Abstract
The interaction of selected endotoxin preparations (lipid A from Erwinia carotovora and LPS Re and Ra from Salmonella enterica sv. Minnesota strains R595 and R60, respectively) with selected bile acids was investigated biophysically. Endotoxin aggregates were analyzed for their gel-to-liquid crystalline phase behavior, the type of their aggregates, the conformation of particular functional groups, and their Zeta potential in the absence and presence of the bile acids by applying Fourier-transform infrared spectroscopy, differential scanning calorimetry, measurements of the electrophoretic mobility, and synchrotron radiation X-ray scattering. In addition, the ability of the endotoxins to induce cytokines in human mononuclear cells was tested in the absence and presence of varying concentrations of bile acids. The data show that the endotoxin:bile acid interaction is not governed by Coulomb forces, rather a hydrophobic interaction takes place. This leads to an enhanced formation of the inherent cubic aggregate structures of the endotoxins, concomitant with a slight disaggregation, as evidenced by freeze-fracture electron microscopy. Parallel to this, the addition of bile acids increased the bioactivity of lipid A and, to a lower degree, also that of the tested rough mutant LPS at lower concentrations of the endotoxin preparation, a finding similar as reported for the interaction of other agents such as hemoglobin. These data imply that there are general mechanisms that govern the expression of biological activities of endotoxins.
Introduction
Bile salts are a family of physiological detergents that are synthesized in a primary form from cholesterol in the liver of mammalian species and are important in the production of bile flow, excretion of cholesterol as well as in intestinal digestion and absorption of fats and fat-soluble vitamins. The term ‘bile acids’ is frequently used synonymously with ‘bile salts’, even though the free acid and the monovalent sodium salt forms have different physicochemical properties.1,2 Structurally, the bile salts/acids have a steroid nucleus composed of four fused hydrocarbon rings with polar hydroxyl functions and an acidic side chain that is enzymically linked via an amide bond to the amino acids glycine or taurine in hepatic bile production. Largely as a result of the unilateral (α)-orientation of the hydroxy groups in the steroid core, bile salts represent a particular class of water-soluble amphiphiles comprising of a hydrophilic (polar) and a hydrophobic (nonpolar) surface, in addition to an acidic head group. It is known that bacteria from the gut metabolically convert primary bile acids/salts, i.e. predominantly cholate and chenodeoxycholate in human bile, into secondary bile acids/salts, namely deoxycholate and lithocholate within the human enterohepatic circulation. In addition, tertiary bile acids/salts are formed by further metabolic modification of secondary bile acids/salts by the intestinal flora or hepatocytes; in humans these are ursodeoxycholate, the 7 β-epimer of chenodeoxycholate, and the sulfate ester of lithocholate. 1
In a series of studies on animal models of endotoxaemia, the clearance of bacterial LPS from the circulation has been well documented to occur mainly by excretion into the gut via the liver-bile pathway. 3 Moreover, investigations on bile-deprived rats have indicated that bile salts prevent the gastrointestinal translocation of endotoxin from commensal Gram-negative bacteria. 4 Concerning the LPS-directed effects of bile salts, electron microscopic and ultracentrifugational investigations employing sodium deoxycholate have revealed a strong and reversible dispersion of LPS aggregates by the bile salt reagent in vitro, leading to the formation of smaller-sized LPS-Na-deoxycholate complexes of about 10 and 20 kDa for smooth (S) and rough (R) LPS chemotypes respectively. These values were calculated on the basis of only two molecules of LPS per Na-deoxycholate-complexed particle. 5 – 8 In vivo studies on the effects of bile salts on immunoactivation by LPS in rabbits, the pyrogenic and adjuvant activities of endotoxin have been described to be prevented by preincubation with higher concentrations of deoxycholate,5,7,9 and the LPS-induced cytokine release by human monocytes was also initially reported to be inhibited by higher concentrations of deoxycholate and chenodeoxycholate in vitro.10,11 However, subsequent in vitro studies revealed that these inhibitory activities were as a result of general cytotoxic effects of bile salts in the higher dose range in cell culture, whereas lower non-toxic concentrations of bile salts did not influence LPS-induced cytokine production in human monocytes and murine Kupffer cells. 12
We investigated the effects of bile salts/acids on endotoxins in more detail by employing a panel of biophysical methods and a standard in vitro assay of cytokine induction in human mononuclear cells (MNC). Free lipid A, the ‘endotoxic principle’ of LPS, from Erwinia carotovora, as well as Re- and Ra-type LPS from Salmonella enterica sv. Minnesota, were the endotoxins analyzed, and the bile salts/acids examined for their effect on the ultrastructures and cytokine-inducing activities of lipid A and LPS preparations were Na-cholate and Na-deoxycholate and, also in some cases, Na-dehydrocholate, lithocholic acid and Na-chenodeoxycholate.
Materials and methods
Chemicals
The LPS from rough mutants Re and Ra from Salmonella enterica sv. Minnesota strains R595 and R60, respectively, were extracted from bacteria according to standard procedures. 13 Highly purified bisphosphoryl lipid A sample was isolated from LPS from E. carotovora as described. 14 Bile salts and acids Na-cholate, Na-deoxycholate, Na-dehydrocholate, lithocholic acid and Na-chenodeoxycholate were purchased from Sigma-Aldrich (Deisenhofen, Germany).
