Abstract
Clinical data indicate that inflammatory responses differ across sexes, but the mechanisms remain elusive. Herein, we assessed in vivo and ex vivo cytokine responses to bacterial endotoxin in healthy men and women to elucidate the role of systemic and cellular factors underlying sex differences in inflammatory responses. Participants received an i.v. injection of low-dose endotoxin (0.4 ng/kg body mass), and plasma TNF-α and IL-6 responses were analyzed over a period of 6 h. In parallel, ex vivo cytokine production was measured in endotoxin-stimulated blood samples obtained immediately before in vivo endotoxin administration. As glucocorticoids (GCs) play an important role in the negative feedback regulation of the inflammatory response, we additionally analyzed plasma cortisol concentrations and ex vivo GC sensitivity of cytokine production. Results revealed greater in vivo pro-inflammatory responses in women compared with men, with significantly higher increases in plasma TNF-α and IL-6 concentrations. In addition, the endotoxin-induced rise in plasma cortisol was more pronounced in women. In contrast, no sex differences in ex vivo cytokine production and GC sensitivity were observed. Together, these findings demonstrate major differences in in vivo and ex vivo responses to endotoxin and underscore the importance of systemic factors underlying sex differences in the inflammatory response.
Keywords
Introduction
Clinical observations have provided compelling evidence that humoral and cell-mediated immune responses markedly differ across sexes, with women showing more pronounced inflammatory and innate immune responses during bacterial and viral infections than men.1,2 The augmented inflammatory response is thought to be beneficial by limiting pathogen spread and by accelerating pathogen clearance, resulting in lower infection-related mortality rates in women compared with men.3,4 This is supported by findings from population-based studies indicating that female sex is associated with a better prognosis after sepsis or severe trauma.5–10 However, there seems to be a downside of this heightened inflammatory responsiveness as chronic inflammatory and autoimmune diseases are more prevalent in women than in men.11–13
The biological mechanisms underlying sex-related differences in human inflammatory responses remain elusive and are difficult to address in the clinical setting, where many confounding factors (e.g. differences in disease onset, medication, comorbidities) exist. To overcome this problem, experimental studies in the past used whole blood or isolated peripheral blood mononuclear cells from healthy donors to investigate whether immune cells from men and women respond differently to stimulation with PAMPs such as bacterial endotoxin (i.e. LPS). However, most likely because of methodological differences, these studies yielded inconsistent results. While some studies reported greater endotoxin-induced ex vivo or in vitro cytokine production in men compared with women, others failed to reproduce these findings or even found the opposite.14–19 A different approach was recently taken in a handful of in vivo studies in which administration of endotoxin in healthy individuals was used as model of acute systemic inflammation.20–24 In line with clinical observations, these studies consistently reported greater pro-inflammatory cytokine responses (e.g. TNF-α, IL-6, IL-8) in women compared with men. However, in vivo endotoxin administration not only results in immune activation but also triggers the nervous and endocrine systems that provide important feedback regulation of the inflammatory response, making it difficult to separate cellular from systemic effects. This calls for comparative studies in which both cellular and systemic responses to endotoxin are assessed in the same individuals in parallel.
Against this background, we herein analyzed for the first time endotoxin-induced production of pro-inflammatory cytokines both in vivo and ex vivo in a sample of healthy male and female volunteers. As endogenous glucocorticoids (GCs) play an important role in the negative feedback regulation of the inflammatory response, 25 we additionally investigated potential sex differences in the LPS-induced cortisol response and, in a separate experiment, ex vivo GC sensitivity of cytokine production.
Materials and methods
Study participants
Healthy male and female volunteers were recruited via public advertisement and underwent an extensive screening consisting of a personal interview, a physical examination, and the assessment of blood and clinical chemistry parameters [i.e. complete blood cell count, C-reactive protein (CRP), coagulation factors, liver enzymes and renal parameters]. General exclusion criteria were pre-existing or current medical conditions, body mass index (BMI) < 18 or ≥ 29 kg/m2, current medication, smoking and regular alcohol use (>4 drinks/wk). To exclude pregnancy, only women using hormonal contraceptives were included and a pregnancy test was conducted on the day of the experiment. The study protocol was approved by the Ethics Review Board of the University of Duisburg-Essen (approval no. 09-4271). All subjects provided written informed consent and received financial compensation for their participation in the study.
