Abstract
We have recently reported that neutrophils play a pivotal role in innate defense against Streptococcus pneumoniae (Spn) during mouse acute otitis media (AOM). However, the underlying mechanism remains unclear. By constructing models of pneumococcal AOM in C57BL/6 mice and using a specific inhibitor in vivo, we investigated the role of myeloperoxidase (MPO), one of the most important protein components of neutrophils. Experiment results showed a significant increase in MPO production of the recruited neutrophils in Spn-infected mice. Neutrophils killed Spn in a MPO-dependent manner. MPO facilitated the generation of reactive oxygen species (ROS), and consequently promoted Spn clearance at an early stage and exacerbated tissue damage. Moreover, MPO induced neutrophil apoptosis and necrosis, which, in turn, worsened tissue damage. In summary, our study demonstrates that neutrophil MPO plays a paradoxical role in bacterial clearance and tissue damage in pneumococcal AOM.
Introduction
Acute otitis media (AOM) is one of the most common infectious diseases among children aged ≤ 2 yr, and about 40% of them will have six or more recurrences up to the age of 7 yr. 1 Furthermore, AOM may cause a number of serious complications, such as mastoiditis, facial paralysis and bacterial meningitis.2–4 Reports have indicated that pathogenic bacteria exist in the middle-ear fluid of up to 70% of patients with AOM, and 50% of the infections are due to Streptococcus pneumoniae (Spn). 5 With the widespread use of antibiotics, the incidence of AOM and its complications have been under control, but regretfully concomitant drug resistance can progress the condition to prolonged or recurrent otitis media (OM). 6 Once progressing into chronic OM, AOM will likely raise the risk of hearing loss. 7 Given the pressing demand for new treatment targets, it is necessary to decipher the pathogenesis of AOM.
Neutrophils are the first line of innate immune defense system. For pneumococcal infections, whether in asymptomatic nasopharyngeal colonization or invasive pulmonary infection, neutrophils are the first effector cells recruited to sites of infection. Moreover, it has been shown that colonized Spn will develop into invasive infections in neutrophil-depleted mice, suggesting the vital role of neutrophils in defending against Spn. 8 Our prior study has found that neutrophils are the predominant effector cells (> 98) infiltrating the middle-ear cavity during pneumococcal AOM. 9 However, to date, the underlying mechanism of Spn clearance by neutrophils during AOM remains largely unknown.
Myeloperoxidase (MPO) is a major granule constituent of neutrophils, accounting for 5% of total neutrophil protein. 10 MPO performs its essential function by carrying out antimicrobial activity against intracellular microorganisms, using H2O2 and chloride ions to catalyze the formation of highly cytotoxic oxidant HOCl and free radicals with oxidation ability, thus generating the effective MPO–H2O2–chloride microbicidal system. 11 A variety of studies has revealed that MPO not only kills microbes swallowed into the cells, but also can be released extracellularly to cause damage to multiple target organs. In agreement with this notion, studies have documented that high-level production of MPO is detected in many inflammatory diseases, such as infection, ischemia, atherosclerosis and acute myeloid leukemia.11–14 Consistently, clinical data have also shown that MPO exists in abundance in middle-ear lavage fluid (MELF) 15 and middle ear mucosa 16 of patients with AOM. However, the precise role of MPO in pneumococcal AOM is still poorly characterized.
In the present study, we investigated the potential roles of MPO in defending against Spn and in inducing tissue damage in AOM, utilizing a specific MPO inhibitor, 4-aminobenzoic acid hydrazide (4-ABAH), in vivo. Results suggest that MPO plays a crucial role in bacterial clearance mediated by several pathways, but MPO also causes middle-ear tissue injury.
Materials and methods
Bacteria
Spn clinical isolate 31207 (serotype 6B) was purchased from the National Center for Medical Culture Collections (CMCC, Beijing, China). Bacteria were inoculated in casein hydrolysate plus yeast extract (C + Y) medium after overnight (12–16 h) incubation on a blood agar plate at 37℃ in 5% CO2 until cultures reached log-phase (OD600 = 0.6). Spn were then harvested by centrifugation at 3500 g for 15 min, washed twice and re-suspended in sterile pyrogen-free PBS. The bacterial concentration was determined by plating 10-fold serial dilutions on agar plates.