Lipid sample preparation
The lipid samples were prepared as aqueous dispersions at varying buffer (20 mM HEPES, pH 7.4) content, depending on the sensitivity of the Fourier-transform infrared spectroscopy (FTIR) experiments. In all cases, the lipids were suspended directly in buffer, sonicated and temperature-cycled several times between 5 °C and 70 °C, and then stored at 4 °C before measurement. The bile acids were dispersed in buffer and added to the lipids at the indicated molar ratios, and the measurements were done with these freshly prepared dispersions.
Fourier-transform infrared spectroscopy (FTIR)
The infrared spectroscopic measurements were performed on an IFS-55 spectrometer (Bruker, Karlsruhe, Germany). For phase transition measurements, the lipid samples were placed in a CaF2 crystal disk with a 12.5 µm Teflon® spacer. Temperature-scans were performed automatically between a low initial temperature of around 10 °C to 70 °C with a heating rate of 0.6 °C/min. For measurement of hydrated lipid samples, these were spread on an ATR ZnSe plate and free water was evaporated to dryness. Every 3 °C, 50 interferograms were accumulated, apodized, Fourier-transformed, and converted to absorbance spectra.
For the evaluation of the gel-to-liquid crystalline phase behavior, the peak position of the symmetric stretching vibration of the methylene band νsCH2 around 2850 cm−1 was taken, which is a sensitive marker of lipid order. Furthermore, vibrational bands from the interface region (ester carbonyl stretch band at around 1725 to 1740 cm−1) and the head group (phosphate antisymmetric stretch at 1220 to 1260 cm−1) were analysed, along with sugar ring vibrations (around 1160 and 1080 cm−1). 15
Differential scanning calorimetry
Differential scanning calorimetry measurements were performed with a calorimeter VP-DSC (MicroCal, Inc., Northampton, MA, USA) with a sample cell volume of 0.5127 ml. The lipid A samples were dispersed in 10 mM phosphate buffer (pH 7.0) by 1 min vortex mixing and ultrasonification at 65 °C for 10 min. The concentration of the bisphosphoryl lipidA samples were adjusted to 0.5 mM by assuming a MM in the bi-sodium salt forms (hexaacyl: MM = 1814). Different lipid A to bile acids ratios were prepared by pipetting the corresponding amount of the bile acid solution into aliquots of the lipid A dispersion, 1 min vortex mixing, ultrasonification for 5 min at 65 °C and, finally, cooling down to 4 °C. The sample dispersions were degassed directly before measurement using a ThermoVac diaphragm vacuum pump (MicroCal) for 3 min at 25 °C. The DSC curves were obtained at a heating scan-rate of 45 °C h−1 from 5 to 80 °C under an applied air pressure of around 1.8x105 Pa. Twenty consecutive heating scans, after cooling of the sample in the calorimeter, were done to check the reproducibility of the DSC experiments. The accuracy of the DSC experiments was ±0.1 °C for the main phase transition temperatures and ±1.0 kJ mol−1 for the main phase transition enthalpy. The reference cell was filled up with the same phosphate buffer. The phase transition enthalpy was calculated by integration of the heat capacity curve of the 20th scan.
Zeta potential
For the determination of the Zeta-potential of the samples, the electrophoretic mobility of the lipid dispersions in the absence and presence of the bile acids was monitored by measuring the electrophoretic mobility in an electric field of 21.4 V/cm with a Zetasizer 2000 (Malvern Instruments, Malvern, UK) by Laser-Doppler anemometry at a scattering angle of 90°. The Zeta potential was then calculated according to theHelmholtz-Smoluchovski equation. The lipid A-bileacid mixed solutions were prepared in 10 mM phosphate buffer (pH 7.0) at a 0.03 mM lipid A concentration. Measurement was done at 25 °C after the ultrasonification step similar to the DSC measurements.
X-Ray diffraction
X-Ray diffraction measurements of lipid:bile acid mixtures were performed at the European Molecular Biology Laboratory (EMBL) outstation at the Hamburg synchrotron radiation facility, HASYLAB, using the double-focusing monochromator-mirror camera X33. Diffraction patterns in the range of the scattering vector 0.001 < s < 0.08 Å−1 (s = 2 sin θ/λ, 2θ scattering angle and λ the wavelength = 1.5 Å) were recorded at 20, 40 and 60 °C with exposure times of 1 min, using an image plate detector withonline readout (MAR345, MarResearch, Norderstedt, Germany).
16
The s-axis was calibrated with Ag-Behenate, which has a periodicity of 58.4 Å. The diffraction patterns were evaluated as described previously,
17
assigning the spacing ratios of the main scattering maxima to defined three-dimensional structures. The lamellar and cubic structures are most relevant here and are characterized by the following features:
Lamellar: The reflections are grouped in equidistant ratios, i.e. 1, 1/2, 1/3, 1/4, etc. of the lamellar repeat distance dI. Inverted cubic Q: These are structures with three-dimensional geometry. The relation between reciprocal spacing shk and lattice constant a is:
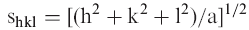
Cytokine assay (MNC)
Human mononuclear cells (MNC) were prepared as following. Heparinized (20 IU/ml) blood was mixed with an equal volume of Hank's balanced salt solution, layered over Ficoll, and centrifuged for 40 min (21 °C, 500 g). The interphase layer of MNC was collected and washed twice in Hank's medium and then resuspended in RPMI 1640 supplemented with 2 mM L-glutamine, 100 U/ml penicillin, 100 µg/ml streptomycin, and 4% heat-inactivated human serum type AB from healthy donors. The MNC (200 µl/well, 5x106 cells/ml) were transferred into 96-well culture plates. Twenty micro liters of a mixture containing the endotoxin samples (1 to 2000 ng/ml) and the bile acids in different concentration ratios was added to each well. Supernatants were harvested after 4 h incubation at 37 °C under 5% CO2, and tumor TNF-α production was measured in a sandwich-ELISA. Production of TNF-α was determined in duplicate at two different dilutions and the values from two independent experiments were averaged.