In vivo LPS challenge
The in vivo experiment was conducted in a medically equipped room and was supervised by an emergency physician. Upon arrival at the study site, a catheter was inserted into a forearm vein for repeated blood collection and endotoxin application. After a rest period of 30 min, body temperature, heart rate and blood pressure were measured, and blood samples for in vivo baseline measures and ex vivo testing were obtained. Fifteen min later, subjects received an i.v. injection of 0.4 ng LPS/kg of body mass (reference standard endotoxin from Escherichia coli O113:H10, lot H0K354; United States Pharmacopeia, Rockville, MD, USA). The LPS (2000 EU/ml) had been subjected to a microbial safety testing routine by the German Federal Agency for Sera and Vaccines (Paul-Ehrlich Institute, Langen, Germany) and was stored in endotoxin-free borosilicate tubes (Pyroquant Diagnostik, Mörfelden-Waldorf, Germany) at −20℃ until use. Additional blood samples were collected 1, 2, 3, 4 and 6 h after endotoxin injection in EDTA-treated tubes. Plasma was separated by centrifugation and was stored at −80℃ until analysis.
Ex vivo LPS stimulation
For ex vivo testing, blood was collected in endotoxin-free heparinized tubes. Five microliters of whole blood were mixed in 96-well flat-bottom microtiter plates with 240 µl medium (RPMI-1640; Life Technologies, Darmstadt, Germany) and 15 µl of either endotoxin-free physiological saline (negative control) or saline containing different doses of LPS (12.5, 25, 50, 100, 200, 400 pg/ml). The LPS used for ex vivo stimulation was the same as in the in vivo experiment. After incubation (24 h, 37℃, 5% CO2), culture supernatants were collected and stored at −80℃ until analysis.
GC sensitivity of ex vivo cytokine production
In a separate experiment, heparinized diluted blood was stimulated with 25 ng/ml LPS and cultured in absence or presence of different doses (10–9, 10–8, 10–7, 10–6, 10–5 M) of the GC receptor agonist dexamethasone (DEX; Sigma-Aldrich, Munich, Germany). After incubation (24 h, 37℃, 5% CO2), culture supernatants were collected and stored at −80℃ until analysis. As an index for GC sensitivity of LPS-induced cytokine production, the half maximal inhibitory concentration (IC50) for each individual dose–response curve was calculated using GraphPad Prism 6 (GraphPad Software, La Jolla, CA, USA).
Hematological analyses
Complete blood counts, including white blood cell (WBC) differential and red blood cell (RBC) counts, as well as hematocrit (HCT) levels, were obtained with an automated hematology analyzer (KX-N21; Sysmex Europe, Norderstedt, Germany).
Cytokine analyses
Concentrations of TNF-α and IL-6 in plasma and culture supernatants were measured by ELISA (Human Quantikine or DuoSet ELISA; R&D Systems, Minneapolis, MN, USA). The sensitivity of the assays was 0.11 pg/ml for TNF-α and 0.70 pg/ml for IL-6.
Cortisol analyses
Plasma concentrations of cortisol were measured using a competitive binding ELISA (Cortisol ELISA; IBL International, Hamburg, Germany). The sensitivity of the assay was 0.08 ng/ml and cross-reactivity with other relevant steroids was 7.0% (11-deoxycortisol), 4.2% (cortisone), 1.4% (corticosterone), 0.35% (progesterone) and < 0.01% (estrone, estradiol, estriol, testosterone).
Statistical analysis
Data analysis was performed using SPSS 22.0 (IBM, Armonk, NY, USA) and the level of significance was set at P < 0.05. Normality of residuals was examined using the Shapiro–Wilk test and data were log-transformed when necessary. Demographic and clinical data were compared using independent samples t-tests. Body temperature, WBC counts, cytokine and cortisol data were analyzed by repeated-measures ANOVA with ‘time’ (in vivo), ‘LPS dose’ (ex vivo) or ‘DEX dose’ (GC sensitivity) as within-subject factors and ‘sex’ as between-subject factor. Greenhouse–Geisser correction was performed when the assumption of sphericity was violated. If ANOVA revealed a significant sex × time interaction and/or significant sex effect, independent t-tests were computed for group comparisons at the different sampling points. The area under the curve (AUC) was calculated using the trapezoid formula. 26
Results
Sample characteristics
Clinical characteristics.
