Abstract
CCR-2 signaling regulates recruitment of monocytes from the bone marrow into the bloodstream and then to sites of infection. We sought to determine whether CCL-2/CCR-2 signaling is involved in the killing of Staphylococcus aureus by murine bone marrow cells (BMCs). The intermittent link of reactive oxygen species (ROS)–NF-κB/p38–MAPK-mediated CCL-2 production in CCR-2 signaling prompted us to determine whether neutralization of CCR-2 augments the response of murine fresh BMCs (FBMCs) after S. aureus infection. It was observed that anti-CCR-2 Ab-treated FBMCs released fewer ROS on encountering S. aureus infection than CCR-2 non-neutralized FBMCs, also correlating with reduced killing of S. aureus in CCR-2 neutralized FBMCs. Staphylococcal catalase and SOD were also found to play a role in protecting S. aureus from the ROS-mediated killing of FBMC. S. aureus infection of CCR-2 intact FBMCs pre-treated with either NF-κB or p-38-MAPK blocker induced less CCL-2, suggesting that NF-κB or p-38-MAPK is required for CCL-2 production by FBMCs. Moreover, blocking of CCR-2 along with NF-κB or p-38-MAPK resulted in elevated CCL-2 production and reduced CCR-2 expression. Inhibition of CCR-2 impairs the response of murine BMCs to S. aureus infection by attenuation ROS production and modulating the cytokine response.
Introduction
The recruitment of inflammatory cells to sites of injury is mediated by chemoattractive chemokines. There are four different subfamilies of chemokines (CC, CXC, CX3C and C), based on their biochemical, structural and functional properties. Chemokine (C-C motif) receptor 2 (CCR-2) is the receptor for a number of C-C motif chemokines, including CCL-2, which is also known as monocyte chemotactic protein 1 (MCP-1). 1 CCR-2 is also known to be a receptor for additional chemokines, including CCL-7, CCL-8, CCL-13 and CCL-16, 2 and is expressed on the surface of leukocytes from multiple lineages, including monocytes, macrophages, lymphocytes and granulocytes, and its expression is tightly regulated, differing from one cell type to another. 3 CCR-2 is considered a pro-inflammatory mediator in many inflammatory diseases (e.g. rheumatoid arthritis). However, mice lacking CCR-2 develop exacerbated collagen-induced arthritis. 4 CCL-2/CCR2 signaling is involved in several inflammatory diseases such as cresentric glomerulonephritis, 5 pulmonary hypertension, 6 pulmonary fibrosis, 7 atherosclerosis, 8 multiple sclerosis 9 and rheumatoid arthritis, 10 characterized by macrophage infiltration. It has been reported that mice deficient in CCR-2 fail to mobilize monocyte/macrophage populations in response to infections by intracellular bacteria and have impaired bacterial clearance. 11 In mice that lack the Ccr2 gene, the recruitment of macrophages to sites of injury is also impaired. 12 CCL-2 has been shown to be associated with monocytic infiltration in several inflammatory diseases. 13 Its receptor, CCR-2, is essential in mediating macrophage recruitment and host defense against bacterial pathogens. 14 CCL-2, like many chemokines, can be induced by diverse bacterial agents and pro-inflammatory cytokines. 15 The regulation of its receptor CCR-2 was much poorly understood until recent reports of inhibition of CCR-2 expression in human monocytes in vitro by LPS and several other microbial agents, as well as cytokines.16–18
Earlier studies showed that bone marrow cells (BMCs) showed the strongest capacity in both proliferation and phagocytosis among the three populations of macrophages (splenic macrophage, peritoneal macrophages and bone marrow-derived macrophages) under resting conditions. 19 In macrophage studies, macrophage cell lines, including J774A.1, RAW264.7, P388D1 and U937, 20 have been used in earlier studies; however, continuous subculture of these cell lines found loss of gene and impaired macrophage immune functions. Therefore, macrophages from bone marrow, spleen and peritoneum in primary culture are more commonly used in earlier studies. 21 To date, macrophage studies have been performed and validated extensively using BMCs,22,23 but less so with fresh BMCs (FBMCs).
Although there are many advantages to using BMCs in immunological studies, such as their high yield, homogeneity and long lifespan, 22 the features of FBMCs are not fully characterized. Murine macrophage response from the peritoneal cavity requires signals mediated by chemokine receptor CCR-2 during early Staphylococcus aureus infection. 24 Also, the CCR-2 signal pathway is reported to play a functional role in peripheral blood mononuclear cell trafficking in the early phase of S. aureus mastitis. 25 S. aureus is one of the most prominent causes of bacterial infections in humans worldwide. Emergence of methicillin-resistant S. aureus (MRSA) strains has become a global challenge for first-line antibiotic treatment regimens of infections with the pathogen. 26 Failure of vaccine development programs poses an additional threat to public health and urges to a better understanding of the host–pathogen interaction. 27 A key host defense mechanism against S. aureus is phagocyte-mediated killing. In return, the pathogen is well adapted to the human host, evading host immune responses at multiple levels using an arsenal of virulence factors. 28
Precise regulation of chemokine expression, receptor association and subsequent cellular responses likely optimizes protection and minimizes deleterious effects of uncontrolled microbial growth and invasion. Several studies indicated the involvement of CCL-2 in bacterial clearance in a model of peritoneal sepsis. 29 The important role of CCL-2 in monocyte/macrophage-mediated host defense against bacterial infection has been documented by previous studies. CCL-2 overexpression augments bacterial clearance during Streptococcus pneumoniae infection. 30 CCL-2 is also shown to be essential for the survival of mice and macrophage-mediated bacterial killing during Pseudomonas aeruginosa and Salmonella enteric serotype Typhimurium infections. 31 CCL-2 is capable of regulating the pathobiology of various inflammatory disease models of humans such as asthma, multiple sclerosis and atherosclerosis.32,33 Additionally, its potent chemoattractive effect on monocytes and macrophages are enhanced in response to certain antigenic challenge. 34
CCR-2 is the only known functional receptor for CCL-2 expressed in monocytes,15,35 and also has a vital role in host defense to a number of pathogens.36,37 Likewise, the lack of CCR-2 impairs the control of infectious diseases such as from Mycobacterium tuberculosis, Toxoplasma gondii and infection of Leishmania major.37,38 Early immune defense against Listeria monocytogenes infection requires CCR-2-mediated recruitment of inflammatory monocytes from the bone marrow,11,39 and differentiation of recruited monocytes into TNF-α- and inducible NO synthase (iNOS)-producing dendritic cells at sites of infection. In mice lacking CCR-2 or CCL-2, TNF-α-/iNOS-producing dendritic cell recruitment is markedly diminished in spleen, resulting in enhanced in vivo bacterial growth that overwhelms the host. 40
CCR-2/CCL-2 signaling controls the movement of monocytes from the bone marrow into the bloodstream and from the circulatory system into sites of inflammation after injury. 41 The CCR-2/CCL-2 axis is positively associated with osteoclast recruitment, bone resorption and orthodontic tooth movement. 42 Bone loss due to older age and disuse can be attenuated by modulation of the CCR-2 and/or its ligands, which may otherwise contribute to osteoporosis and increases fracture risk. 43 Also, in the absence of estrogen, CCR-2 was demonstrated to be involved in mediating bone loss. 44 In addition, both CCR-2 and CCL-2 were previously shown to be induced during osteoclast differentiation, but little was known about the effects of CCR-2-dependent signaling on bone loss. 45 Ccr2−/− osteoclasts had reduced bone resorption capacity compared with wild type cells and Ccr2 mutant mice are osteoporotic owing to the decreased function and number of osteoclasts in bone. 44 CCR-2 has thus been recognized as an attractive potential target inhibition in the treatment of macrophage-mediated chronic diseases.
However, there is no current study of the involvement of CCL-2/CCR-2 signaling in the intracellular survival of S. aureus by murine fresh BMCs of wild type Swiss albino mice. To clarify the role of CCL-2/CCR-2 signaling in the cellular innate immune response against pathogenic microbes, we here examined the involvement of CCL-2/CCR-2 signaling in the phagocytosis of S. aureus by fresh BMCs and its intracellular survival. CCL-2 interacts with CCR-2 to activate different MAPK cascades that are important for the physiological function of CCL-2 and have been recently shown to be involved in integrin activation and chemotaxis triggered by CCL-2. 46 The MAPKs are recognized to be among the most versatile signaling kinases, which play an essential role in the regulation of cellular functions in multiple cell types in response to a wide range of stimuli. In addition, NF-κB is one of the well-studied transcription factors that mediate the induction of numerous inflammatory cytokines and chemokines. 47 However, the role of different pro-inflammatory cytokines, chemokines, and reactive oxygen and nitrogen species in the activation of different signaling molecules specifically p38-MAPK and NF-κB during acute staphylococcal infection in fresh BMCs remains questionable. As there is an interlink among reactive oxygen species (ROS)–NF-κB/p38–MAPK–CCL-2 production and CCR-2 signaling, it prompted us to determine whether neutralization of CCR-2 regulates the response of murine fresh BMCs after S. aureus infection, utilizing ROS production and cytokine profile in regard to killing of S. aureus. Here, we report that inhibition of CCR-2 exacerbates the murine fresh BMCs response to S. aureus infection via two distinct mechanisms: (i) after utilizing ROS production, and (ii) by regulating the cytokine response on the other. We also suggest the involvement of CCR-2/CCL-2 signaling via NF-κB and/or the p38 MAPK pathway in host defense during acute staphylococcal infection.
Materials and methods
Maintenance of experimental animals
All experiments involving Swiss albino mice were conducted according to the protocols that had been approved by the Institutional Animal Ethics Committee (IAEC), Department of Physiology, University of Calcutta, under the guidance of CPCSEA (Approval Number: IAEC/IV/Proposal/BB-1/2014 dated 26.08.2014), Ministry of Environment and Forest, Government of India. Wild type male Swiss albino mice were used throughout the study.
Preparation of bacteria
The S. aureus strain AG-789 was obtained from Apollo Gleneagles Hospital, Kolkata, West Bengal, India, and was maintained in our laboratory. This bacterial strain is of catalase and coagulase-positive. S. aureus (AG-789) grown overnight in Luria Bertani broth, and was diluted with fresh broth and cultured until mid-logarithmic phase of growth. Bacteria were adjusted to the desired inoculum spectrophotometrically before infection (OD620 = 0.2 for 5.0 × 107cells/ml for S. aureus) and the colony-forming unit (CFU) count of the desired inoculum was confirmed by serial dilution and culture on blood agar. 48 Among the three clinical isolates (AG-886, AG-591 and AG-789) used in earlier studies from our laboratory, it was observed that all of the three strains of S. aureus (AG-886, AG-591 and AG-789) showed the expression of both the TSST-1 and coagulase genes in all phases of their growth, and induction was maximum during the log and stationary phases of their growth. Furthermore, from the biochemical analysis and biotyping for characterization of the S. aureus (AG-789) isolate, it was found that S. aureus (AG-789) was catalase-positive, coagulase-positive, could coagulate bovine plasma and was resistant to methicillin. This MRSA was found to be sensitive to ciprofloxacin, chloramphenicol and azithromycin. The results of the checker board assay showed no synergistic interaction between the antibiotics tested. Antagonism was observed with methicillin interacting with ciprofloxacin or azithromycin. In our laboratory, several other clinical isolates of S. aureus also have been extensively studied in the mouse model of arthritis with short term but non-lethal infection.
Isolation and stimulation of murine FBMCs
The Swiss albino mice were used at 6–8 wk of age and fed standard laboratory chow and water. Murine resident FBMCs were isolated from femurs of Swiss albino mice according to the Marim et al. 23 The femurs from Swiss albino mice were dissected using scissors, cutting through the tibia below the knee joints, as well as through the pelvic bone close to the hip joint. Muscles connected to the bone were removed using clean gauze, and the femurs were placed into a polypropylene tube containing sterile PBS on ice. In a tissue culture hood, the bones were placed in 70% ethanol for 1 min, washed in sterile RPMI 1640 and then both epiphyses were removed using sterile scissors and forceps. Briefly, bone marrow was flushed into a 50-ml conical tube with RPMI 1640, 100 IU/ml penicillin with 100 µg/ml streptomycin, 20% (v/v) FBS a using a 10-ml syringe and a 26-G needle. Cells were then passed through a 23-G needle and cultured in 20 ml RPMI in 95-mm diameter Petri dishes at 37℃ in humidified 5% CO2 in air. 49 FBMCs were cultured 200 µl of culture medium (RPMI-FBS) in a well and incubated under different conditions (with S. aureus, inhibitors, Abs, etc.).
Cell viability assay
Cell viability was determined by the MTT assay. FBMCs were cultured 200 µl culture medium in a well and incubated under different conditions (with S. aureus, inhibitors, Abs, etc.). Ten µl MTT (10 mg/ml) were added to each well, and the mixture was incubated for 4 h at 37℃. SDS was then added to the mixture at 10% and incubated for 3 h. Culture medium was then replaced with DMSO to dissolve formazan crystals. After shaking at room temperature (23–25℃) for 10 min, absorbance of each well was determined at 570 nm using a microplate reader (Bio-Rad, Richmond, CA, USA). Samples were measured in three replicates and each experiment was repeated at least twice. 50
Immunophenotyping of BMCs by FACS analysis
The cell suspension generated thereafter was considered as FBMC. CD11b, known as the integrin alpha M chain, is implicated in different adhesive interactions of monocytes, macrophages and granulocytes. 51 It has been shown that the spleen and peripheral blood monocyte progenitors share phenotypic markers with bone marrow progenitors, but differ in their expression of CD11b, which was low in bone marrow but high in the periphery. It was also reported that in the bone marrow populations, these peripheral populations contain common progenitors for osteoclast, macrophages and dendritic cells. These progenitors share phenotypic markers with the bone marrow progenitor, but differ in their expression of CD11b. 52 Therefore, characterization of FBMC by CD11b staining seems valid. Further phenotypic studies based the expression of F4/80 and CD11b could suggest that they might present a monocyte/macrophage phenotype; however, we did not do it in this case. Therefore, these FBMCs were definitely monocyte/macrophages and devoid of neutrophils. FBMCs were used, when > 90% of cells were positive for CD11b, a monocyte/macrophage marker.
FBMCs, pooled from six mice were used for immunolabelling. Trypsinized cells, washed with PBS, were incubated for 30 min with FITC-conjugated IgG2b rat anti-mouse CD11b (1 µg/ml, FITC-conjugated; BD Bioscience, Oxford, UK) on ice. After washing twice with PBS containing 1% FCS, cells were filtered through a 70-µm cell strainer (BD Biosciences) and were analyzed on a fluorescence-activated cell sorter (FACS AriaIII; BD Biosciences) for measurement of cell size (forward scatter) and complexity or granularity (side scatter). Results were considered positive if 20% or more of the cells expressed a particular Ag. 19 Importantly, it has been shown earlier that bone marrow-derived macrophages obtained from fresh or frozen BMCs did not differ in expression of surface molecules, such as CD80 and CD86, either in response to LPS or live intracellular bacteria Legionella pneumophila; therefore, description of CD80/CD86 surface molecules after S. aureus infection is not relevant. Samples of the cell cultures were tested for viability by MTT assay and were >93 % viable.
Blocking Ab reagents and culture conditions
For CCR-2/CCL-2R blocking assays, rabbit polyclonal Abs against CCR-2 (Cat. No: orb 10487; Biorbyt, Cambridge, UK) or control IgG isotype polyclonal Ab (Cat. No: ab 37355; Abcam, Cambridge, UK) were added at 5 µg/ml to isolated BMCs and were incubated for 1 h at 37℃ in a 5% CO2 incubator.53,54 This anti-CCR-2 polyclonal Ab is reactive against human (Swissprot: P25024), mouse, rat, cow, dog and rabbit, and was recommended as quite homologous to murine CCR-2. Then, both anti-CCR-2 Ab-treated or untreated murine fresh BMCs (5 × 106 cells/ml) were infected with S. aureus (5 × 106 CFU/ml) for 30 min, 60 min and 90 min at 37℃ in 5% CO2 incubator. The same rabbit polyclonal Abs against CCR-2 (Cat. No: orb 10487; Biorbyt) were also used in a separate in vivo study for neutralization of endogenous CCR-2 in Swiss albino mice.
