Abstract
Reactive oxygen species (ROS) are part of the weapons used by the immune system to kill and degrade infecting microorganisms. Bacteria can produce macromolecules, such as polysaccharides, that are able to scavenge ROS. Species belonging to the Burkholderia cepacia complex are involved in serious lung infection in cystic fibrosis patients and produce a characteristic polysaccharide, cepacian. The interaction between ROS and bacterial polysaccharides was first investigated by killing experiments, where bacteria cells were incubated with sodium hypochlorite (NaClO) with and without prior incubation with cepacian. The results showed that the polysaccharide had a protective effect towards bacterial cells. Cepacian was then treated with different concentrations of NaClO and the course of reactions was followed by means of capillary viscometry. The degradation products were characterised by size-exclusion chromatography, NMR and mass spectrometry. The results showed that hypochlorite depolymerised cepacian, removed side chains and O-acetyl groups, but did not cleave the glycosidic bond between glucuronic acid and rhamnose. The structure of some oligomers produced by NaClO oxidation is reported.
Introduction
Reactive oxygen species (ROS) are produced by neutrophils following the oxidative burst. The cascade of enzyme-driven processes leads to the formation of different reactive species terminating with the formation of the highly oxidising hypochlorite anion. 1 ROS are part of the weapons of the innate immune system and are involved in the destruction of phagocytised bacteria. Thus, a defence against ROS is part of the bacteria resistance in infection processes. Studies carried out on Pseudomonas aeruginosa, a nosocomial bacterium involved in serious lung infections, including those affecting cystic fibrosis (CF) patients, showed that alginate, the exopolysaccharide (EPS) produced by this bacterial species, acts as scavenger of ROS throughout the degradation of the polysaccharidic backbone.2,3 An investigation on a clinical Burkholderia cenocepacia isolate, a species belonging to the B. cepacia complex (BCC), also demonstrated that its EPS possesses ROS scavenging ability. 4 BCC is composed of at least 17 different species and some of them cause lung infections in CF patients that are resistant to eradication.5,6 Occasionally, BCC infections develop into the so called ‘cepacia syndrome’, a necrotising pneumonia followed by patient death.
The ability of BCC species to biosynthesise different EPS was thoroughly investigated, resulting in the identification of at least six polysaccharides with very diverse primary structures.
7
–
9
Among them, cepacian was isolated from the majority of the BCC species and it was generally produced in larger amounts than other EPS. Therefore, it was considered as the typical EPS of the BCC. Cepacian exhibits a rather complex structure, having a repeating unit characterised by a trisaccharide in its backbone and three lateral chains (Figure 1). The glucuronic acid in the backbone is fully substituted, leading to a hindered environment which contributes to chain rigidity.
10
The native polymer also contains 1–3 acetyl substituents, depending on the bacterial isolate, and whose locations were recently investigated.
11
In addition to this, light scattering, capillary viscosity and atomic force microscopy measurements showed that cepacian chains can form duplexes, thus probably contributing to the formation of a polysaccharidic network enveloping and protecting bacteria against external threats.
12
Chemical structure of de-acetylated cepacian repeating unit.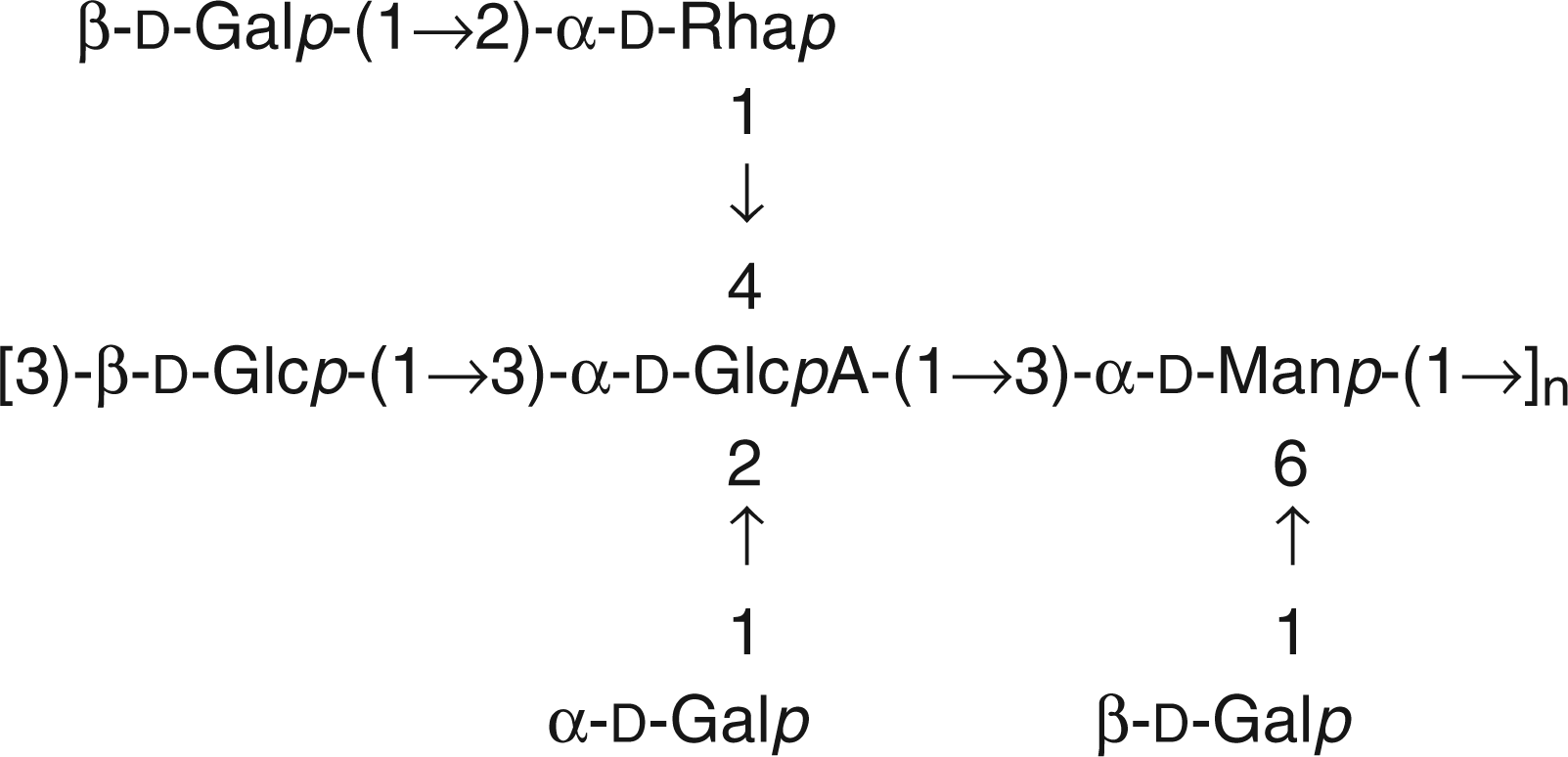
Many investigations on the reactions between hypochlorite and amino acids 13 or nucleic acids components 14 have been described in the literature. On the contrary, very few have focussed on the effect of this oxidising agent on polysaccharides,2,3 although the latter are exposed on bacterial cells as capsular polysaccharides, EPSs and LPSs, thus constituting the interface with the environment.
The investigation of the protective effect of cepacian was first carried out by determining the survival of Burkholderia pyrrocinia BTS7 clinical isolate after treatment with sodium hypochlorite (NaClO), with and without pre-incubation with cepacian. Then, in order to better understand the molecular changes induced by NaClO on cepacian, the EPS was treated in vitro with the oxidising agent. The time course of the reactions was followed by capillary viscometry; the reaction products were analysed by size-exclusion chromatography, NMR spectroscopy and mass spectrometry.