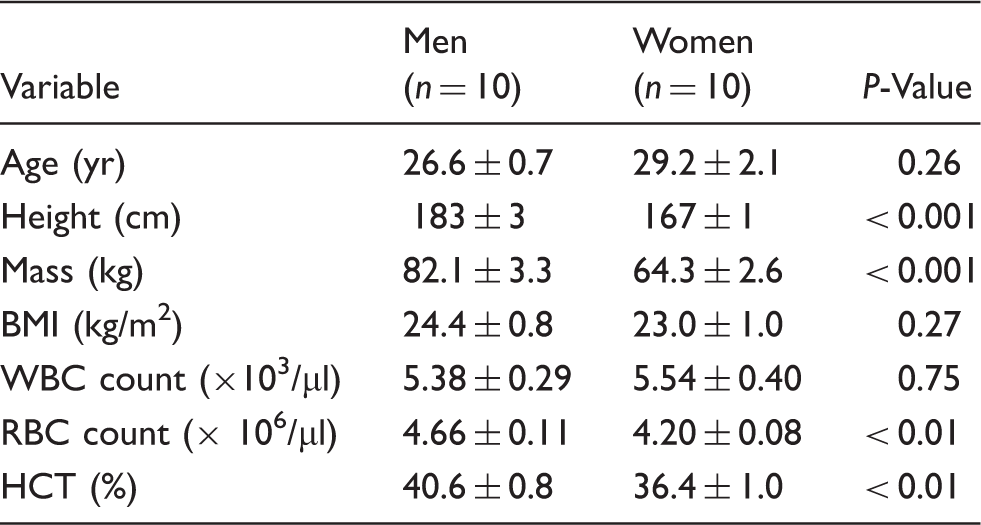
Data are mean ± SEM.
BMI: body mass index; HCT: hematocrit; RBC: red blood cell; WBC: white blood cell.
In vivo response to endotoxin
Intravenous administration of LPS elicited in all subjects an acute systemic inflammatory response as evident from a significant elevation in body temperature (time effect: F = 37.9, P ≤ 0.001; Figure 1a), a profound neutrophilia (time effect: F = 86.5, P ≤ 0.001; Figure 1b) and monocytopenia (time effect: F = 41.4, P ≤ 0.001; Figure 1c), as well as significant increases in plasma levels of TNF-α (time effect: F = 40.2, P ≤ 0.001; Figure 2a) and IL-6 (time effect: F = 60.5, P ≤ 0.001; Figure 2c). The rise in body temperature occurred earlier and was greater in women compared with men (time × sex interaction: F = 2.4, P < 0.05; sex effect: F = 4.5, P < 0.05). Women also mounted a stronger pro-inflammatory cytokine response than their male counterparts, with significantly higher plasma increases in TNF-α (time × sex interaction: F = 7.2, P ≤ 0.01; sex effect: F = 14.3, P ≤ 0.001) and IL-6 (time × sex interaction: F = 6.7, P ≤ 0.01; sex effect: F = 8.0, P ≤ 0.01), as well as a significantly higher total release of TNF-α (AUC: t = 4.1, P ≤ 0.001) and IL-6 (AUC: t = 2.7, P ≤ 0.01) over time, calculated as AUC from baseline to 6 h post-LPS injection (Figure 2a, c, inserts). In contrast, men showed a greater increase in neutrophil counts and the initial monocytopenia was followed by a significant increase in monocyte counts relative to baseline. The systemic inflammatory response was accompanied by a significant increase in plasma cortisol concentration (time effect: F = 14.2, P ≤ 0.001; Figure 1d) that was significantly higher in women than in men (sex effect: F = 16.9, P ≤ 0.001).