Assays for colony-forming ability of engulfed bacteria and determination of survival index
For the in vitro study, cells were combined from a minimum of six mice to obtain the requisite amount of individual cells (5 × 106/ml), counted using a hemocytometer and were used for each experiment. 55
Murine FBMCs (5 × 106 cells/ml), were mixed with S. aureus (5 × 106 CFU/ml) in a 1:1 cell:bacterium ratio and incubated for 30 min, 60 min and 90 min at 37℃ in a cell culture incubator in the presence or absence of anti-CCR-2 Ab, as well IgG isotype control Ab. Extracellular S. aureus were removed by washing the suspension four times in RPMI by centrifugation at 250 g at 4℃ for 5 min. After centrifugation, cell culture supernatants were collected and stored at –80℃ for further assay. Then, the pellets were disrupted in sterile water containing 0.01% BSA by vigorously vortexing to release intracellular bacteria in the lysate. The lysate–containing bacteria were plated at serial dilutions on mannitol agar plates. The plates were incubated at 37℃ for 1–2 d and the number of colonies was determined. 49
In addition to the percentage of viable intracellular bacteria at time t was calculated according to the formula: Survival index = (CFU/ml)t/(CFU/ml)0 × 100. 56 It has been previously demonstrated that extracellular bacteria are sufficiently removed by washing and that at least 98% of the remaining bacteria are intracellularly located.
Labeling of S. aureus with FITC
Briefly, bacteria were grown overnight (16–18 h) at 37℃ in nutrient broth, then diluted 1:100 in fresh nutrient broth and grown at 37℃ with shaking for 3 h. Bacteria were then collected by centrifugation and washed twice in PBS and then bacterial density was adjusted spectrophotometrically. Bacteria were then labeled by incubation with 0.1% FITC (Cat. No: ab 145325; Abcam) in 50 mM sodium carbonate buffer, pH 9.0, for 1 h at 37℃, protected from light. 24
Immunofluorescence study for engulfment of bacteria by resident FBMCs
FBMCs were suspended in RPMI 1640 medium supplemented with 10% FBS, 100 IU/ml penicillin and 100 µg/ml streptomycin A at prior to infection and 5 × 105 cells were seeded in six-well plates (total volume: 3 ml) (Invitrogen, Carlsbad, CA, USA). Two hundred µl FITC-labeled S. aureus was mixed with equal volumes of murine FBMCs and incubated for 90 min at 37℃. However, extracellular S. aureus were removed by washing the suspension in RPMI (note that we were unable to use antibiotics to kill bacteria present outside of macrophages because engulfed bacteria died quickly during the period necessary for the action of antibiotics). After the invasion procedure, 2–4% paraformaldehyde fixative solution was added and incubated for 20 min at room temperature. Then, the coverslips containing fixed cells were washed twice with PBS. After rinsing, FBMC cell nuclei were stained using Nuclear yellow (Hoechst S769121; ab13903) at 250 µg/ml dilution for 2 h. Finally, specimens were mounted on slides in 10% glycerol and visualized under a 60 × oil immersion lens. The excitation and emission filters for FITC were 495/519 nm and for Hoechst were 355/495 nm, respectively. Images were acquired by an Olympus Laser Confocal Scanning Microscope (Spectral type inverted Microscope IX81) and then analyzed with Olympus FV1000 viewer software. Co-localization was accessed by Pearson’s correlation coefficient. Higher co-localization indicates more S. aureus engulfed within the BMCs. 24
Assay of hydrogen peroxide production
Hydrogen peroxide (H2O2) released in the supernatant and lysate at 30, 60 and 90 min of phagocytosis by BMCs infected with S. aureus pre-treated with or without exogenous catalase (catalogue no SC-50508) (166.7 µg/ml) was measured according to the method as described earlier, with slight modification.48,57 Briefly 70 µl lysate of different groups of BMCs, 20 µl HRP (500 µg/ml) and 70 µl of phenol red (500 µg/ml) were added in each of the microtiter plates and was allowed to react at 37℃. The reaction was stopped by adding 25 µl 2 N NaOH and the absorbance was read at 620 nm. A standard H2O2 curve was plotted and the amount of H2O2 released in lysate was evaluated and expressed in µM/106 cells.
Assay of superoxide anion production
Superoxide anion (O2–) release assay measures the change in color of cytochrome C (cytC), when reduced by O2– released from the stimulated BMCs infected with S. aureus pre-treated with or without exogenous SOD (catalogue no SC-11407) (2.78 µg/ml). The difference between the amounts of cyt C reduced in the presence and absence of SOD represents the amount of O2– generated during the incubation. The instrument was blanked on the samples containing cytC plus SOD and instructed sequentially to read the absorbance of all the samples at 550 nm in reference to the blank. The absorbances read in this manner are closest to the true SOD inhibitable cytC reduction. Cell supernatant and lysate obtained after the time-dependent phagocytosis by BMCs, infected with S. aureus, were incubated in the presence of cytC (100 µl at 2 mg/ml). The production of O2– was monitored spectrophotometrically at 550 nm with reference to the blank.49,57 The amount of O2– production was calculated by the following formula: micromoles of superoxide anion = (mean absorbance at 550 nm × 15.87).
Assay of NO production
The amount of NO released in the supernatant and lysate was determined by the Griess reagent assay. Supernatant and lysate (50 µl), obtained after dependent phagocytosis by BMCs infected with S. aureus, were incubated with 40 µM Tris (pH 7.9) containing 40 µM of the reduced form of NADP, 40 µM flavin adenine dinucleotide and 0.05 U/ml nitrate reductase at 37℃ for 15 min. Then, reduced samples were incubated with an equal volume of Griess reagent consisting of sulfanilamide [0.25% (w/v)] and N-1-naphthylethylenediamine [0.025% (w/v)] for 10 min and the absorbance at 550 nm was measured. The total nitrate/nitrite concentration was determined by comparison with a reduced NaNO3 standard curve. 53
Intracellular ROS production by flow cytometry
Flowcytometric data acquisition and analysis were performed on a BD FACS Verse. The flow cytometer was equipped with an iron laser with excitation at 488 nm and 15-mW output power. A single cell suspension was prepared with a density of 105–106 cells/ml and a minimum of 10,000 cells were measured. Non-fluorescent 2’,7’-dichlorofluorescin diacetate (DCFH-DA) that upon esterase activity within the cells become DCFH, which, after oxidation by ROS, converted to fluorescent product (DCF). The amount of DCF was proportional to the amount of ROS present inside the cell.
Briefly, 20 mM DCFH-DA stock solution prepared in DMSO was diluted in the cultured medium from different groups of BMCs, to yield a 20-µM working solution. Then the cells were incubated with for 30 min, in the dark, at 37℃. Finally, cells were suspended in PBS and ROS generation was measured by determining the fluorescence intensity at 530 nm. 53
TNF-α, IFN-γ, IL-6, IL-10, IL-1β and CCL-2 ELISA assays
Sandwich ELISA was used to determine cytokine concentrations from cell culture supernatant. Supernatants from different groups were normalized to the protein content by the method of Lowry et al. 55 before the assay, and the levels of major pro-inflammatory cytokines—TNF-α, IFN-γ, IL-6, IL-1β and CCL-2 along with an anti-inflammatory cytokine IL-10—were determined as per the manufacturer’s guidelines, in a BioRad ELISA Reader at 450 nm (Raybiotech, Norcross, GA, USA). The reproducibility of cytokine kits are intra-assay coefficient of variation (CV) < 10% and interassay CV < 12%.
Western blot analysis for CCR-2 expression
Western blot analysis for CCR-2 expression was performed by standard methods. In brief, the cell lysate from different groups of BMCs, were lysed with RIPA-NP40 and normalized to the protein content following the method of Lowry et al. 55 Sixty µg of the lysate was separated on a 10% SDS-PAGE gel and blotted onto nitrocellulose membrane. The membranes were blocked with 5% BSA in TBST for 3 h at room temperature, washed and incubated with primary anti-mice CCR-2 Abs in 1/500 dilution overnight at 4℃. The membranes were washed with TBST and incubated with the appropriate HRP-conjugated secondary Ab in 1/5000 dilution for 1 h at room temperature. Detection of antigen was performed using the enhanced chemiluminescent detection method (Thermo Scientific, Waltham, MA, USA). We used β-tubulin as loading control for Western blot to ensure equal loading throughout the gel as it is a housekeeping gene that exhibits high level, constitutive expression in the sample. It also has a different molecular mass than our protein of interest, i.e. CCR-2, to help distinguish between both bands. 24
Preparation of whole staphylococcal cell lysate for demonstration of staphylococcal catalase and SOD enzyme activity
The bacteria that survived after 30, 60 and 90 min of phagocytosis by FBMCs, as obtained from the plates of respective incubation times, were washed twice in PBS, vortexed and finally diluted to an OD at 600 nm of 0.6 and re-suspended in lysis buffer. 58 One hundred µl of this crude bacterial lysate was used to estimate the whole staphylococcal cell catalase and SOD enzyme activity. Catalase activity was determined spectrophotometrically by measuring the decrease in H2O2 concentration at 240 nm. Catalase activity was expressed in terms of mmol/min/mg protein.
For, SOD enzyme activity assay, 100 µl of the bacterial crude lysate was mixed separately with 1.5 ml of a Tris-EDTA-HCl buffer (pH 8.5), then 100 µl of 7.2 mmol/l pyrogallol was added and the reaction mixture was incubated at 25℃ for 10 min. The reaction was terminated by the addition of 50 µl of 1 M HCl and measured at 420 nm. The activity was expressed as unit/mg protein. 59
Assay of catalase activity of 3 amino-1,2,4-triazole pre-treated or non-treated FBMC infected with S. aureus with or without anti-CCR-2 Ab
One mM 3 amino-1,2,4-triazole (ATZ) solution, an intracellular BMCs catalase inhibitor, was added to the FBMCs, incubated at 37℃ for 30 min and washed thoroughly with PBS to remove excess ATZ from the medium. Then, catalase enzyme activity in supernatant or in lysate of ATZ pre-treated or non-treated FBMCs after infection with the recovered S. aureus from different groups was determined spectrophotometrically by measuring the decrease in H2O2 concentration at 240 nm, as described in an earlier section. Catalase activity was expressed in terms of mmol/min/mg protein. 60
Assay of SOD activity of diethyl-dithiocarbamic acid pre-treated or non-treated FBMCs infected with S. aureus with or without anti-CCR-2 Ab
One mM diethyl-dithiocarbamic acid (DDC) solution, an intracellular SOD inhibitor of BMCs was added to the fresh BMCs, incubated at 37℃ for 30 min and washed thoroughly with PBS to remove excess DDC from the medium. Then, SOD activity in supernatant, as well as in the lysate of DDC pre-treated or non-treated FBMCs after infection with S. aureus, was determined as mentioned earlier. 59 The activity was expressed as unit/mg protein.
Immunoblot analysis for SOD
Bacterial suspensions were lysed with 1 mM disodium EDTA, pH 7.2, 0.5 mM PMSF and sonicated on ice at 8 µm for 90 s (six 15-s bursts with 15-s cooling periods) and then centrifuged at 16,000 g for 45 min at 4℃. One hundred µl of this crude bacterial lysate was used to estimate the whole staphylococcal cell SOD expression. Bacterial cell lysate samples were re-suspended in SDS-PAGE sample loading buffer, subjected to SDS-PAGE and transferred onto BioTrace PVDF membrane, as described elsewhere. 61 Membranes were probed at room temperature with primary Abs against catalase (Cat. No. SC50508; Santa Cruz Biotechnology, Santa Cruz, CA, USA) and SOD (Cat. No. SC11407; Santa Cruz Biotechnology) at a 1:3000 dilution, and immunoreactivity was detected with a goat anti-rabbit IgG HRP-conjugated Ab (Biorbyt). Blots were developed with a chemiluminescent substrate (Thermo Scientific) according to the manufacturer’s instructions. We used β-tubulin as a loading control to ensure equal loading throughout the gel as it is a housekeeping gene that exhibits high-level, constitutive expression in the sample.
RNA extraction and PCR
S. aureus strains were extracted by the cetyltrimethylammonium bromide method after pre-treatment of bacteria with lysostaphin (1 mg/ml) for 1 h at 37℃ in Tris/EDTA/sucrose. 62 Total RNA was prepared by the method of Kornblum et al. 63 Finally, RNA samples were dissolved in 30 µl RNase-free water. RNA integrity was tested with electrophoresis on 1% agarose gel. RNA quantification was performed measuring the absorbance at 260 nm. Nucleic acid purity was assessed measuring A260/A280 ratio (acceptable ratio was between 1.8 and 2.0).
cDNA synthesis using RT-PCR
A sufficient quantity of RNA thus obtained was used to synthesize first-strand cDNA by using the RevertAid H Minus First Strand cDNA synthesis kit (Thermo Scientific) (lot no. 00110899, Cat. No. K1632). The 20 µl reaction mixture, according to the manufacturer’s instructions, contained 11 µl RNA templates, 1 µl primers (equivalent to 15–20 pmol), 4 µl 5 × reaction buffer, 1 µl RiboLock RNase inhibitor (20 U/µl), 2 µl 10 mM deoxynucleoside triphosphate mixture and 1 µl reverse transcriptase enzyme (200 U/µl). The nucleotide sequence of the forward primer for the SODM (sodM) was CCG GAA GCG ATG AGG ATG TCA GTC, and the sequence for the reverse primer was TGC CCC ACT GCG CTT TGA TGT C. 64 The nucleotide sequence of the forward primer for the SODA (sodA) was TGC ACG CTT TGG TTC AGG TTG GG, and the sequence for the reverse primer was GCG CCA ATG TAG TCA GGG CGT TTG. 64 For the catalase gene (katA) a primer set comprising the nucleotide sequence of the forward primer for the (katA) 5′-ATGTCACAACATGATAAAAA-3′ and the sequence for the reverse primer 5′-TTATTTTTTAAAGTTTTCGTA-3′ were used. 65 The primers were designed using S. aureus MSHR1132 (GenBank accession number FR821777.2) catalase as a reference.
PCR was carried out in a 50 µl volume containing 50 ng cDNA with reagents and protocols supplied by the manufacturer. Thermocycler reaction conditions were 1 min at 94℃, 1 min at 52℃, and 1 or 1.5 min at 72℃ for 30 cycles. All PCR amplifications included preliminary denaturation at 94℃ for 10 min and a final incubation at 72℃ for 10 min. 66 The PCR products were run on 2% agarose gels containing 0.01% ethidium bromide and viewed in a Bio-Rad Gel Doc Imager. The images were analyzed using the Image Lab software from Bio-Rad Laboratories.
Immunoblot analysis of CCR-2 expression, NF-κB expression and CCL-2 chemokine production after blocking of intracellular NF-κB and p38 MAPK
To elucidate the roles of NF-κB and p38 MAPK on CCR-2 expression, as well as CCL-2 release, pyrrolidine dithiocarbamate [PDTC; Cat No. SC-203224 (Santa Cruz Biotechnology)] an inhibitor of NF-κB activation, and pyridinyl imidazole (Cat. No. SB203580; Santa Cruz Biotechnology) an inhibitor of p38 MAPK activation, were separately added to the cultured FBMCs 30 min prior to S. aureus infection in the presence of anti-CCR-2 Ab.67,68 After 30 min, 60 min and 90 min of phagocytosis, followed by centrifugation, the cell supernatants were used for CCL-2 ELISA assaym 69 and cell lysates were prepared for CCR-2 and NF-κB immunoblot analysis using primary Abs using anti-CCR-2 and anti-NF-κB (p65) (Abcam) as mentioned previously. 70
Transwell migration assay
This assay was performed in 6.5-mm Transwell plates with 8-µm pore inserts. FBMCs (1 × 105 cells) were re-suspended in 100 µl chemotaxis buffer (RMPI 1640 plus 0.02% BSA). In this experiment, cells were first pre-treated with IgG isotype or anti-CCR-2 Ab for 30 min at 37℃ prior to S. aureus infection. Cells were added to the upper chamber, and 600 µl migration medium, with or without CCL-2 as chemotactic factors (10 ng/ml), 71 was added to the lower chamber. Cells were allowed to migrate through the insert membrane for 3 h at 37℃ under a 5% CO2 atmosphere. The inserts were then washed with PBS, and non-migrating cells remaining on the upper surface of the insert were removed with a cotton swab. The migrated cells on the insert were fixed, stained with Giemsa, and mounted on glass slides. Migration was measured visually by counting of cells using a light microscope at 40 × magnification. The mean number of cells in 10 randomly chosen microscopic fields was calculated for each treatment. Results were expressed as the mean number of migrated cells/10 microscopic fields at 40 × magnification. 72
In vivo S. aureus infection of either anti-CCR-2 Ab-treated or IgG isotype Ab pre-treated mice and clearance of bacterial load from blood, spleen and synovial tissue
Most of the earlier studies used MC21 as the neutralizing Ab to CCR-2 in vivo,73,74 but we used the same rabbit polyclonal Ab against CCR-2 (Cat. No. orb 10487; Biorbyt) to neutralized CCR-2 in vivo, as well as in vitro study to demonstrate whether this Ab could also reduce inflammation due to S. aureus infection by determination of bacterial burden in blood, spleen and synovial tissue. Swiss albino mice were pre-treated with either anti-CCR-2 Ab or control IgG isotype (500 µg per mouse) in PBS the day before S. aureus infection. 73
We did not find any reference in favor of the dose of the antiCCR-2 Ab purchased from Biorbyt (Cat. No. orb 10487), and we used 500 µg per mouse of this Ab as opposed to the in vivo dosing of the control IgG isotype. This approach was based on the earlier in vivo study of Vergunst et al., 75 where treatment with CCR2-blocking Ab reduced the levels of free CCR2 on CD14+ monocytes by at least 57% and up to 94%, demonstrating the biologic activity of the compound in patients with active rheumatoid arthritis. 75 A separate dose-dependent study using this antiCCR-2 Ab purchased from Biorbyt (Cat. No. orb 10487) will give a better indication for endogenous neutralization of CCR-2 after determining the CCL-2 level.