Materials and methods
Bacterial strains and solutions
The water used in all experiments was purified using an Elix system (Millipore, Billerica, MA, USA). Ps. aeruginosa strain PAO1 was a kind gift of Professor J. Govan (UK), B. pyrrocinia strain BTS7 was isolated from a CF patient in care at the Regional Centre for Cystic Fibrosis located in Trieste, Italy. 15 NaClO was purchased from Sigma-Aldrich (St Louis, MO, USA) as a solution (available chlorine 10-13%) and diluted to the desired concentration by measuring the absorbance at 290 nm. Reactions were carried out at pH 7.0 in 20 mM phosphate buffer (PB) as: (i) the HClO pKa is 7.54; (ii) acid catalysed hydrolysis could be avoided; and, (iii) Segal et al. reported that after ingestion of foreign microorganisms the pH in phagosomes increases up to 7.75. 16
Evaluation of cepacian production by B. pyrrocinia strain BTS7 in different media
B. pyrrocinia strain BTS7 is a clinical isolate recovered from the sputum of a CF patient and is a good cepacian producer. 15 It displays a mucoid phenotype on yeast extract mannitol (YEM) agar plates (20 g mannitol, 2 g yeast extract, 15 g agar per litre) but a non-mucoid phenotype on Luria broth (LB) agar plates. Cepacian production in both media was evaluated by growing BTS7 in 50 ml broth at 30°C for 24 h with orbital shaking. Bacteria were separated from the supernatant by centrifugation and the EPS was precipitated from the supernatant with four volumes of isopropanol, dissolved in water, and freeze-dried.
Killing of BTS7 by ClO−
BTS7 grown in YEM medium, as described above, was divided in two parts. One part was treated directly with NaClO, whereas the other part first had the EPS removed by centrifugation (25,000 g for 30 min). The harvested bacteria were re-suspended in an equal volume of PBS and treated with NaClO. Bacterial suspensions (0.25 ml) were added to 2.25 ml NaClO (60 µM final) in PBS, mixed briefly and allowed to stand for 15 min. Then, 1 ml was serially diluted and counted on agar plates to evaluate surviving cells. In another experiment, BTS7 grown in LB medium was centrifuged and re-suspended as described above. Bacterial suspension (0.25 ml) was added to 2 ml of purified cepacian (100 µg/ml or 1 mg/ml final) in PBS, mixed briefly and allowed to stand for different times before adding NaClO and incubating for further 15 min. Surviving cells were evaluated as described above.
Growth of bacterial strains and EPS purification
Ps. aeruginosa strain PAO1 was grown directly on Pseudomonas Isolation Agar plates at 37°C for one night, followed by 2 d of incubation at room temperature (22°C). Burkholderia pyrrocinia strain BTS7 was grown for 16 h at 30°C in 5 ml of LB with gentle shaking. Cell suspension, diluted 1:1000, and 100 µl of the diluted suspension were distributed on each solid YEM agar plate. Bacteria were cultured at 30°C for 3 d and at room temperature for 1 d. For both strains, the mucoid layer was harvested with NaCl 0.9%, NaN3 was added and the bacteria were stirred for 18 h at 10°C. Bacterial cells were separated by centrifugation at 35,000 g for 30 minutes at 4°C. The supernatant was collected and the polysaccharide was precipitated with four volumes of cold isopropanol. The polysaccharide was then dissolved in water, dialysed first against 0.1 M NaCl and then distilled water. The solution was treated with NaOH to get a pH of about 7 and filtered with 0.45 µm membranes. The absence of nucleic acids and proteins was checked by UV spectroscopy using a Ultraspec 2000 (Pharmacia Biotech, Uppsala, Sweden) spectrophotometer. Samples were recovered by freeze-drying. De-acetylation was performed by dissolving the polysaccharide in 10 mM NaOH at 1% w/v concentration and stirring the solution under a nitrogen flow for 5 h at room temperature.
Capillary viscometry measurements
Viscometric measurements were carried out using automatic Schott-Geräte equipment with a Cannon-Ubbelohde suspended level capillary viscometer (diameter 0.53 mm) immersed in a CT 1150 Schott-Geräte water thermostat (30.0 ± 0.1°C). Polysaccharide solutions were prepared in 20 mM PB pH 7.0 and filtered on 0.45 µm membranes prior to analysis. The concentrations chosen for the two investigated polysaccharides were 0.4 mg/ml (alginate) and 0.8 mg/ml (cepacian). At the beginning of each experiment, a volume of 12 ml of polysaccharide solution was used to measure the flowing time in the capillary. Subsequently, 1 ml of hypochlorite solution at a given concentration was added directly to the polysaccharide solution and flowing time was recorded continuously. Different NaClO concentrations were tested: 0.0, 0.2, 0.3, 1.0 and 1.8 mM on alginate solutions and 0.0, 0.4, 1.0 and 2.0 mM on cepacian solutions. For each experiment, data were collected continuously for the first 3 h and then the value of the plateau was measured after 20 h of incubation.
Determination of hypochlorite consumption
Consumption of NaClO was determined spectrophotometrically. The absorbance of 0.86 mg/ml cepacian solutions in 0.2 mM and 2 mM NaClO was monitored at 290 nm for 30 min. The consumption of hypochlorite was calculated using a molar extinction coefficient equal to 300 M−1cm−1. The same measurement was performed on a 0.43 mg/ml alginate solution in 0.2 mM NaClO.
Size-exclusion chromatography of cepacian after ClO− treatment
At the end of viscometric analysis, cepacian solutions degraded with NaClO were freeze-dried and subjected to size-exclusion chromatography on a Sephacryl S-300 column (1.6 cm i.d. × 90 cm). Samples were dissolved in eluent, filtered and degassed 50 mM NaNO3, at a final concentration of ≈15 mg/ml. Separations were performed with 6 ml/h flow rate and fractions were collected every 20 min with a Frac-100 (Pharmacia Biotech) collector. The detector was a WGE Dr. Bures (LabService Analytica, Anzola Emilia, Italy) refraction index connected to a paper recorder and interfaced with a computer via PicoLog software.
Calibration of the size exclusion Sephacryl S-300 column was performed using the chromatogram of cepacian after treatment with 2 mM ClO−. Selected fractions were permethylated 17 and subjected to MALDI-MS analysis.
Degradation of polysaccharides with ClO− for MALDI and NMR experiments
Some samples of cepacian were treated with ClO− without monitoring the reaction by capillary viscometry. In particular: (i) 100 mg of native cepacian was reacted with 15 mM ClO−; (ii) 42 mg of native cepacian was treated with 60 mM ClO−; (iii) two batches of 74 mg each of de-acetylated cepacian were reacted with 60 mM ClO−. Cepacian solutions were prepared at 0.8 mg/ml concentration in 20 mM PB, pH 7. Hypochlorite was added to reach the desired final concentration and the solutions were stirred at 30°C for 20 h. Degraded samples were then dialysed against water using a membrane with a molecular mass cut-off of 1000 u. After dialysis, the samples were adjusted to pH 7 with NaOH and freeze dried. The samples were then subjected to size-exclusion chromatography on a Sephacryl S-300 column, with the same set up and experimental conditions reported above. From all these separations, only fractions in the tail of the peaks were used for subsequent experiments. NMR analysis was performed after desalting of the sample via dialysis, as reported above. Better MALDI mass spectra are obtained on samples with low polydispersity. 18 Therefore, the tail of the size exclusion chromatographic peak of the deacetylated cepacian degraded with 60 mM ClO−, corresponding to oligosaccharides in the mass range 1100–6900 u, was desalted and fractionated on the same Sephacryl S-300 column which was previously equilibrated in water. Fractions were collected every 15 min and the low molecular mass ones were analysed by MALDI-MS in the negative ion mode. For cepacian treated with 15 mM ClO−, fractions within the molecular mass range 5000–10,000 u were selected for NMR analysis.