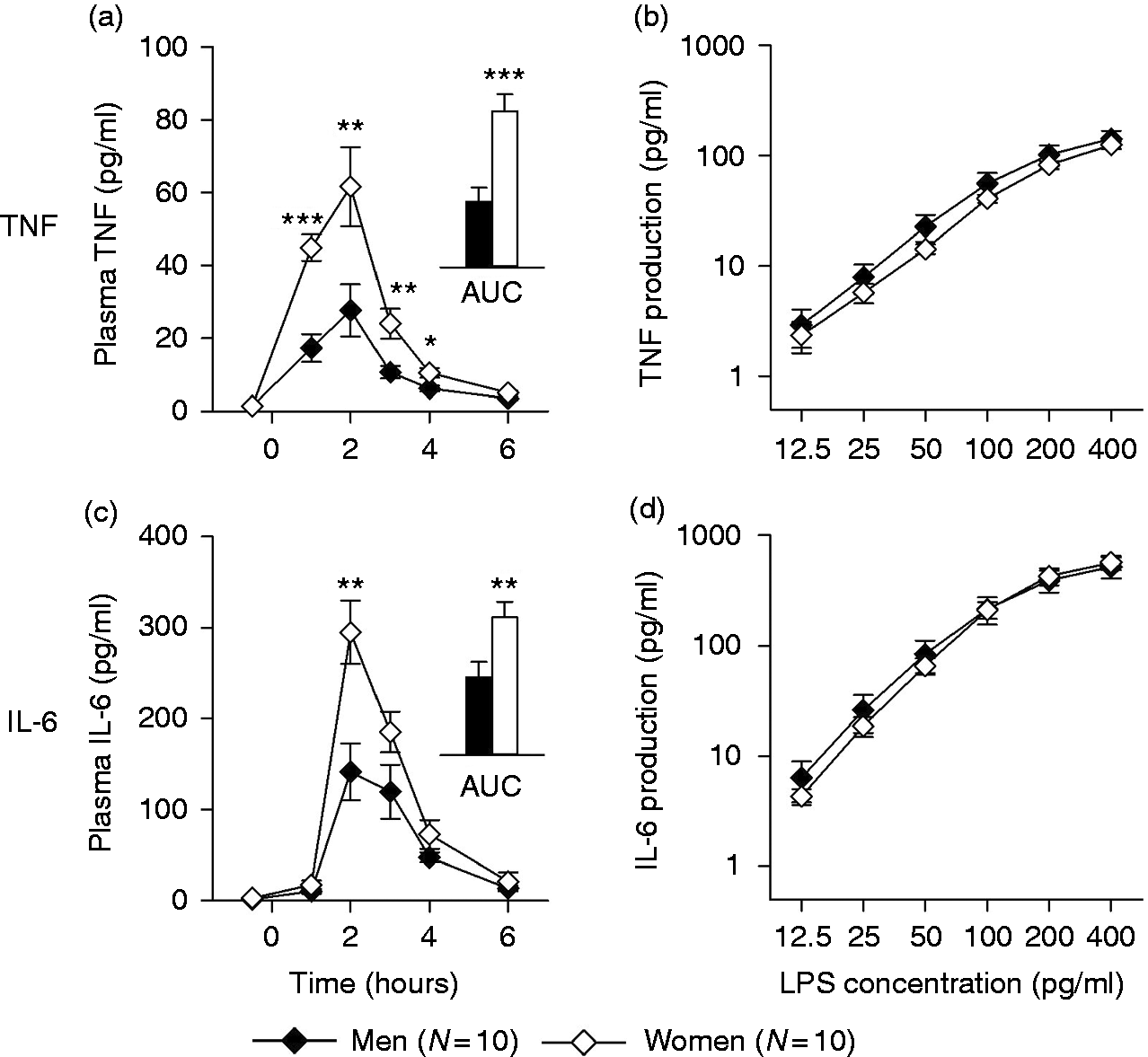
(a) Body temperature, (b) neutrophil counts, (c) monocyte counts and (d) plasma cortisol concentration in men (n = 10) and women (n = 10) before and after in vivo administration of 0.4 ng/kg of endotoxin. Means ± SEM are shown. *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001. In vivo and ex vivo cytokine responses to endotoxin. (a, c; in vivo): plasma concentrations of TNF-α and IL-6 in men (n = 10) and women (n = 10) before and after i.v. administration of 0.4 ng/kg endotoxin. Figure inserts depict the AUC from baseline to 6 h post-injection [TNF-α: 65.0 ± 13.8 pg/ml (men) vs. 154.7 ± 17.0 pg/ml (women); IL-6: 348.5 ± 68.1 pg/ml (men) vs. 615.8 ± 69.6 pg/ml (women)]. (b, d; ex vivo): concentrations of TNF-α and IL-6 in supernatants of diluted whole blood cultures from the same subjects after ex vivo stimulation with indicated doses of LPS. Means ± SEM are shown. *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001.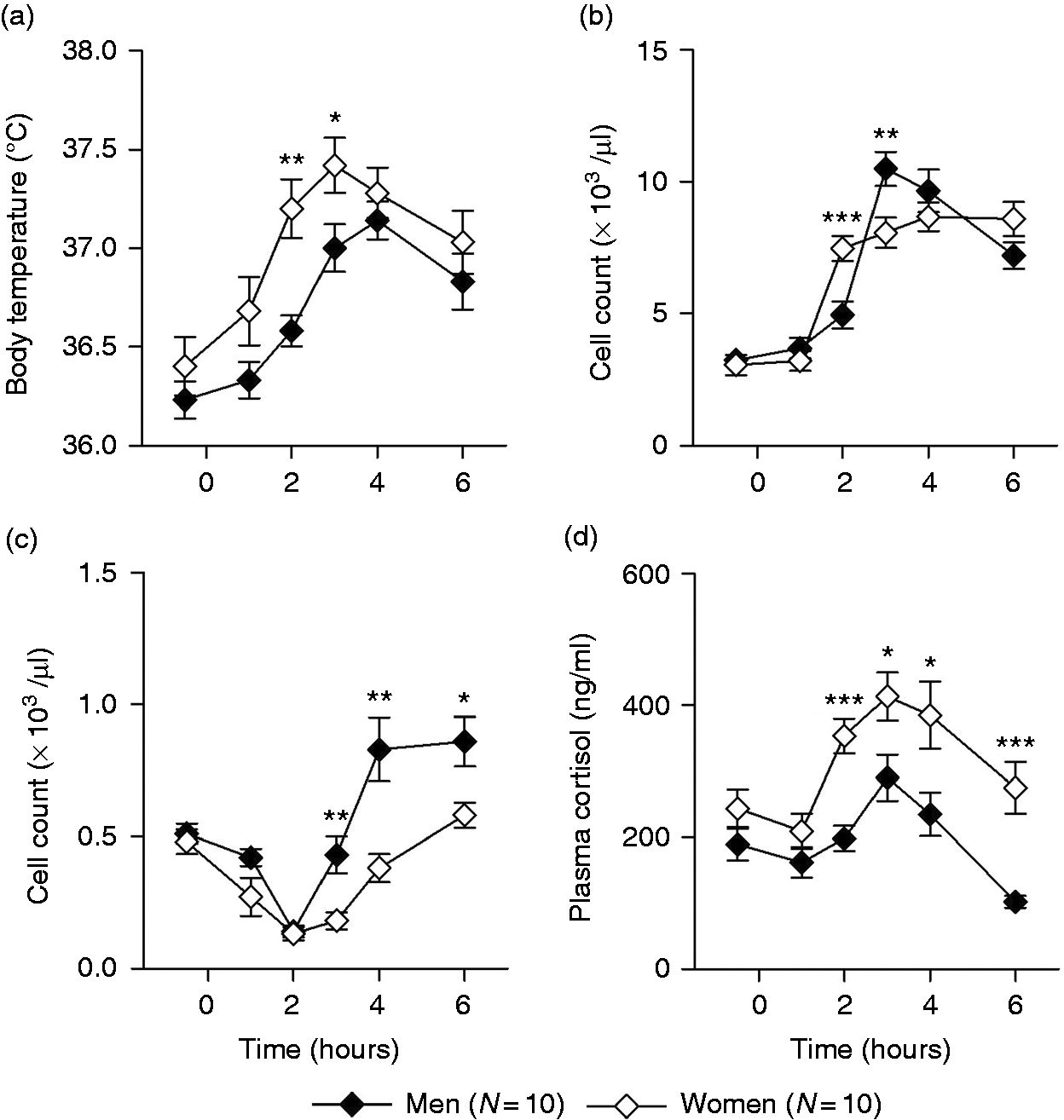
Ex vivo cytokine production