Results
Gel-to-liquid crystalline phase transition of the acyl chains of LPS
According to the fluidity of the hydrocarbon chains, LPS and lipid A can form two major temperature-dependent states, the gel and the liquid crystalline phases, between which a phase transition at a temperature Tm takes place. For endotoxins from enterobacterial origin, Tm has been shown to lie around 30–36 °C depending on the length of the saccharide chains and around 43 °C to more then 50 °C for lipid A.
14
The influence of bile acids on this parameter was monitored by applying FTIR and DSC. For FTIR, the peak position of the symmetric stretching vibrational band of the methylene groups has values around 2850 cm−1 in the gel, and 2852.5–2853.0 cm−1 in the liquid crystalline phase. In Figure 1A, the interaction of the bile acids Na-cholate and Na-deoxycholate with lipid A from E. carotovora results in a shift of Tm, lying at 52 °C for the pure lipid, to lower temperatures, which is indicative of a bile acid-induced fluidisation of the acyl chains. With DSC, the specific excess heat of the phase transition for the same samples was elucidated (Figure 1B). It can be seen that the melting of the hydrocarbon chains has a peak around 50 °C in the absence of the bile acids, which shifts to lower temperatures in their presence. However, there is no decrease in the phase transition enthalpy ΔH; in particular, even an increase takes place in the presence of Na-deoxycholate. The enthalpy change of E. carotovora lipid A was 36.9 kJ mol−1 with Tm = 49.6 °C, while the bile acid mixtures showed only a small increase in ΔH value (see legend of Figure 1B).
The phase transition behavior was also monitored for LPS from S. enterica sv. Minnesota strain R60 in the presence of Na-deoxycholate by FTIR (Figure 1C). Similar to the findings with lipid A, a bile acid-induced decrease of Tm can be seen, corresponding to a fluidization of the acyl chains.
Influence of bile acids on lipid A functional groups
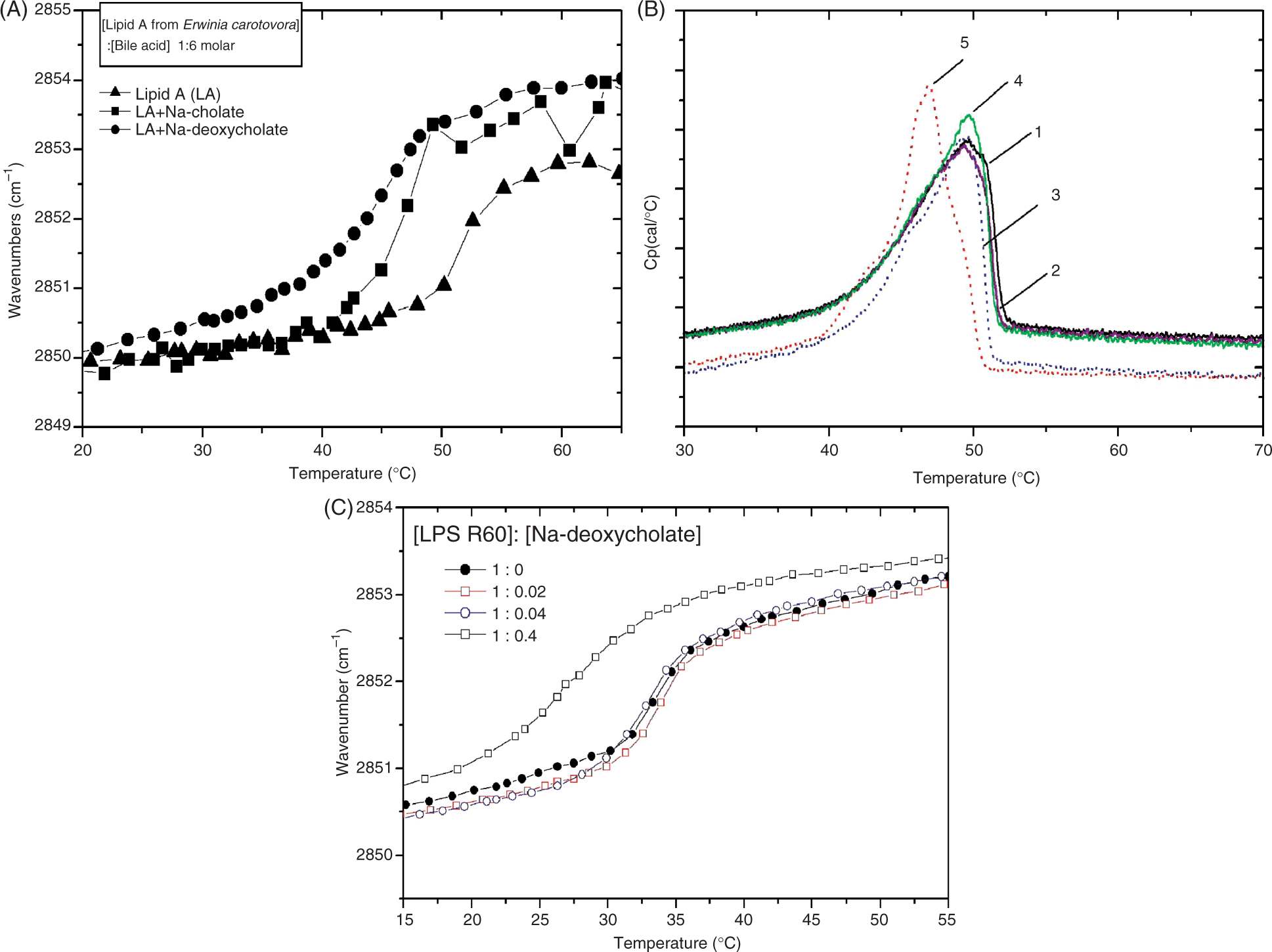
To investigate the possible interaction of bile acids with various molecular parts of lipid A, the infrared spectra in the ‘fingerprint region’ 1400–900 cm−1 were investigated (Figure 2). Lipid A shows vibrational bands of the diglucosamine rings around 1040 and 1160 cm−1, and phosphate bands as shoulders in the range 1300–1200 cm−1.