MALDI-MS
Permethylated samples were dissolved in methanol and mixed in a 1:1 (vol/vol) ratio with the matrix solution (2,5-dihydroxybenzoic acid (DHB) in methanol 30 mg/ml). A volume of 1 µl of the final solution was then deposited onto the MALDI plate and left to co-crystallize at room temperature. MALDI-MS analyses were recorded in positive linear mode using a Perseptive (Framingham, MA, USA) Voyager STR instrument equipped with delayed extraction technology. Ions formed by a pulsed UV laser beam (nitrogen laser, λ = 337 nm) were accelerated through 24 kV.
The sample Cep-deAc-60 was dissolved in trifluoroacetic acid 0.1% and MALDI spectra were performed using DHB 50 mg/ml in trifluoroacetic acid 0.1%–acetonitrile 80:20 (v/v) as matrix solution. Reflectron MALDI-MS and MS/MS analyses were performed in negative polarity with a MALDI-TOF/TOF instrument (4800 Proteomic Analyzer, Applied Biosystems). The system is equipped with Nd:YAG laser at a wavelength of 355 nm with <500-ps pulse and 200-Hz firing rate. Approximately 2000 laser shots were accumulated for each spectrum in the MS experiments and 4000–6000 shots were summed for the MS/MS data acquisitions.
NMR spectroscopy
NMR spectra were recorded on a Varian 500 MHz spectrometer. Samples (about 5 mg) were dissolved in 99.9% D2O and freeze dried three times before final solubilisation in 600 µl 99.96% D2O. For native polymers, a preliminary step of sonication was applied in order to reduce their molecular mass. A Branson sonifier equipped with a microtip at 2.8 Å was used, applying 5 pulses of 1 min each with 1 min intervals and keeping the sample in an ice bath. 1H-NMR, correlation spectroscopy (COSY), total correlation spectroscopy (TOCSY) and heteronuclear single quantum correlation (HSQC) experiments were performed at 50°C with the following specific parameters: TOCSY mixing time was 100 ms and HSQC one-bond coupling constant JCH was 140 Hz.
Results and discussion
Killing of B. pyrrocinia BTS7 isolate by ClO−: protection conferred by cepacian
Killing of Burkholderia pyrrocinia BTS7 isolate by NaClO and the effect of cepacian addition.
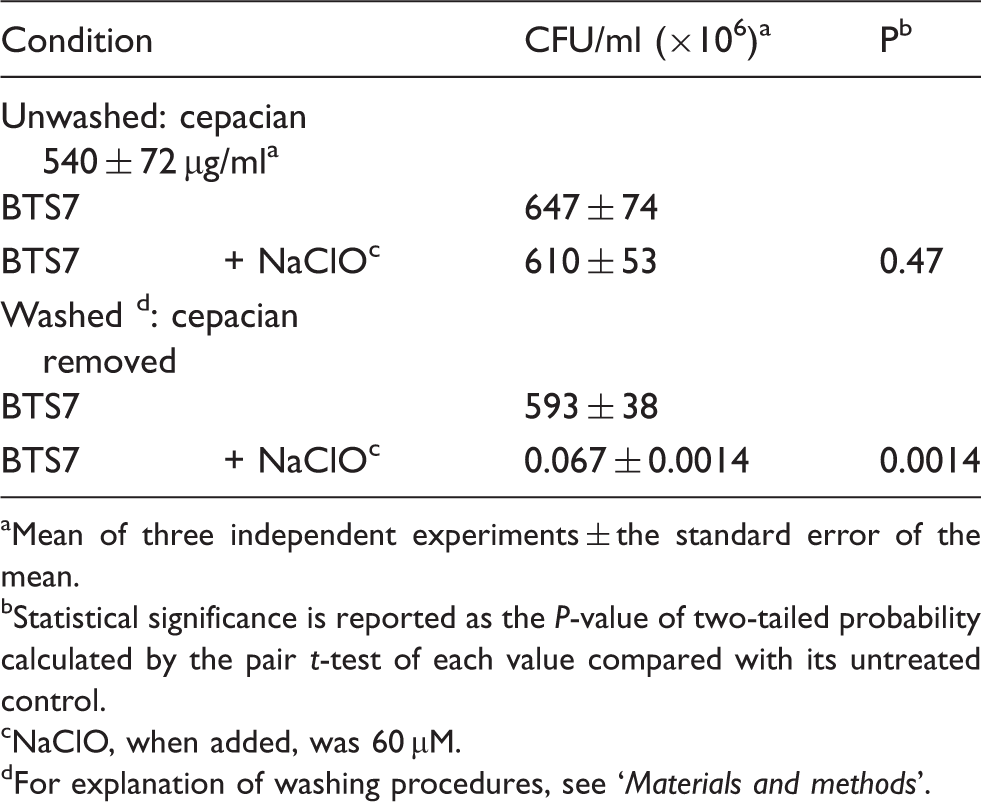
Mean of three independent experiments ± the standard error of the mean.
Statistical significance is reported as the P-value of two-tailed probability calculated by the pair t-test of each value compared with its untreated control.
NaClO, when added, was 60 µM.
For explanation of washing procedures, see ‘Materials and methods’.
To better evaluate the protective role of cepacian, bacteria were grown in LB medium where the EPS is not produced, harvested and re-suspended in PBS before adding different quantities of the purified cepacian. In this way, it was found that a concentration of 100 µg/ml of cepacian did not significantly affect BTS7 resistance to NaClO killing, but 1 mg/ml of EPS markedly improved BTS7 survival in the presence of the oxidant (data not shown). The latter concentration was used in the following experiment, where the effect of bacteria pre-incubation with the EPS together with the protective efficacy of cepacian were evaluated. Treatment of the cells with NaClO showed a three-log decrease in survival with respect to the control (Figure 2: bars NC and B respectively), while simultaneous addition of NaClO and cepacian conferred protection to the cells, resulting in only a one-log decrease (Figure 2: bars B and t = 0 respectively). Moreover, the protective effect of cepacian increased by increasing the incubation time of the cells with EPS before NaClO addition, as shown in Figure 2, resulting in an almost complete scavenging of NaClO after an incubation time of 2 h. Probably, a suitable incubation time of added cepacian with bacteria cells restored a distribution of EPS around the cells resulting in protection from oxidation almost identical to that one given by cepacian produced in situ.