Next, live S. aureus (106 CFU/mouse of average body mass of 20 g) were administered in 0.1 ml saline via the tail vein. The mice were monitored daily. Blood was obtained on d 3, 9 and 15 after infection by retro-orbital sinus bleeding before sacrifice. The blood from each infected mouse was plated on mannitol salt agar selective media. Then, the mice were sacrificed, and spleen tissues were aseptically removed and homogenized with 3 ml sterile RPMI-1640. Synovial tissues from joints of each mouse were removed, weighed and homogenized in RPMI-1640 medium (1 ml per 100 mg joint mass). After homogenization, all tissue samples were diluted and plated in triplicate on mannitol agar, and the results were expressed as the number of CFU per ml of blood and per whole organ. To avoid false-positive results due to contamination, an isolate was considered positive when 15 or more S. aureus colonies were present. 59
Sample preparation for estimation of serum CCL-2
Blood was obtained on d 3, 9 and 15 after infection by retro-orbital sinus bleeding before sacrifice. The blood (0.2 ml) was transferred to micro-centrifuge tubes and allowed to clot at 4℃. Blood samples were then centrifuged at 300 g for 5 min at 4℃. The supernatant, pale yellow-colored serum, was pipetted out carefully with the help of micropipettes into fresh micro-centrifuge tubes, labeled and stored at –80℃ for CCL-2 analysis. In each experiment, the mice were coded to ensure that the observer was blinded. Serum from different groups were normalized to the protein content by the Bradford method before the assay and levels of CCL-2 were determined by Sandwich ELISA, according to the manufacturer’s instructions, in a BioRad ELISA Reader. 76
Statistical analysis
Isolated FBMCs from six mice were combined to obtain the requisite number of macrophages (5 × 106 cells/ml) and the different parameters were measured. This was repeated three times for each parameter and then the mean values of these triplicate experiments were taken for calculation. Data are expressed as mean ± SD. One-way model 1 ANOVA was performed between the groups. In ANOVA, observed variance is partitioned into components owing to different explanatory variables. A level of P < 0.05 was considered significant. Significant differences of the means between the groups were performed by one-way ANOVA. Scheffe’s F-test was done as a post-hoc test for multiple comparisons of means of different groups when a significant F value was found. A Scheffe’s F-, post-hoc test for multiple comparisons of the different groups was done when significant P-values were obtained. A P-value < 0.01 was considered significant. All analyses were done using OriginPro 8 software (Origin Lab, Northampton, MA, USA)
Results
Immunolabeling of BMCs by FACS analysis
BMCs were studied for surface antigen expression using monoclonal Abs directly conjugated with fluorochrome. CD11b-positive immune phenotype was observed in > 80% of the gated area (Figure 1). FBMCs were tested for viability by MTT assay and > 93% were found to be viable. It is reported that the spleen and peripheral blood monocyte progenitors share phenotypic markers with bone marrow progenitors, but differ in their expression of CD11b, which is low in bone marrow but high in the periphery. It is also reported that in the bone marrow populations, these peripheral populations contain common progenitors for osteoclasts, macrophages and dendritic cells. As these progenitors share phenotypic markers with the bone marrow progenitor, but differ in their expression of CD11b, CD11b staining of FBMCs definitely indicates monocyte/macrophages lineages and no neutrophils.
Immunophenotyping of BMCs by surface antigen marker. BMCs were immunolabeled by their specific surface antigen marker, conjugated with a fluorochrome. A gate was selected in the combination of forward scatter (FSC) and side scatter (SSC), and analysis was performed on cells with the most appropriate promyelocyte gate (intermediate-to-high SSC and intermediate-to-high FSC).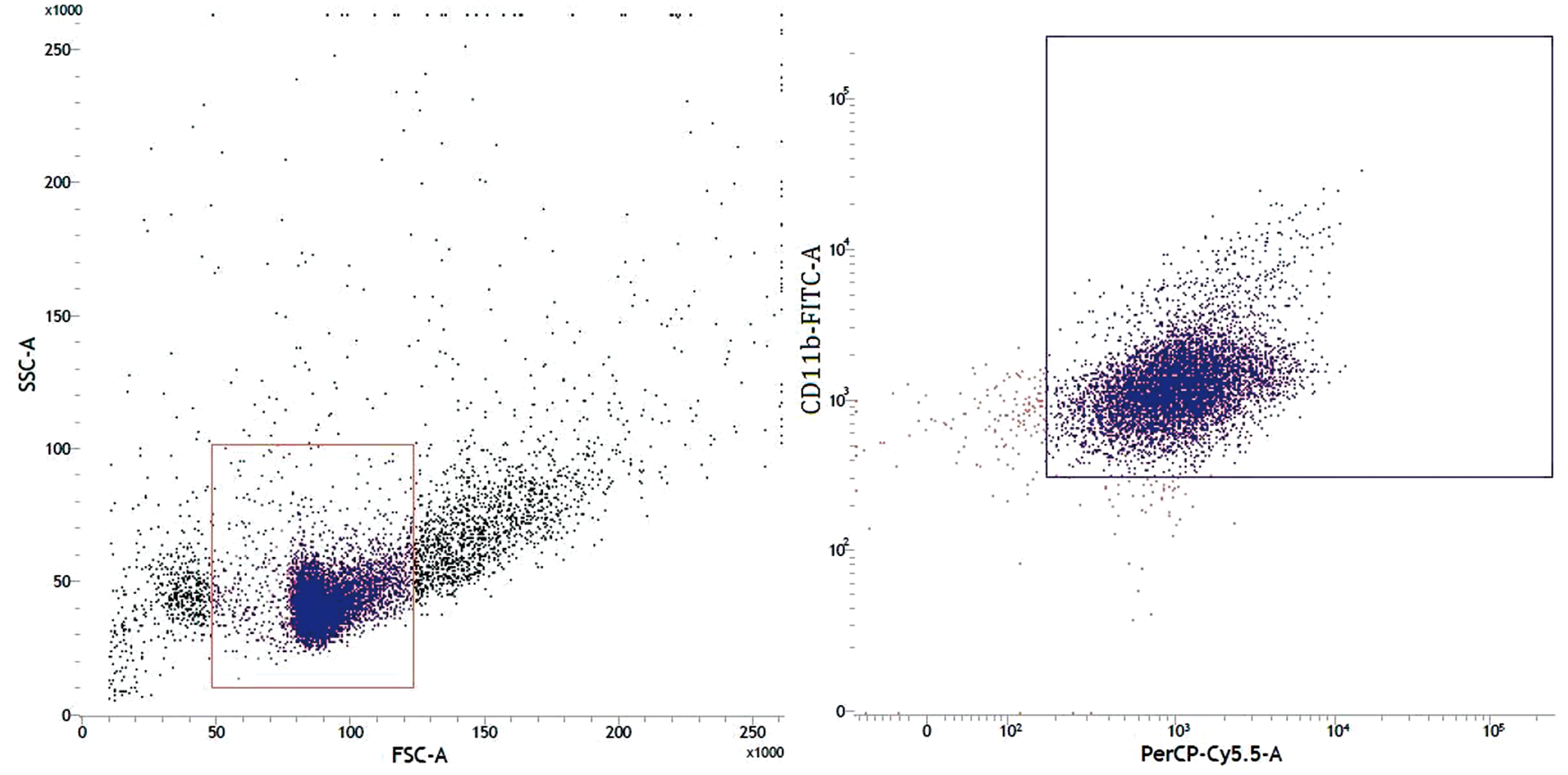
Phagocytic activity of murine FBMCs after neutralization of CCR-2 during acute S. aureus infection
Phagocytic activity of murine FBMCs with or without neutralization of cell surface CCR-2 during acute S. aureus infection by plate count method.
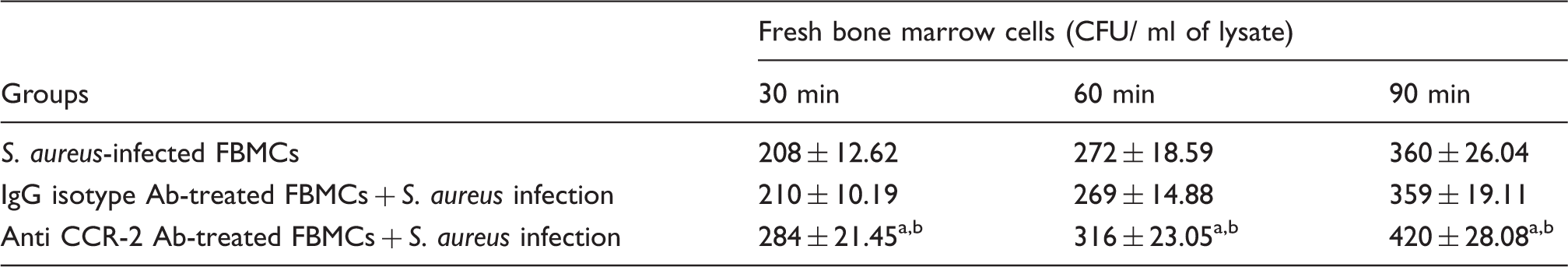
S. aureus was allowed to interact with murine FBMCs in the presence or absence of anti-CCR-2 Ab and incubated for 30, 60 and 90 min at 37℃. Cells were then lysed, plated and incubated overnight to obtain the CFU of the intracellularly surviving bacteria the next day. Values are expressed as mean ± SD of three independent experiments. aP < 0.05 with respect to S. aureus-infected group. bP < 0.05 with respect to IgG isotype pre-treated fresh BMCs prior to S. aureus infection.
Determination of intracellular killing of S. aureus
Intracellular killing of S. aureus within murine FBMCs with or without neutralization of cell surface CCR-2 during acute S. aureus infection.

Murine FBMCs (5 × 106 cells/ml) were allowed to adhere to the glass slides for 1 h at 37℃. S. aureus (5 × 106 cells/ml) were allowed to interact with the adhered BMCs initially for 1 h and were then re-incubated in HBSS–BSA for the time indicated. The results represent the survival index (%) (mean ± SD) of three independent experiments. aP < 0.05 with respect to S. aureus-infected group. bP < 0.05 with respect to IgG isotype pre-treated fresh BMCs prior to S. aureus infection.
Immunofluorescence study
In order to investigate the internalization of S. aureus into murine FBMCs pre-treated with or without anti-CCR-2 Ab, we performed an immunofluorescence study. S. aureus cells were found to be efficiently engulfed by CCR-2-intact BMCs at all time points, as indicated by a time-dependent increase in co-localization signals in this group. However, low co-localized signals in CCR-2-blocked FBMCs after time-dependent phagocytosis of S. aureus indicate less engulfment in this group (Figure 2). Also, co-localized signals in CCR-2 FBMCs containing engulfed S. aureus were found to be significantly higher at 30, 60 and 90 min of phagocytosis as compared with CCR-2-blocked FBMC.
Immunofluoroscence analysis of engulfment of S. aureus by murine FBMCs. Immunofluoroscence analysis showed that S. aureus are engulfed by FBMCs 30 min post-infection. Panel [1] shows the merged image (where FBMCs are in blue and internalized bacteria are in green) where the internalized bacteria were brightly fluorescent after engulfment within FBMCs. Panel [2] shows the co-localization image in a scatter plot. Panel [3] shows the fold changes in fluorescent activity in terms of Pearson’s coefficient from different sets of groups. (a, c, e) CCR-2-intact FBMCs infected with S. aureus at 30, 60 and 90 min, respectively; (b, d, f): CCR-2 blocked FBMCs infected with S. aureus at 30, 60 and 90 min, respectively. Results are shown as mean ± SD of three independent experiments. #P < 0.05 with respect to S. aureus-infected group.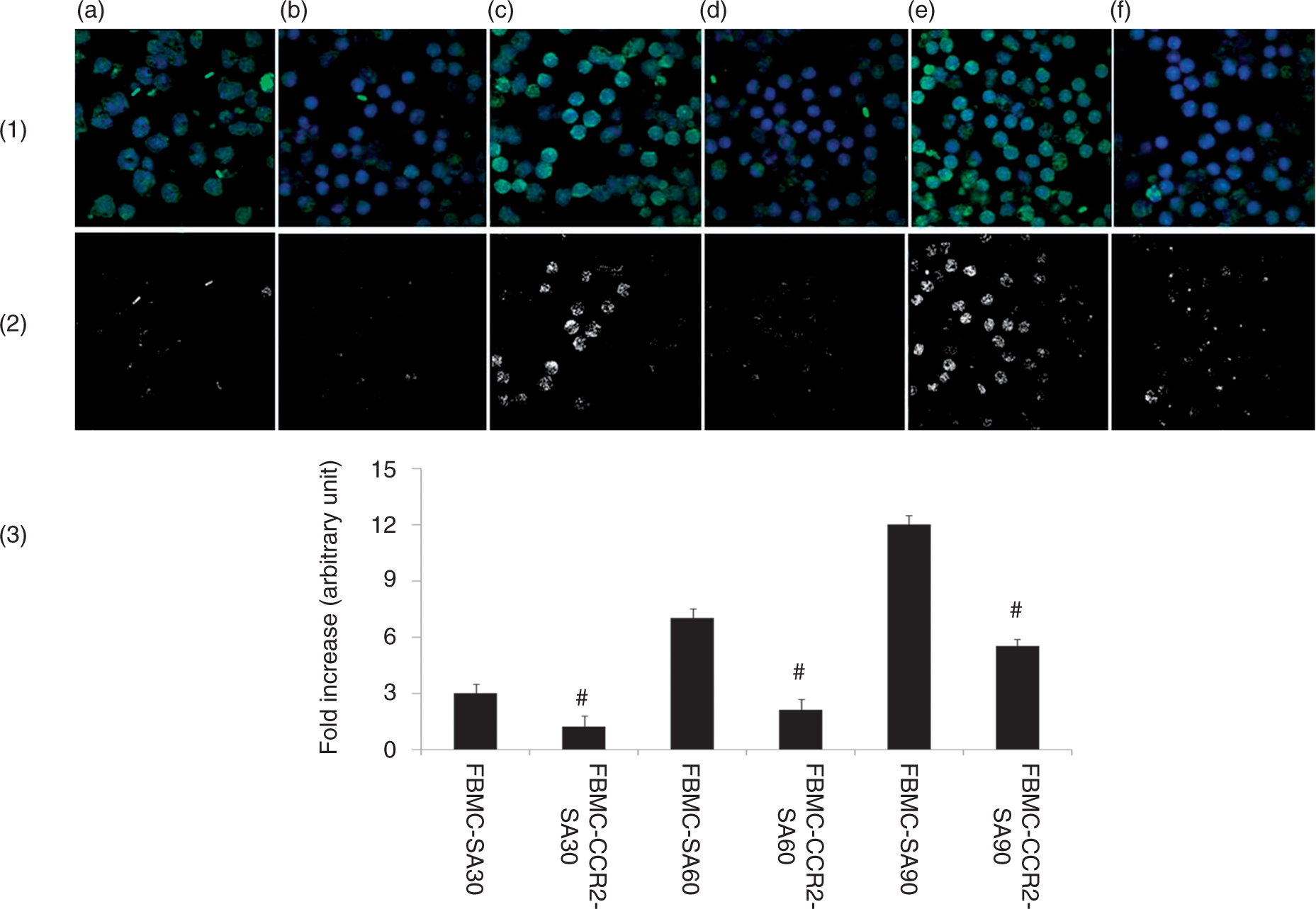
Alteration in H2O2 release by murine FBMCs infection with S. aureus due to CCR-2 blocking
We tested whether H2O2 produced by FBMCs in supernatant or in cell lysate was CCR-2-dependent during short-term S. aureus infection. Both anti-CCR-2 Ab-untreated and anti-CCR-2 Ab-treated BMCs were exposed to S. aureus and incubated for 30, 60 and 90 min at 37℃. Anti-CCR-2 Ab treatment significantly (P < 0.05) inhibited H2O2 production, both in supernatant and lysate, by S. aureus-infected BMCs at all three time points after infection compared with the Ab-untreated and infected FBMCs (Figure 3a, b). However, no such differences in H2O2 production were observed in S. aureus-infected BMCs with or without IgG isotype control Ab treatment. However, S. aureus-infected BMCs, pre-treated with anti-CCR-2 Ab, showed a significant decrease (P < 0.05) in H2O2 production at all three time points, with respect to IgG isotype-treated BMCs during S. aureus infection.