15
The data shown in Figure 2 clearly illustrate that the spectra of the mixed compounds correspond more or less only to additions of the spectra of the pure compounds; therefore, no significant interaction with these functional groups of lipid A takes place.
Infrared spectrum in the ‘fingerprint’ region 1400-900 cm−1 for lipid A from E. carotovora alone and in the presence of different concentrations of Na-deoxycholate.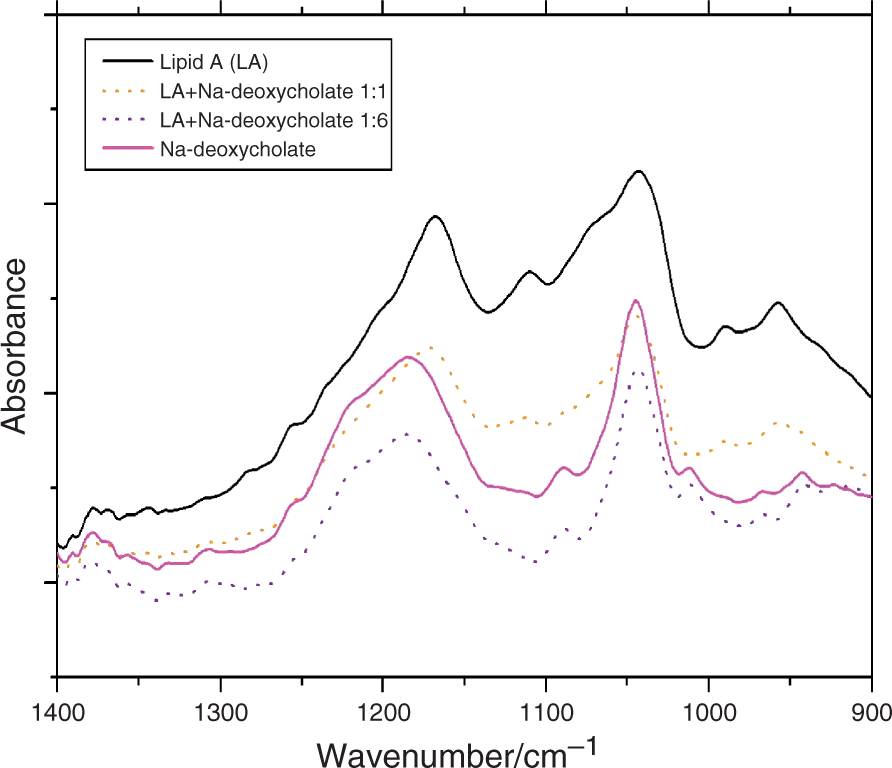
Zeta potential of lipid A aggregates
Zeta potential of lipid A from E. carotovora in the absence and presence of bile acids
Supramolecular structure and morphology of LPS aggregates
The type of aggregate structure of endotoxins and their morphology has been shown to be of fundamental importance for understanding their ability to express biological activity. 21 For the determination of the supramolecular aggregate structure of LPS, synchrotron radiation small-angle X-ray scattering (SAXS) was applied. The LPS Re and Ra from S. enterica sv. Minnesota strains R595 and R60, respectively, were taken as relevant endotoxins. For pure LPS R595, SAXS patterns are usually indicative of a broad reflection between s = 0.1 to 0.35/nm (‘form factor’) consistent with the existence of a lipid bilayer and small additional reflections indicative for a structure with cubic symmetry (‘structure factor’) (data not shown 22 ).
In Figures 3A and B, SAXS patterns are presented for LPS R595 in the presence of Na-cholate (A) and Na-deoxycholate (B). For both bile acids, the diffraction patterns of LPS R595 exhibit a peak at 11–11.5 nm, which is consistent with the cubic periodicity. The patterns in the presence of Na-deoxycholate show two more sharp peaks at 20 °C and 40 °C, for example at 40 °C at 6.60 and 3.76 nm, which can be assigned to the 1/√3 and 1/√9 values of the periodicity at 11.4 nm (Figure 3B). Thus, an aggregate structure with cubic symmetry can be deduced. However, an assignment to a particular space group is not possible because of the lack of evaluable sharp reflections.