Protective effect of purified cepacian against killing of BTS7 by NaClO. BTS7 grown in LB was harvested and re-suspended in PBS + cepacian 1 mg/ml. NaClO 60 µM was added at different times indicated below the bars. After 15 min of incubation, bacteria were diluted to evaluate CFUs. B: bacteria only (no cepacian, no NaClO); NC: bacteria + NaClO, no cepacian; T0 - T120: incubation time (min) in 1 mg/ml cepacian before addition of NaClO.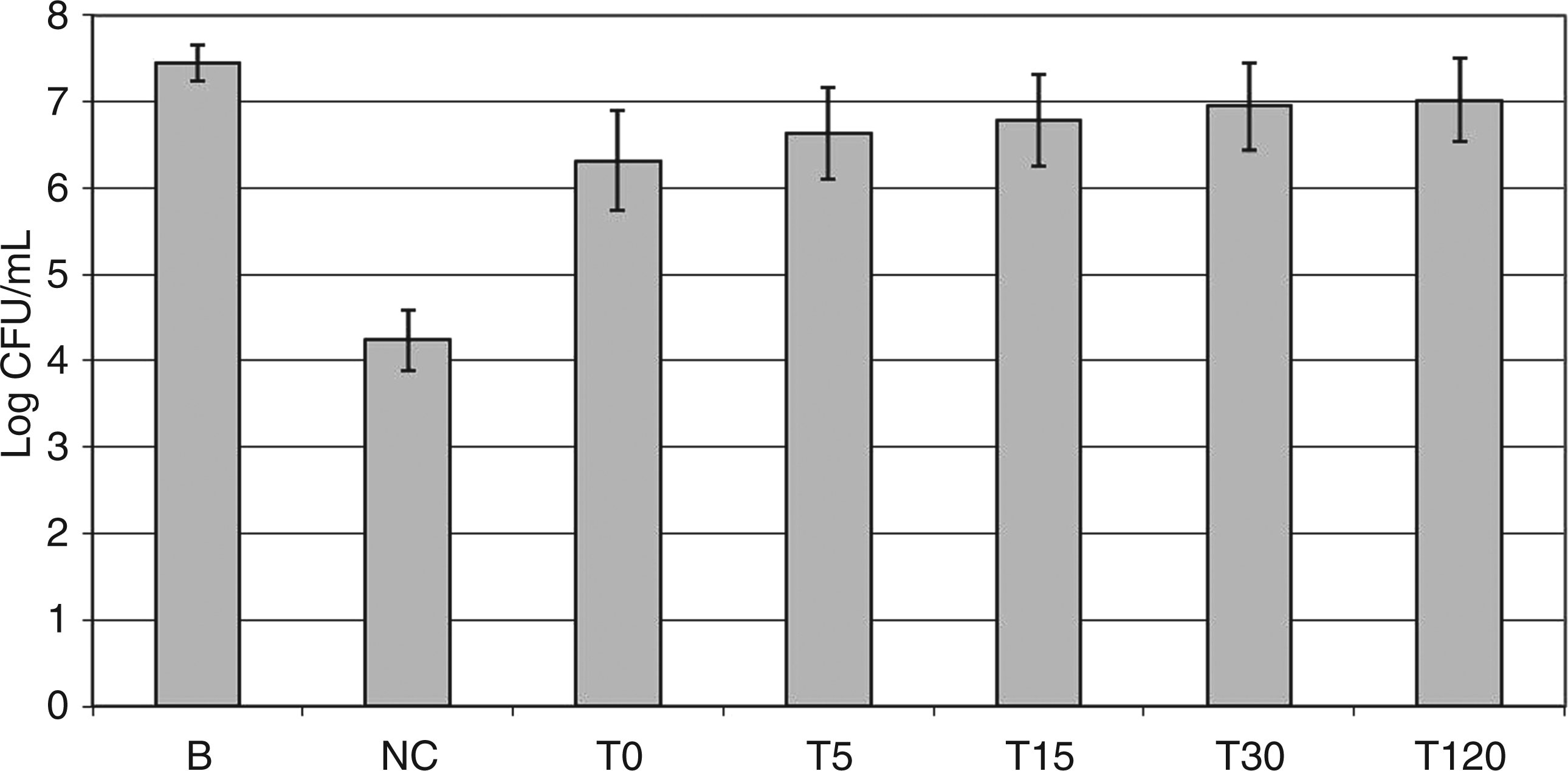
Macromolecular and structural properties of cepacian after ClO− treatment
Although ClO− concentration in biological systems is difficult to determine, some values have been reported in the literature. For example, in phagolysosomes during the oxidative burst, a concentration of ClO− as high as 100 mM was estimated. 1 Therefore, ClO− concentrations in the range 0–60 mM were used in the present study in order to stay within an interval of biological values.
The macromolecular properties of cepacian after treatment with NaClO at different concentrations were investigated by means of capillary viscosity and size exclusion chromatography. Degradation of alginate, a known scavenger of ROS,2,3 was also evaluated by viscosity measurements in order to verify the correctness of the experimental set up. Both polymers were treated with NaClO in the range 0.0–2.0 mM and the reaction was followed by measuring the flowing time of the solutions in a capillary viscometer. For both polymers, a decrease of flowing time as a function of the reaction course was observed, indicating a reduction in molecular mass (Figure 3). Under the experimental conditions adopted, 80% of the total effect of NaClO treatment was obtained within 2 h of reaction. Consumption of NaClO was determined spectrophotometrically, measuring the decrease in absorbance at 290 nm in the first 30 min of reaction. For the oxidation of cepacian (2 ml solution, 0.86 mg/ml) with 2 mM and 0.2 mM NaClO, 190 nmoles and 33 nmoles were consumed respectively. When alginate (2 ml solution, 0.43 mg/ml) was treated with 0.2 mM NaClO, 27 nmoles were consumed.
Capillary flowing time of alginate (A) and cepacian (B) solutions upon treatment with NaClO at different concentrations as a function of the reaction time. Alginate was treated with NaClO 0.0 (▽), 0.2 (□), 0.3 (△), 1.0 (○) and 1.8 (⋄) mM. Cepacian was treated with NaClO 0.0 (▽), 0.4 (□), 1.0 (△) and 2.0 (○) mM.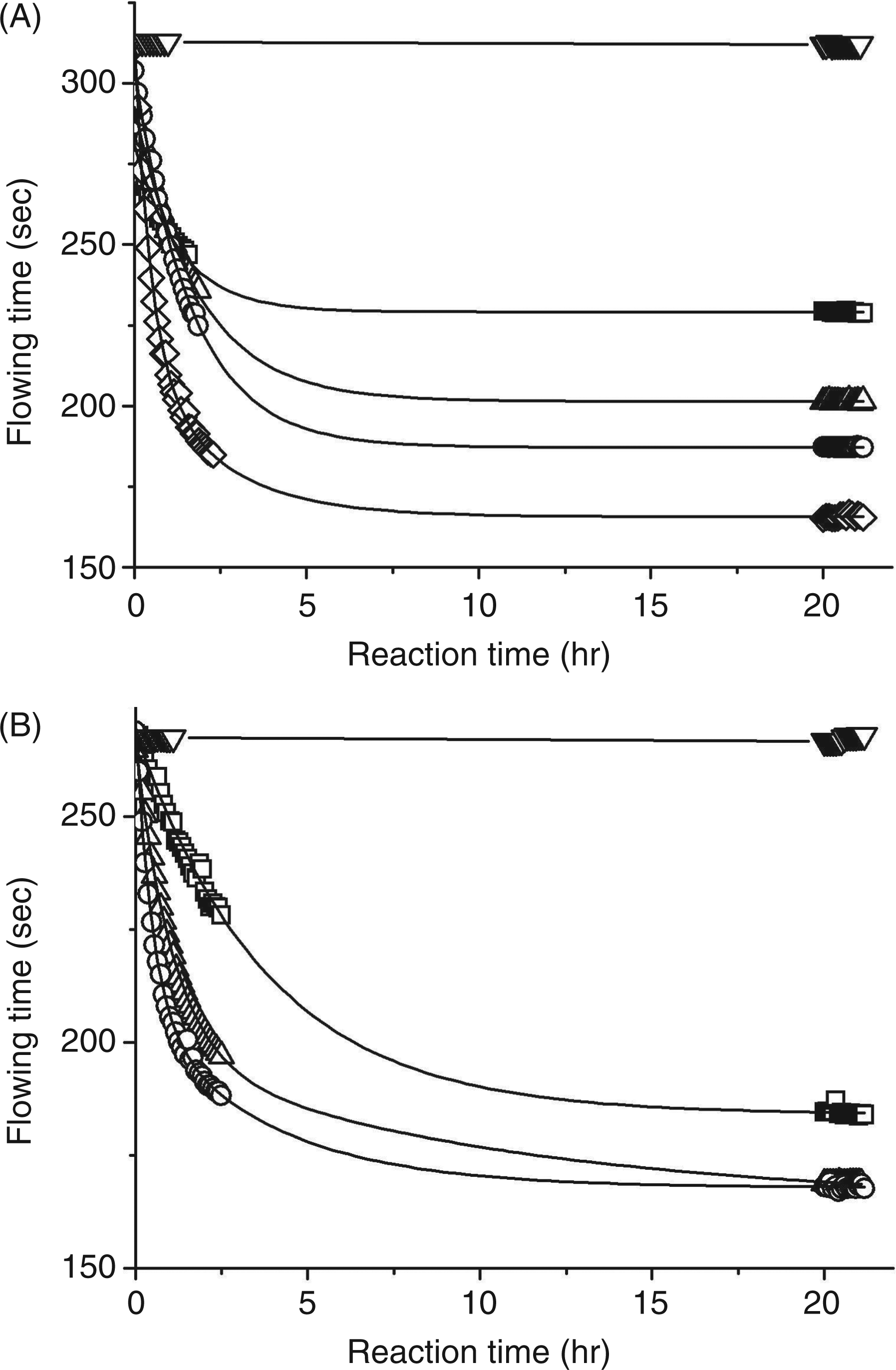
The decrease in mean molecular mass of cepacian treated with NaClO from 0.0 mM to 60.0 mM was investigated by means of size-exclusion chromatography on a Sephacryl S-300 column. The elution profiles obtained are reported in Figure 4A and they clearly indicate that cepacian molecular mass decreased upon increasing of hypochlorite concentration.
