactoferrin.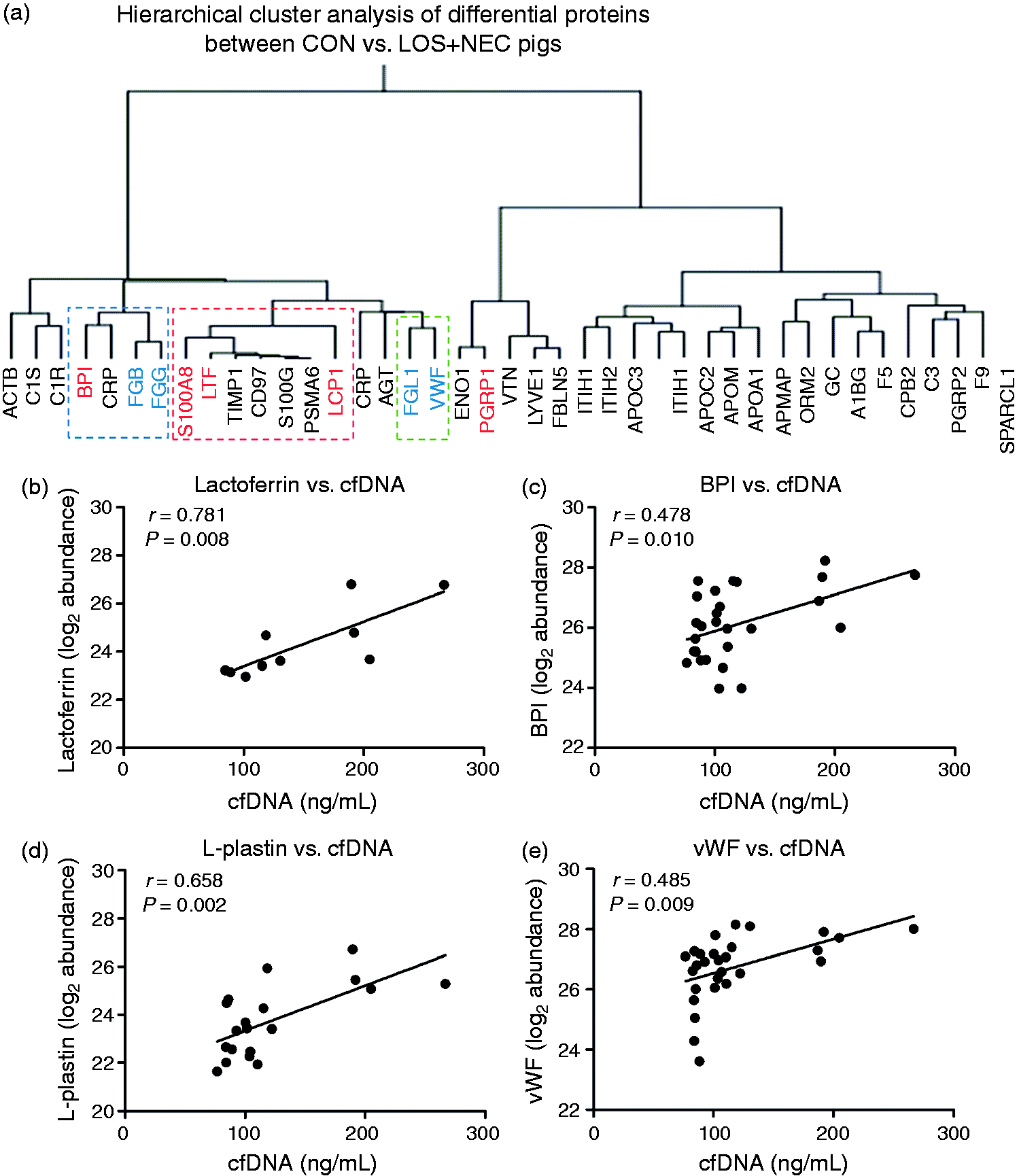
Circulating cfDNA and splenic bacteria in sepsis-induced preterm pigs and neonatal mice
Next, we sought to examine if elevated levels of circulating neutrophil components also occur in neonatal sepsis without the presence of NEC. We verified cfDNA changes in neonatal sepsis using two sepsis models with systemic challenge of S. epidermidis in preterm pigs (PND4) and neonatal mice (PND9 and PND14). Sepsis-induced preterm pigs (SE pigs) showed 1.5-fold higher circulating cfDNA levels than control pigs [130 vs. 85 ng/ml (P < 0.01); Figure 4a]. For both PND9 and PND14 mice, circulating cfDNA showed a strong tendency to be elevated in SE vs. control saline-injected mice (P = 0.05; Figure 4b). When combining data from PND9 and 14 (n = 9–12), SE mice had significantly elevated cfDNA levels [two-fold (P < 0.05); Figure 4b]. All nine control mice were negative for splenic bacteria, while the spleen of 10/12 SE mice contained > 105 CFU/mg (Figure 4c).