Mice
Male or female C57BL/6 mice aged 6–8 wk were obtained from Chongqing Medical University (Chongqing, China) and housed in a specific-pathogen-free environment. All animal experiments were performed in accordance with the Institutional Animal Care and Use Committee’s guidelines of Chongqing Medical University.
Mouse model of AOM
The mouse models of AOM were established via direct bilateral transtympanic inoculation of the middle ears, as previously described. 17 In brief, mice were anesthetized with i.p. injections of ketamine hydrochloride (20 mg/kg) and xylazine (5 mg/kg). AOM was then induced by direct bilateral transtympanic injection of approximately 5 μl bacterial suspension containing 1 × 107–1 × 108 CFUs of Spn in sterile pyrogen-free PBS. After being challenged with Spn, mice were subsequently treated with equal volume of saline or 4-ABAH (40 mg/kg; Sigma-Aldrich, St. Louis, MO, USA), diphenyleneiodonium (DPI) (1 mg/kg; Sigma-Aldrich), i.p., and the following injections were given every 12 h. To inhibit the caspase activity, mice were inoculated with Spn and the general caspase inhibitor carboxybenzyl-Val-Ala-Asp-fluoromethylketone (Z-VAD-FMK, 50 µM in PBS; R&D Systems, Minneapolis, MN, USA) simultaneously. A mock control cohort of mice was inoculated with 5 µl PBS alone. All experiments were carried out using 4–5 mice per group.
Collection of MELF
Mice were anesthetized and sacrificed at designated time points postchallenge. MELF were collected by lavaging middle-ear space six times with 10 µl sterile pyrogen-free PBS containing 1% BSA each time. Five µl MELF from each ear was used for bacterial quantification and the rest was then centrifuged at 500 g for 10 min; aliquots of the supernatant were stored at –80℃. The cell pellets were washed twice after lysis of red blood cells and re-suspended in PBS for inflammatory cell quantification by a hemocytometer and for subsequent experiments.
Bacterial load determination
Pneumococcal loads in MELF were determined by plating 10-fold serial dilutions of MELF on blood agar plates. Viable colonies were counted after incubation at 37℃ in 5% CO2 overnight.
Quantification of MPO activity in MELF
MPO activity was evaluated by the MPO kinetic-colorimetric assay (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). Briefly, 10 μl supernatant was transferred into PBS (pH 6.0) containing 0.17 mg/ml 3,3′-dimethoxybenzidine and 0.0005% H2O2, and mixed and incubated for 30 min at 37℃ in a water bath. MPO activity of the supernatant was determined by detecting the H2O2-dependent oxidation of 3,3′-dimethoxybenzidine by measuring the absorbance at 460 nm.
ROS determination by flow cytometry
ROS generation was measured by 2,7-dichlorofluorescein diacetate (DCFH-DA; Beyotime, Shanghai, China) staining. DCFH-DA was cell permeable and the diacetate was cleaved to DCFH by intracellular esterases. DCFH was impermeable and oxidized to dichlorofluorescein (DCF), which emits green fluorescence in the presence of ROS. According the manufacturer's instructions, cells (1 × 106/ml) were incubated in the dark with 2 μM DCFH-DA at 37℃ for 20 min, washed three times with PBS to remove extracellular DCFH-DA, then re-suspended in 200 µl PBS and detected by flow cytometry (FCM).
Histopathologic analysis
Mice were sacrificed after general anesthesia at designated time points. Subsequently, the bullae were carefully isolated and then fixed in 4% paraformaldehyde at 4℃ for 24 h, decalcified in 10% EDTA at 4℃ for approximately 1 mo and embedded in paraffin. Serial sections were cut to a thickness of 5 µm by a Leica Microtome (Leica Biosystems, HD, Germany). Representative slides in each group were stained with hematoxylin and eosin. Digital micrographs were taken at standardized positions of the middle ears of each specimen. The thickness of middle-ear mucosa was analyzed with NIS-Elements BR 4.10.00 software by two pathologists in a blinded manner.