Size-exclusion chromatography data obtained for cepacian degraded with NaClO at different concentrations (from bottom to top 0.0, 0.4, 1.0, 2.0, 15.0 and 60.0 mM) (A). Calibration of the size-exclusion chromatogram obtained by treating cepacian with 2.0 mM NaClO by means of MALDI-MS (B).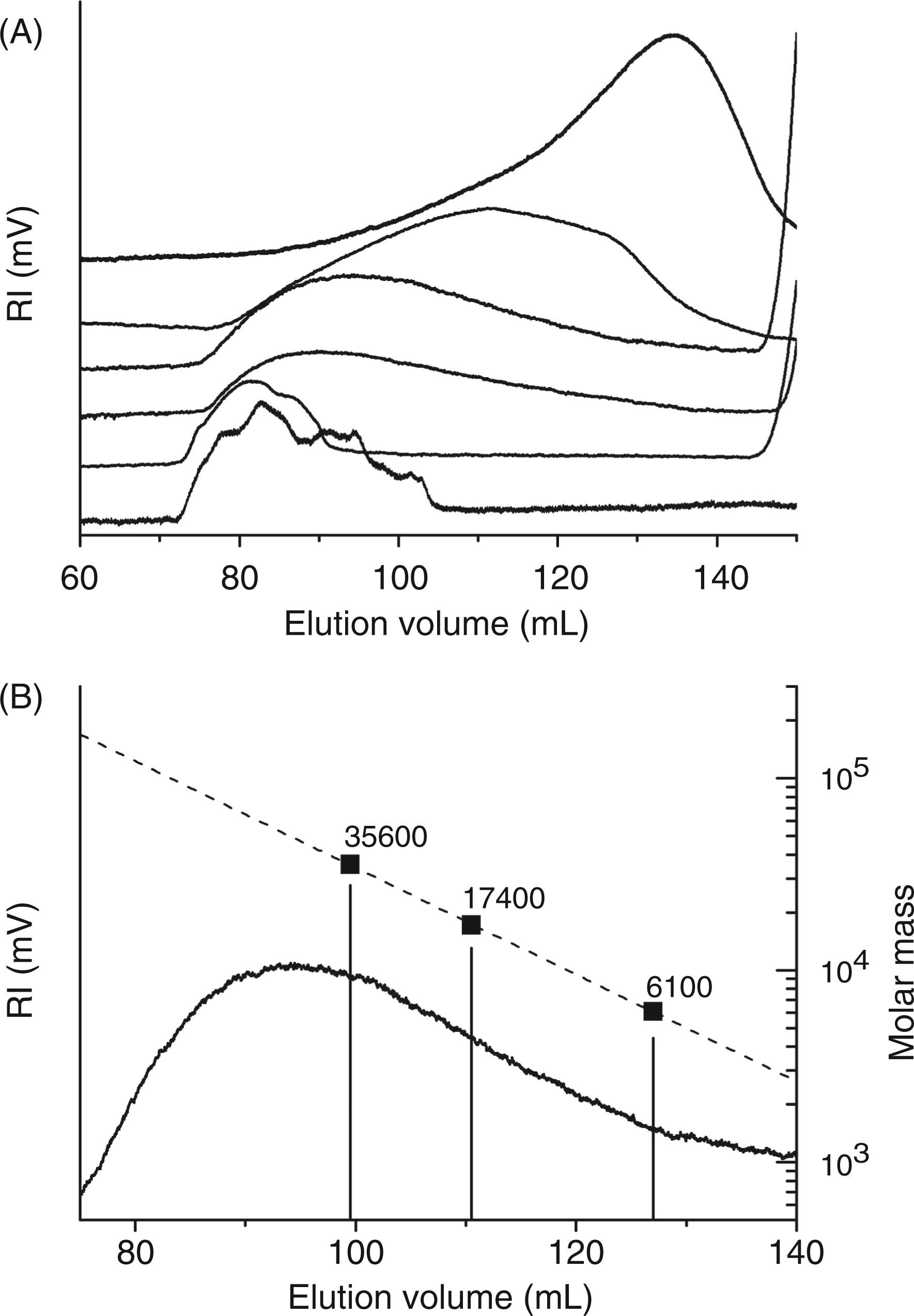
The precise evaluation of cepacian molecular mass after treatment with 2.0 mM NaClO was achieved by means of MALDI-MS. To do this, three fractions named A, B and C (the position of the fractions in the elution profile is indicated in Figure 4B) were collected from the size-exclusion chromatography and, after permethylation, they were subjected to MALDI-MS analysis. The mass values of the permethylated fractions resulted to be A = 41,200 u, B = 20,100 u and C = 7100 u. From these data, the mass values for the same fractions before permethylation were calculated as follows: A = 35,600 u, B = 17,400 u and C = 6100 u. The number of O-acetyl substituents was also included in the calculation and their amount was obtained from 1H-NMR spectra (see below). In this way, a precise calibration of the size-exclusion chromatographic column was obtained and it was used to select fractions with the desired molecular mass for MALDI-MS and NMR experiments.
Acetylation degree after reaction with NaClO.

O-Ac/Rha: molar ratio of acetyls/Rha residues.
In order to have an estimation of the selectivity between acetyl removal and chain cleavage, a plot of the decrease of acetylation and molecular mass as a function of NaClO concentration was obtained (Figure 5). The molecular masses were calculated at the top of each eluting peak, reported in Figure 4, using the MALDI–MS calibration of the S-300 chromatographic column. The data clearly showed that most of acetyls cleavage occurs before an extensive degradation of the polysaccharide backbone.
