Abstract
Immature immunity may predispose preterm neonates to infections and necrotizing enterocolitis (NEC). Intravenous antibiotics are frequently given to prevent and treat sepsis, while oral antibiotics are seldom used. We hypothesized that oral antibiotics promote maturation of systemic immunity and delay gut bacterial colonization and thereby protect preterm neonates against both NEC and bacteremia in the immediate postnatal period. Preterm pigs were given formula and administered saline (CON) or broad-spectrum antibiotics orally (ORA) or systemically (SYS) for 5 d after birth. Temporal changes in blood parameters and bacterial composition in the intestine, blood and immune organs were analyzed. Newborn preterm pigs had few blood neutrophils and a high frequency of progenitor cells. Neutrophils gradually matured after preterm birth with increasing CD14 and decreasing CD172a expressions. Preterm neutrophil and monocyte TLR2 expression and TLR2-mediated blood cytokine responses were low relative to adults. ORA pigs showed enhanced blood neutrophil maturation with reduced cell size and CD172a expression. Only ORA pigs, but not SYS pigs, were protected from a high density of gut Gram-positive bacteria, high gut permeability, Gram-positive bacteremia and NEC. Neonatal oral antibiotics may benefit mucosal and systemic immunity via delayed gut colonization and enhanced blood neutrophil maturation just after preterm birth.
Introduction
Neonatal immunity depends, to a large degree, on innate cells and soluble mediators for protection against environmental antigens and pathogens.1,2 At birth, the innate immune system is immature, 3 and blood neutrophils and monocytes develop during late gestation and after birth, reflected by their increased expression of the surface receptors CD14, MD2, TLR2, and TLR4.1,4–6 In pigs, these markers have similar trends during cell maturation,1,4,5 whereas CD172a expression decreases postnatally. 7 Neutrophils also decrease in size during maturation, 8 and monocytic CD14 expression increases when activated by bacteria. 9
In preterm infants, insufficient time for immune maturation likely plays a key role in their susceptibility to infection, sepsis and necrotizing enterocolitis (NEC).10–12 Compared with term, preterm infants have markedly fewer leukocytes, and lower expression of CD14, MD2, TLR2 and TLR4 in blood neutrophils and monocytes.1,13 NEC and sepsis have been shown to be associated with neutropenia, monocytopenia, thrombocytopenia and/or suppression of erythropoiesis,14–17 although it remains unclear whether systemic immunity is functionally connected with these conditions.
NEC pathogenesis is incompletely understood, although premature birth, excessive bacterial colonization and aggressive enteral feeding are important predisposing factors. 12 Breast milk is believed to protect against NEC, relative to infant formula,18,19 but is often absent during the first week after preterm birth. Modulation of initial gut colonization by anti-, pre- and probiotics may reduce NEC risk but the optimal efficacy, time and dose of such treatments are unclear.20–25 Although antibiotics have been reported to decrease NEC incidence in several small studies, 25 there is a well-justified reluctance to use prophylactic antibiotics owing to insufficient mechanistic evidence for benefits and risk of antibiotic resistance. In addition, it is important that prolonged postnatal administration of systemic antibiotics is associated with altered gut bacterial colonization and increased risk of NEC in preterm infants. 26
The preterm pig is the only described animal model that incorporates the normal clinical signs of prematurity (e.g. respiratory defects, hypothermia, dysmetabolism, elevated infection risk) and spontaneously develops NEC following feeding with infant formula during the postnatal period. 27 Preterm pigs also allow parenteral and enteral feeding protocols, repeated blood sampling and detailed sample collection from the gut lumen and mucosa to monitor parameters related to infection and NEC. 28 We have previously shown that broad-spectrum antibiotics (ampicillin, gentamicin and metronidazole) provided simultaneously via the oral and systemic routes for 5 d completely prevented NEC in formula-fed preterm pigs. 24 In this study, it was not clear if systemic immunity was affected or if both oral and systemic administration was required for NEC protection. Without antibiotics and standard clinical care, gut colonization rapidly progresses to reach a colonic bacterial density of 1010–1011 bacteria after birth. 24 Preterm newborns are susceptible to translocation of bacteria, or bacterial products, from the gut and other epithelial surfaces into the blood stream.1,2 Conversely, oral antibiotics may delay neonatal colonization of the gut and thereby maintain the immature intestine in a state that more resembles the bacteria-deficient in utero condition. In turn, this may facilitate immune maturation during the critical period just after preterm birth.
We hypothesized that systemic immunity is immature in newborn preterm pigs with functional deficits in specific leukocyte populations and that leukocyte maturation occurs during the first few days after preterm birth when enteral feeding volumes are still low and NEC symptoms are absent. We speculate that only oral, not systemic, administration of antibiotics enhances neonatal blood leukocyte maturation, delays gut colonization and thereby protects against bacteremia and formula-induced NEC.
Materials and methods
Experimental animal procedures
The animal protocol was approved by the Danish National Committee of Animal Experimentation. Cesarean-delivered preterm pigs (105–106 d of gestation, 90–92%) from three sows received parenteral nutrition (4–6 ml/kg/h) via umbilical catheters and minimal enteral nutrition (3 ml/kg/6 h) with formula via orogastric tubes for the first 2 d, followed by 2 d with total enteral nutrition (15 ml/kg/3 h) with formula. The composition of the formula was specifically designed for the piglets’ need. Pigs were randomized into three groups: those receiving saline (CON, n = 17); those receiving broad-spectrum antibiotics via the oral route (ORA, n = 18); or those receiving antibiotics via the systemic route via umbilical catheters (SYS, n = 17). Antibiotics (ampicillin: 30 mg/kg three times daily; gentamicin: 2.5 mg/kg twice daily; metronidazole: 10 mg/kg three times daily) were given in identical doses to ORA and SYS pigs based on the commonly used i.v. antibiotic regimes used for pediatric patients with suspected NEC or sepsis. On d 5, pigs were euthanized and five gastrointestinal regions (stomach, proximal, middle and distal small intestine, and colon) were macroscopically evaluated for pathological changes by a NEC scoring system (score 1–6), indicative of the severity of inflammation and necrosis. An animal was designated as NEC-positive when a minimum score of 3 in at least one region was observed. Further details of pig handling, housing, feeding, NEC scoring and clinical assessment were described previously.24,29 Other specific details of the protocol are described in the Supplementary Material.
Blood was collected on d 1 (cord), 2 and 3 (umbilical catheter), and at euthanasia (d 5, cardiac puncture). For bacterial characterization, samples of blood, spleen and femur-derived bone marrow were aseptically collected at euthanasia. Furthermore, cord blood from four Cesarean-delivered term pigs (116–117 d of gestational age) and blood from 11 healthy adult pigs (∼1 yr old) were collected for comparison with blood from preterm pigs.