Immunofluorescence assay
Middle-ear sections were deparaffinized, rehydrated and subsequently incubated in 0.01 M sodium citrate buffer for antigen retrieval. Sections were blocked with 5% donkey serum at 4℃ overnight after being permeablized by 0.1% Triton X-100, then incubated with primary Ab Rabbit anti-MPO (1:100; Boster, Wuhan, China), and secondary Abs DyLight 594-conjugated donkey anti-rabbit IgG and Alexa Fluor 488-conjugated donkey anti-rabbit IgG (1:1000; Jackson ImmunoResearch Laboratories, West Grove PA, USA). Nuclei were counterstained with DAPI (1:1000; Invitrogen, Carlsbad, CA, USA). Immunofluorescence images were obtained using a digital microscope.
Immunohistochemistry
Middle-ear sections were deparaffinized, rehydrated and then incubated in 0.125% trypsin solution to unmask antigens. Endogenous peroxidase was removed by 0.3% H2O2. The sections were subsequently blocked with 5% BSA for 1 h at room temperature (20–25℃), incubated with rabbit anti-MPO primary Ab (1:100; Boster, Wuhan, China) at 4℃ overnight and HRP-labeled goat anti-rabbit secondary Ab (Zhonghshanjinqiao, Beijing, China), which was developed by a diaminobenzidine substrate kit for peroxidase and then counterstained with hematoxylin. Immunohistochemical images were obtained using a digital microscope.
Lactate dehydrogenase assay
The activity of lactate dehydrogenase (LDH) in supernatant of MELF released by damaged cells was measured by LDH assay kit (Beyotime), according to the manufacturer’s instructions. In brief, the supernatant of MELF was mixed with the detection solution and incubated at room temperature in the dark for 30 min, measuring absorbance at a wavelength of 490 nm.
ELISA
Levels of TNF-α and IL-1β in MELF of each group were measured with commercially available ELISA kits (Biolegend, San Diego, CA, USA) following the manufacturer’s protocol.
Apoptosis assessment
Apoptosis was assessed using Apoptosis Detection Kit (BD Biosciences, San Jose, CA, USA). Briefly, MELF were combined from 4–5 mice as described above, cells were centrifuged at 500 g for 10 min, the cell pellets were washed twice with cold PBS, re-suspended in 1 × binding buffer to a concentration of 1 × 106 cells/ml. Cells were incubated with annexin V in combination with propidium iodide (PI) at room temperature for 15 min in the dark. The events of apoptotic cells were analyzed by FCM within 1 h.
FCM analysis of neutrophils
MELF were combined from 4–5 mice as described above, cells were centrifuged at 500 g for 10 min, the cell pellets were treated with RBC lysis buffer (Biolegend). Non-specific binding was blocked with Mouse BD Fc Block in staining buffer (BD Pharmingen, Franklin Lakes, NJ, USA). A cocktail of fluorophore-conjugated rat anti-mouse cell surface Abs included APC–CD45, FITC–Ly6G and PE–CD11b (BD Pharmingen), and isotype Abs were added to the suspension and incubated for 45 min at 4℃ in the dark. Cells were then washed three times and re-suspended in staining buffer. The events of neutrophils were analyzed by FCM.
Statistical analysis
All data are presented as mean ± SEM. GraphPad Prism software version 5.01 was applied for statistical analysis. Unpaired t-tests were performed for data with normal distribution between groups, and Mann–Whitney U-tests were performed for data with non-normal distribution. Differences were considered statistically significant if P < 0.05.
Results
Spn induces high-level production of neutrophil MPO in AOM
Our earlier study found that neutrophils are the predominant effector cells (> 98) infiltrating the middle-ear cavity in both Spn- and PBS-treated groups.
9
Compared with the PBS group, the MPO production of the recruited neutrophils in Spn-infected mice was markedly up-regulated (Figure 1a, b). Meanwhile, the activity of MPO in MELF supernatant was found to peak on d 3, before gradually declined thereafter (Figure 1c). The results indicate that Spn notably induces high-level production of MPO in recruited neutrophils during AOM. By immunofluorescence and immunohistochemistry staining of middle-ear tissue sections, large numbers of MPO-positive neutrophils were found to localize at the surface membrane of epithelial cells (Figure 1d, e).