Small-angle X-ray scattering of LPS Re from S. enterica sv. Minnesota R595 aggregates in the presence of Na-cholate (A) and Na-deoxycholate (B) at different temperatures. The logarithm of the scattering intensity log I is plotted versus the scattering vector s (s = 1/d, d = spacing of the reflections).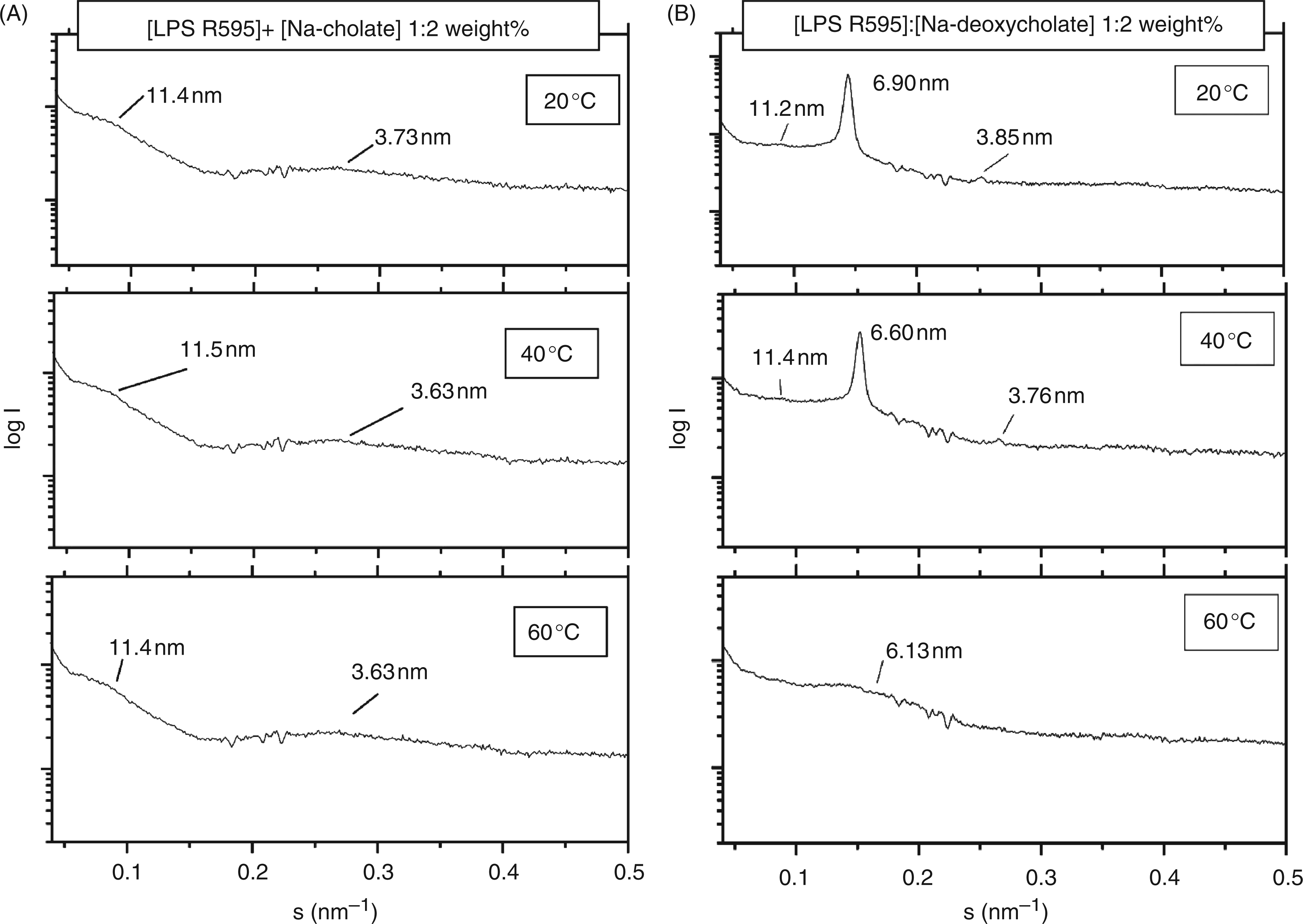
Similarly, the diffraction patterns of LPS R60 (Figure 4) can be interpreted. Thus, for the pattern at 45 °C, the peaks at 21.2, 9.43 and 4.76 nm correspond to the cubic periodicity and the 1/√5 and 1/√20 reflections. At first glance, the two further reflections at 3.25 and 2.65 nm could be assigned to the third and fourth order of a main reflection at 9.43 nm as a result of multilamellar stacking. However, the 3- and 4-fold of these values would lead to values of 9.75 and 10.6 nm, thus making the above interpretation obsolete. As controls, the pure bile acids were measured. Na-Cholate exhibits a weak reflection around 5.0 to 5.6 nm, and Na-deoxycholate one sharp reflection at 8.0 nm at 20 °C, and one weak reflection at 6.20 nm at 50 °C, which do not interfere with the patterns of the mixtures.