Blood phenotyping
Blood cell counting was analyzed by an automatic cell counter (Advia 2120i Hematology System; Siemens, Munich, Germany). For further phenotyping, whole blood (100 µl) was used and erythrocytes were lysed by 1 × BD FACS Lysing solution (BD Biosciences, Franklin Lakes, NJ, USA). Lysed blood was washed by PBS supplemented with FBS and NaN3. Fc receptors on leukocytes were then blocked by porcine serum [10 min, room temperature (20℃), in the dark] prior to leukocyte staining with the following Abs: CD14–FITC (MIL2; AbD Serotec, Kidlington, UK), CD172a-PE (74-22-15A; BD Biosciences), TLR2–AF647 (CD282, 11G7; AbD Serotec). IgG2b-FITC (AbD Serotec) and IgG2b-PE (eBioscience, San Diego, CA, USA) were used as isotype controls. Cells were incubated with surface Abs (15 min, room temperature, in the dark), and washed twice prior to fixation in 4% formalin. Cells were analyzed on a BD FACS Canto II flow cytometer (BD Biosciences). Staining profiles were analyzed using FlowJo (Tree Star, Inc., Ashland, OR, USA).
Gating strategy was performed as previously described in pig leukocytes, 30 and further described in the Supplementary Material and Supplementary Figures S1 and S2. Briefly, based on CD172a, CD14 and side scatter (SSC), neutrophils were CD172a+CD14+SSChigh, and monocytes were CD172a+CD14+SSCmedium. Small cells (low forward scatter; FSC) with variable granularity were judged to be progenitor cells. TLR2 expression was low in both monocytes and neutrophils in blood from preterm pigs.
Blood stimulation with TLR2 and TLR4 agonists
At Cesarean section in each of three separate litters, a pooled cord blood sample from all preterm pigs and sow blood were collected. Blood was stimulated with TLR agonists (Invivogen, San Diego, CA, USA): freeze-dried heat-killed Listeria monocytogenes (HKLM, TLR2 agonist, 102–106 cells/ml), Pam3CSK4 (bacterial lipopeptide, TLR1/2 agonist, 10−4–1 ng/ml) and LPS (TLR4 agonist, 10−2–102 ng/ml). After stimulation at 37oC, 5% CO2 for 5 h, blood was diluted fivefold with RPMI 1640 medium (Life Technologies, Carlsbad, CA, USA), centrifuged at 2500 g at 4oC for 10 min. Supernatants were analyzed for IL-6 and TNF-α by ELISA (R&D Systems, Abingdon, UK).
TLR2 gene expression in whole blood
RNA from blood samples was extracted using MagMAX 96 total RNA Isolation Kit (Life Technologies) and converted into cDNA as previously described. 31 Quantitative real-time PCR (qPCR) was performed using TaqMan Universal Master Mix II (Life Technologies) to quantify TLR2 gene expression in blood on d 1 and 3. Primers of TLR2 and the housekeeping gene (β2 microglobulin; B2M) were designed by PrimerExpress sofware (Applied Biosystems, Foster City, CA, USA) and synthesized by Applied Biosystems: TLR2 (F: TCGAAAAGAGCCAGAAAACCAT and R: CTTGCACCACTCGCTCTTCAC), and B2M (F: ATCCGCCCCAGATTGAAATT and R: TGCTCCGCGTTCATCTTCT). Relative gene expression of TLR2 was calculated by normalization to levels of the reference gene B2M.
Bacterial adherence to intestinal mucosa by fluorescence in situ hybridization
Bacterial abundance on three intestinal regions (proximal, middle and distal) was evaluated by fluorescence in situ hybridization (FISH) on a paraffin-embedded cross section (3 µm) using Alexa fluor 555-label oligonucleotide probes (Eurofins MWG Operon, Ebersberg, Germany) targeting the 16S rRNA of general bacteria (5′-GCTGCCTCCCGTAGGAGT-3′), 32 and visualized by an fluorescence microscope. The tissue sections were scored from 0 to 6 based on the fluorescence signal, where 0 = no bacteria, 1 = very few bacteria, 2 = few spread bacteria, 3 = bacteria spread in the whole tissue section, 4 = minor colonies spread in the whole tissue section, 5 = numerous larger colonies, and 6 = widespread overgrowth with large colonies. Values are means ± SEM across intestinal regions.
Bacterial identification and quantification
Blood, homogenized spleen and bone marrow were cultured on blood agar (5% bovine blood supplemented-Blood Agar Base CM0055; Oxoid, Basingstoke, UK) at 37oC for 24 h. After enumeration, different bacterial colonies were counted, isolated, transferred and incubated in Brain–Heart Infusion Broth (Difco, Brøndby, Denmark) at 37oC for 24 h. Bacterial identification from cultured pure isolates were performed by a MALDI-TOF Mass spectrometer (Vitek MS, Biomérieux, France). 33
Total DNA from colon contents was extracted and total bacterial load was quantified by qPCR (see Supplementary Material). Colonic prokaryotic microbiota composition was determined by tag-encoded 16S rRNA gene MiSeq-based (Illumina, San Diego, CA, USA) high-throughput amplicon sequencing.34,35
Intestinal permeability
Three h prior to euthanasia, pigs received an oral bolus of 5% lactulose and 5% mannitol (15 ml/kg). The ratio of urinary lactulose and mannitol levels at euthanasia was determined as the indicator of intestinal permeability, 24 as mannitol and lactulose are markers of transcellular absorption and paracellular permeability, respectively.
Statistics
Cell count comparisons among newborn preterm, term and adult pigs, NEC and FISH scores, and bacterial load were analyzed by the Kruskal–Wallis test (JMP 10; SAS Institute, Cary, NC, USA). NEC incidence was analyzed by Fisher’s exact test, and cytokines from blood assays were tested in a linear mixed model using pig (preterm or adult) and agonist concentration as fixed factors (JMP). Other parameters were fitted to a linear mixed model with treatment, day and litter as fixed factors, and gender and pig code as random factors using the ‘lmer’ function (R; www.R-project.org). 36 The significance of treatment and day was tested by the Tukey test using an ANOVA function. Differences among groups on each day were tested using the ‘glht’ function in the package ‘multcomp’. 37
Results
Oral antibiotics prevent NEC
No pigs from the ORA group developed NEC lesions (incidence 0%, 0/16), compared with 63% (10/16) in CON and 59% (10/17) in SYS pigs (P < 0.001; Figure 1A). No difference in NEC incidence was observed between CON and SYS pigs. The average NEC severity score of five gastrointestinal regions was lower in ORA than SYS and CON pigs (P < 0.01; Figure 1B). NEC lesions were relatively mild in both CON and SYS pigs, as the lesions were present mainly in the colon with limited differences in the small intestine (Figure 1C, D).