Decrease of acetylation degree (Table 2) (▪) and of log of molecular mass (▾) as a function of NaClO concentration.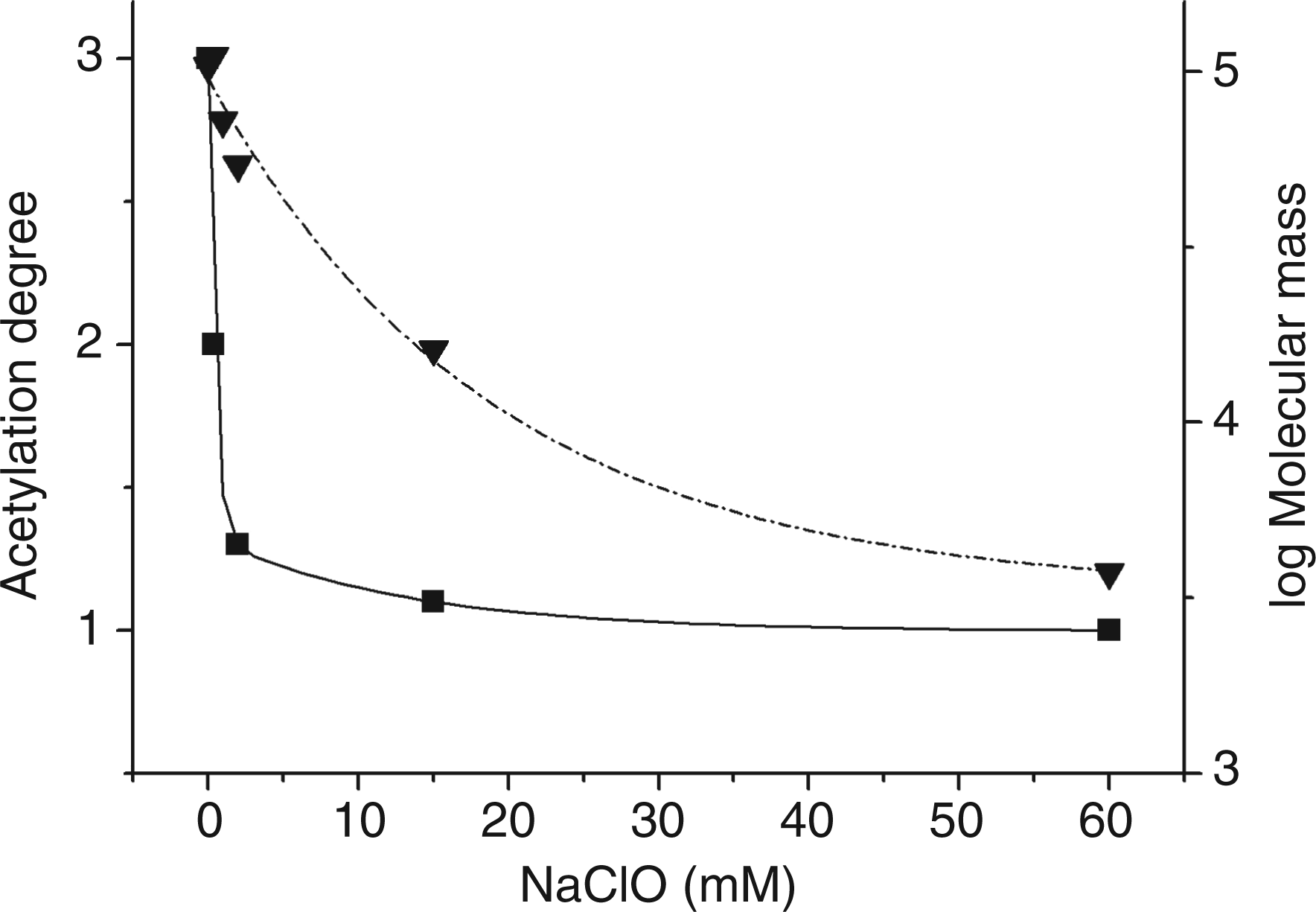
Considering that NaClO 60 mM was in the range of physiological values, and size-exclusion chromatography indicated that it produced suitable amounts of low molecular mass saccharides, detailed NMR and MS analyses were performed on de-acetylated cepacian samples treated with this hypochlorite concentration. The use of de-acetylated polysaccharide resulted in less signals, both in mass spectrometry and NMR experiments. Although de-acetylation inevitably led to loss of information, it was necessary to permit the identification of structural details of the oligosaccharides produced.
MALDI-MS of single fractions isolated by size-exclusion chromatography
Assignment of the ions generated upon MS/MS of the parent ion at 957.61 u.
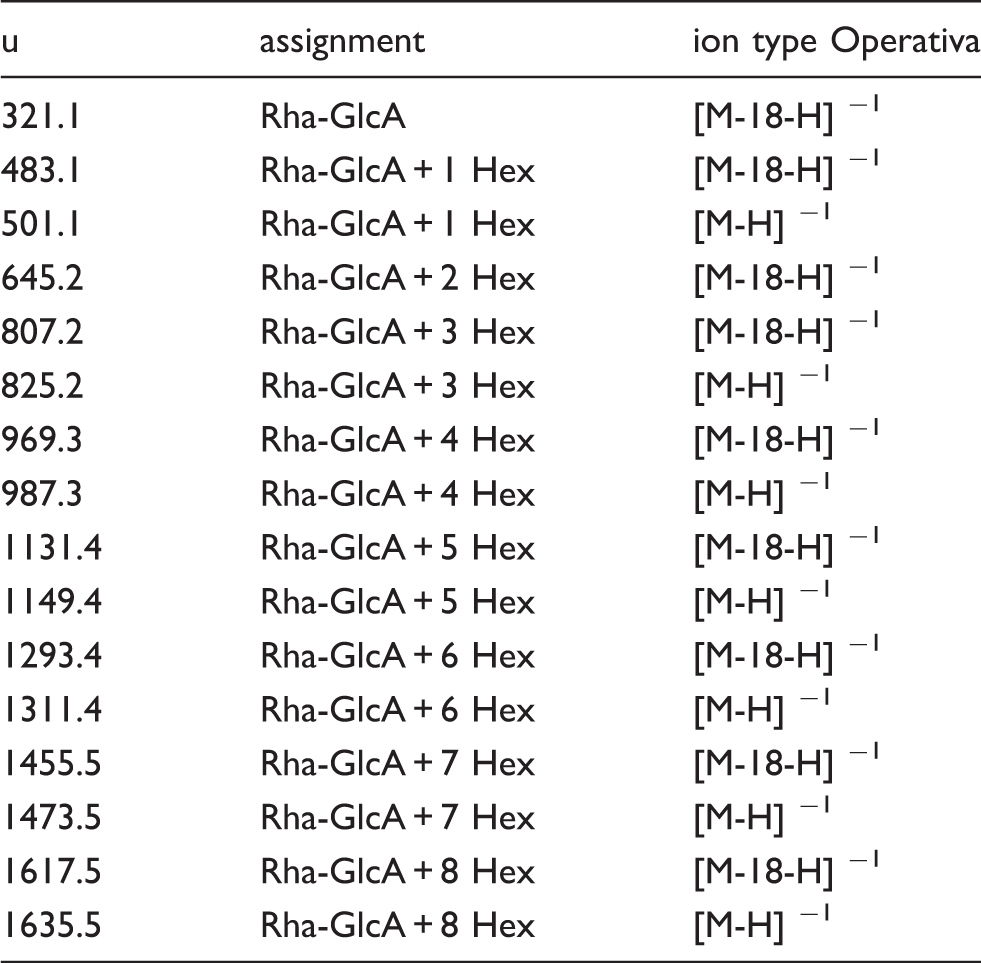
Assignment of oligosaccharides in Cep-deAc-60.