Blood samples obtained immediately before in vivo LPS administration were cultured ex vivo in absence and presence of LPS. Endotoxin stimulation induced dose-dependent increases in the production of TNF-α (dose effect: F = 58.6, P ≤ 0.001; Figure 2b) and IL-6 (dose effect: F = 52.5, P < 0.001; Figure 2d), without evidence for sex differences (sex effect: TNF-α: F = 0.76, P = 0.39; IL-6: F = 0.02, P = 0.88; sex × dose interaction: TNF-α: F = 0.32, P = 0.89; IL-6: F = 0.19, P = 0.96). As monocytes are the main cytokine producing cells in LPS-stimulated blood cultures, we also quantified blood monocyte numbers. However, no significant differences in monocyte counts were found (t = 0.32, P = 0.75) between blood samples obtained from men (0.50 ± 0.04 × 103 cells/µl) and women (0.48 ± 0.04 × 103 cells/µl).
Correlational analyses
Partial correlations controlling for sex revealed a significant positive association between endotoxin-induced TNF-α and IL-6 responses in vivo (AUC: r = 0.762, P ≤ 0.001), as well as a significant positive association between LPS-stimulated TNF-α and IL-6 production ex vivo (LPS 100 pg/ml: r = 0.599, P ≤ 0.01). However, no significant correlations between in vivo and ex vivo cytokines responses were found.
GC sensitivity