Pathological evaluation. (a) NEC incidence, (b) average NEC severity score throughout the gastrointestinal tract, and average NEC score in the (c) colon and (d) small intestine (n = 12–17 per group). Values (means ± SEM) in the same region not sharing common letters are significantly different (P < 0.05).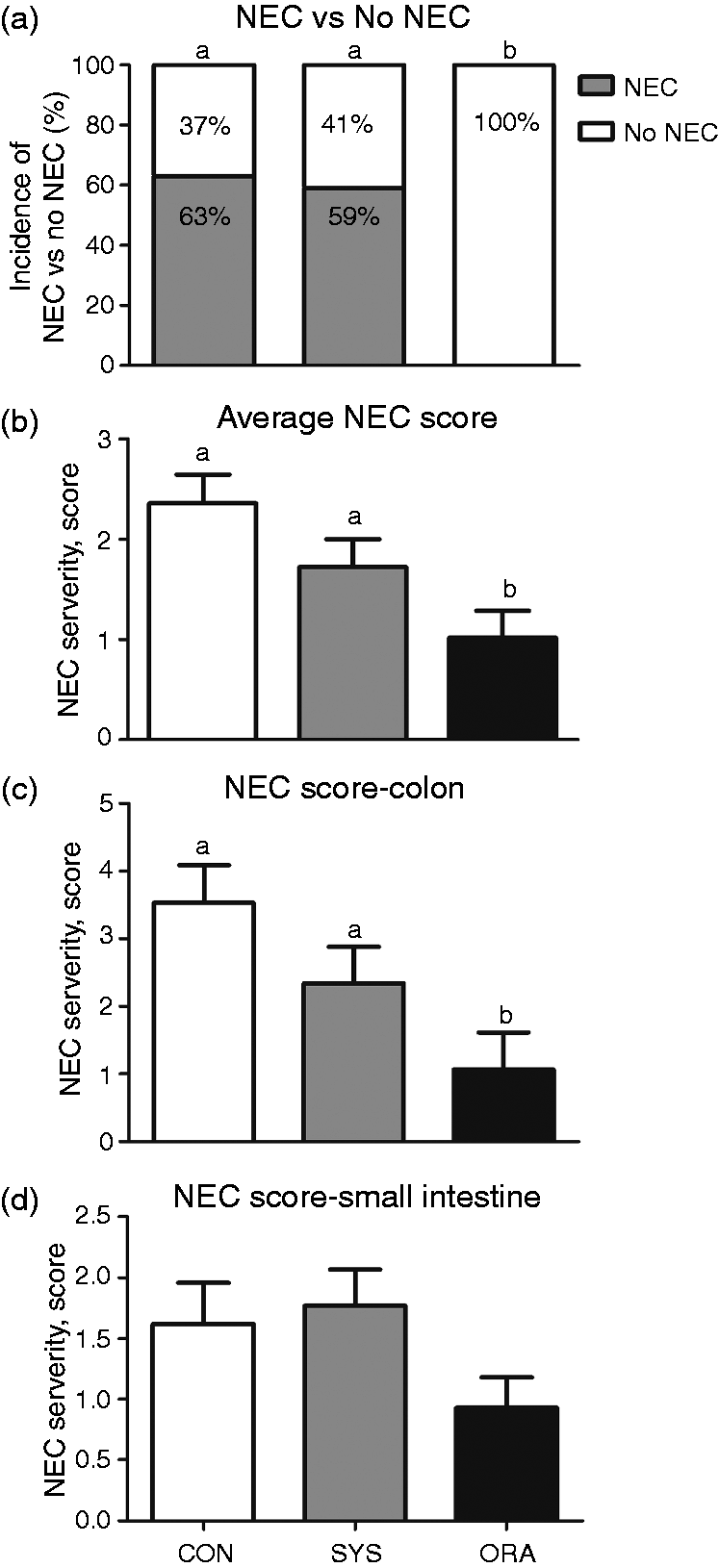
Immature blood leukocytes in newborn preterm pigs
Hematological profile of newborn preterm (n = 51), newborn term pigs (n = 4) and healthy adult pigs (n = 11).

Means ± SEM. Values in the same row not sharing the same letters are significantly different (P < 0.05)
Preterm blood neutrophil and monocyte development during the first 3 d of life
Leukocyte development was characterized from d 1 to d3 in preterm pigs (all treatment groups were combined owing to similar developmental trends), prior to the increases in enteral feeding volume and before any clinical signs of NEC. 38 Total leukocyte and neutrophil counts peaked on d 2 and returned to basal levels on d 3, whereas other cell counts remained constant during d 1–3 (Supplementary Figure S4).
The expression of CD172a, CD14 and TLR2 and cell size (reflected by FSC) were used as markers of maturation and development of neutrophils and monocytes (Figure 2A–C). CD14 intensity and frequency of CD14high cells increased, and frequency of CD14low cells decreased (P < 0.001) in both neutrophils and monocytes on d 2–3, relative to d 1. In parallel, neutrophil and monocyte CD172a expression decreased over time (P < 0.001), similar to their postnatal development in term pigs.
7
Over time, neutrophil size decreased, whereas monocyte granularity increased (P < 0.001). These changes indicate a gradual maturation and development of blood neutrophils and monocytes in preterm pigs. In contrast, TLR2 intensity was low and did not change in both cell types during d 1–3, suggesting impaired development of TLR2 responses. TLR2 presence was confirmed by qPCR analysis of blood RNA. Of note, the frequency of progenitor cells decreased over time (Figure 2D).