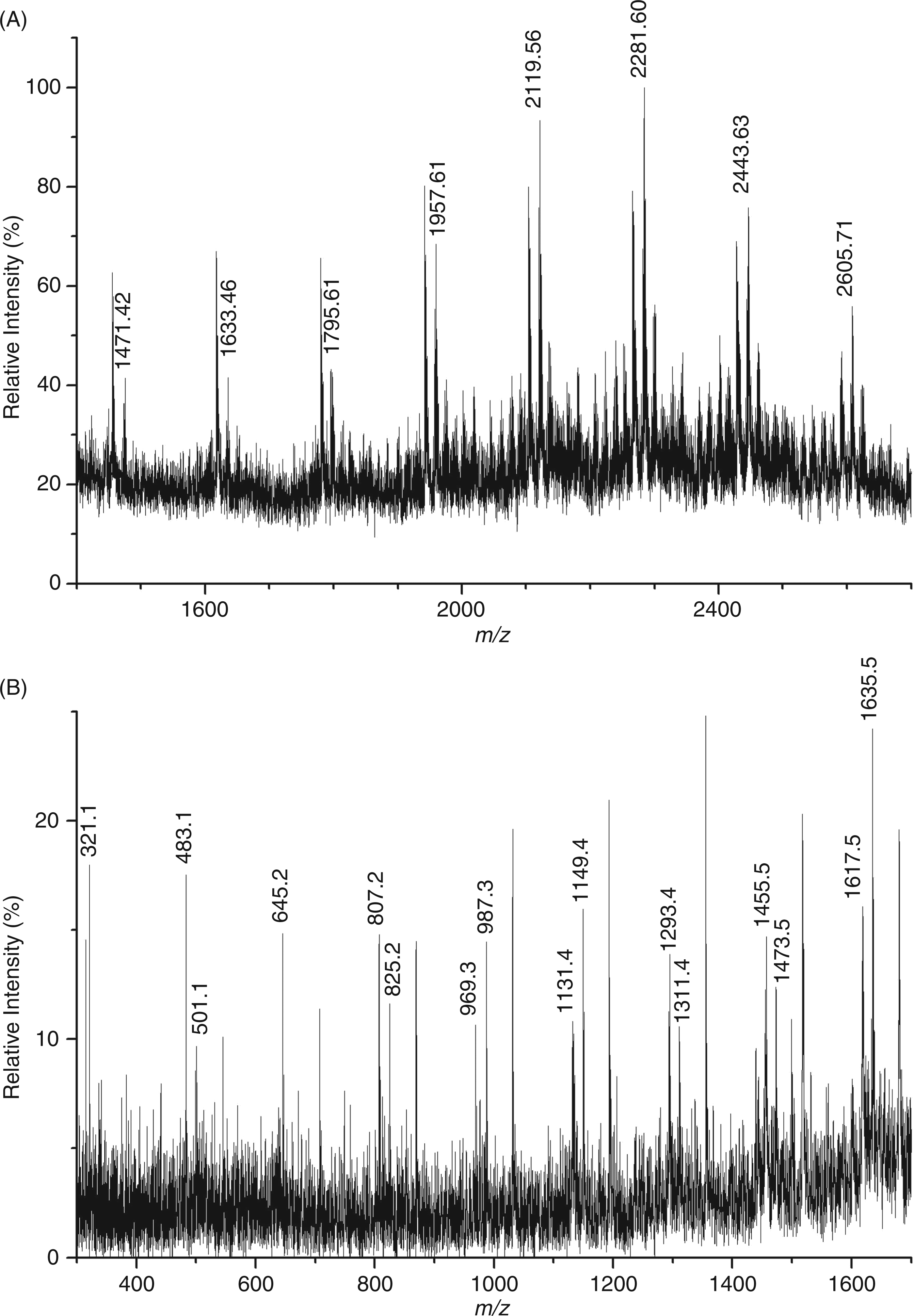
MALDI-MS of Cep-deAc-60 (A) and MALDI-MS/MS of Cep-deAc-60 (B) recorded in the negative mode.
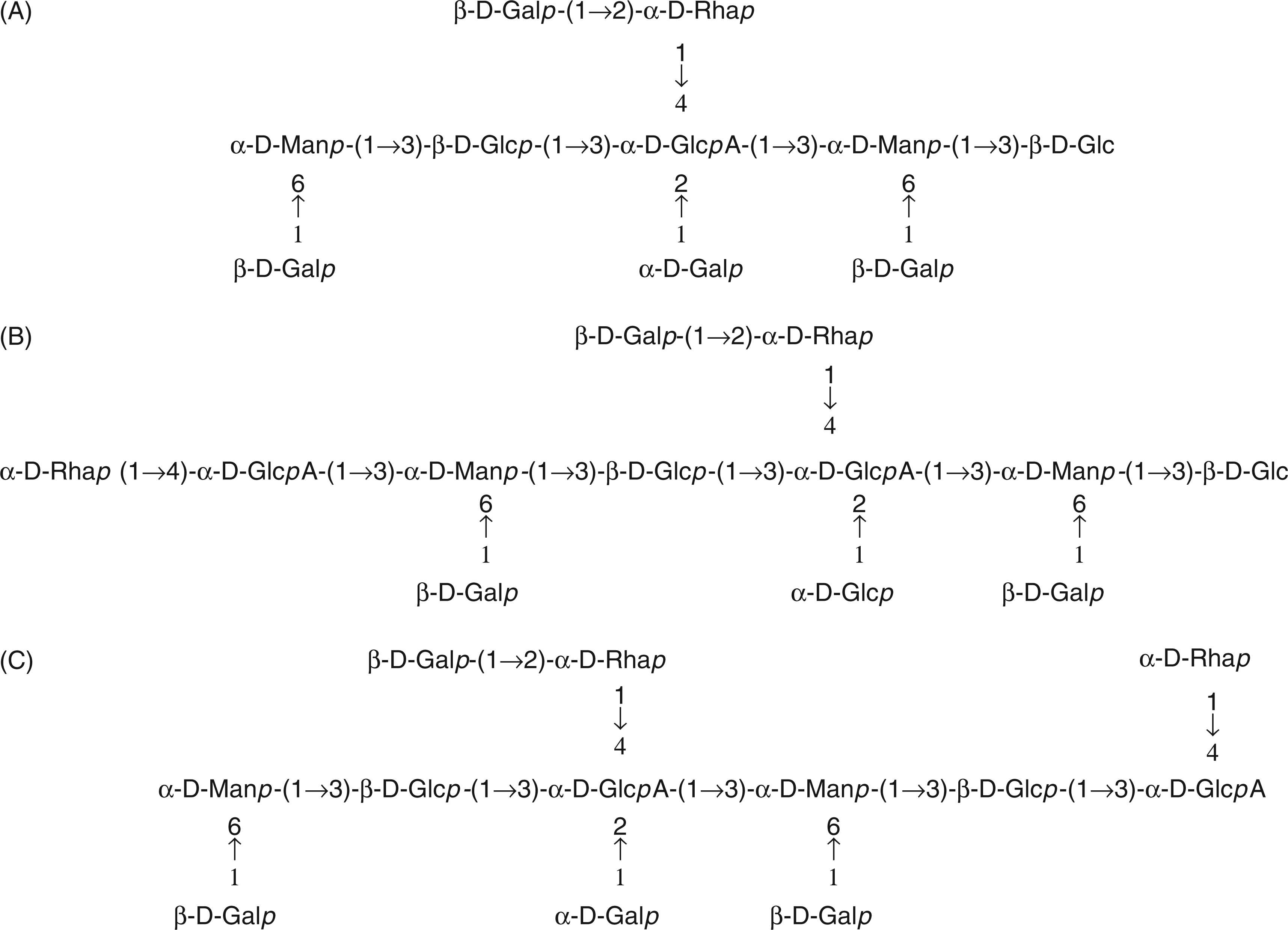
Structures of oligosaccharides deduced from MALDI-MS and present in Cep-deAc-60. (A) Structure of oligosaccharide representing the fragment ion at m/z 1635.6 u. (B) Structure of an oligosaccharide compatible with the ion at m/z 1957.61 u. (C) Structure of another oligosaccharide compatible with the ion at m/z 1957.61 u.
NMR investigation on de-acetylated samples in fractions 70–77 after 60-mM NaClO treatment
For NMR spectroscopy, a sample of de-acetylated cepacian degraded with 60 mM NaClO was separated by size-exclusion chromatography on a Sephadex S-300 column using 0.05 M NaNO3 as eluent and only the fractions (sample named Fr 70–77) with a molecular mass range of 1000–2500 u, very close to that of the sample Cep-deAc-60 used for MALDI-MS, were analysed. In this way, the data obtained from MALDI-MS studies could be used for NMR interpretation. The 1H-NMR spectrum obtained was well resolved owing to the low molecular mass of the analysed species. An expansion of the 1H-NMR anomeric region is compared with that of de-acetylated, non-oxidised cepacian in Figure 8. The complexity of the spectrum of the degraded sample is caused by its composition—a mixture of oligosaccharides differing in size and structure—as demonstrated by MALDI-MS analysis for a single fraction. Nevertheless, a thorough inspection of the data gave interesting structural information. The following discussion is also based on the data obtained investigating COSY, TOCSY and HSQC plots (data not shown).