GCs have profound anti-inflammatory properties and provide negative-feedback regulation of the inflammatory response. Thus, we examined in a follow-up study with an independent cohort of blood donors (12 men, 12 women on hormonal contraceptives) whether sex differences in DEX-induced inhibition of LPS-stimulated TNF-α and IL-6 production exist. Sample characteristics in the follow-up study [age: 26.5 ± 1.2 yr (range 20–41 yr); BMI: 23.1 ± 0.6 kg/m2 (range 17.7–27.6 kg/m2)] were highly comparable with the first study. In line with the initial study, no significant sex differences in endotoxin-induced production of TNF-α (t = 0.60, P = 0.55) and IL-6 (t = 1.32, P = 0.20) were found. DEX dose dependently inhibited the LPS-stimulated production of TNF-α (dose effect: F = 160.0, P ≤ 0.001; Figure 3a) and IL-6 (dose effect: F = 885.8, P < 0.001; Figure 3b). However, GC-induced suppression of pro-inflammatory cytokine production did not significantly differ between men and women (sex effect: TNF-α: F = 2.09, P = 0.16; IL-6: F = 1.48, P = 0.24; sex × dose interaction: TNF-α: F = 1.36, P = 0.26; IL-6: F = 1.13, P = 0.34), with similar IC50 values for both sexes (TNF-α: t = 0.88, P = 0.39; IL-6: t = 1.57, P = 0.39).