Alteration in H2O2, O2– anion and NO release by murine FBMCs infected with S. aureus after CCR-2 blocking. FBMCs, incubated with or without anti-CCR-2 Abs for 30 min and infected with S. aureus for 30, 60 and 90 min. Supernatant and lysate were prepared as mentioned in the ‘Materials and methods’. H2O2 release in (a) supernatant and (b) lysate, O2– anion release in (c) supernatant and (d) lysate, and NO release in (e) supernatant and (f) lysate are expressed in terms of μM/106 cells. Results are shown as mean ± SD of three independent experiments. *P < 0.05 compared with uninfected control group; #P < 0.05 with respect to S. aureus-infected group; $P < 0.05 with respect to IgG isotype pre-treated FBMCs prior to S. aureus infection.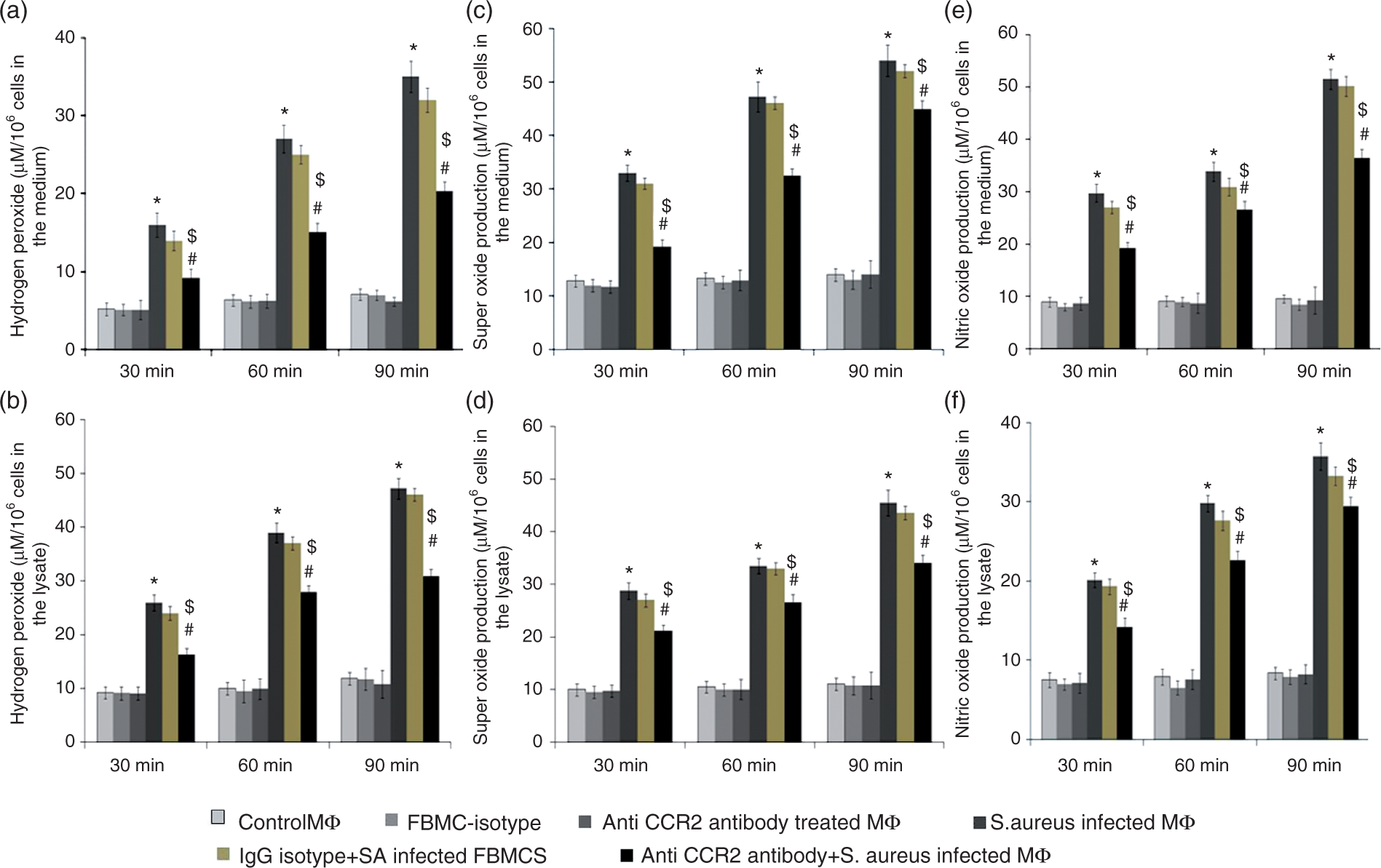
Alteration in O2– free radical release by murine FBMC infection with S. aureus due to CCR-2 blocking
Murine FBMCs were infected in vitro with S. aureus, as described earlier, or left uninfected. At 30, 60 or 90 min after infection, the amount of O2– produced was estimated. Both in culture supernatant (Figure 3c) and in lysate (Figure 3d), S. aureus-infected BMCs produced significantly (P < 0.05) higher amounts of O2– than uninfected BMCs after 30, 60 and 90 min infection, whereas infection of CCR-2-neutralized BMCs after S. aureus infection produced lower levels of O2– than BMCs infected with S. aureus alone at also 30, 60 and 90 min in culture medium, as well as in lysate. However, no such differences in O2– production were observed in S. aureus-infected BMCs with or without IgG isotype control Ab treatment. However, S. aureus-infected BMCs, pre-treated with anti-CCR-2 Ab showed significant decrease (P < 0.05) in O2– production at all three time points, with respect to IgG isotype-treated BMCs during S. aureus infection.
Alteration in NO production by murine FBMCs infection with S. aureus due to CCR-2 blocking
To investigate the crucial role of CCR-2 in acute in vitro infection of FBMCs with S. aureus for 30, 60 and 90 min, we determined the NO production in the supernatant and lysate (Figure 3e, 3f) at 30, 60 and 90 min in the presence or absence of anti-CCR-2 Ab. A significant decrease in NO production in the lysate and supernatant (P < 0.05) was found, owing to CCR-2 blocking in S. aureus-infected FBMCs after 30, 60 and 90 min compared with only S. aureus-infected BMCs. However, no such differences in NO production were observed in S. aureus-infected BMCs with or without IgG isotype control Ab treatment. However, S. aureus-infected BMCs that had been pre-treated with anti-CCR-2 Ab showed a significant decrease (P < 0.05) in NO production at all three time points, with respect to IgG isotype-treated BMCs during S. aureus infection.
Alteration in H2O2 and O2– free radical release by adding exogenous pure SOD and catalase in the medium
To find out the specificity of reaction by H2O2 and O2–, we investigated H2O2 and O2– production during acute staphylococcal infection in the presence of exogenous catalase and SOD enzyme, respectively. A significant (P < 0.05) decrease in absorbance values was observed for H2O2 production in CCR-2-neutralized S. aureus-infected FBMCs pre-treated with exogenous catalase compared with both S. aureus-infected FBMCs and IgG isotype pre-treated S. aureus-infected FBMCs along with catalase treatment suggested the specificity of the reaction by H2O2 only. A significant (P < 0.05) decrease in absorbance values observed for O2– production in CCR-2 neutralized S. aureus-infected FBMCs pre-treated with exogenous SOD compared with both S. aureus-infected FBMCs and IgG isotype pre-treated S. aureus-infected FBMCs along with SOD treatment suggested the specificity of the reaction by O2– only (Figure 4a, b).
Determination of H2O2 and O2– anion released by CCR-2-neutralized murine FBMCs after S. aureus infection in the presence of exogenous catalase and SOD. (a) H2O2 and (b) O2–anion production by CCR-2-blocked FBMCs infected with S. aureus are expressed in terms of µM/106cells in the presence of exogenous catalase and SOD, respectively. Results are shown as mean ± SD of three independent experiments. #P < 0.05 with respect to S. aureus-infected group; $P < 0.05 with respect to IgG isotype pre-treated FBMCs prior to S. aureus infection.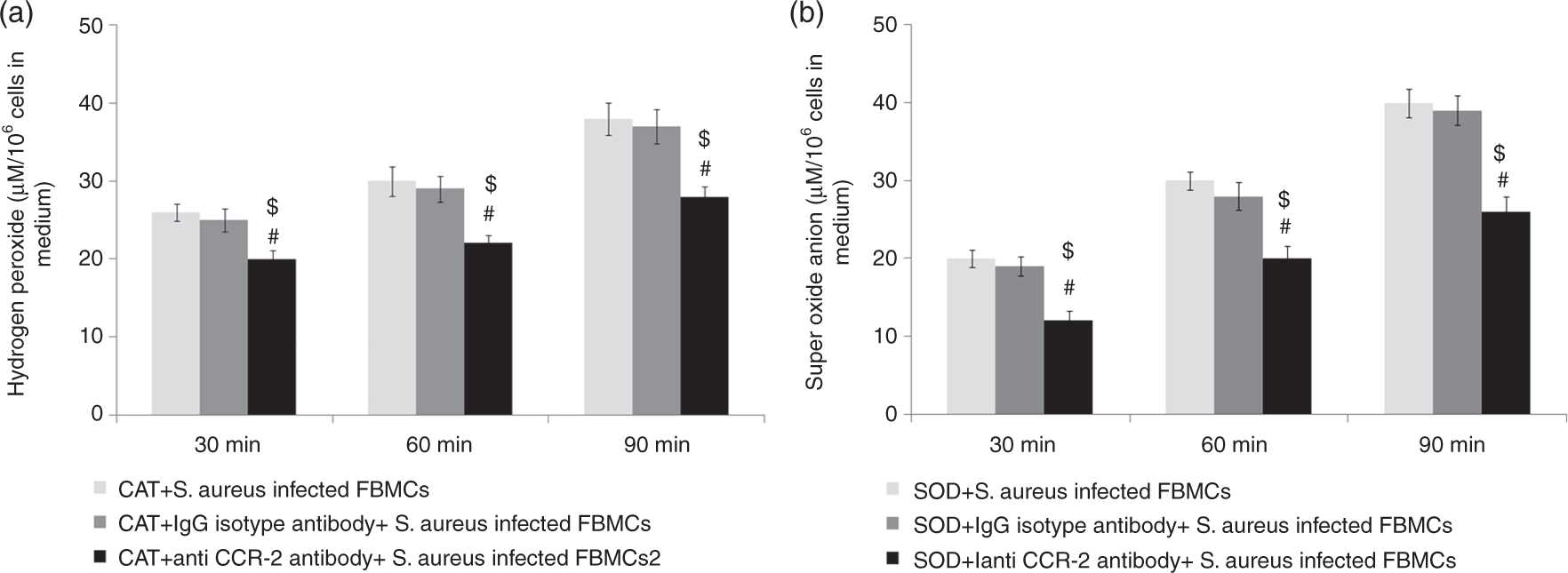
Alteration in cellular ROS production by murine FBMC infection with S. aureus due to CCR-2 blocking
As it has been found that infection of BMCs with S. aureus from anti CCR-2-treated BMCs group led to a significant decrease (P < 0.05) compared with anti-CCR-2-untreated BMCs in H2O2, O2– and NO production, we further determined the intracellular ROS production by flow cytometry at 30, 60 and 90 min post-infection. Following incubation with DCF-DA, it has been found that the macrophages infected with S. aureus from CCR-2 neutralized groups showed a significant decrease (P < 0.05) in ROS production after only 60 and 90 min post-infection than that of their respective CCR-2 non-neutralized groups (Figure 5a, b).
Alteration in intracellular ROS generation by murine FBMCs infected with S. aureus after CCR -2 blocking. FBMCs pre-treated with or without anti-CCR-2 Ab, were infected with S. aureus for 30, 60 and 90 min and then incubated with 20 mM DCHF-DA for 30 min. Fluorescence from cells was measured by FACS and indicated as control (a), S. aureus-infected (b), anti-CCR-2 Ab-treated (c) and anti-CCR-2 Ab-pre-treated FBMCs infected with S. aureus (d). ROS generation in different groups after (A, D) 30 min, (B, E) 60 min and (C, F) 90 min of phagocytosis is shown in the fluorescence intensity. Values are expressed as median ± SD of three independent experiments. *P < 0.05 compared with uninfected control group; # P< 0.05 with respect to the S. aureus-infected group.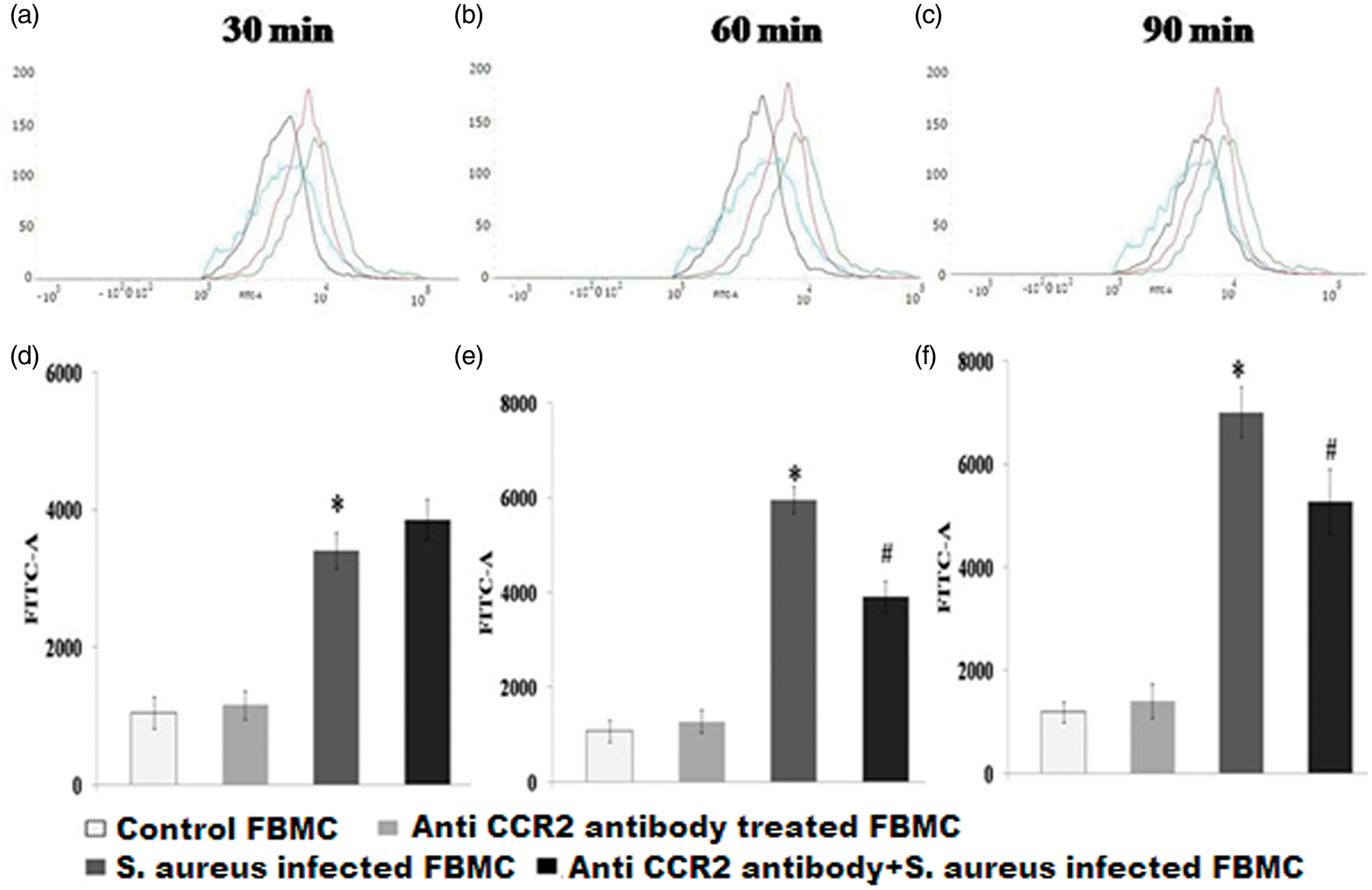
Alteration in cytokine (TNF-α, IFN-γ, IL-6, IL-1β, CCL-2 and IL-10) production by murine FBMCs after infection with S. aureus due to CCR-2 blocking
Acute infection of BMCs for 30, 60 and 90 min with S. aureus in presence of anti-CCR-2 Ab resulted in a significant decrease (P < 0.05) in IFN-γ and IL-6 release (Figure 6b, c) compared with that amount secreted by the CCR-2-untreated BMCs groups. However, S. aureus-infected BMCs, pre-treated with anti-CCR-2 Ab, showed a marked increase (P < 0.05) in CCL-2 and IL-10 production at 30, 60 and 90 min compared with the Ab-untreated but S. aureus-infected group (Figure 6e, f). A similar scenario was also observed in case of TNF-α and IL-1β (Figure 6a, d) where anti-CCR-2 Ab pre-treated BMCs infected by S. aureus had insignificantly decreased TNF-α and IL-1β production with respect to Ab-untreated but S. aureus-infected BMCs. However, no such differences in cytokine or chemokine production were observed in S. aureus-infected BMCs with or without IgG isotype control Ab treatment. S. aureus-infected BMCs, pre-treated with anti-CCR-2 Ab, showed a significant decrease (P < 0.05) in the release of TNF-α and IFN-γ but increase the release of CCL-2 and IL-10 at all three time points, with respect to IgG isotype-treated BMCs infected with S. aureus.