Small-angle X-ray scattering of LPS Ra from S. enterica sv. Minnesota R60 aggregates in the presence of Na-cholate at 20 and 45 °C. The logarithm of the scattering intensity log I is plotted versus the scattering vector s (s = 1/d, d = spacing of the reflections).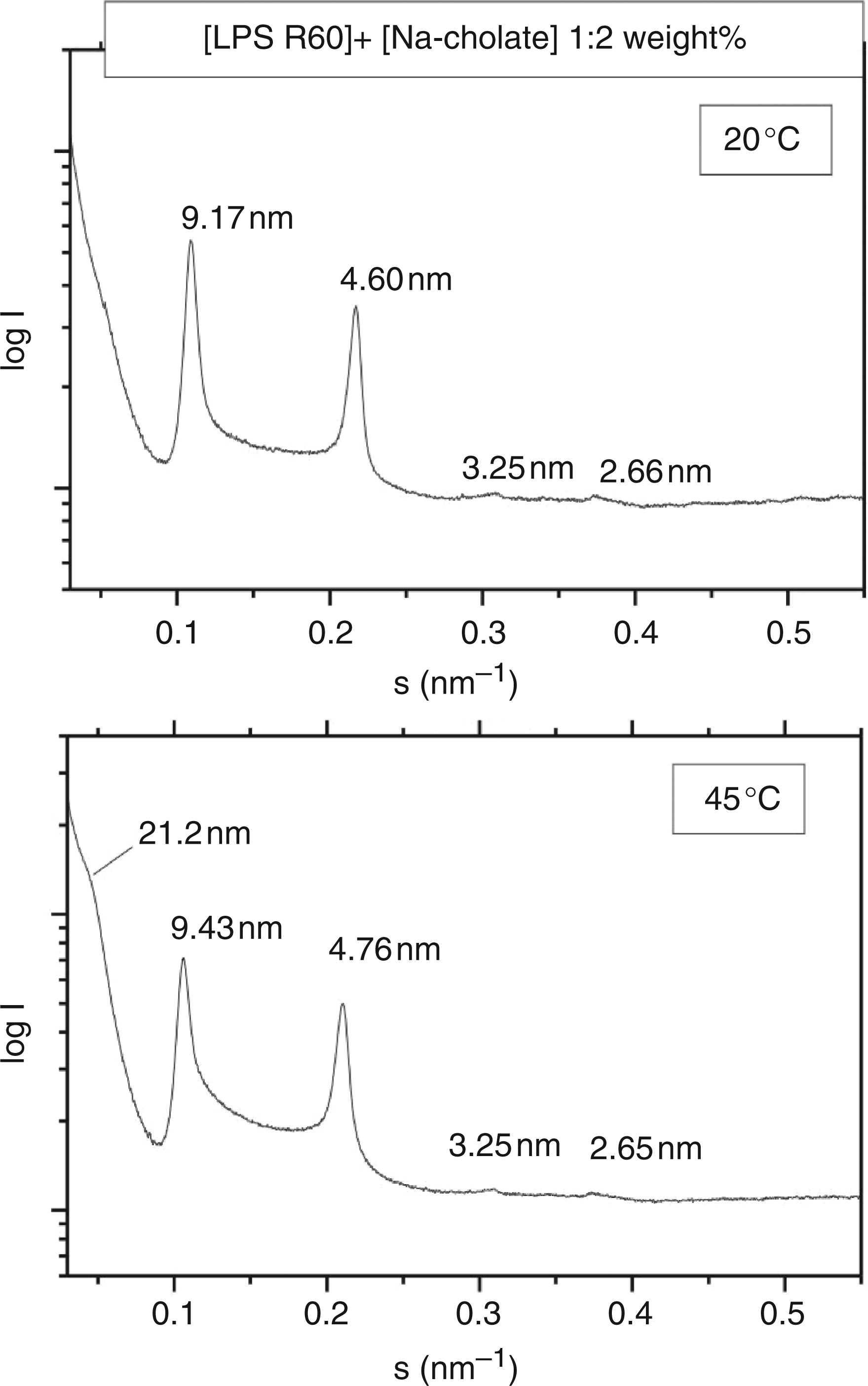
Cryo- and freeze-fracture electron microscopy was applied for the elucidation of the aggregate morphology of LPS R595 and LPS R60 samples in the presence of Na-cholate (Figure 5). Previously, for the former LPS spherical, but not closed particles, in the size-range of 100–300 nm (‘open egg-shells’) have been described,
23
while for the latter LPS ribbon-like structures with long filaments and diameters of 8–12 nm were found.
24
As can be seen, for LPS R595, much of the spherical character is retained, except for the additional occurrence of small disc-like and spherical micellar structures (Figure 5A). LPS R60 reacts stronger to the admixture of Na-cholate, as observed by the appearance of only very few small vesicle structures, and predominantly micellar structures (Figure 5B). Thus, for both endotoxins a decrease in the aggregate sizes takes place.
Freeze-fracture micrograph of LPS Re from S. enterica sv. Minnesota R595 (A) and LPS Ra from S. enterica sv. Minnesota R60 (B) in the presence of Na-cholate.