Development of leukocyte subsets during the first 3 d of life across all three groups of preterm pigs. Developmental and maturational status of neutrophils and monocytes measured by (a, b) expression of markers CD172a, CD14 and TLR2, (c) cell size and granularity. (d) Changes in frequency of blood progenitor cells. Values (means ± SEM, n = 55) across d 1–3 in the same blood cell subsets not sharing the same letters are significantly different (P < 0.05).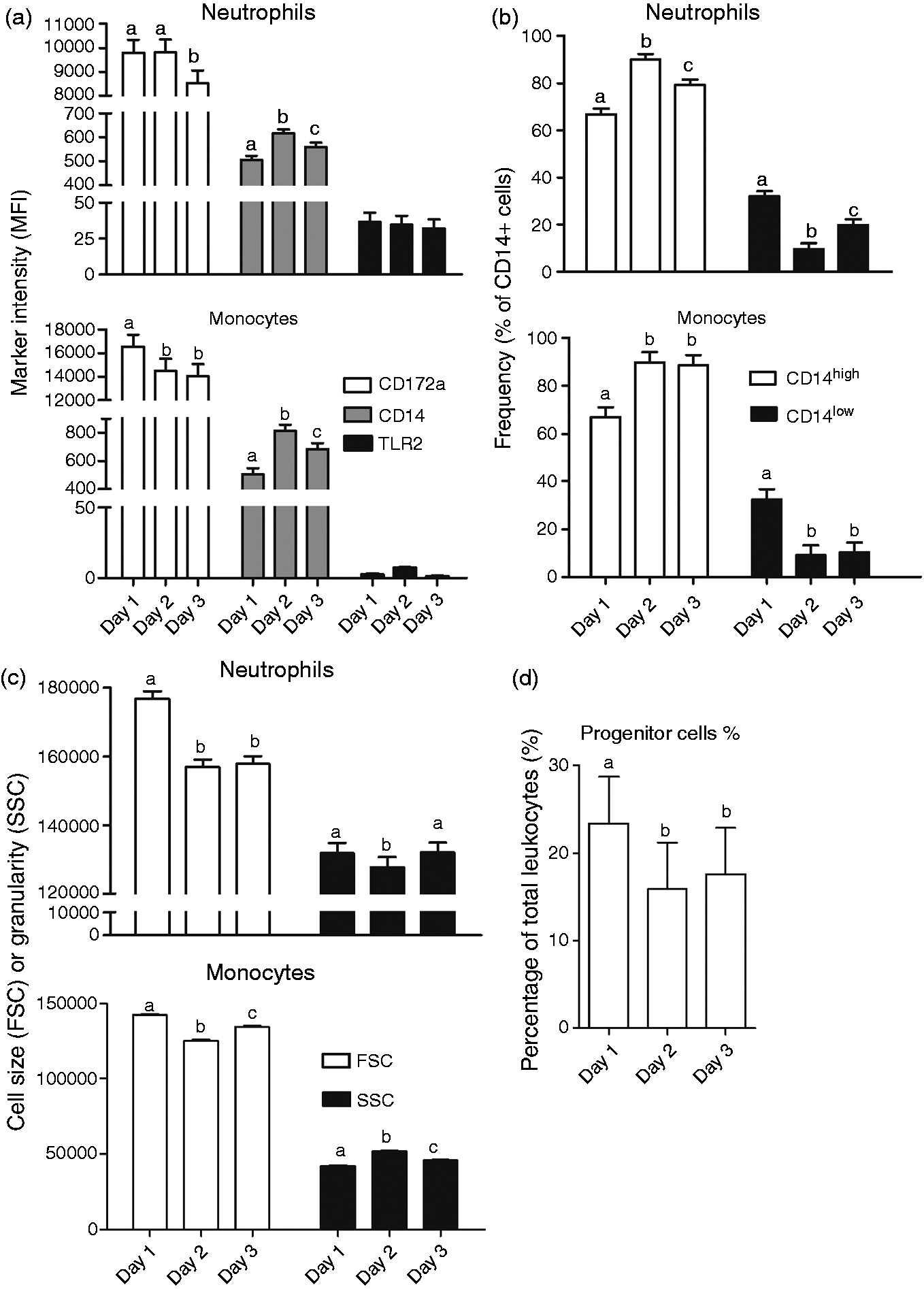
Impaired TLR2-mediated cytokine production from blood leukocytes in preterm pigs
To assess the functional correlates of TLR2 expression, we measured TLR2 agonist-induced pro-inflammatory cytokine production in whole blood. TLR2-mediated TNF-α and IL-6 production (via Pam3CSK4 and HKLM cells) were markedly lower in newborn preterm vs. adult pig blood (P < 0.01; Figure 3A–D). Preterm blood leukocytes did not produce IL-6 at any of the TLR2 agonist concentrations, in contrast to the 200–500 pg/ml IL-6 found in adult blood. TLR2 agonist-induced TNF-α levels in preterm blood were either negligible or less than half of that in adult blood. These findings suggest a negligible expression and poor function of surface TLR2 in innate blood leukocytes of preterm pigs. In contrast, TLR4-mediated TNF-α and IL-6 production in newborn preterm blood was more similar to that in adult blood (Figure 3E, F).
Newborn preterm whole blood demonstrated impaired TLR2-mediated cytokine production. Pro-inflammatory cytokine responses (TNF-α and IL-6) following whole blood stimulation with (a, b) TLR2 agonist (HKLM cells), (c, d) TLR1/2 agonist (Pam3CSK4) and (E, F) TLR4 agonist (LPS) at increasing concentrations