Alteration in cytokine (TNF-α, IFN-γ, IL-6, IL-1β, CCL-2 and IL-10) production by murine FBMCs infected with S. aureus after CCR-2 blocking. FBMCs pre-incubated with or without anti-CCR-2 Ab/isotype control Ab for 30 min were infected with S. aureus for the time indicated. Levels of (a) TNF-α, (b) IFN-γ, (c) IL-6, (d) IL-1β, (e) CCL-2 (f) and IL-10 in the supernatants were determined by ELISA, according to the manufacturer’s recommendations, and are expressed from triplicate experiments. Values are expressed as mean ± SD of three independent experiments. *P < 0.05 compared with uninfected control group; #P < 0.05 with respect to S. aureus-infected group; $P < 0.05 with respect to IgG isotype-pre-treated fresh BMCs prior to S. aureus infection.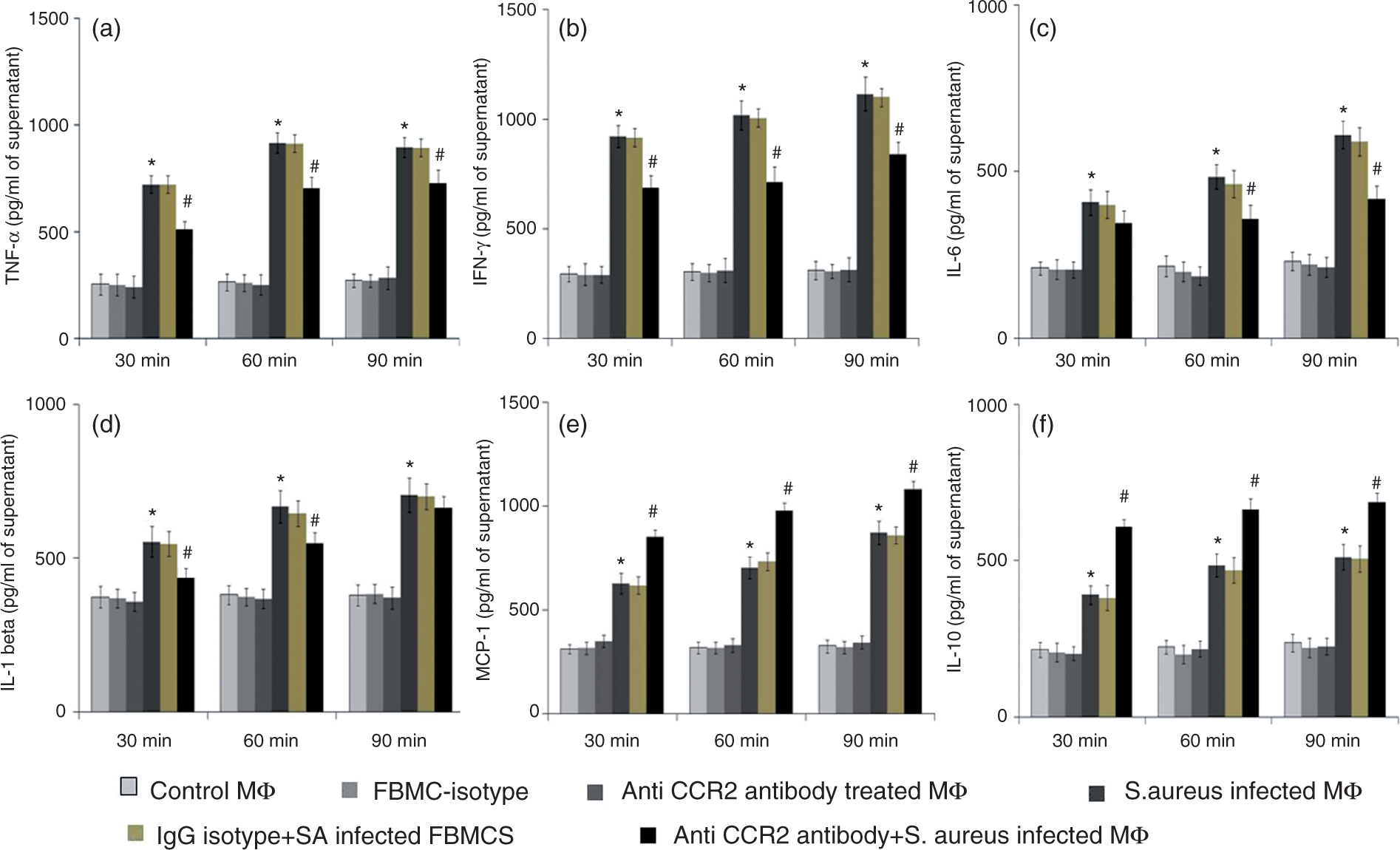
Western blot analysis for CCR-2 expression of FBMCs during acute S. aureus infection
Receptor expression was studied in murine FBMCs infected with S. aureus for 30, 60 and 90 min in the presence or absence of anti-CCR-2 Ab, to get a wider overview of the end product. The data presented in Figure 7 show that stimulation of anti-CCR-2 Ab-treated BMCs infected by S. significantly reduced expression of CCR-2 (P < 0.05), particularly after 60 and 90 min post-infection compared with CCR-2 non-neutralized BMCs. Additionally, no such reduction in CCR-2 expression was observed in IgG isotype Ab-treated, S. aureus-infected BMCs. However, there was a significant decrease (P < 0.05) in CCR-2 expression in anti-CCR-2 Ab pre-treated BMCs prior to S. aureus infection compared with IgG isotype pre-treated BMCs prior to S. aureus infection.
Expression of CCR-2 receptor by murine fresh bone marrow cells (FBMCs) infected with S. aureus in presence or absence of anti CCR-2 antibody/isotype control antibody Whole cell lysate was prepared for analysis of CCR-2 expression by western blot. All the samples were probed with β-tubulin to show equal protein loading. (a) Lane 1 (FBMC) represented CCR-2 expression on fresh bone marrow cells; lane 2 (FBMC-IgG isotype) represented CCR-2 expression on control IgG isotype treated FBMCs; lane 3 (FBMC-Anti CCR-2 antibody) represented CCR-2 expression on anti CCR-2 antibody treated FBMCs; lane 4 (FBMC+SA) represented CCR-2 expression on S. aureus infected FBMCS; lane 5 (FBMC+IgG isotype+SA) represented CCR-2 expression on control IgG isotype pretreated FBMC after S. aureus infection and lane 6 (FBMC+ Anti CCR-2 antibody +SA) represented CCR-2 expression on anti CCR-2 antibody pretreated FBMC after S. aureus infection for 30 min, 60 and 90 minute respectively. (b) Fold changes of CCR-2 expression were expressed as mean ± SD of three independent experiments. '*' indicates significant difference (p < 0.05) compared to uninfected control group; '#' indicates significant difference p < 0.05) with respect to S. aureus infected group; '$' indicates significant difference (p < 0.05) with respect to IgG isotype pretreated fresh bone marrow cells prior to S. aureus infection.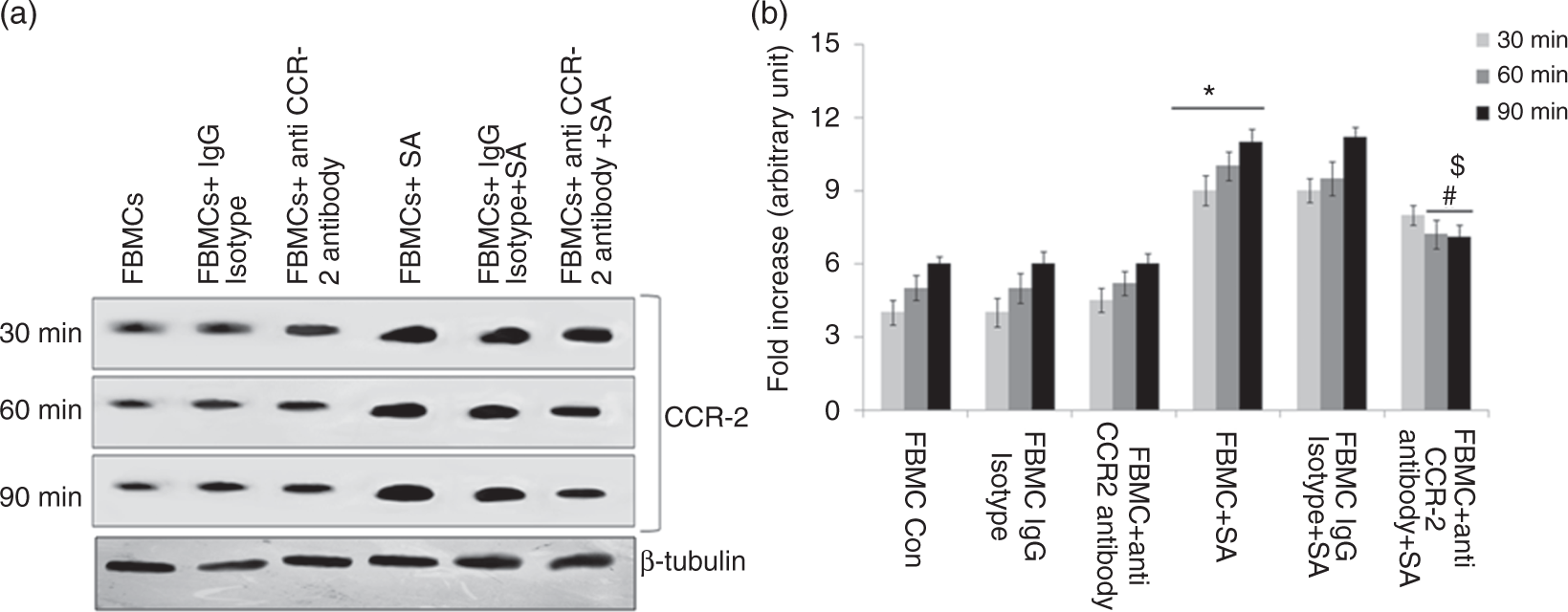
Alteration in the bacterial catalase activity from the recovered S. aureus after time-dependent phagocytosis
Bacterial catalase activity from the recovered S. aureus after time-dependent phagocytosis.

Catalase activity was determined in the presence of 15 µmol H2O2 ml phosphate buffer and expressed in terms of mmol/min/mg bacterial protein Results are presented as mean ± SD of three independent experiments. aP < 0.05 with respect to S. aureus strain (AG-789); bP < 0.05 with respect to S. aureus recovered from CCR-2 unblocked group.
Alteration in the bacterial SOD activity from the recovered S. aureus after time-dependent phagocytosis
Bacterial SOD activity from the recovered S. aureus after time-dependent phagocytosis.
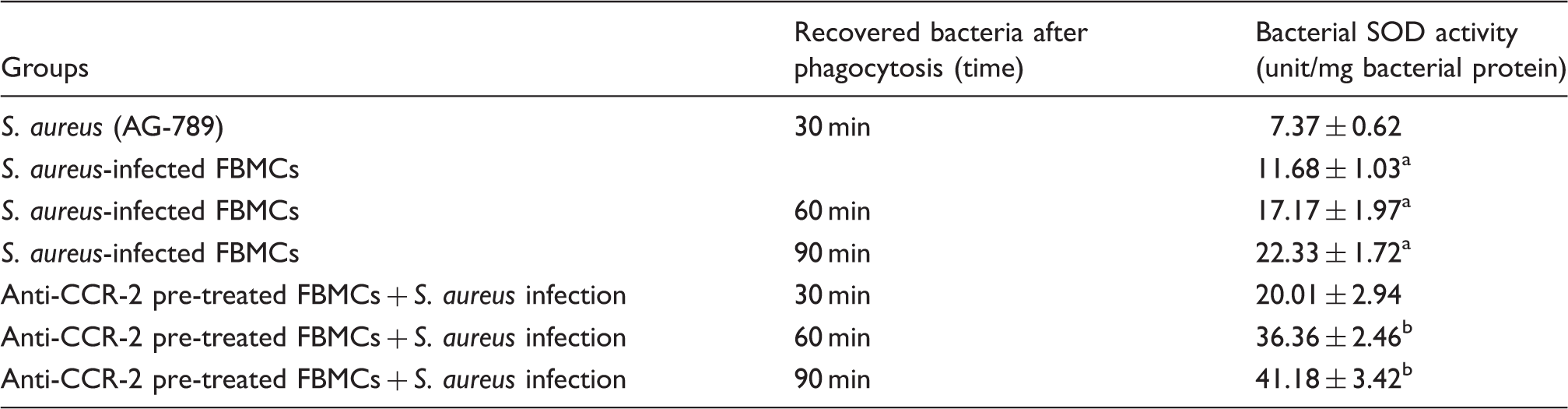
SOD activity was determined and expressed in terms of unit/mg bacterial protein. Results are presented as mean ± SD of three independent experiments. aP < 0.05 with respect to S. aureus strain (AG-789); bP < 0.05 with respect to S. aureus recovered from CCR-2 unblocked group.
Alteration in catalase activity of ATZ-pretreated and -non-treated BMCs infected with S. aureus in the presence or absence of anti-CCR-2 Ab
Catalase activity of the ATZ-pre-treated or -untreated FBMCs infected with S. aureus in the presence or absence of anti-CCR-2 Ab.
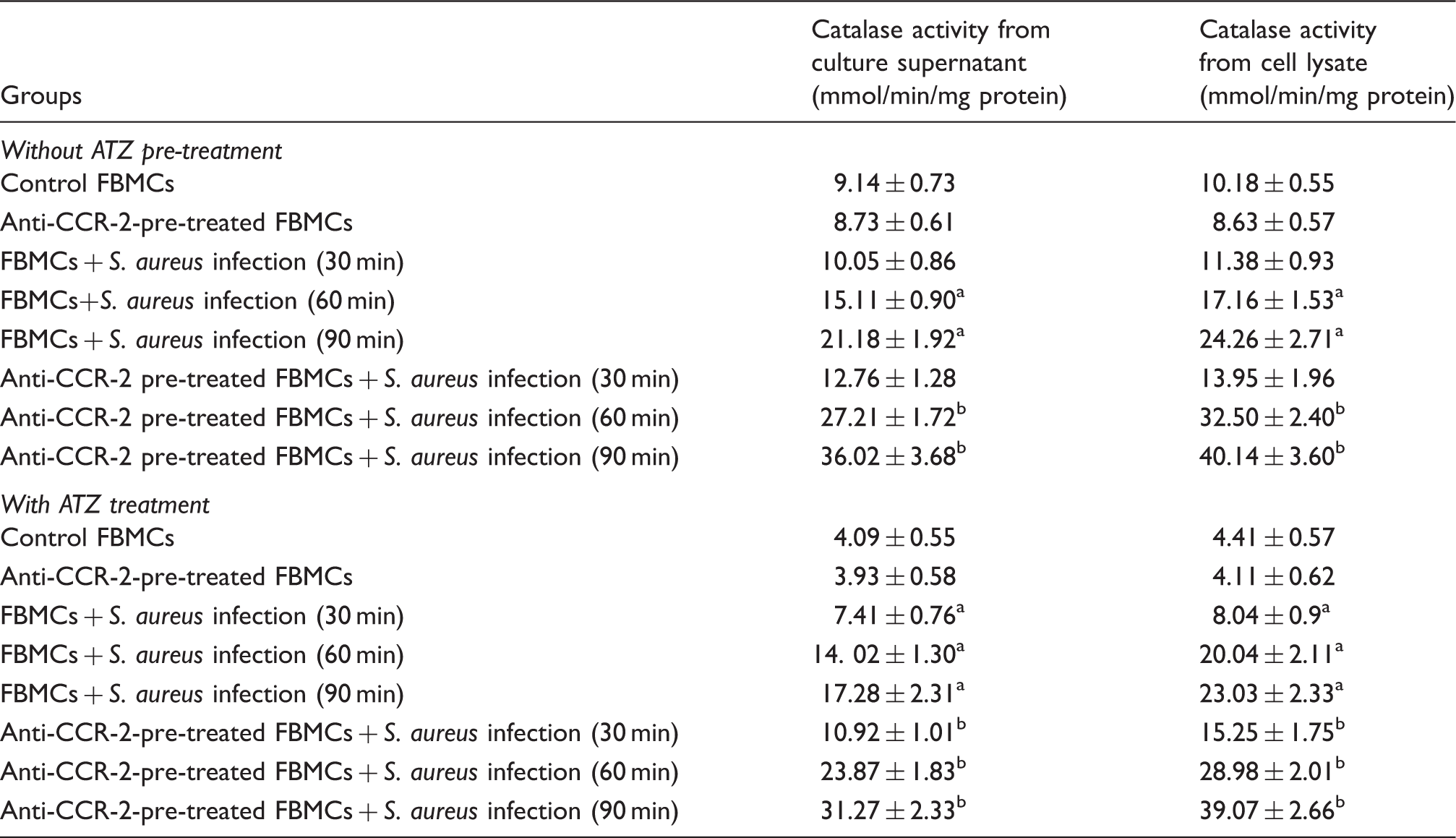
Catalase activity from culture supernatant and lysate of either ATZ pre-treated or untreated FBMCs after S. aureus infection was measured as mentioned earlier and expressed in terms of mmol/min/mg bacterial protein. Values are expressed as mean ± SD of three independent experiments. aP < 0.05 compared with uninfected control group; bP < 0.05 with respect to S. aureus-infected group.