Spn induces high-level expression of neutrophil MPO in AOM. (a) Representative immunofluorescence images of middle-ear sections on d 1 after PBS or Spn inoculation. Cells were stained with MPO (red) and DAPI (blue). Scale bar, 25 µm. (b) Immunohistochemical staining of MPO on middle-ear sections on d 1 after PBS or Spn inoculation. Control stains were performed with omission of rabbit anti-MPO primary antibody. Scale bar, 50 µm. (c) MPO activity in combined MELF supernatant at indicated time points after PBS or Spn inoculation. Values represent mean ± SEM (n = 4–5). *P < 0.05. Cumulative data from three independent studies are shown. (d) Immunofluorescent staining of MPO (green) and DNA (blue) on middle-ear sections on d 1 after Spn inoculation. Arrow, MPO-positive neutrophils. Scale bar, 50 µm. (e) Immunohistochemical staining of MPO on middle-ear sections on d 1 post-inoculation with Spn (left), isotype control staining with omission of rabbit anti-MPO primary Ab (right). Arrow, MPO-positive neutrophils. Scale bar, 100 µm.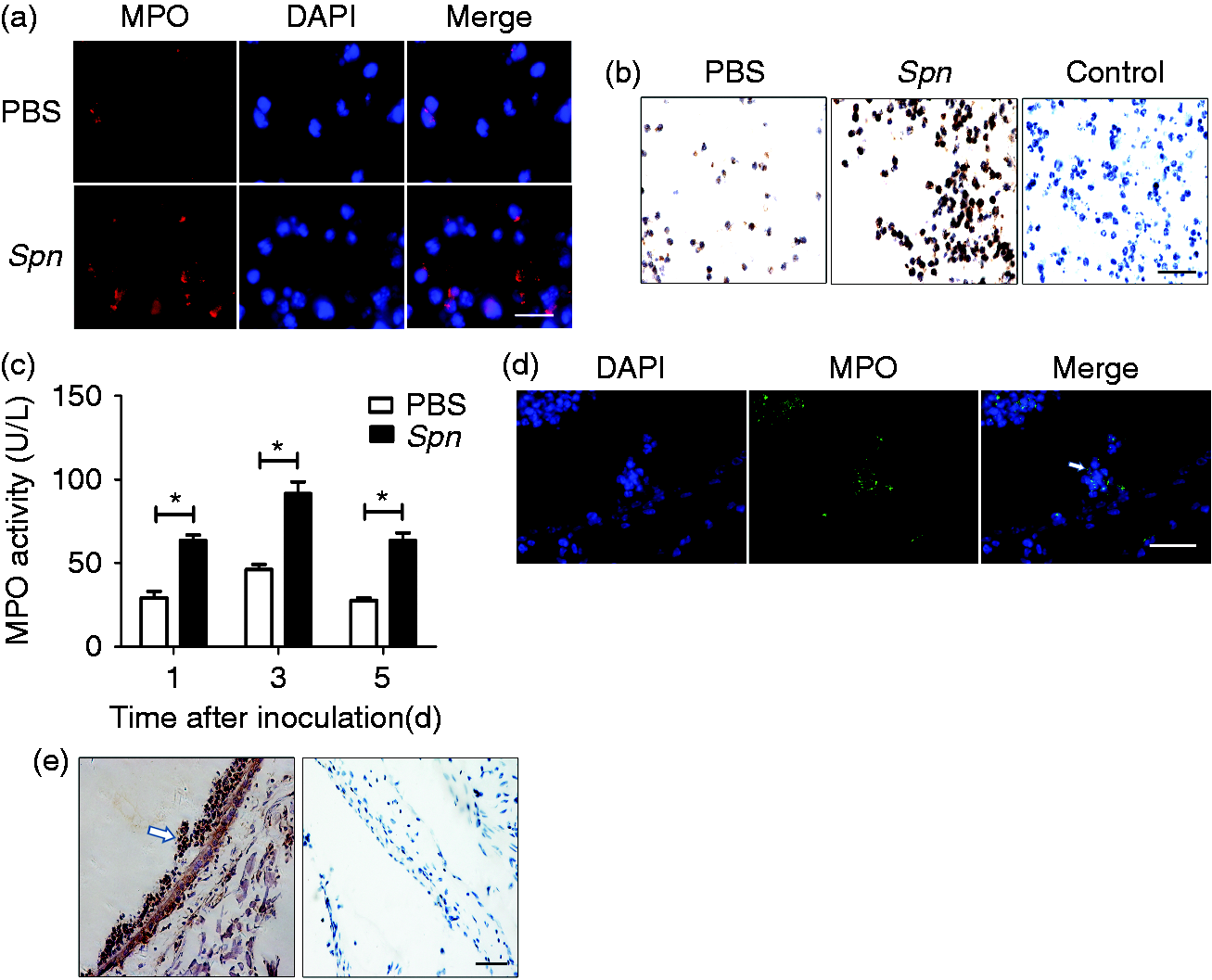
Neutrophils kill Spn in a MPO-dependent manner in AOM