in preterm pigs at birth (pooled blood from 20–22 pigs each litter, n = 3 from three litters) compared with adult pigs (n = 3 sows). Values represent means ± SEM, and curves with increasing concentrations of agonists not sharing the same letters are significantly different (P < 0.01).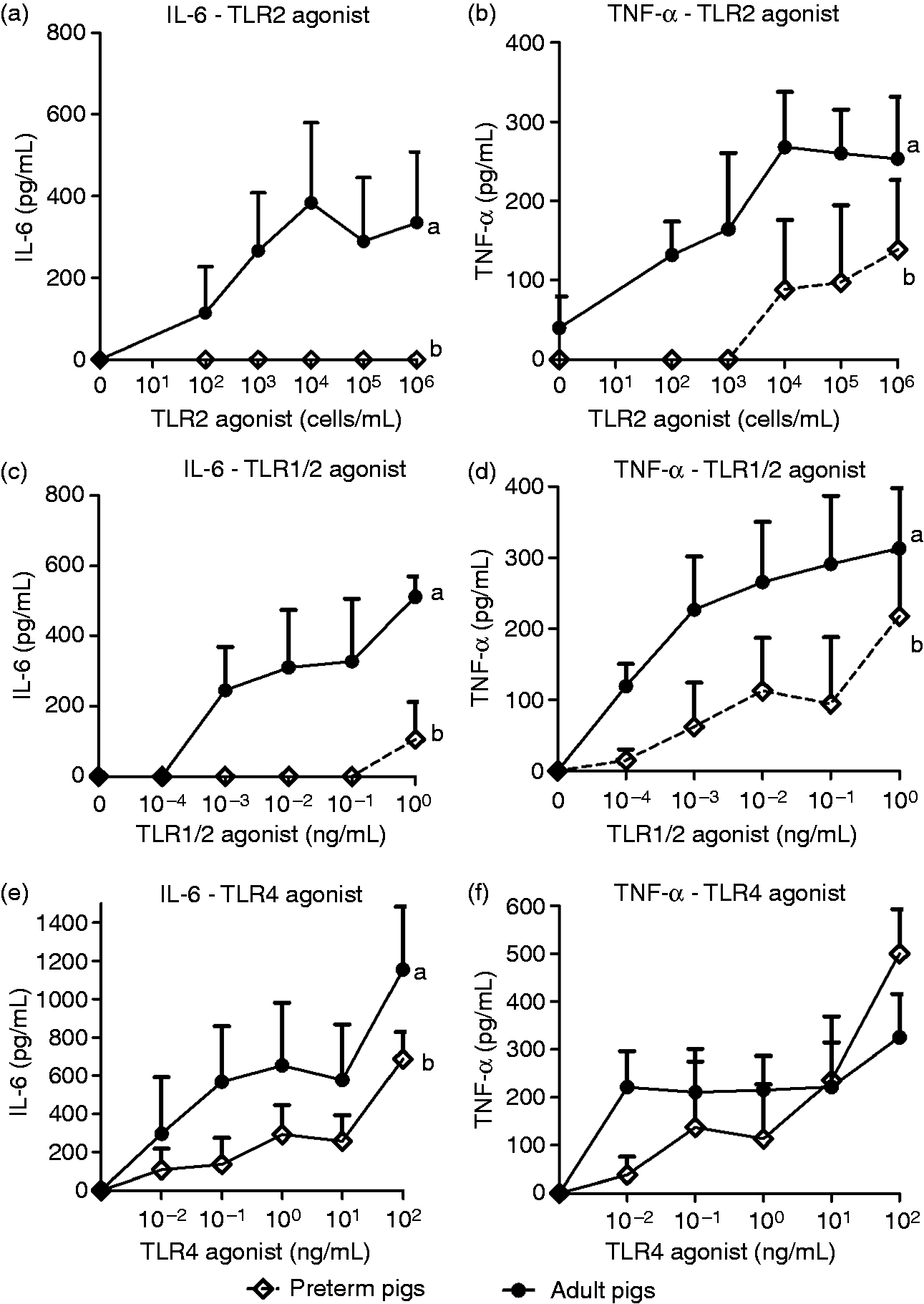
Oral antibiotics enhance neutrophil maturation
ORA pigs showed more advanced neutrophil maturation, shown by lower cell size and CD172a expression on d 2 and 3, compared with CON and SYS pigs (P < 0.05; Figure 4A). In parallel, progenitor cells showed a strong trend for a lower percentage in ORA pigs on d 3 (P = 0.05; Figure 4B). These findings suggest that oral administration of antibiotics accelerates blood neutrophil maturation. In contrast, monocytes in CON pigs were more granular (P < 0.05) and tended to have higher CD14 expression than ORA and SYS pigs on d 3 (P = 0.08; Figure 4B), indicating more inflammatory monocytes in CON pigs.
Effects of antibiotics on the development of blood leukocytes during the first 3 d of life in preterm pigs (CON, n = 17; ORA, n = 18; SYS, n = 17). (a) neutrophils; (b) monocytes and progenitor cells. Values (means ± SEM) from each day not sharing common letters are significantly different (P < 0.05).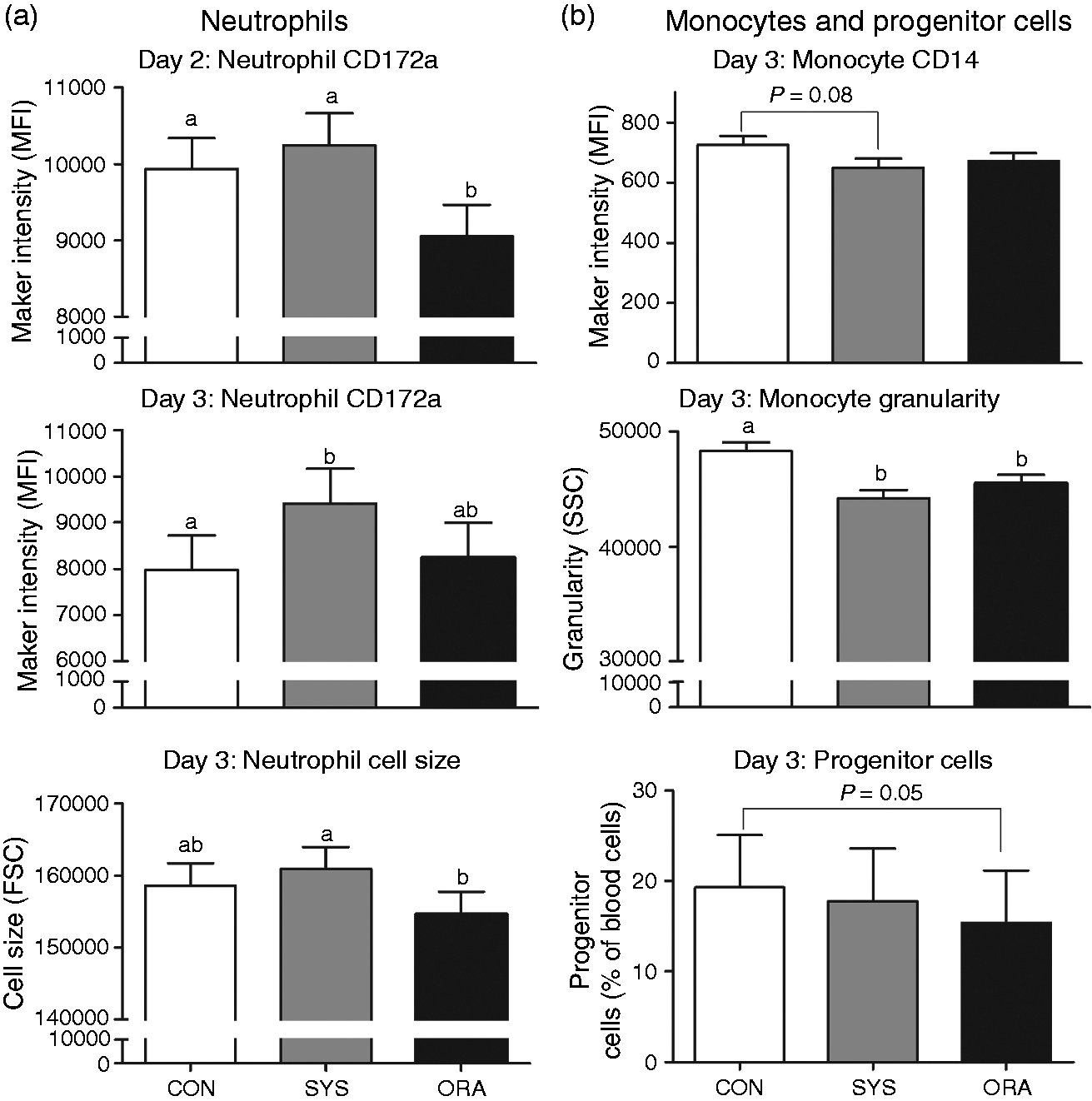
Effects of antibiotics and NEC on blood leukocytes at euthanasia on d 5
At euthanasia, ORA pigs had lower monocyte number and granularity relative to CON pigs (P < 0.05; Supplementary Figure S5). Additionally, healthy pigs without NEC on d 5 also had greater total leukocyte and lymphocyte counts, and lower monocyte granularity than pigs with NEC (P < 0.05; Supplementary Figure S5).