Circulating cfDNA and splenic bacteria in preterm pigs and neonatal mice following S. epidermidis challenge. (a) Plasma cfDNA in sepsis-induced preterm pigs (SE pigs) vs. saline controls (SAL pigs). (b, c) Plasma cfDNA and accumulated splenic bacteria in sepsis-induced neonatal mice (SE) vs. saline controls (SAL), including a pooled group with data from mice at both PND9 and 14 that received the same treatments. Values are means ± SEM. *P < 0.05, **P < 0.01 and ***P < 0.001.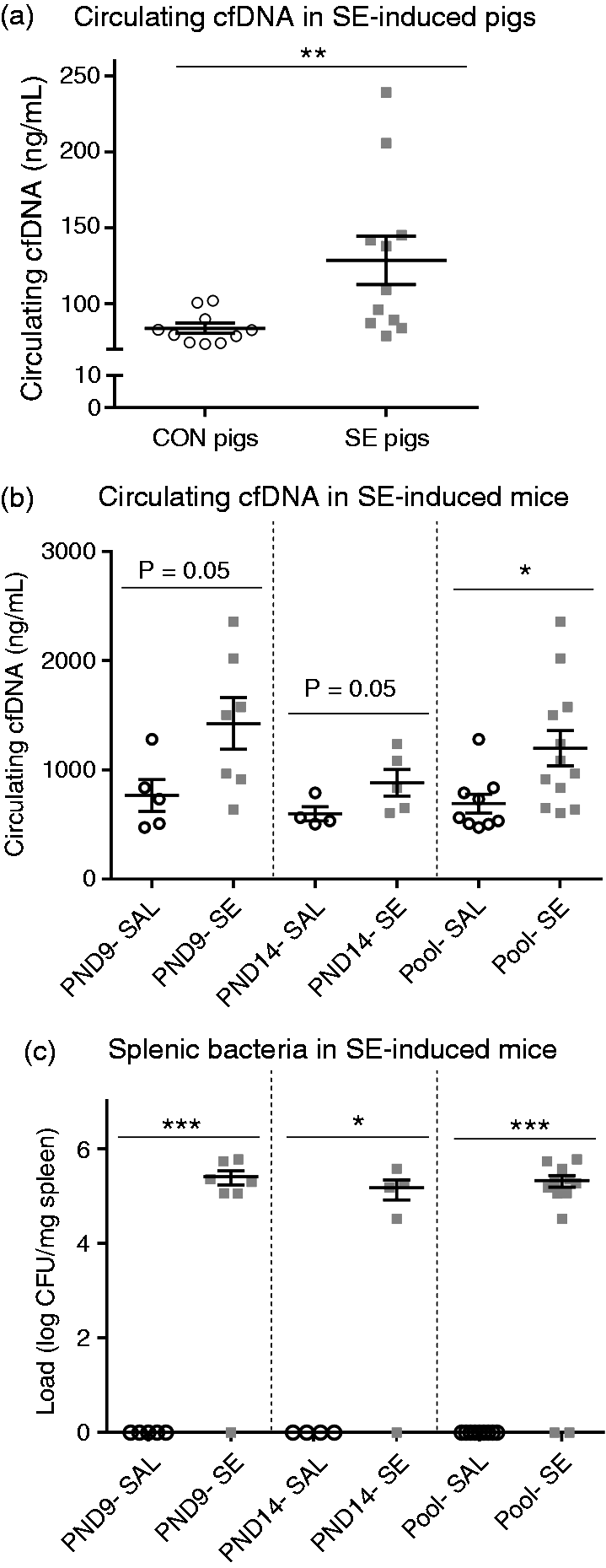
Circulating cfDNA in preterm infants with LOS and/or NEC
Patients demographic for the pilot retrospective case–control study.