Alteration in SOD activity of DDC-pretreated and -non-treated murine BMCs infected with S. aureus in the presence or absence of anti-CCR-2 Ab
SOD activity of the DDC-pretreated or -untreated FBMCs infected with S. aureus in the presence or absence of anti-CCR-2 Ab.
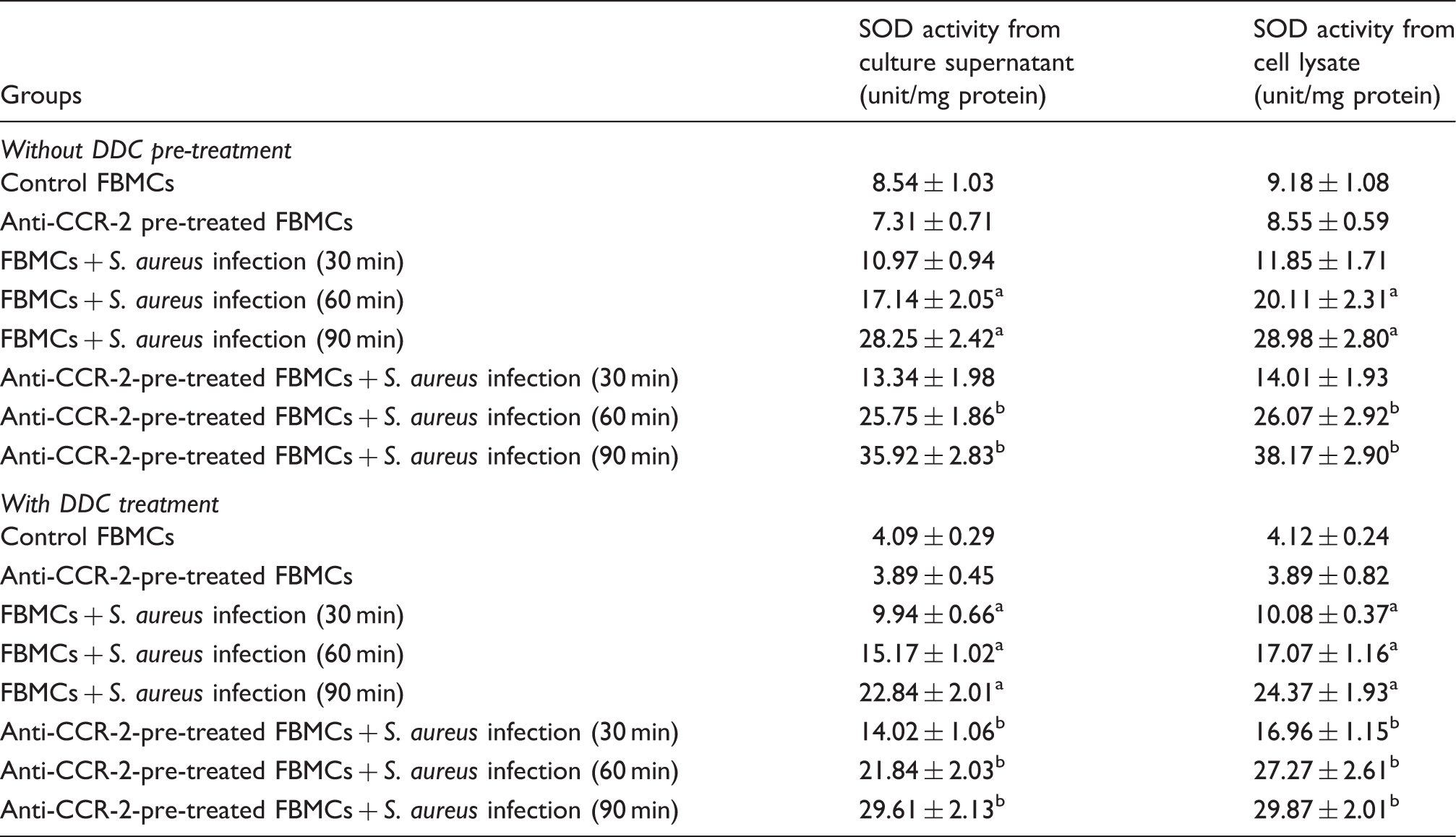
SOD activity from culture supernatant and lysate of either DDC-pretreated or -untreated FBMCs after S. aureus infection was measured as mentioned earlier and expressed in terms of unit/mg bacterial protein. Values are expressed as mean ± SD of three independent experiments. aP < 0.05 compared with uninfected control group; bP < 0.05 with respect to S. aureus-infected group.
Alteration in the expression of SOD from recovered S. aureus after time-dependent phagocytosis
Crude bacterial lysate from the recovered bacteria after time-dependent phagocytosis in the presence or absence of anti-CCR-2 Ab-mediated receptor blocking was prepared as described earlier. A marked increase (P < 0.05) in the expression of bacterial SOD protein was observed in the cell-free lysate of bacteria recovered from anti-CCR-2-pre-treated BMCs at 30, 60 and 90 min of phagocytosis, compared with that of Ab-untreated BMCs (Figure 8a, b).
Expression of SOD protein from recovered bacteria after time dependent phagocytosis All the samples were probed with β-tubulin to show equal protein loading. (a) Lane 1 (SA-AG-789) represented SOD expression in S. aureus (AG-789) strain; Lane 3, 5 and 7 (FBMC-RSA) represented SOD expression in S. aureus which has been recovered from fresh bone marrow cells after 30, 60 and 90 minute of phagocytosis; Lane 2,4 and 6 (FBMC-CCR2-RSA) represented SOD expression in S. aureus which has been recovered from anti CCR-2 antibody pretreated fresh bone marrow cells after 30, 60 and 90 minute of time dependant phagocytosis. (b) Fold changes in SOD expression were expressed as mean ± SD of three independent experiments. ‘ϕ' indicates significant difference (p < 0.05) with respect to mother S. aureus strain (AG-789); ‘#' indicates significant difference (p < 0.05) with respect to S. aureus recovered from CCR-2 unblocked group.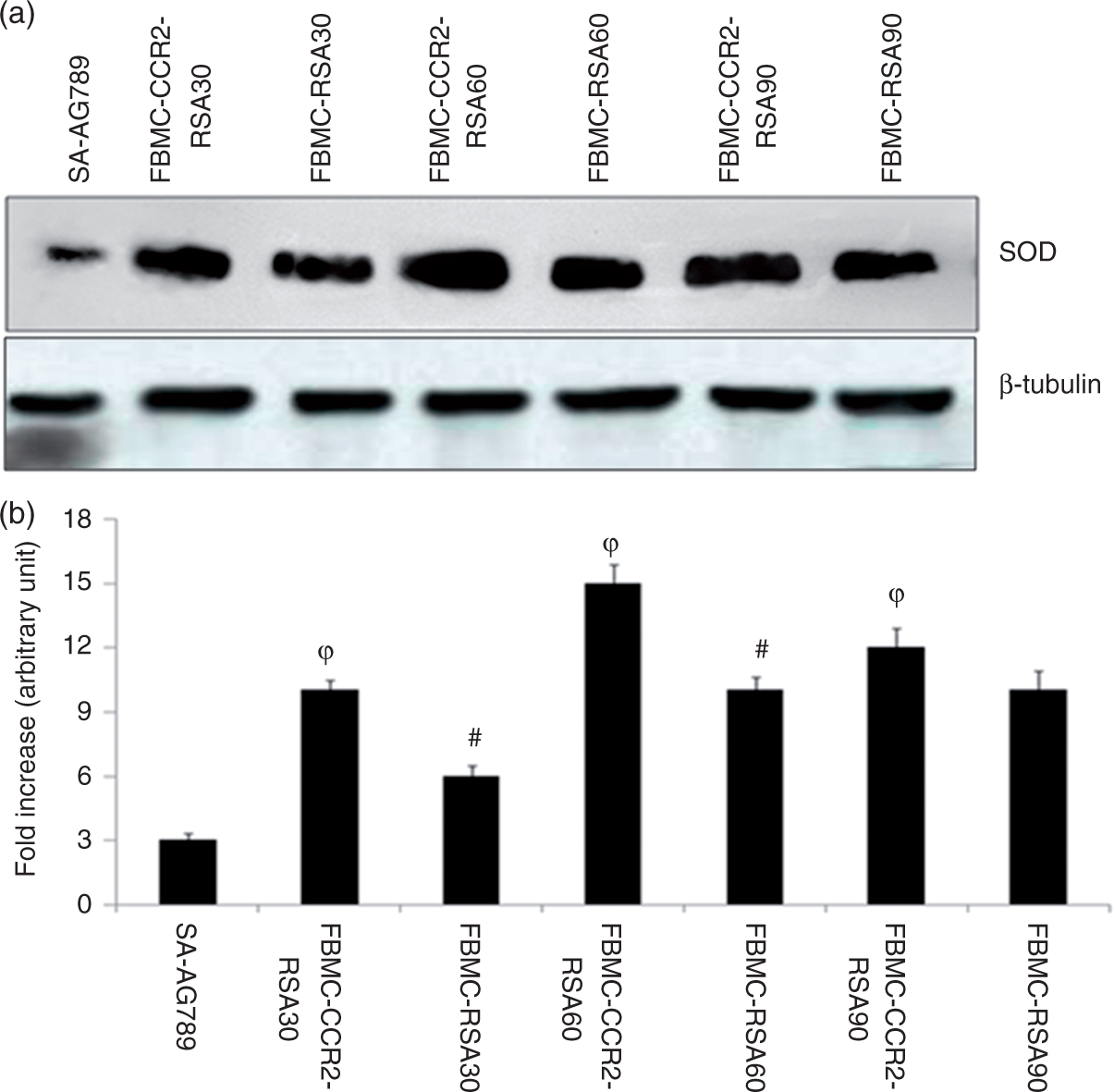
Alteration in S. aureus-specific gene expression (mRNA) of sodA, sodM and katA from the recovered bacteria after time-dependent phagocytosis in presence or absence of anti-CCR-2 Ab
In order to assess if the changes in SOD activity take place at the transcription level, we performed estimation of sodA and sodM gene expression levels in S. aureus recovered after time-dependent phagocytosis. Both sodA and sodM gene expression levels were found to be significantly higher in S. aureus recovered from CCR-2-blocked BMCs after 60 and 90 min of phagocytosis compared with S. aureus recovered from CCR-2-unblocked BMCs (Figure 9a). Similar patterns of results were also observed when we measured katA expression in S. aureus recovered from FBMCs in the presence or absence of anti-CCR-2 Ab after time-dependent phagocytosis (Figure 9b).
Transcription of the sodA, sodM and katA genes Northern analysis of the (a) soda and sodM, (b) katA transcripts in S. aureus which has been recovered from fresh bone marrow cells after 30, 60 and 90 minute of phagocytosis; 'ϕ' indicates significant difference (p < 0.05) with respect to mother S. aureus strain (AG-789); '#' indicates significant difference (p < 0.05) with respect to S. aureus recovered from CCR-2 unblocked group. Lane 1 (SA-AG-789) represented sodA, sodM and katA expression in S. aureus (AG-789) strain; Lane 2, 4 and 6 (FBMC-RSA) represented sodA, sodM and katA expression in S. aureus which has been recovered from fresh bone marrow cells after 30, 60 and 90 minute of phagocytosis; Lane 3,5 and 7 (FBMC-CCR2-SA) represented sodA, sodM and katA expression in S. aureus which has been recovered from anti CCR-2 antibody pretreated fresh bone marrow cells after 30, 60 and 90 minute of time dependant phagocytosis.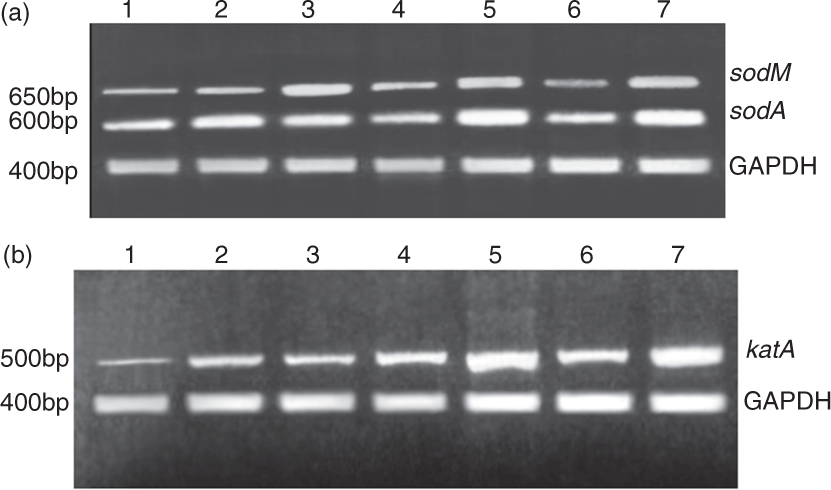
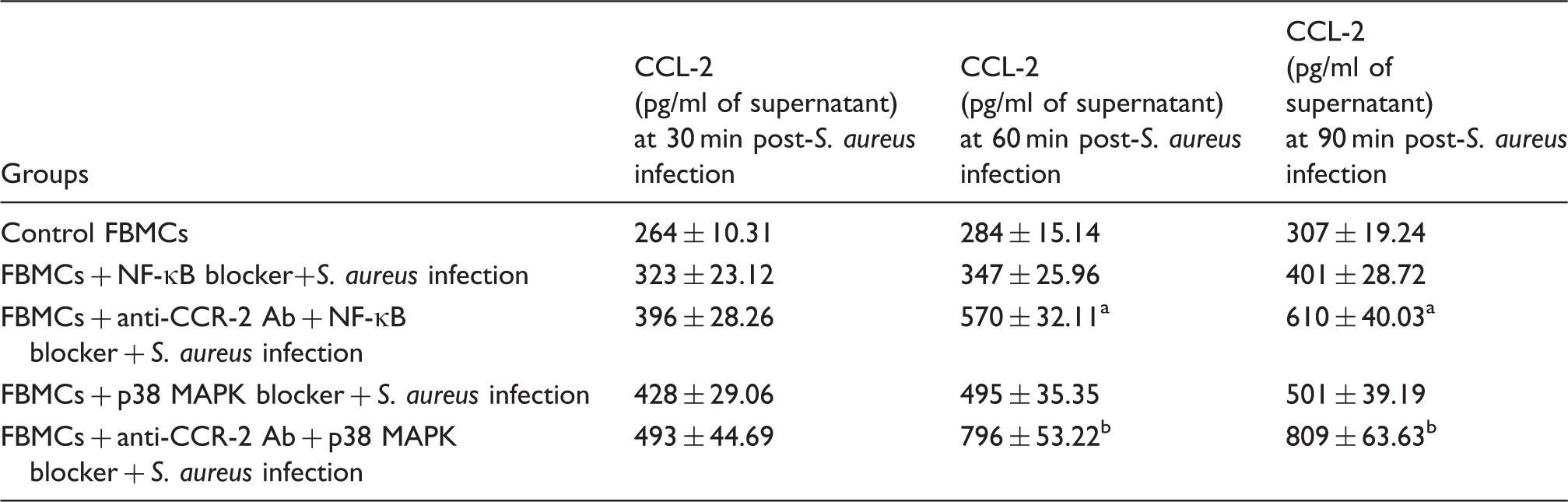
Alteration in CCR-2 expression, NF-κB expression and CCL-2 chemokine production in BMCs due to intracellular NF-κB and p38 MAPK blocking during acute S. aureus infection in the presence or absence of anti-CCR-2 Ab
CCR-2 receptor expression (Figure 10a, b), NF-κB expression (Figure 10c) and CCL-2 chemokine production (Table 7) were studied in the lysate and supernatant of anti-CCR-2 Ab-pre-treated or untreated murine BMCs infected with S. aureus for 30, 60 and 90 min during NF-κB and p38 MAPK blocking. Figure 10 shows that NF-κB, as well as p38 MAPK blocker-treated BMCs with S. aureus infection in the presence of anti-CCR-2 Ab, significantly reduced (P < 0.05) the expression of CCR-2 and NF-κB, particularly 60 and 90 min post-infection compared with the CCR-2-non neutralized condition. However, Table 7 shows that the production of CCL-2 was markedly increased (P < 0.05) after 60 and 90 min of phagocytosis in the case of anti-CCR-2 Ab-treated BMCs in presence of both NF-κB and p38 MAPK blocker than that of their only anti-CCR-2 Ab-treated one during acute S. aureus infection.