GC sensitivity of ex vivo cytokine production. Inhibition of LPS-induced (a) TNF-α and (b) IL-6 production by treatment with indicated doses of the GC receptor agonist DEX. Data are expressed as percentage of cytokine production in the absence of DEX. Means ± SEM are shown. IC50 values indicate the concentration of DEX required for 50% inhibition of cytokine production.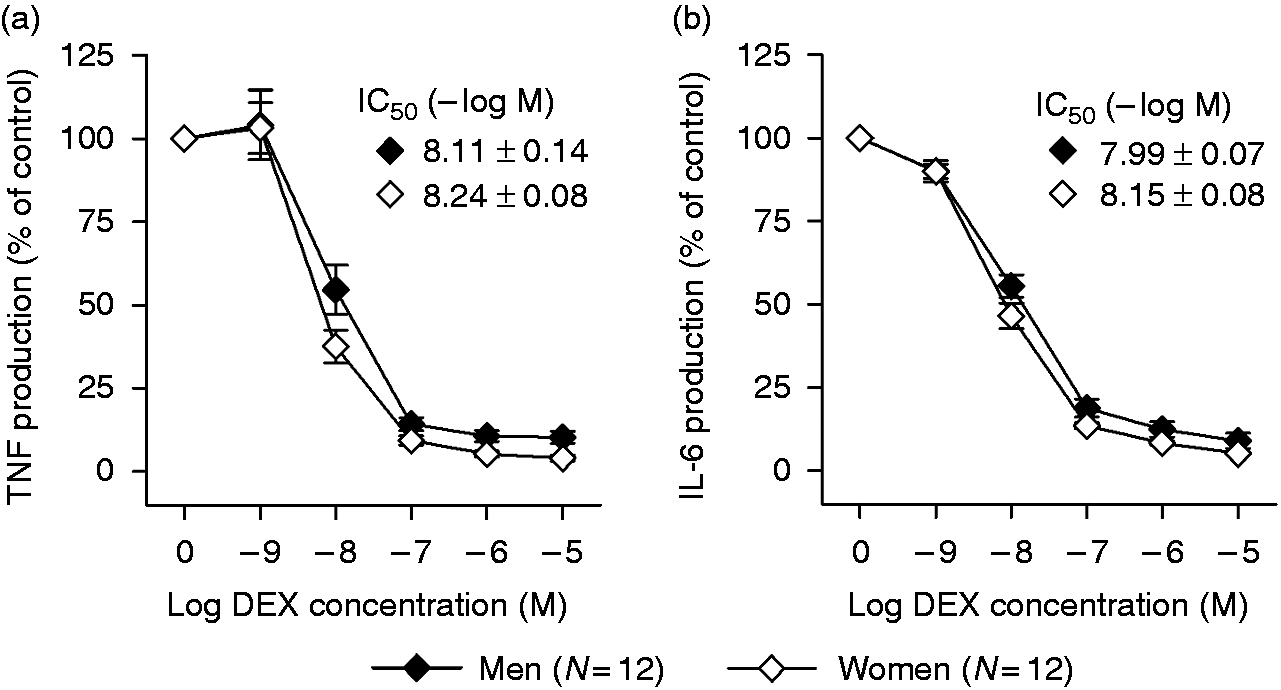
Discussion
Our study revealed pronounced sex differences in endotoxin-induced cytokine production in vivo that did not unfold ex vivo. Intravenous administration of LPS provoked in both men and women an acute systemic inflammatory response that was characterized by a transient rise in plasma concentrations of pro-inflammatory cytokines. Importantly, female participants mounted a substantially stronger in vivo cytokine response, with significantly greater increases in TNF-α and IL-6. This not only confirms and extends the findings from earlier endotoxin studies in healthy humans,21–24 but is also consistent with clinical observations in septic patients. 5 In contrast, ex vivo LPS stimulation of blood samples taken from the same individuals immediately prior to the in vivo challenge did not reveal any differences in pro-inflammatory cytokine production between men and women, suggesting that the observed in vivo differences were related to sex differences in the systemic rather than the cellular response.
One critical step in the response to endotoxin is the LPS sensing process. In vivo, this process starts with the detection of LPS in the circulation by LPS-binding protein (LBP), a hepatically derived acute-phase protein. LBP binds LPS with high affinity and transfers it to CD14, a PRR that exists in both soluble and membrane-bound forms. CD14, in turn, presents the LPS/LBP complex to TLR4, expressed on the surface of monocytes/macrophages, triggering the synthesis and release of cytokines by these cells. 27 Although not essential for TLR4 activation, LBP enhances inflammatory responses by facilitating the transfer of LPS to CD14. 28 As women exhibit substantially higher serum levels of LBP compared with men, as recently shown by us and others,21,24 the augmented in vivo pro-inflammatory response to endotoxin in our female study participants may have resulted from enhanced LPS sensing in women. This would also explain the absence of sex differences in LPS-induced ex vivo cytokine production. Owing to the high dilution of the blood samples, LBP levels in the cell culture were low and had, if at all, only a minor impact on endotoxin-induced ex vivo TLR4 activation. Hence, the potentially enhancing effect of higher serum LBP levels in women was eliminated in our ex vivo setting. Thus, future studies may either use undiluted whole blood or addition of autologous serum to the cell culture to further explore the relevance of LBP and/or other serum factors as a potential mechanism underlying sex differences in the inflammatory response.