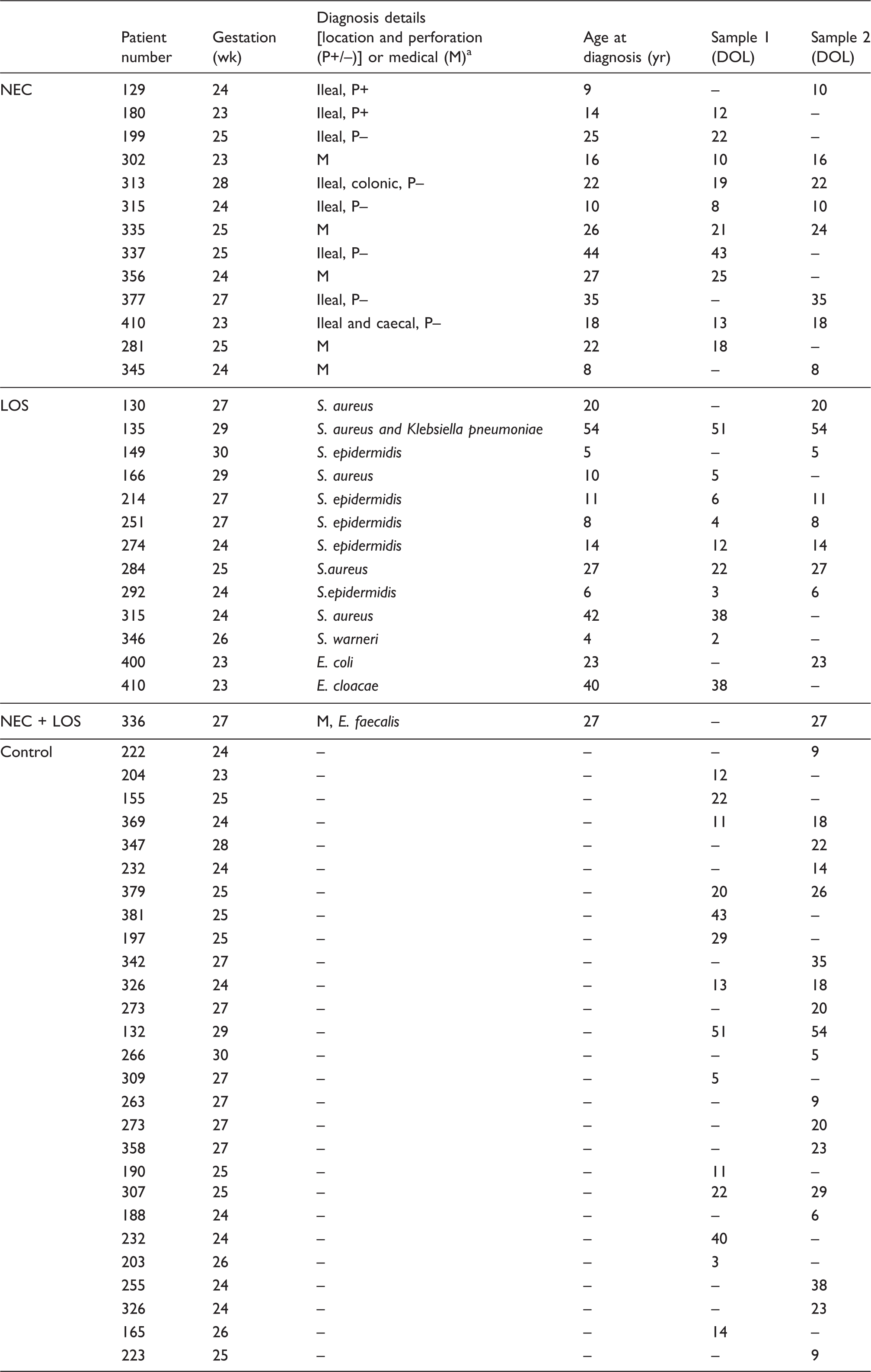
DOL: day of life. aFor NEC: if surgically managed, location and perforation (P+/P-) or medical (M). For LOS: organism.