Endotoxin-induced TNF-α production in human MNC
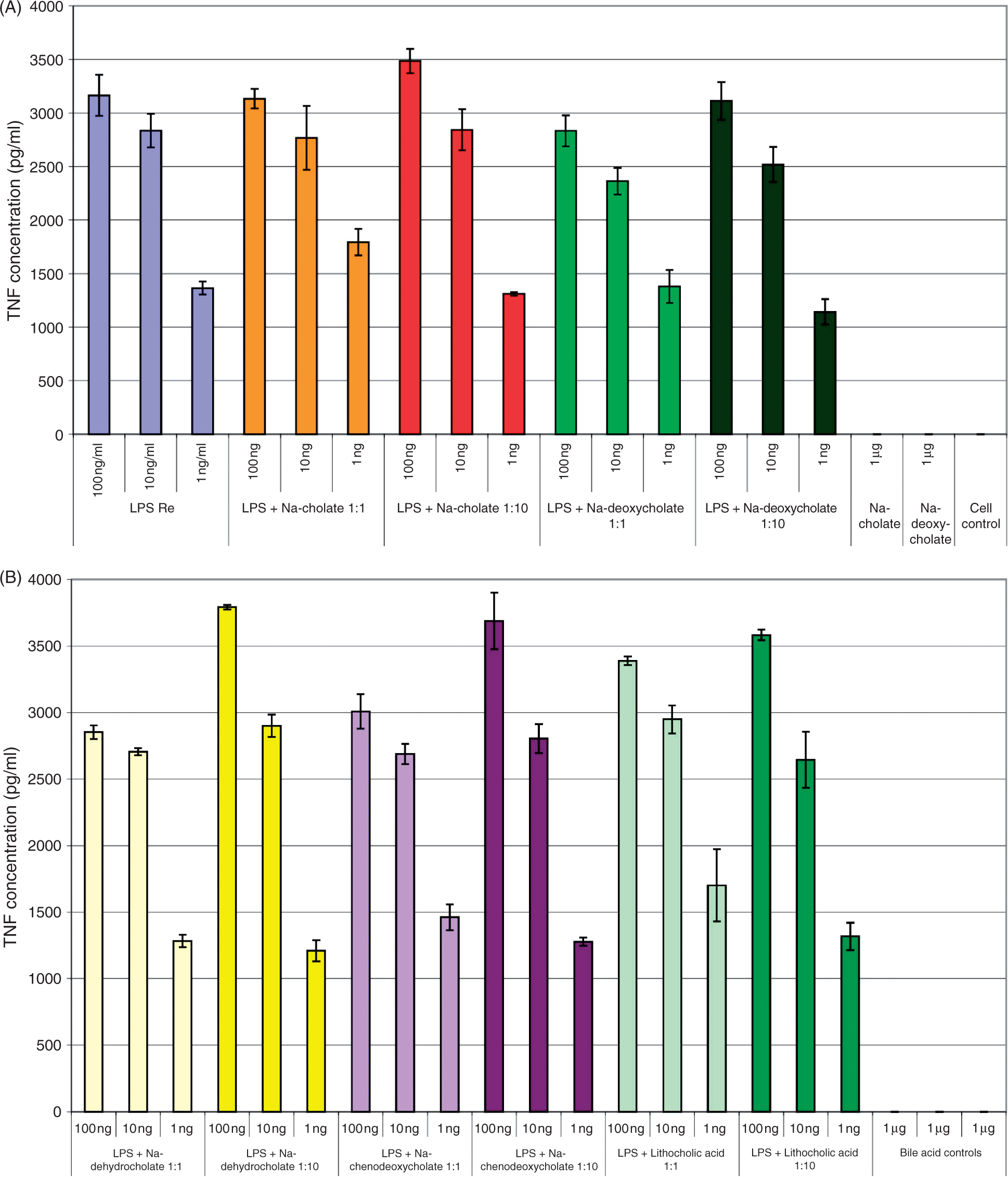
The influence of the bile salts Na-cholate, Na-deoxycholate, and Na-dehydrocholate on the lipid A-induced cytokine secretion (TNF-α) in human MNCs was analyzed at three different lipid A concentrations (100, 10 and 1 nM) and two lipid A:bile salt molar ratios of 1:1 and 1:6. As shown in Figures 6A and 6B, respectively, particularly at the lower lipid A concentrations tested, Na-deoxycholate, and Na-dehydrocholate induced a significant increase in lipid A-induced TNF-α release at both the 1:1 and 1:6 molar ratios. This enhancing effect on lipid A in vitro activity was also observed to a lesser extent for Na-cholate at the lipid A:bile salt ratio of 1:6. By themselves, the bile acids did not induce any cytokine production. In contrast, in similar experiments performed with Re-LPS from S. enterica sv. Minnesota strain R595 and the complete set of bile salts/acids of this study at molar LPS:bile salt/acid ratios of 1:1 and 1:10 (Figures 7A and 7B), only a slight effect of these bile salts on LPS-induced TNF-α production in human MNCs was observed, in particular, a weak enhancement for dehydrocholate, chenodeoxycholate and lithocholic acid (Figure 7B). Analogous results were obtained for Ra-type LPS from S. enterica sv. Minnesota strain R60 (data not shown).
Lipid A-induced secretion of TNF-α of human MNC in the absence and presence (A) 1:1 molar and (B) 1:6 molar concentrations of various bile acids. LPS Re (from S. enterica sv. Minnesota R595)-induced secretion of TNF-α of human MNC in the absence and presence at 1:1 molar and 1:10 molar concentrations of various bile acids.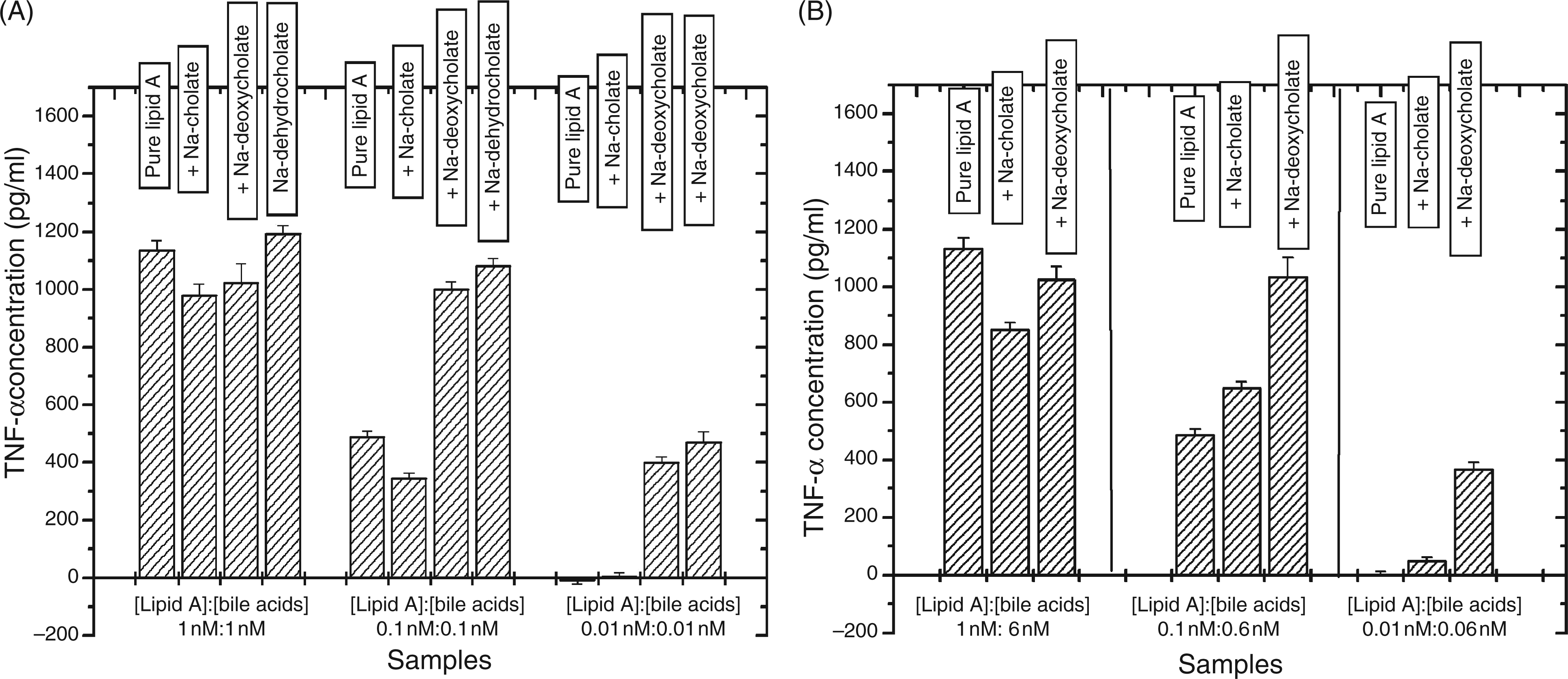
Discussion
The interaction of bile acids with bacterial metabolic products of high physiological relevance, is still poorly understood. Therefore, we have studied this interaction biophysically by using well-defined endotoxins (lipid A, Re- and Ra-LPS) and various bile acids. The presented data are clearly indicative of a hydrophobic interaction of the bile acids with the acyl chains of lipid A, leading to fluidization in both phase states (Figures 1A and 1C), and a small increase of the enthalpy change (Figure 1B). This interpretation is confirmed by the data from the infrared ‘fingerprint’ region, comprising the phosphate and sugar backbone vibrations (Figure 2), and the Zeta potential data (Table 1), which show the absence of any interaction of the bile acids with charged or polar molecular groups within the endotoxin molecules. Rather, the main interaction of the bile acids with endotoxin molecules consists of a considerable change of their aggregate structures, regarding the type of aggregate, as well as the morphologies. There is an essential enhancement of the expression of cubic aggregate structures (Figures 3 and 4), which are already inherently found in enterobacterial endotoxins, 21 and a reduction of their aggregate sizes from sphere-like or ribbon-like types to disc-like and spherical micellar structures (Figure 5).