Oral antibiotics reduce bacterial adherence, gut colonization, gut permeability and bacteremia
The FISH scores in the small intestine, reflecting the relative abundance of bacteria that adhere to the intestinal mucosa, were highest (2.4 ± 0.3) in CON, followed by SYS (1.9 ± 0.3) and lowest in ORA pigs (1.1 ± 0.3; P < 0.01). Intestinal permeability shown by the urinary lactulose/mannitol ratio did not differ in pairwise group comparisons but was significantly reduced in ORA pigs, relative to the combined value from CON and SYS pigs (0.027 ± 0.022 vs. 0.094 ± 0.016; P < 0.05).
ORA pigs had 2 log (100-times) lower bacterial load in colon contents than CON and SYS pigs (P < 0.05 and 0.01; Figure 5A). In blood, bacteria were not detected in ORA pigs and were most abundant in CON pigs. In bone marrow, ORA pigs showed markedly reduced bacterial load relative to the other two groups (Figure 5B; P < 0.01). There was no significant difference in bacterial load among the three groups in the spleen although numerically, values remained lowest in the ORA group.
Microbiology. (a, b) Total bacterial load and (c, d) predominant genera (c-d) in the gut (n = 7–13/group), and in blood, spleen and bone marrow (n = 16–17/group). Values (means ± SEM) for bacterial load in gut, blood, bone marrow or spleen not sharing common letters are significantly different (P < 0.05).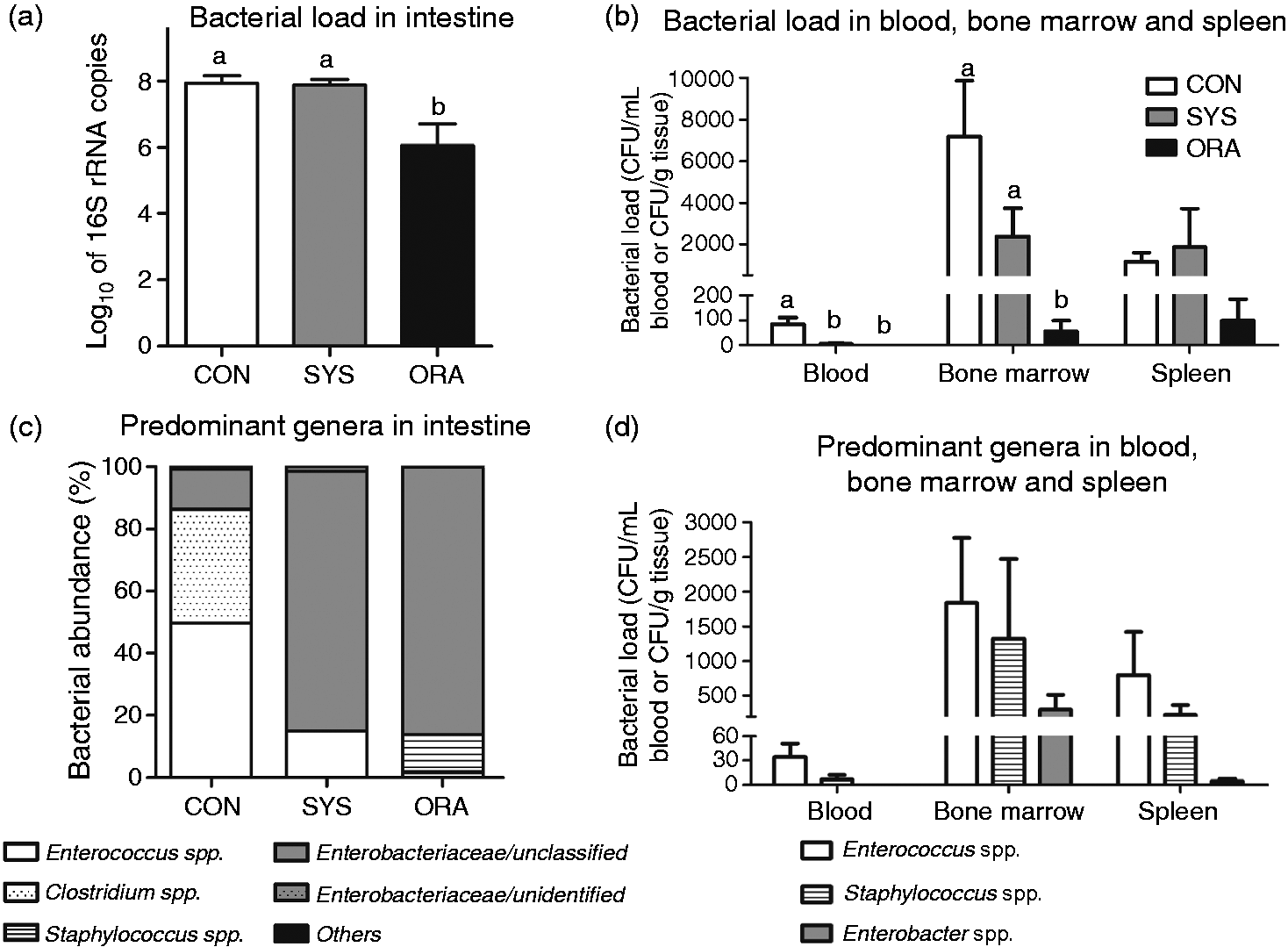
Gram-positive bacteria (Enterococcus, Clostridium and Staphylococcus spp.) were predominant in the colon content of CON pigs, whereas most ORA pigs were dominated by Gram-negative bacteria (Enterobacteriaceae; Figure 5C). Principal component analysis and analysis of similarities (ANOSIM) from sequencing data also showed that the colonic microbiota in CON and ORA pigs differed significantly (R = 0.38, P < 0.05). For those pigs with detected bacteria in blood and organs, Enterococcus was the predominant genus in blood, whereas both Enterococcus and Staphylococcus were predominant groups in bone marrow and spleen (Figure 5D). Enterobacter was not found in blood, and only present in spleen and bone marrow in a few pigs. Together, these data suggest that oral antibiotics efficiently eliminated most gut Gram-positive bacteria and thereby reduced translocation and bacteremia.