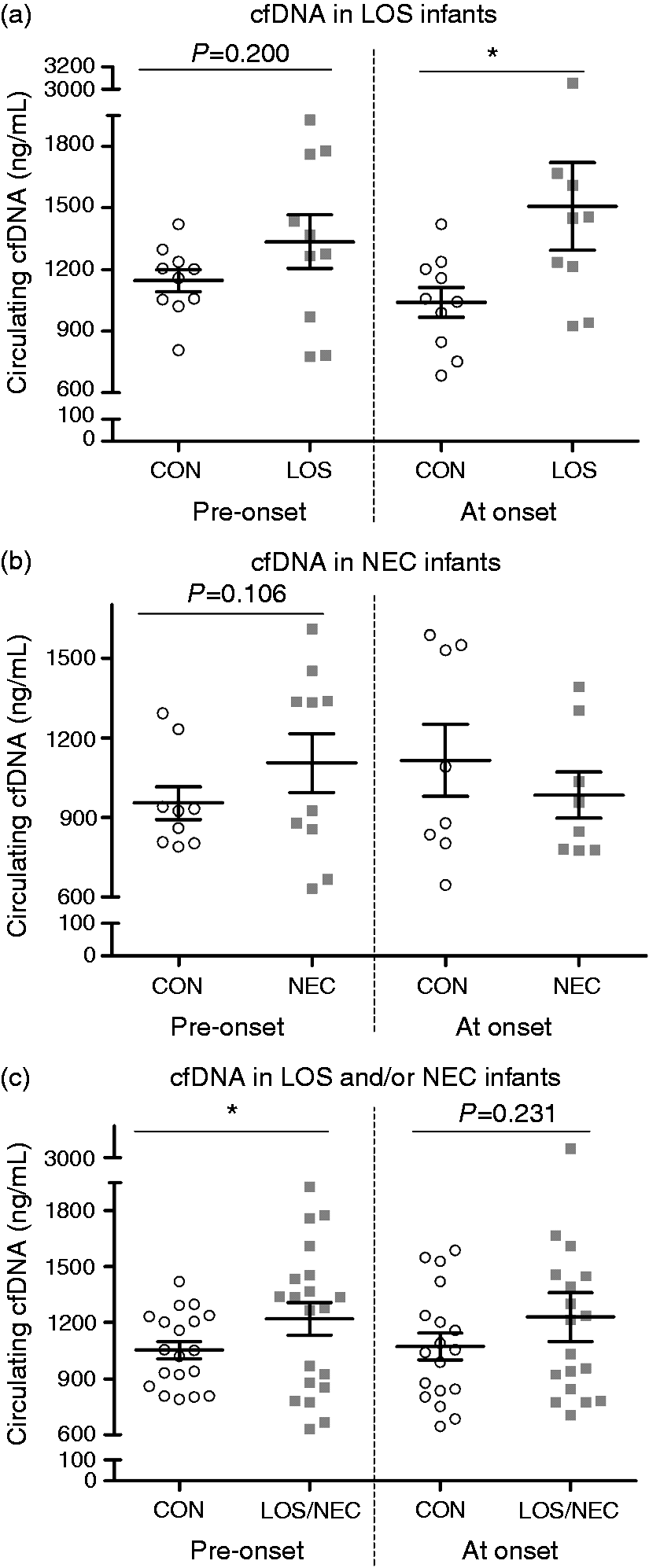
Circulating cfDNA in preterm infants with LOS and/or NEC before and at disease onset. (a) Only LOS cases (n = 13). (b) Only NEC cases (n = 13). (c) Cases with LOS and/or NEC (n = 27). CON in each comparison included control infants with matched gestational age and day of life. Serum samples from pre-onset of LOS or NEC were obtained 1–6 d prior to clinical diagnosis. Values are means ± SEM, *P < 0.05.