Concomitantly, we have observed an enhancement in the cytokine-inducing activity of lower lipid A concentrations in MNCs by bile salts (Figures 6 and 7) that may be as a result of improved solubility of lipid A in bile salt complexes, in turn providing an increase in lipid A binding to proteins such as LPS-binding protein, CD14 and to the TLR4-MD-2 receptor on the cell surface. This effect appeared to be rather lipid A-specific, as all other bile salt/acid preparations tested here provided only a slight enhancing effect in Re- and Ra-LPS-induced cytokine production of human MNC. The latter observation is well in line with the findings of Bergamini and colleagues who showed that lower, non-cytotoxic, concentrations of bile salts only slightly influenced LPS-induced cytokine production in MNCs and murine Kupffer cells. 12 Thus, as compared to earlier studies analyzing LPS preparations, we have shown here, for the first time, that bile salts provide an amplification of lipid A-mediated immunostimulation in vitro. This interaction of endotoxin and its lipid A moiety with bile acids is physiologically highly relevant considering that bile acids are known to commute several times between intestine and liver in the ‘enterohepatic circulation’.1,4
In former reports we have investigated the interaction of LPS-binding compounds, for example lactoferrin, albumin and hemoglobin, with endotoxins. We have found that proteins such as lactoferrin, which lead to a decrease of LPS immunostimulatory activity, concomitantly cause a change into a multilamellar aggregate structure connected to an increase in aggregate size.25,26 Proteins which do not change the LPS-stimulatory activity do not change the endotoxin aggregates, neither the type nor the size. 27 Finally, proteins leading to an increase in LPS bioactivity cause an increase in the expression of the cubic aggregate structure connected with a decrease in aggregate size, 28 – 30 which holds true particularly for hemoglobin and part structures.
Change of LPS-induced bioactivity and biophysical parameters when external compounds are added to LPS

ENP, recombinant form of Limulus-anti-LPS factor; LF, lactoferrin; Alb, recombinant human albumin; Hb, hemoglobin.
Footnotes
Acknowledgements
The authors are indebted to the German ministry BMBF (project 01GU0824) for financial support. Part of this work was supported by FY2010 Researcher Exchange Program between JSPS and DAAD, Japan-Germany Research Cooperative Program, Grant-in-Aid for Scientific Research (KAKENHI 21550168), and Clinique La Prairie Research (Clarens, Switzerland).