Expansion of the anomeric region of the 1H NMR spectrum recorded at 50°C of the combined fractions 70–77 obtained from size-exclusion chromatography of de-acetylated cepacian degraded with 60 mM NaClO (lower trace) and untreated (upper trace).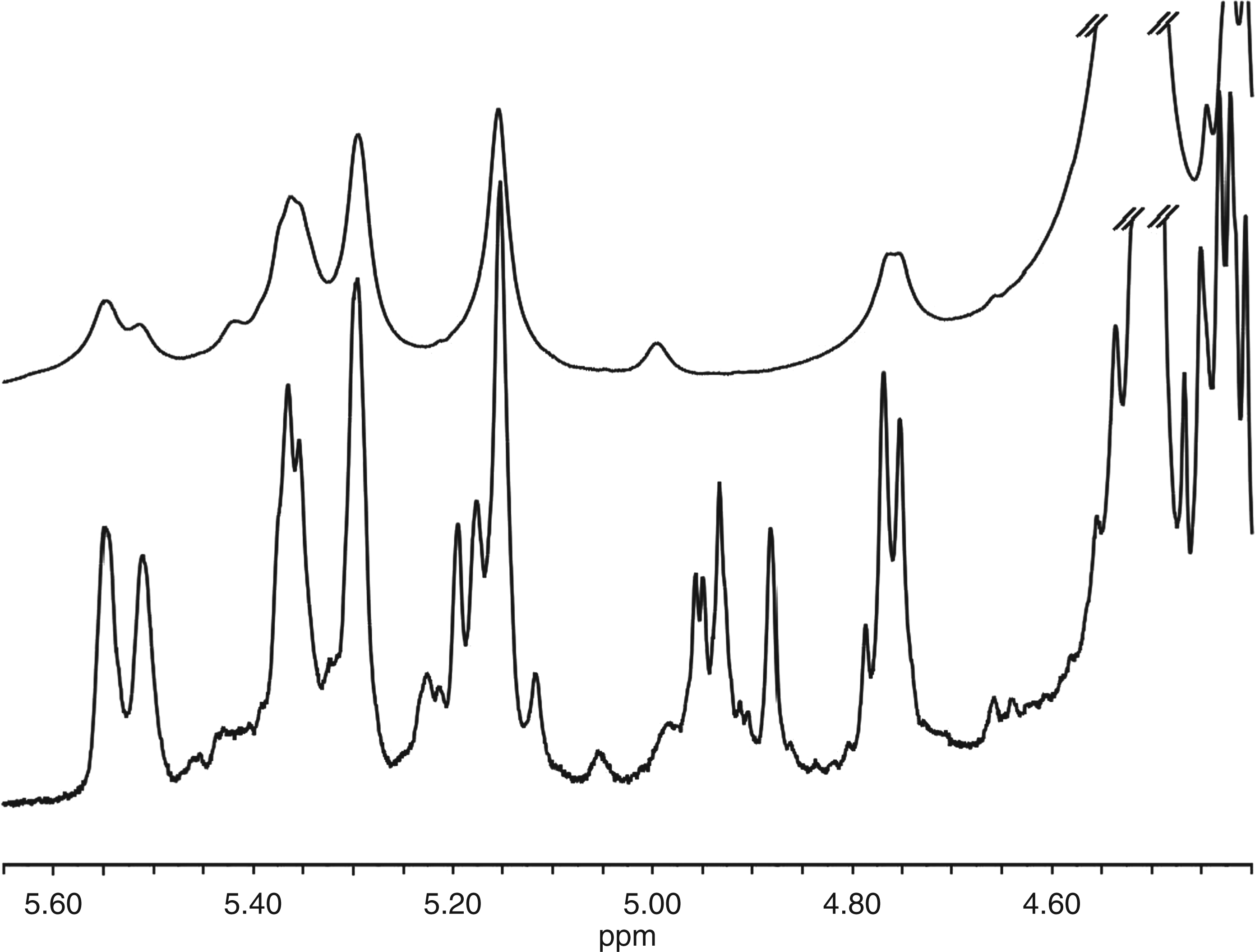
The main features of the 1H-NMR spectrum of NaClO treated cepacian sample, with respect to the native one, are hereafter reported and are discussed on the basis of the MALDI-MS findings. In fact, the oligomers structures depicted in Figure 7 (B, C) refer to species with a size comparable with those present in the chromatographic fractions used for NMR analysis.
The H-1 GlcA signal at 5.51 and 5.55 ppm is twinned. The H-1 Rha signal is also twinned. The twinning of both signals was caused by the presence of GlcA residues differently substituted (see Figure 7C) owing to some loss of terminal Gal residues from the Gal-Rha lateral chain. The H-1 t-α-Gal signal is still present; however, the H-1 Man/Gal ratio increased to 2 from 1 in the native polymer. Actually, the loss of several Gal residues from cepacian lateral chains increased the Man/Gal ratio. The H-1 Man resonance was split into multiple H-1 signals. Parallel to GlcA and Rha anomeric protons, also Man anomeric protons experience different chemical environments, thus resulting in multiple resonances.
Other NMR signals present in the 1H-NMR spectrum of the NaClO treated cepacian sample are rather difficult to interpret owing to the heterogeneities of the species produced by ROS action and were not assigned. Nevertheless, the good agreement between MS and NMR data fully justify the structural details proposed for the cepacian oligomers obtained.
Conclusions
The interaction between pathogenic bacteria and the host immune system is a very complex process, involving different molecules of both the invading microbe and of the host. A better understanding of the mechanisms involved in this interaction and of the molecular reactions occurring will help in characterising the virulence factors of the microbial invaders and in evaluating their importance in infection maintenance. The focus of this investigation was on the biological activity of EPSs and their interactions with ROS. In particular, the strain considered is a clinical isolate of the B. cepacia complex—opportunistic pathogens of CF patients. Using killing experiments, it was demonstrated that the EPS cepacian, which is the characteristic EPS of the BCC, has, indeed, a protecting role towards NaClO action. These findings are in agreement with data reported for a rugose variant of Vibrio cholerae O1 El Tor, which showed an association between EPS production and acquired chlorine resistance, compared with the smooth variant. 25 Viscosity and size-exclusion chromatography experiments were able to detect the rapid degradation of the cepacian backbone, thus indicating its role in ROS scavenging. In addition, mass spectrometry and NMR experiments permitted the assignment of structures to some oligosaccharides obtained by NaClO action.
One of the structural effects of NaClO, besides degradation, was cleavage of acetyl substituents which occurred before extensive polymer degradation. Acetyl removal greatly affects the aggregation ability of cepacian chains, as it has been previously demonstrated that acetylation is involved in the formation of chain duplexes. 12 Therefore, cleavage of O-acetyl groups decreases the capacity of chains to aggregate and, as a consequence, renders the reaction sites on cepacian chains more accessible to the action of NaClO, eventually facilitating the production of low molecular mass oligosaccharides. It could be interesting to investigate if these oligosaccharides might have other biological role. In fact, preliminary experiments (data from our laboratory) on the complexation of alginate with antimicrobial peptides of the innate immune system showed that low molecular mass species might have a larger complexation ability than the native high molecular mass polymer, thus better contributing to the inhibition of peptide activity.
Footnotes
Funding
This work was supported by the Italian Cystic Fibrosis Foundation (Project #11/2006 with the contribution of CF delegation of Belluno, Italy); the Italian Ministry of University and Research (PRIN 2007); and the FVG regional project R3A2 within L.R.26/2005.
Conflicts of interest
The authors have no conflicts of interest to declare.