Discussion
Colonization of the gut, skin and lungs with billions of bacteria during the first days after birth is a challenging event for newborns, especially preterm neonates that show immature mucosal and systemic immune systems. Consequently, a large proportion of preterm infants (20–40%) experience one or more episodes of infection and sepsis during the first few weeks after birth. 1 Systemic entry of bacteria often arises from the indwelling catheters that are required to allow intensive medical and nutritional treatments. 39 Systemic treatment with broad-spectrum antibiotics is common practice for infants born from mothers with prenatal infections, 40 or for infants with suspected NEC. 41 Gut bacterial overgrowth, coupled with low species diversity and high intestinal permeability, are associated with NEC in both infants and animal models, 28 but it remains unclear if bacterial translocation from the immature gut plays an important role in sepsis and impaired immunity. We used preterm pigs as a model for preterm infants as they are highly sensitive to NEC and sepsis and show many of the normal signs of physiological immaturity. 27 We described the development of blood leukocyte subsets during the first days of life and documented that only oral, and not systemic, antibiotics, delayed gut colonization, reduced gut permeability and bacteremia, and was associated with enhanced maturation of systemic innate immunity. Our results help to understand the important role of the gut microbiota for the high sensitivity to sepsis and NEC within the first few weeks after preterm birth.
First, we showed that systemic immunity was immature in newborn preterm pigs as shown by their reduced number of blood leukocytes and higher number of progenitor cells, compared with newborn term or adult pigs. This is in line with the common knowledge that quantitative and qualitative leukocyte deficiencies predispose preterm infants to infections, sepsis,42,43 and potentially also to NEC.14–16 Regardless, the possible direct link between NEC and systemic immunity is not clear, neither in animal model studies nor in studies of human preterm infants.
In human and porcine newborns, severe NEC lesions are rare before enteral feeding is approaching full volumes (>120 ml/kg/d) and NEC development is highly diet-dependent, with infant formula being associated with the highest NEC sensitivity. Consequently, our observations on blood cell maturation during the first 3 d after birth with minimal enteral nutrition represent the development during the immediate postnatal period in the absence of NEC processes. Consistent with the known ontogeny of systemic immunity in both humans and pigs,1,4,5,7 preterm pigs demonstrated increasing postnatal maturation during d 1–3, with decreasing cell size and CD172a expression, and increasing CD14 expression in blood neutrophils and monocytes. However, compared with CON and SYS pigs, ORA pigs showed enhanced neutrophil maturation, although these effects may be indirectly induced via changes in the gut microbiota. ORA pigs also showed limited bacterial adherence to the intestinal mucosa (low FISH score) and reduced bacteremia (with no bacteria detected in blood), probably because of reduced gut bacterial density and permeability in response to oral antibiotics. These combined effects of oral antibiotics may benefit NEC resistance, as well as the systemic defense against infections after preterm birth. In contrast, less mature neutrophils, more inflammatory monocytes (higher CD14 expression) and extensive translocation may be the systemic consequences of a more normal bacterial colonization in the gut of CON pigs. Parameters in SYS were intermediate between those of ORA and CON, probably indicating both a systemic and a marginal luminal effect of systemic antibiotics to the SYS pigs.
The gut microbiota was dominated by Enterococcus and Clostridium (CON pigs) or Enterobacteriacae (SYS and ORA pigs), but not by Staphylococcus. This suggests that a small proportion of the Staphylococcus present in the blood and immune organs may be derived from catheter-associated bacteremia, rather than bacterial translocation across the leaky gut, similar to the frequent observation of systemic infections with Staphylococcus in human preterm infants. 39 Conversely, the high Enterococcus load in blood, spleen and bone marrow of CON and SYS pigs are likely gut-derived bacteria translocated from the gut. We speculate that absence of bacteremia in ORA pigs may be explained by the reduced bacterial translocation from the gut due to delayed gut colonization and low permeability. The resulting blood neutrophil maturation may follow from these effects and future studies using germ-free, NEC-resistant preterm pigs would be important to confirm these underlying mechanisms.
CD14, MD2 and TLR4 mediate detection of LPS from Gram-negative bacteria, whereas CD14 and TLR2 mediate signaling by bacterial lipoproteins from Gram-positive bacteria. Expression of these receptors on blood neutrophils and monocytes increases with gestational age from very preterm to term infants.1,13,44 Bacteremia caused by Gram-positive bacteria is also associated with increased mononuclear cell CD14 and TLR2 expression.9,45 Consistent with these observations, we observed in the current study increased CD14 intensity in preterm blood monocytes and neutrophils with advancing age. However, TLR2 intensity was low and did not change over time on neutrophils and monocytes from any of the three groups, even though Gram-positive bacteria dominated in the gut lumen of CON pigs and also the blood in CON and SYS pigs. The low TLR2 expression may explain the impaired TLR2-mediated cytokine production in preterm blood. In the preterm pig intestine, TLR2 gene expression, but not TLR4, increases following enteral feeding.46,47 This is in line with our data showing a predominance of Gram-positive bacteria in the gut of CON pigs, as this may trigger the development of intestinal TLR2 expression. This does not appear to induce a parallel increase in blood leukocyte TLR2 expression and suggest a deficient systemic immunity to Gram-positive bacteria in preterm pigs during the first few days of life. In contrast, TLR4-mediated preterm blood cytokine responses were more pronounced and Gram-negative bacteria were negligible in blood of all three groups of pigs. This suggests that a relatively mature blood leukocyte response to Gram-negative bacteria reduces bacteremia caused by these pathogens in newborn preterm pigs. ORA pigs showed no evidence of bacteremia, implying that oral broad-spectrum antibiotics may prevent Gram-positive bacteremia in preterm neonates, partly via a reduced gut permeability, bacterial colonization and translocation.
With regard to NEC prevention, oral was superior to systemic antibiotic treatment, probably because only oral treatment depressed the rapid gut bacterial colonization and reduced gut permeability. Conversely, a high gut bacterial load, combined with an immature systemic innate immune response against Gram-positive bacteria, may predispose to bacteremia and NEC. More inflammatory monocytes with high CD14 expression on d 3 and increased monocyte counts on d 5, as shown in CON pigs, may also play a role in NEC pathogenesis. This is in line with the observed higher monocyte CD14 expression and monocyte granularity in NEC pigs. When the bacterial translocation occurs in SYS pigs, systemic antibiotics may help to clear the blood from intact bacteria but not from the products released from dead bacteria. These may then contribute to the systemic inflammation often associated with NEC.