Discussion
In preterm infants, LOS and NEC often develop together, although the direct links between LOS and NEC has been difficult to demonstrate. Currently, no single circulating markers for LOS and/or NEC are specific and sensitive enough for clinical use although many candidates have been assessed.4,5 In adults, neutrophil-associated components (including cfDNA and proteins) have been suggested as reliable diagnostic markers for sepsis, 19 but no knowledge is available about these components in the bloodstream in association with neonatal infectious diseases. In the current study, we demonstrated consistent up-regulation of circulating cfDNA and neutrophil-associated proteins at or shortly before the onset of neonatal LOS and/or NEC in three different species (human, pig and mouse). First, using the pig model of spontaneous LOS + NEC, we showed elevated cfDNA levels in LOS + NEC pigs with good sensitivity and AUC in ROC analysis. Importantly, the plasma proteomic analyses demonstrated consistent up-regulation of neutrophil-associated proteins including lactoferrin, S100A8, BPI, L-plastin and PGRP1, as well as proteins associated with neutrophil-platelet interaction during systemic inflammation, including fibrinogen chains and vWF in LOS + NEC pigs. These proteins clustered well in the hierarchical clustering analysis and their abundances were highly correlated with circulating cfDNA levels. We also demonstrated that circulating cfDNA levels were also elevated during LOS (without NEC occurrence) as shown in preterm pigs and neonatal mice challenged with S. epidermidis. Finally, data in the preterm infant cohort revealed a significant elevation in cfDNA levels at LOS diagnosis and a tendency to higher cfDNA levels shortly before NEC diagnosis, compared with controls.
Elevated circulating cfDNA levels in preterm infants, preterm pigs and mice with LOS and/or NEC may reflect the release of DNA derived from neutrophils in the blood in response to the systemic inflammation associated with LOS and NEC in the form of NETs, as suggested by previous studies.20–22 Increased NET formation and accumulation may occur before, or close to the disease onset without proper clearance mechanisms, as shown for traumatized or septic adults.39,40 NET formation in the intestine with released DNA and neutrophil-derived calprotectin has been shown to be associated with NEC, 26 but it was not clear if circulating NETs may play a role. In the present study, elevated circulating cfDNA levels 1–6 d before NEC onset in preterm infants suggest that sub-clinical systemic inflammation at an early stage of NEC may stimulate neutrophils to release DNA and antimicrobial proteins in the circulation, which may add further inflammatory insults that could advance NEC progression.
Among circulating neutrophil-associated proteins (Figure 2c) with up-regulated levels in LOS + NEC pigs, lactoferrin, S100A8, BPI and L-plastin are among the key proteins present in neutrophil granules.13–16 Lactoferrin and S100A8 were proposed as potential single circulating biomarkers of sepsis in infants and children, but their neutrophil derivation was not studied.41,42 S100A8, which was highly up-regulated (48.2-fold) in LOS + NEC pigs in the current study, is a subunit of calprotectin that was shown to be the key protein in the extruded NET structures in the intestine of patients with NEC. 26 Further, BPI and lactoferrin are among potent antimicrobial proteins released from neutrophils that may help to eliminate trapped bacteria. 13 Of note, up-regulated fibrinogen chains and vWF were among the nine circulating proteins involved in platelet activation and blood coagulation that differed between control and LOS + NEC pigs. Fibrinogens and vWF are both involved in the scaffolds that include neutrophil-derived DNA, platelet aggregates and fibrin during systemic inflammation.19,37,38 Our data support the previously described evidence of strong interaction between NET structures and activated platelets during endothelial injury and sepsis in adults. 19 Importantly, cfDNA levels showed strong correlations (r = 0.48–0.78) with neutrophil-associated proteins, especially with lactoferrin, BPI, L-plastin and vWF, suggesting that these components may be co-localized in the circulating NET structure released during LOS and NEC, although no immune staining was performed for confirmation of co-localization. Some other important low-molecular-mass neutrophil proteins (10–15 kDa), such as histones and neutrophil defensins, were not shown in the raw data protein list, likely due the filtration with approximately 10 kDa spin filter in the initial steps of sample processing procedures prior to MS analysis. Still, our proteomic data with multiple neutrophil-associated proteins and proteins involved in the neutrophil-platelet interaction during systemic inflammation in LOS + NEC pigs strongly supported cfDNA data in infants with LOS and/or NEC, and in preterm pigs and neonatal mice with S. epidermidis-induced LOS.