Differential expression of CCR-2 receptor and NF-κB in S. aureus-infected FBMCs after NF-κB and p38 MAPK blocking in presence of anti-CCR-2 Ab. The figure shows CCR-2 and NF-κB expression in NF-κB-blocked FBMCs after S. aureus infection either in presence of anti-CCR-2 Ab from a set of triplicate experiments. All samples were probed with β-tubulin to show equal protein loading. Lanes 1, 3 and 5 (FBMC-blocker-SA) represent CCR-2 expression on anti-CCR-2-pre-treated FBMCs after S. aureus infection for 30, 60 and 90 min, respectively. Lanes 2, 4 and 6 (FBMC-CCR2-blocker-SA) represent CCR-2 expression on (a) NF-κB and (b) p38 MAPK blocker-pre-treated FBMCs in presence of anti-CCR-2 Ab. (c) NF-κB expression in presence of NF-κB blocker. Fold changes of NF-κB expression fold changes of CCR-2 expression after (d) NF-κB and (e) p38 MAPK blocking are expressed as mean ± SD of three independent experiments. (f) Fold changes of NF- κB expression expressed as mean ± SD of three independent experiments. $P < 0.05 between anti-CCR-2 Ab-pre-treated FBMCs after S. aureus infection and NF-κB blocker-pre-treated FBMCs infected with S. aureus in the presence of anti-CCR-2 Ab. γP < 0.05 between anti-CCR-2 Ab-pre-treated FBMCs after S. aureus infection and p38 MAPK blocker-pre-treated FBMCs infected with S. aureus in presence of anti-CCR-2 Ab. CCL-2 chemokine production in FBMCs due to intracellular NF-κB and p38 MAPK blocking during acute S. aureus infection in presence of anti-CCR-2 Ab. CCL-2 release was measured in the supernatant by the FBMCs, which were pre-treated with either NF-κB or p38 MAPK blocker during S. aureus infection in the presence or absence of anti-CCR-2 Ab by utilizing ELISA according to the manufacturer’s recommendations and expressed as pg/ml of supernatant. Values are expressed as mean ± SD of three independent experiments. aP < 0.05 compared with NF-κB-blocker-treated FBMCs after S. aureus infection; bP < 0.05 compared with p38 MAPK blocker-treated FBMCs after S. aureus infection.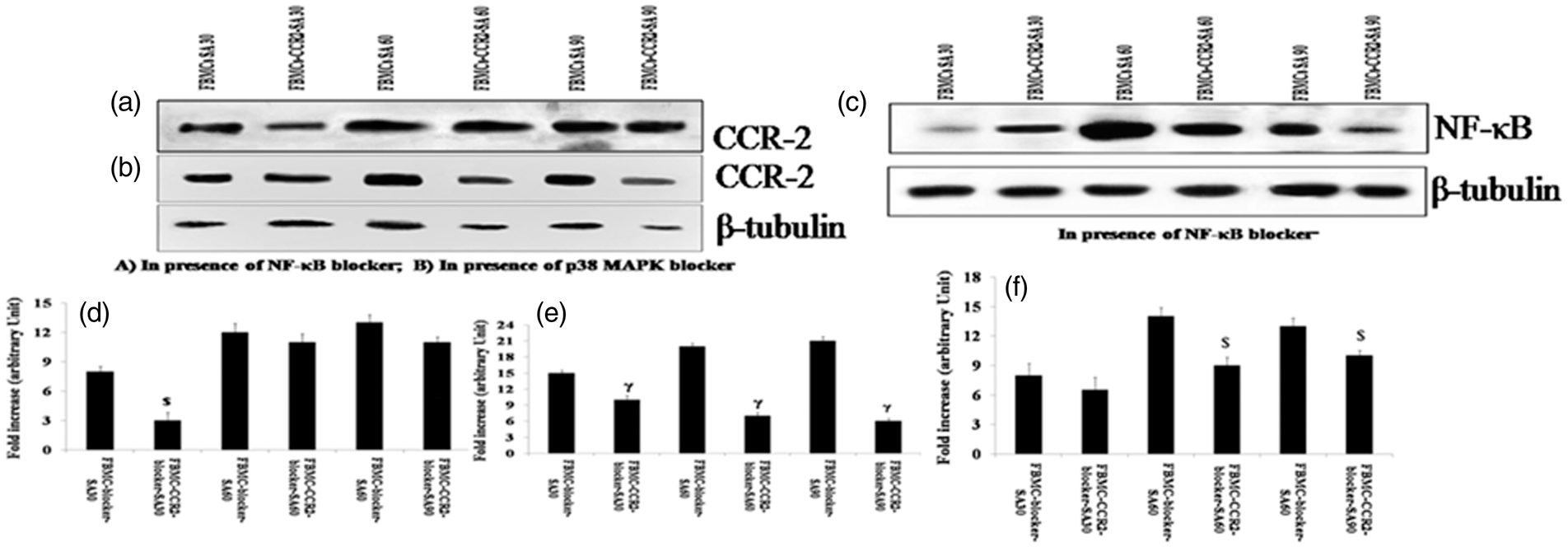
Anti-CCR-2 Ab suppresses BMC migration in response to S. aureus infection
To investigate the contribution of CCR-2 to S. aureus-infected BMC chemotaxis to CCL-2, in vitro transwell migration assays were performed following pre-incubation with neutralizing anti-CCR-2 Ab and control isotype Ab. Cell migration assays with CCL-2 and control serum-free medium were performed and demonstrated that CCL-2 has a pronounced recruitment effect on the migration of FBMCs. A significant increase (P < 0.05) in migration was observed in S. aureus-infected BMCs compared with uninfected BMCs. However, S. aureus-infected BMCs pre-treated with anti-CCR-2 Ab showed a significant decrease (P < 0.05) in migration when compared with only S. aureus-infected BMCs. Additionally, we observed no significant increase in the migration of BMCs with or without S. aureus infection but pre-treated with control IgG isotype Ab as compared with control group or S. aureus-infected BMCs, respectively. However, S. aureus-infected BMCs pre-treated with anti-CCR-2 Ab showed a significant decrease (P < 0.05) in migration when compared with only S. aureus-infected BMCs pre-treated with IgG isotype control (Figure 11).
Transwell migration assay. Monocyte chemotactic protein-1 (CCL-2) induces in vitro migration of FBMCs. CCL-2 chemotaxis of FBMCs was studied with Transwell Permeable Supports with 10 ng/ml CCL-2 protein (experimental group) or without protein (control group) in the bottom chamber. All cells were seeded on the upper chamber. After the assay was performed, the migratory cells on the bottom side of the membrane were stained and counted. Values are expressed as median ± SD of three independent experiments. *P < 0.05 compared with uninfected control group; #P < 0.05 with respect to S. aureus-infected group. $P < 0.05 with respect to IgG isotype-pre-treated FBMCs prior to S. aureus infection.
Recovery of bacteria from the blood, spleen and synovial tissue of mice after intravenous injection of S. aureus
Bacterial density in blood, spleen and synovial tissue of mice infected with S. aureus (AG-789) either prior to CCR-2 neutralization or treatment with control IgG isotype.
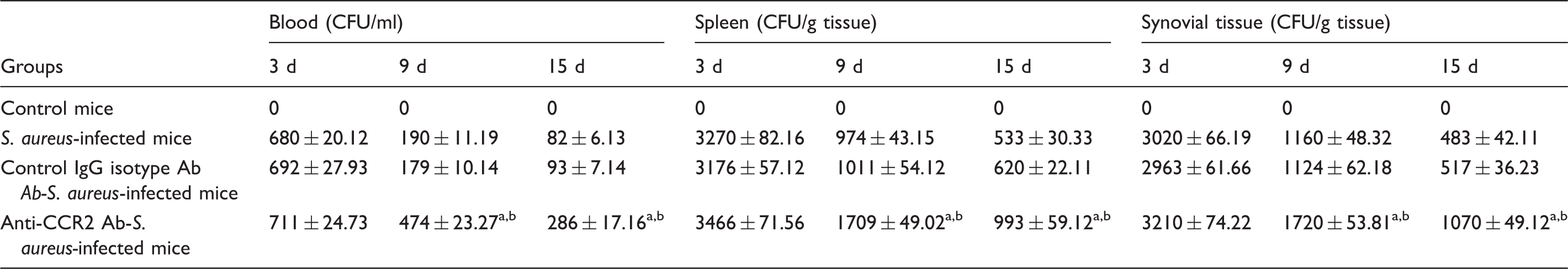
Bacterial burden was determined in terms of CFU count, as mentioned earlier. Results are shown as mean ± SD of three independent experiments. aP < 0.05) with respect to S. aureus-infected group; bP < 0.05 with respect to IgG isotype pre-treated fresh BMCs prior to S. aureus infection.
Alteration in serum CCL-2 production in mice after intravenous injection of S. aureus
Serum CCL-2 chemokine production in mice infected with S. aureus (AG-789) either prior to CCR-2 neutralization or treatment of control IgG isotype.
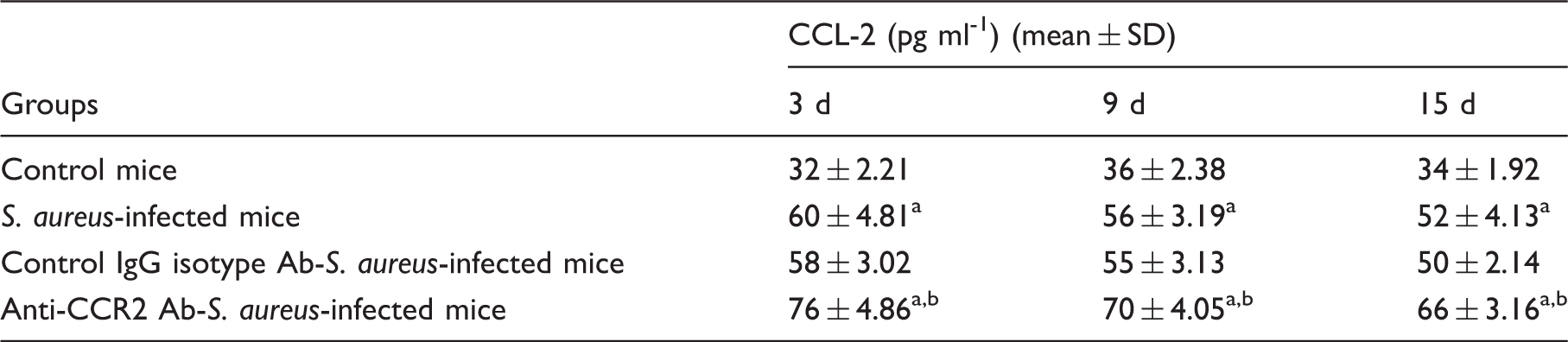
Blood samples from different groups were centrifuged at 300 g for 5 min at 4℃ and serum was prepared for estimation of CCL-2 production. Levels of CCL-2 in serum were determined by ELISA, according to the manufacturer’s recommendations, and were calculated from triplicate experiments. Values are expressed as mean ± SD of three independent experiments. aP < 0.05 compared with uninfected control group; bP < 0.05 with respect to S. aureus-infected group; cP < 0.05 with respect to IgG isotype-pre-treated FBMCs prior to S. aureus infection.
Discussion
The regulated interaction of chemokines with their respective receptors is thought to mediate the controlled recruitment of specific subpopulations required during host defense and inflammation.77,78 The specific biologic functions of chemokines and their receptors have been difficult to predict, as most receptors recognize more than one chemokine, and several chemokines bind to more than one receptor in vitro. Among the β-chemokine receptors, CCR-2 appears to be rather specific for ligands that belong to the MCP family. Even though CCL-2 binds only CCR-2 with high affinity, CCR-2 also serves as receptor for MCP-3 and MCP-5.79–81 Both CCR-2 and CCL-2 were previously shown to be induced during differentiation of BMCs, 82 but little was known about the effects of CCR-2-dependent signaling on resident FBMCs, particularly in relation to S. aureus infection. As there is an interlink among ROS–NF-κB/p38-MAPK–CCL-2 production and CCR-2 signaling, it prompted us to determine whether neutralization of CCR-2 regulates the response of murine FBMCs after S. aureus infection utilizing ROS production and cytokine profile in regard to the killing of S. aureus.