The suggested mechanisms based on our study of neonatal antibiotics to preterm pigs are summarized in Figure 6. We demonstrate that oral administration of antibiotics during the first few days after preterm birth enhances maturation of blood neutrophils, diminishes gut colonization and permeability, and prevents bacterial translocation. These combined effects may appear to protect formula-fed preterm neonates against bacteremia and NEC-like lesions in the period immediately after birth. In contrast, control formula-fed neonates are associated with a higher gut bacterial load, increased blood monocyte CD14 expression, increased bacterial translocation and inflammation, damage of epithelial cells and greater sensitivity to NEC (Figure 6). Future investigations comparing preterm and term pigs at different time points after birth and fed different enteral diets are important to further elucidate the mechanisms of immune maturation in pigs, used as models for human preterm and term infants. A very high sensitivity to both NEC and sepsis in preterm pigs indicate that the gut and immune systems may be even more immature in preterm pigs vs. infants,
27
but this also makes pigs very sensitive models to study factors that influence the early maturation of these systems.
Summary of the suggested mechanisms that relate to neonatal antibiotics treatment of preterm pigs. The diagram illustrates blood leukocyte development, gut colonization, translocation and NEC development (CON, SYS and ORA pigs) during the first 5 d after preterm birth. (a) Gut colonization occurs rapidly after birth with a high load of Gram-positive bacteria in CON pigs and Gram-negative bacteria in SYS pigs, whereas colonization is delayed with few bacteria in ORA pigs (b). Oral antibiotics delay colonization and (c) enhance blood neutrophil maturation, as reflected by reduced cell size and CD172a expression on d 2–3. High bacterial load in the gut of CON and SYS pigs facilitates (c) Gram-positive bacterial adherence to the intestinal mucosa and (d) translocation of bacteria and bacterial products into the blood stream. The combined effects of high bacterial load, translocation and less neutrophil maturation contribute to NEC development in CON and SYS pigs with (e) damage to epithelial cells, (f) leukocyte infiltration and increased intestinal permeability.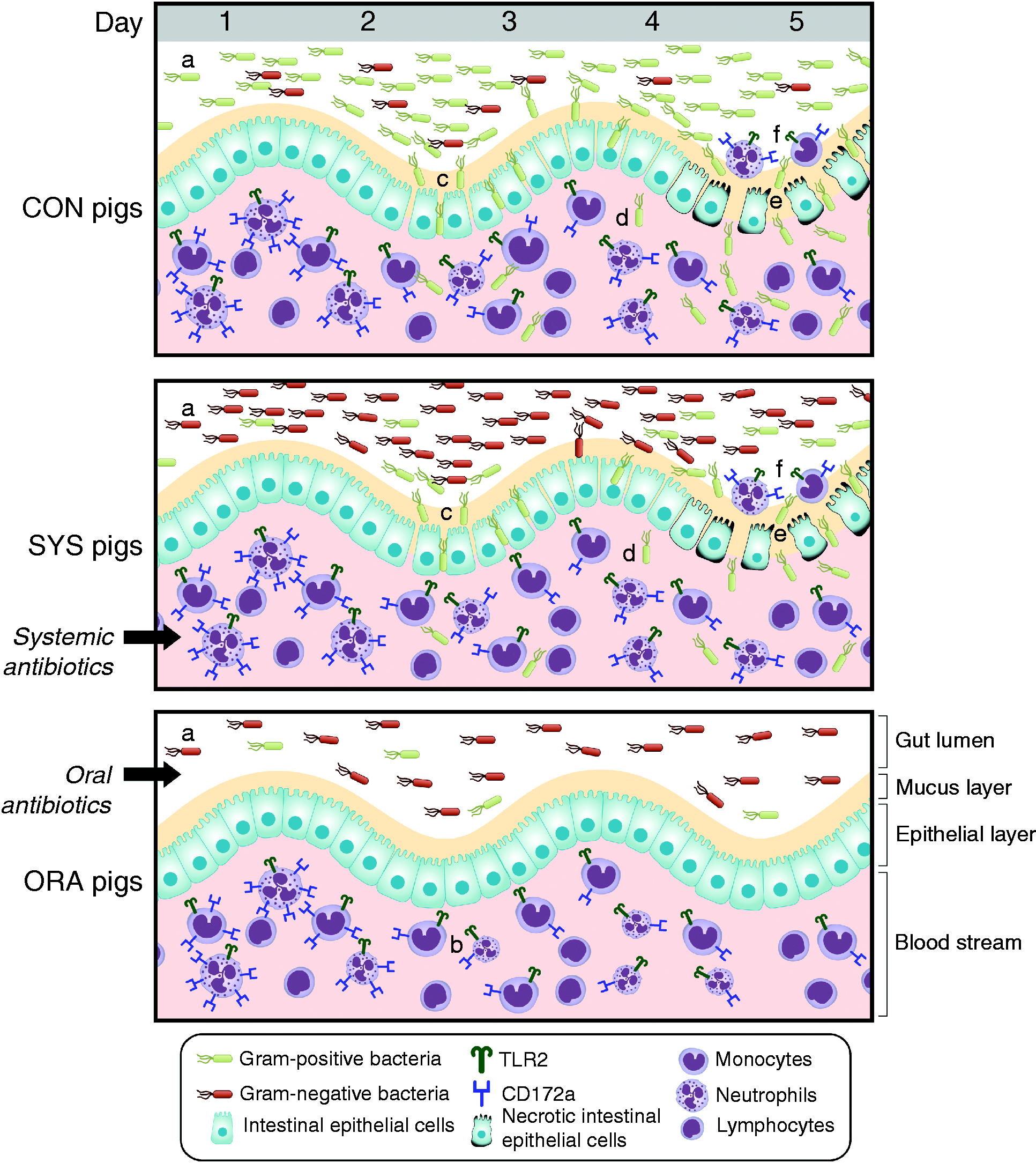
Our study illustrates important principles for the postnatal development of gut bacterial colonization, systemic immunity and NEC sensitivity in response to systemic or oral antibiotics to formula-fed preterm neonates. While concerns about increased microbial resistance and adverse long-term effects may prevent widespread use of neonatal short-term antibiotics, even for preterm newborns, it remains important to investigate novel preventive and therapeutic regimens for these sensitive pediatric patients. Antimicrobial proteins and peptides in natural milk may mimic the effects of oral antibiotics and there is a need to understand the combined effects of early milk and microbiota on the developing gut and immune systems in order to support health of preterm infants in both the short and long term.
Footnotes
Acknowledgements
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Danish Strategic Research Council (NEOMUNE, grant number 12-132401).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