In our cohort of preterm infants, five of 13 LOS cases were detected with S. epidermidis in blood cultures, probably acquired from the environment. 7 In the remaining seven LOS cases, Staphylococcus aureus, E. coli or Enterobacter spp. were detected, and these microbes are frequently found in the stool. 8 Consequently, LOS may partly arise from bacterial translocation across the immature gut, suggesting also that a damaged gut barrier in NEC may precede LOS.32,43 Conversely, LOS may also predispose to NEC, as indicated by the observation of NEC a few wk after LOS diagnosis. 32 Thus, LOS and NEC have partly overlapping pathogenesis and etiology and therefore many biomarkers have been suggested for combined LOS and/or NEC. 2 In addition to the elevated cfDNA levels at LOS diagnosis, we found that circulating cfDNA tended to be elevated shortly before the onset of LOS or NEC, which may show significance with a larger sample size in future studies. Using a Seal Envelope tool for power calculation, we estimated that a sample size of 92 VLBW infants for a future retrospective study is required to obtain a significant elevation in cfDNA levels shortly before NEC onset with a 20% elevation (46 cases, 90% power and 5% significance level) as shown in the current study. To obtain a pre-defined sensitivity of 90% for cfDNA as a diagnostic marker for LOS and NEC (prevalence of 21% and 7% in VLBW infants, 7% of maximum marginal error), a prospective study of 336 infants for LOS or 1008 infants for NEC would be required. 44 Still, our data strongly suggest the involvement of cfDNA and neutrophil-associated proteins in the pathogenesis of LOS and NEC. In infants, LOS was associated with higher cfDNA fold changes than NEC, and we confirmed the association between cfDNA and LOS in both of the used sepsis animal models. It is possible that neonatal bacteremia may initially induce neutrophils to secret their antimicrobial components in the NET structure for bacterial elimination, but impaired clearing mechanisms may aggravate the systemic inflammation that then contributes to LOS and NEC development. Combining neutrophil markers found in this study with other conventional markers (CRP or IL-6) may increase the overall diagnostic sensitivity and specificity.
This is the first study to show elevated levels of multiple circulating neutrophil-associated markers in neonatal LOS and NEC, with consistent data across preterm infants, preterm pigs and neonatal mice. Although our analyses were performed only using frozen plasma/serum without staining of DNA and neutrophil-associated proteins from blood smears to confirm their NET origin, our study suggests that these markers may play a role in the pathogenesis of these neonatal diseases. Reasonable specificity and sensitivity of cfDNA detected in LOS + NEC pigs is encouraging, but whether cfDNA and neutrophil-associated proteins, or a panel of combined markers, may provide good diagnostic markers for LOS and/or NEC, they need to be tested in larger clinical trials. In addition, it is also important to note that the diagnostic cut-off value of cfDNA identified in the pig study was data-driven and this may or may not reflect the cut-off value relevant for clinical applications.
Footnotes
Acknowledgements
Jane Povlsen, Elin Skytte and Kristina Møller are thanked for assistance during pig studies. Obelske Family Foundation, SparNord Foundation and Svend Andersen Foundation are acknowledged for supporting the proteomics analytical platform. Support for the collection of samples from infants was provided by Tiny Lives charity, and the Special Trustees of Newcastle Hospital.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by NEOMUNE grant (grant number 12-132401) from the Danish Strategic Research Council and the STIMMUNE grant from the ARLA Food for Health center. The granting bodies were not involved in the manuscript preparation or any parts of the study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