Bone marrow contains a large variety of stem cells comprising multipotent and restricted progenitors. In the mouse, CD11b (Mac-1 α; integrin αM chain) is expressed on monocytes/macrophages, and to a lower extent on granulocytes, natural killer cells and a subset of dendritic cells. 83 CD11b Ab studies in the current set-up have shown that > 80% FBMCs are CD11b-positive, indicating the presence of monocytes/macrophages. Decreased CFUs in CCR-2-intact FBMCs indicates decreased intracellular survival of S. aureus in the current set-up. However, intracellular survival of bacteria was increased in CCR-2-neutralized FBMCs infected with S. aureus. This is well in line with the results of percentage survival, which indicates diminished killing of S. aureus by CCR-2-neutralized FBMCs when compared with a CCR-2-non-neutralized counterpart. These results suggest that inhibition of CCR-2 impairs host defense and may find relevancy in exhibiting marked adverse effects on the innate immune response to bone health. There are reports that macrophages internalize a significant percentage of their membrane surface area during phagocytosis, implying that they must replenish cell surface membranes from the intracellular pool. 84 Although we currently do not have a FBMC model for CCR-2 membrane trafficking in bacterial phagocytosis that involves the CCR-2 recycling, we can neither eliminate the possibility that the Golgi network, directly or indirectly, contributes to phagocytosis nor can we conclude that decreased CCR-2 is due to receptor recycling or to decreased synthesis. Internalized CCR-2 can be recycled to the membrane or become down-regulated through lysosomal degradation.85,86 Interestingly, in the case of CCR-2 receptor, internalization can be dissociated from functional responses. In line with this, it was also shown by others that CCR-2-dependent migration of human monocytes towards the source dispensing CCL-2 is accompanied by receptor-mediated ligand uptake. However, it has been shown that CCR-2 internalization appears not to reduce the responsiveness of the cells to migrate continuously towards CCL-2, suggesting that the receptors are rapidly recycled or those sufficient non-internalized receptors are available at the cell surface to maintain the responsiveness. 87 There are reports on staphylococcal superantigen-induced down-modulation of chemokine responsiveness via CCR-2, which represents a previously unrecognized strategy by S. aureus to subvert immune responses by affecting the intricate balance between chemokine and chemokine receptor expression and function. This down-regulation occurs 15–30 min following superantigen binding and is specific to chemokine receptors. Therefore, binding of S. aureus and its internalization cannot be ruled out in regard to CCR-2 interaction. 88 It has been reported that murine CCL-2 efficiently down-modulates CCR-2 from the surface of T cells and monocytes. Earlier it has been shown that at a concentration of 100 ng/ml, almost 50% of CCR-2 was internalized within 30 min at 37℃. In general, internalization of chemokine receptors depends on receptor activation and reduced internalization would indicate blockade of the receptor. 73
ROS production is reported to be elevated rapidly during infection, serving to facilitate pathogen clearance, as well as contributing to signaling cascades related to inflammation, cell proliferation and immune responses.89–91 Production of ROS, including O2– and H2O2, were found to be diminished in CCR-2-neutralized FBMCs after S. aureus infection compared with their CCR-2 intact counterpart in the present study. Neutralization of extracellular ROS after using SOD and catalase in the assay medium for O2– and H2O2, respectively, indicates the specificity of the reaction. The results of this study present evidence that FBMCs treated with anti-CCR-2 Ab released less NO on encountering S. aureus infection compared with CCR-2 non-neutralized FBMCs. These data correlate well with the increased intracellular survival of S. aureus in CCR-2-neutralized FBMCs, as evidenced by increased CFU observed in the present study. Therefore, based on these results we speculate that acute S. aureus infection of CCR-2-intact FBMCs possibly stimulates toxic effects of ROS to kill bacteria by elaborating O2– and H2O2. Additionally, NO produced by S. aureus-infected CCR-2-intact FBMCs may interact with O2– to form peroxynitrite, another product with enhanced toxicity, to increase killing and limit the growth of S. aureus inside the FBMCs during an early stage of infection and suggesting that the balance between NO and ROS may help to determine immune polarity.92,93
Bacterial antioxidants serve to combat the production of microbicidal host-derived and environmental oxidants. This led us to focus our study on the microbial antioxidant enzymes catalase and SOD, with the hypothesis that they play a role in protecting S. aureus from the oxidative killing mechanisms within the host, especially in the CCR-2-neutralized condition. S. aureus infection to CCR-2-blocked FBMCs leads to increased SOD and catalase activity. Alteration in S. aureus-specific gene expression (mRNA) of sodA, sodM and katA from the recovered bacteria after time-dependent phagocytosis in the presence or absence of anti-CCR-2 Ab was shown. The role of SODs in virulence has been investigated in several bacterial species. S. aureus has two SODs, encoded by sodA and sodM, which inactivate harmful O2– radicals encountered during host infection. 94 O2– induction of sodA and sodM transcription is Mn-dependent. sodM is most strongly induced in response to externally generated O2– stress, whereas sodA is induced by internal stress. A significant increase in both sodA and sodM gene expression after 60 and 90 min of phagocytosis in S. aureus recovered from CCR-2-blocked FBMCs indicates that SodA and SodM both contribute to the survival of S. aureus in the current set-up. This result is in good agreement with the total SOD activity changes in recovered S. aureus seen during time-dependent phagocytosis. Additionally, increased expression of katA in S. aureus recovered from CCR-2-blocked FBMCs consolidate the hypothesis that catalase activity is critical for S. aureus survival in the CCR-2-neutralized condition. Contribution of bacterial catalase was further verified by the assay of catalase in the presence of ATZ. Previous studies using ATZ, a catalase inhibitor, have marked its role in endogenous catalase inhibition, thereby leading to a moderate rise in the H2O2 production,95,96 and subsequently in greater host microbicidal activity. Further, the observed SOD and catalase are of bacterial origin, as both were measured from lysate of CCR-2-blocked macrophages infected with S. aureus in the presence or absence of ATZ (specific blocker of macrophage catalase) or DDC (specific blocker of macrophage SOD).
Infection of CCR-2-neutralized FBMCs with S. aureus has increased our understanding of how this receptor contributes to immunological events involved with host protection. CCR-2-neutralized FBMCs infected with S. aureus exhibit a diminished protective Th1 response accompanied by a switch to the Th2 response, which is evidenced by less production of pro-inflammatory cytokines such as TNF-α, IFN-γ and IL-6, with a concomitant increase in the anti-inflammatory cytokine IL-10, as compared with CCR-2 intact FBMCs. These results are in line with findings that CCR-2-deficient mice infected in the lungs with Cryptococcus neoformans or in the skin with L. major showed a diminished Th1 response with an increased Th2 response.97,98 The pro-inflammatory cytokines (TNF-α, IFN-γ, IL-1β) have been shown to induce CCL-2 gene expression in a variety of cell types. 98 As TNF-α, IFN-γ and IL-1β have been shown to be critical for the eradication of S. aureus and induces CCL-2 gene expression in leukocytes, and CCR-2 signaling initiates the production of CCL-2 and recruitment of macrophages to the site of infection, the level of CCL-2/CCR-2 signaling might indicate its role during S. aureus infection. Thus, CCR-2 is required for the development of a protective Th1 response in FBMCs during early stage of S. aureus infection. Collectively, this study strongly suggests that CCR-2 expression is required for the development of protective Th1 responses in FBMCs following S. aureus challenge. An earlier study from this laboratory also strongly suggests that CCR-2 expression is required for the development of protective Th1 responses after acute S. aureus challenge in murine peritoneal macrophages. Therefore, this switch in Th response correlates with the prolonged infection and greater dissemination of infecting agent throughout the body. 99 Data from this study indicated that the absence of CCR-2 results in a strong Th2-type response to the acute S. aureus infection that fails to clear the infection. Therefore, the data reported here are consistent with the importance of CCR-2 signaling in regulating events contributing to a protective immune response after acute S. aureus infection and also extends earlier observations. There are reports that macrophages lacking CCR-2 produced normal levels of TNF-α, IL-6 and IL-1β; therefore, we hypothesized that the inability of CCR-2-blocked FBMC to clear S. aureus infection was due to the defective phagocytic nature of CCR-2-neutralized FBMCs. 100 The level of sensitivity of FBMCs after addition of more CCL-2 to the system does not confer augmentation of phagocytosis; as was shown previously, transgenic mice expressing high levels of circulating CCL-2 could not clear intracellular pathogens. 101 Therefore, addition of more CCL-2 to this in vitro model system might lead to receptor (CCR-2) desensitization on the FBMCs by the high levels of CCL-2. In male Swiss Albino mice (28–32 g body mass) small amounts of CCL-2 (100–150 pg/ml) were released from unstimulated peritoneal macrophages cultured up to 24 h; 102 therefore, early release of cytokines and chemokines after in vitro bacterial infection, as evident from our study, is not surprising.
Several studies have shown that macrophages release the pro-inflammatory mediators IL-1, IL-6 and TNF-α to particulate debris in bone tissues.103–105 IL-1 and TNF-α induce CCL-2 gene expression in macrophages, T lymphocytes, fibroblasts and endothelial cells. Conversely, CCL-2 stimulated the release of IL-1 and IL-6 from monocytes. 106 Therefore, from the present study, it can be anticipated that acute S. aureus infection of CCR-2-neutralized FBMCs possibly stimulates chemokine (CCL-2) production by decreased pro-inflammatory cytokines.
Increased expression of CCL-2 is and its rapid release in the bone marrow during inflammation has been reported. 107 Earlier, Ning et al. suggested that S. aureus can activate NF-κB in human osteoblasts, and subsequently regulate the secretion of IL-6 and monocyte chemoattractant protein-1 (CCL-2). 108 In this study, CCL-2 production was found to be increased in CCR-2-blocked FBMCs when compared with a CCR-2-intact group. In contradiction, reactive oxygen intermediates (ROIs) play an important regulatory role in the induction of MCP-1 (CCL-2) in certain cells. 109 We observed a higher CCL-2 level in CCR-2-blocked FBMCs, in spite of lower ROIs in this group. These results suggest that higher ROIs are not involved in the CCL-2 production in FBMCs treated with anti-CCR-2 Ab in the current set-up. It has been reported that among the pro-inflammatory cytokines, IL-1β recruits MyD88 as its adaptor protein and induces NF-κB expression. This may also be due to reduced production of pro-inflammatory cytokines, particularly IL-1β. The role of macrophages in releasing CCL-2 has been shown by a set of earlier in vitro experiments. 110 Previous studies investigating CCL-2 production in inflammatory infection models have mainly focused on the joint. 111
Although the precise mechanism of CCL-2 production in CCR-2-neutralized FBMCs is not clear, it is hypothesized that abrogation of CCL-2/CCR-2 binding leads to impairment of CCR-2-mediated down-regulation of CCL-2 production in macrophages and thereby un-regulated CCL-2 production in the current set-up. Therefore, negative loop in between CCL-2 and CCR-2 may be the plausible explanation for such increased CCL-2 production in CCR-2-blocked FBMCs infected with S. aureus.
Further, a specific role of NF-κB or p-38/MAPK in CCL-2/CCR-2 signaling of FBMCs was also explored in this current set-up. The regulation of CCL-2 in response to pro-inflammatory cytokines has been studied previously but is variable, and the contributions of the MAPK and NF-κB pathways may differ depending on the cell type and stimulus conditions. 112 S. aureus infection of CCR-2-intact FBMCs pre-treated with either NF-κB or p-38/MAPK blocker produced less CCL-2 in this study, suggesting that NF-κB or p-38-MAPK is required for CCL-2 production in FBMCs in response to S. aureus infection. Consistent with our findings with FBMCs, most previous studies also find only a partial inhibition of CCL-2 production when blocking either p38 or JNK. 112
Pyrrolidine dithiocarbamate (PDTC) is an antioxidant and potent NF-kB inhibitor, and it has been used to suppress the activity of NF-κB in some kinds of cells.113,114 As PDTC has been shown to diminish NF-κB activity at multiple points of the IκB kinase (IKK)–IkB–NF-κB cascade in several cell lines,115,116 we investigated whether this preparation could affect NF-κB activity and NF-κB-regulated cellular responses of FBMCs, as well as its mechanisms. Alternatively, blocking of CCR-2 along with NF-κB or p-38/MAPK leads to elevation of CCL-2 production and subsequent down-regulation of CCR-2 protein. Particularly, p38 MAPK has numerous direct and indirect interactions with NF-κB, so an obvious direction for future studies will be to examine the interactions of the MAPK and NF-κB pathway and their roles in CCL-2/CCR-2 signaling in FBMCs. However, we did not check IκB degradation and NF-κB DNA binding activity in mediating NF-κB signaling in the present set-up. Still, we have measured the expression of NF-κB in presence of PDTC after using anti-NF-κB-p65 (Figure 9c).
Further, this study demonstrates that CCL-2 has a recruitment effect on the migration of FBMCs. We have demonstrated that CCL-2 recruits the migration of maximum FBMCs infected with S. aureus in an in vitro cell migration assay as compared with CCR-2-neutralized S. aureus-infected FBMCs. In this study, CCL-2 production in CCR-2-intact FBMCs infected with S. aureus was observed. These findings suggest that FBMC migration may result from a dynamic interplay in which FBMCs secreted cytokines in response to acute staphylococcal infection, leading to chemokine receptor up-regulation on FBMCs, and ultimately resulting in enhanced chemotaxis towards the chemokine ligand. In support of this, we found that anti-CCR-2 Ab treatment markedly suppressed FBMCs migration in response to S. aureus infection, possibly owing, in part, to the lower levels of CCR-2 on FBMCs during acute S. aureus infection.
To cause disease, bacteria have to survive in blood, spread from the bloodstream to the joint and survive there, at least for a while (3 dpi). It has been shown previously that the degree of arthritis and inflammation under normal circumstances is dependent on the amounts of bacteria injected and the ability of the host to clear those bacteria. Typically, the peak of bacterial burden after intravenous staphylococcal inoculation occurs in blood and tissue within the 3 dpi, as evident from the CFU count. Mice neutralized with anti-CCR-2 Ab harbor higher bacterial load in spleen and synovial tissues. A significant higher bacterial load was still observed in in vivo CCR-2-neutralized mice 15 d following infection (Table 8). The role of CCR-2 in controlling infection with S. aureus has been supported by findings demonstrating that the organisms are less commonly found in blood of CCR-2 non-neutralized mice. Efficacy of anti-CCR-2 (in vivo) has been demonstrated by measuring the serum levels of CCL-2 (Table 9). Collectively, these findings suggest that the presence of intact CCR-2 signaling is necessary for a competent innate immune response against S. aureus. However, the role of CCR2 in control of infection and driving inflammation could be addressed using Ccr2–/– mice in the in vivo model of S. aureus infection.
In conclusion, we suggest that inhibition of CCR-2 exacerbates the murine FBMC response to S. aureus infection via two distinct mechanisms after utilizing the ROS production on one hand and also by regulating the cytokine response including TNF-α and IL-1 on the other. S. aureus infection of CCR-2-neutralized FBMCs produces less ROS and NO, and favors Th2 response with concomitant down-regulation of Th1 response. Internalized S. aureus also sets its antioxidant armature (increased SOD and catalase activity, along with increased SOD protein expression) as a strategy to escape intracellular killing. Lower oxidative burst and increased Th2 response due to CCR-2 neutralization further limits the activation of NF-κB and p38 MAPK, and augments CCL-2 production. Further, increased CCL-2 production due to inhibition of NF-κB and p38 MAPK with its specific blocker suggests the contribution of CCR-2 blocking-mediated Th2 response in this process. Alternatively, augmented CCL-2 in this set-up down-regulates CCR-2 expression. In addition, Th2/Th1 balance in the CCR-2-neutralized condition possibly does not elicit RANKL expression and may restrict osteoclastogenesis, which otherwise leads to bone resorption.117–120 Thus, taken together all this signaling contributes to effective elimination of the pathogen inside the bone marrow monocytes/macrophages. Because CCL-2 recruits and activates leukocytes selectively via CCR-2, their induction in direct response to S. aureus infection by FBMCs in vitro may be important to toxic shock, as well as the initiation of an inflammatory signal, such as in bacterial septic arthritis. Although CCL-2 has been associated with many pathological conditions such as rheumatoid arthritis, multiple sclerosis, cardiovascular disease and so on, it plays an important role in routine immune surveillance and immune modulation, and also in clearing acute bacterial burden. Therefore, to achieve beneficial effects in some of the above-mentioned pathological conditions, any attempts to reduce CCL-2 production or to reduce CCR-2 expression should be critically explored because of CCL’s role in host defense.
Therefore, this study may add to a better understanding of pathogen-mediated chronic inflammatory bone diseases and development of new treatments. Increased monocyte/macrophage infiltration plays a role in the disease pathogenesis of infectious arthritis; therefore, the function of FBMCs in patients with septic arthritic with high levels of CCL-2 deserves further attention. Because Ccr2 expression in monocytes can be inhibited by several other microbial agents such as inactivated streptococci, Propionibacterium acnes and pro-inflammatory cytokines such as TNF-α and IL-1 in vitro, 16 it would be interesting to determine whether CCR-2 neutralization has any therapeutic effect in S. aureus infection-induced septic arthritis. Although Ccr2–/– mice are available we were unable to procure them for this set-up. We have provided additional data after endogenous CCR-2 neutralization by injecting CCR-2 Ab or IgG control Ab into the mice before S. aureus infection, and the bacterial burden in blood, spleen and synovial tissues were determined at different days post-infection. These data also determine the role of CCR-2 in control of infection (bacterial clearance) and eventually driving inflammation. In animal models, there is considerable evidence that CCR-2 regulates CCL-2 expression, and that subsequent secretion of CCL-2 down-regulates CCR-2 expression.121,122 With the ample, consistent evidence of elevated levels of CCL-2 in synovial tissues from chronic inflammatory diseases such as septic arthritis, which has been reported independently by several research groups, our current results provide the groundwork for therapeutic strategies targeting the CCL-2/ CCR-2 pathways. We have also previously reported that the differences in clinical outcome between infections with hospital isolates vs. the reference ATCC-25923 strain were due to different replication velocities of the bacteria. Relevant to this subject we found that the doubling time of the bacteria was the same, irrespective of the pathogenic TSST-1/coagulase-positive S. aureus phenotype and that the ATCC-25923 strain differs from the pathogenic strain only regarding the absence of TSST-1/coa gene and antibiotic resistance. As S. aureus AG 789 produces TSST-1 it would be interesting to investigate the role of TSST-1 to CCR-2 on fresh bone marrow, and a role of other toxins, including staphylococcal enterotoxins, from this particular strain cannot be ruled out.
Footnotes
Acknowledgements
We are indebted to the Department of Science and Technology, Government of India, for providing us with the instruments procured under the DST-PURSE programme to the Department of Physiology, University of Calcutta.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are thankful to the Department of Science and Technology, Government of India for providing the DST-INSPIRE fellowship to Mrs. Ajeya Nandi (Grant Number: DST INSPIRE Fellowship/2013/1118, dated 23 June 2014). Part of this work was also supported by the Department of Science and Technology (DST), Science and Engineering Research Board (SERB), and Ministry of Science and Technology, Government of India, New Delhi, India (Grant Number: SR/SO/HS/0013/2012, dated 21.05.2013 to Biswadev Bishayi) for funding to the corresponding author.