An alternative explanation for the absence of sex differences in the ex vivo setting could be the different types of cytokine-producing cells and/or the low amounts of endogenous estrogens in the cell culture. While tissue macrophages in the spleen and liver represent the primary source of circulating TNF-α and IL-6 after systemic LPS administration, 29 monocytes are the main cytokine producers in endotoxin-stimulated blood cultures. Furthermore, human monocytes express lower levels of estrogen receptor (ER) α than macrophages. 30 As estrogens have been shown to promote the expression of inflammatory mediators by LPS-activated tissue resident macrophages through ERα signalling,30,31 estrogen levels in the diluted whole-blood cultures may not have been sufficiently high enough to enhance endotoxin-stimulated cytokine production in monocytes. In addition, cytokine clearance largely differs between ex vivo and in vivo conditions. Cytokines in the circulation have a short half-life and are rapidly metabolized in the kidneys and liver,32,33 whereas under cell culture conditions, in the absence of hepatic or renal clearance mechanisms, elimination occurs only via binding to cytokine receptors. Thus, any relevant sex differences in cytokine metabolism would be have been observable only in vivo but not in the cell culture.
The in vivo production of pro-inflammatory mediators is tightly controlled by immune and non-immune regulatory pathways.34–36 Endogenous GCs play an important role in the neuroendocrine feedback regulation of the systemic inflammatory response. Pro-inflammatory cytokines released by activated immune cells in the periphery signal the brain trough afferent pathways and induce the activation of the hypothalamus–pituitary–adrenal (HPA) axis, ultimately leading the release of cortisol from the adrenal cortex into the circulation. 25 Cortisol, in turn, has potent anti-inflammatory activities, for example, by down-regulating the expression of pro-inflammatory mediators.37,38 Herein, we show that the LPS-induced increase in plasma cortisol was greater and longer lasting in women compared with men. This most likely was a direct consequence of the higher rise in TNF-α and IL-6 which, owing to enhanced signaling to the brain, resulted in stronger HPA axis activation in women. In turn, the subsequent drop in plasma cytokines was much steeper in female compared with male participants, suggesting that the greater release of cortisol could be one key mechanism constraining the heightened inflammatory response in women. In a follow-up study, we additionally examined sex differences in GC sensitivity of the cytokine-producing cells. Results showed that DEX-induced suppression of TNF-α and IL-6 production was highly comparable in blood cultures of both sexes, indicating that circulating monocytes from men and women are equally sensitive to the anti-inflammatory actions of GCs. However, it remains open whether this also applies to resident tissue macrophages, which represent the main cytokine producers in vivo. As the negative-feedback regulation of the inflammatory response involves not only the HPA axis, but also neurotransmitters such as noradrenaline and acetylcholine, future studies need to address whether sex differences in, for example, adrenergic and cholinergic regulation of the inflammatory response exist.
In the current study, we only included women on hormonal contraceptives. Thus, our findings may not be representative for naturally cycling or post-menopausal women. Given the putative role of estrogens in augmenting pro-inflammatory responses in women,3,4,30 future studies are warranted to analyze inflammatory responses to endotoxin also in women during the follicular and luteal phases of the menstrual cycle.
Taken together, the findings of our study underscore the important role of systemic factors in driving human sex differences in the inflammatory response, providing a solid basis for future, more mechanistically oriented studies on the molecular underpinnings.
Footnotes
Acknowledgments
The author(s) thank Bettina Löschner and Alexandra Kornowski for excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded, in part, by the German Research Foundation (DFG; BE 5173/2-1; SE 236/11-1) and the German Federal Ministry for Education and Research (BMBF; 0315709). A.W. was supported by an internal research grant of the Medical Faculty of the University of Duisburg-Essen (IFORES).