To evaluate the bactericidal effect of MPO, we used a specific inhibitor, 4-ABAH, for MPO. Given the obvious efficacy of 4-ABAH (Figure 2a), we compared the bacterial loads in MELF before and after the treatment of 4-ABAH. We analyzed the cell types and amounts in MELF to ensure the difference in bactericidal capacity was caused by the alteration of neutrophil MPO rather than the types of recruited cells and neutrophil amount. FCM analysis of MELF showed that neutrophils were still the predominant effector cells (> 98) infiltrating to the middle-ear cavity in the 4-ABAH-treated group (see Figure S1 in the supplementary material). The recruitment of neutrophils in each group was closely monitored over time, and no statistically significant difference in the amounts of neutrophils was detected (Figure 2b). Next, we examined the bacterial burden in MELF and found that the Spn density in the Spn + 4-ABAH group increased significantly over the observation period by 5- to 10-fold (Figure 2c). Taken together, these results indicate that MPO plays a pivotal role in Spn clearance in AOM.
Neutrophils kill Spn in a MPO-dependent manner. Mice were treated with PBS, Spn or Spn + 4-ABAH (40 mg/kg). (a) MPO activity of MELF supernatant on d 3 after treatment with PBS, Spn or Spn + 4-ABAH. Values represent mean ± SEM (n = 4–5). *P < 0.05. Cumulative data from three independent studies are shown. (b, c) Quantification of (b) neutrophils and (c) Spn in MELF of the Spn group and Spn + 4-ABAH group at designated time points. Values in (b) represent mean ± SEM, values in (c) represent individual ear with mean shown by a horizontal bar (n = 4–5). **P < 0.01; ***P < 0.001. ns: not significant. Data representative of three independent experiments are demonstrated.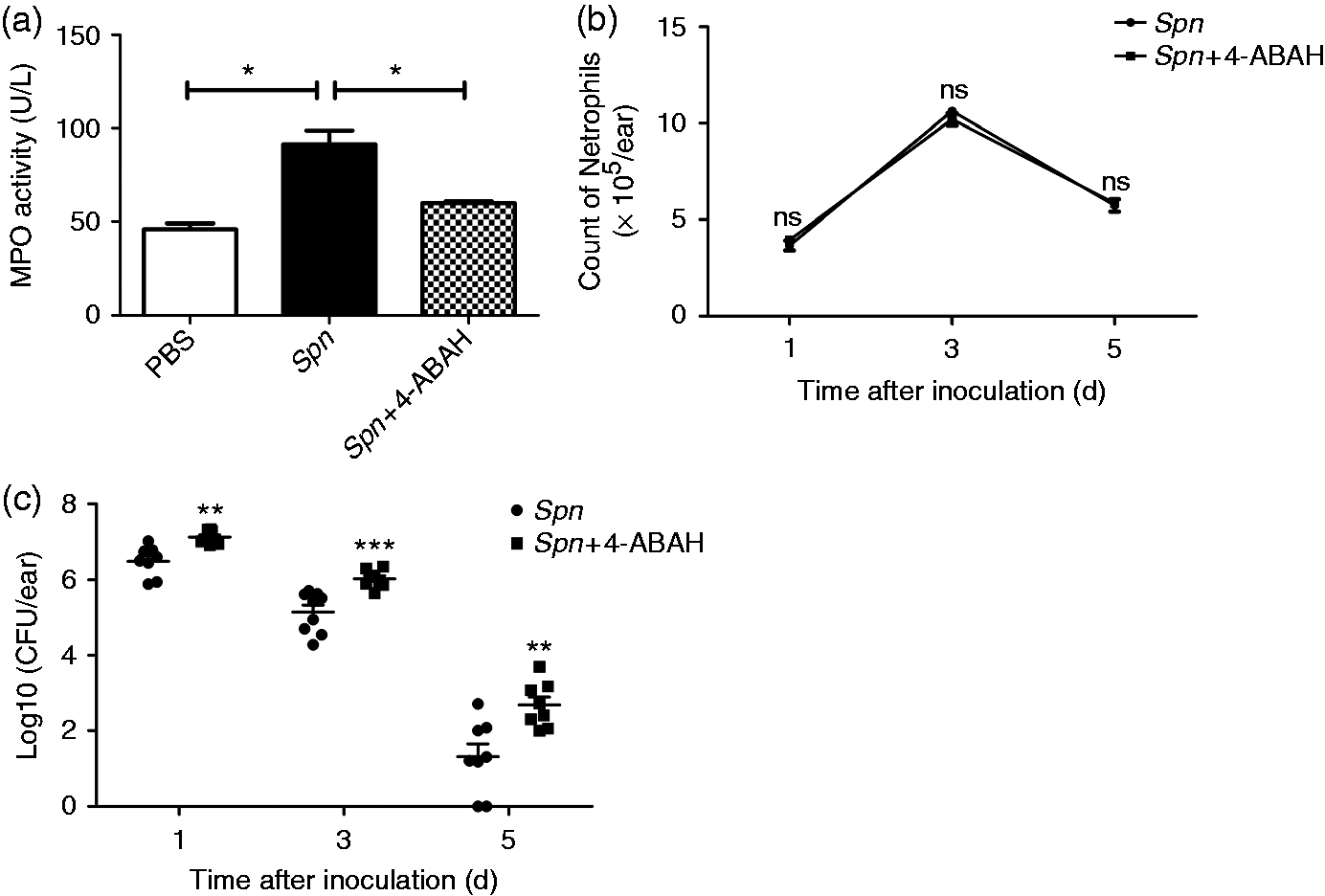
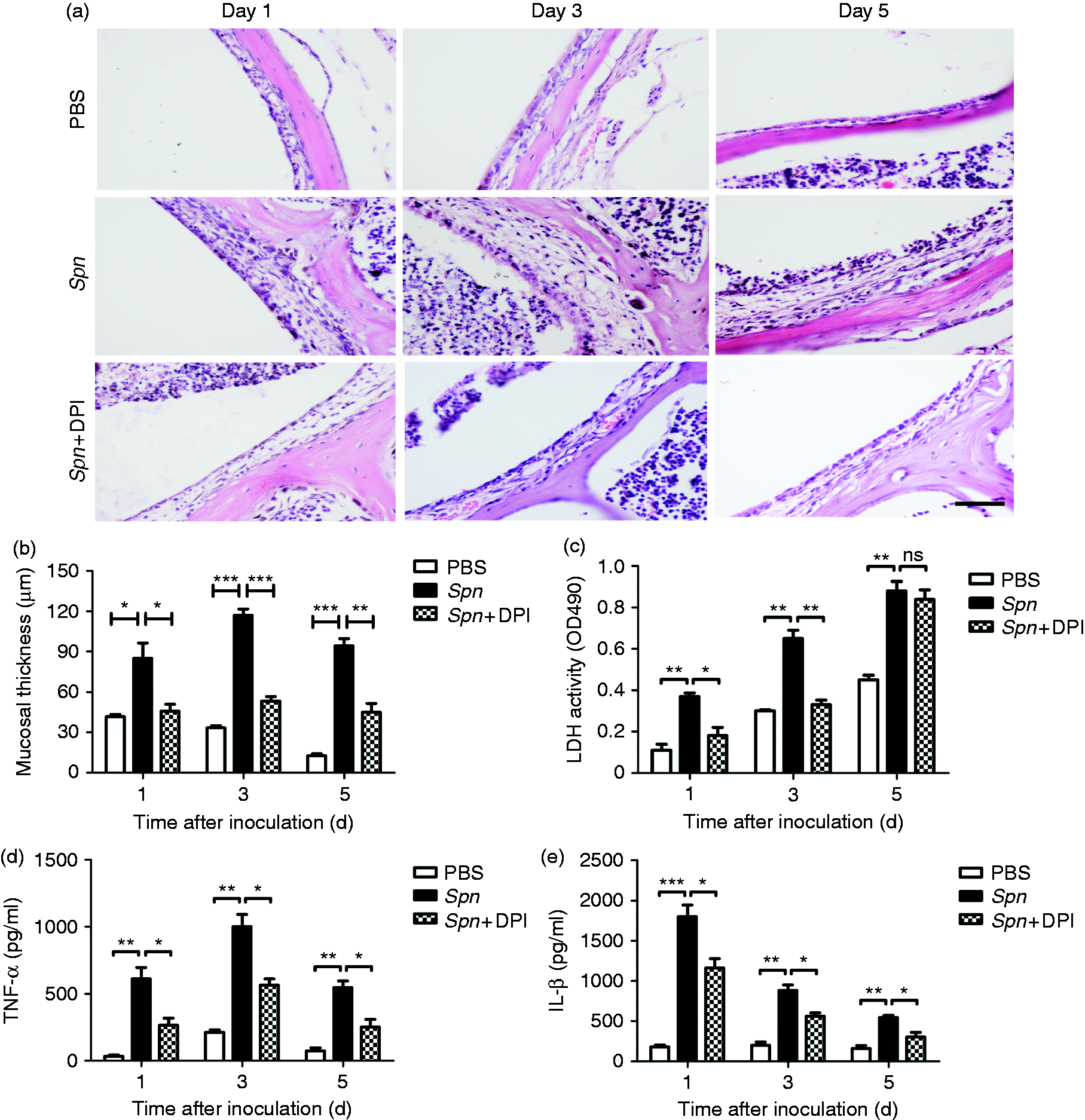
MPO causes tissue damage in AOM
To explore the role of MPO in causing tissue damage in AOM, we examined the extent of tissue damage before and after 4-ABAH treatment. In the PBS group, middle-ear mucosal hyperplasia peaked on d 1, before gradually declined to normal level on d 5. Compared with the PBS group, the Spn group and Spn + 4-ABAH group both manifested remarkably higher mucosal thickness. Further, compared with the Spn group, the Spn + 4-ABAH group was found to have a lesser degree of hyperplasia (Figure 3a, b). The activities of LDH, a marker for general cellular injury,
18
were reduced in 4-ABAH-treated mice on d 1 and 3, and the difference between these two groups on d 5 was not significant (Figure 3c). Meanwhile, the worsening of middle-ear inflammation was usually expressed by enhanced secretion of pro-inflammatory cytokines in MELF. Detection of pro-inflammatory cytokines in MELF showed lower levels of TNF-α and IL-1β in the Spn + 4-ABAH group than in the Spn group (Figure 3d, e). Collectively, these results suggest an important role of MPO in causing tissue damage in pneumococcal AOM.
MPO causes tissue damage. (a) Hematoxylin and eosin staining of middle-ear sections after treatment with PBS, Spn and Spn + 4-ABAH at designated time points. Scale bar, 100 µm. (b) Mucosal thickness quantification of each group. Values represent mean ± SEM (n = 4–5). *P < 0.05; ***P < 0.001. ns: not significant. (c–e) Activities of (c) LDH and levels of (d) TNF-α and (e) IL-1β in MELF supernatant of each group at designated time points. Values represent mean ± SEM (n = 4–5). *P < 0.05; **P < 0.01. ns: not significant. Cumulative data from three independent studies are shown in all panels.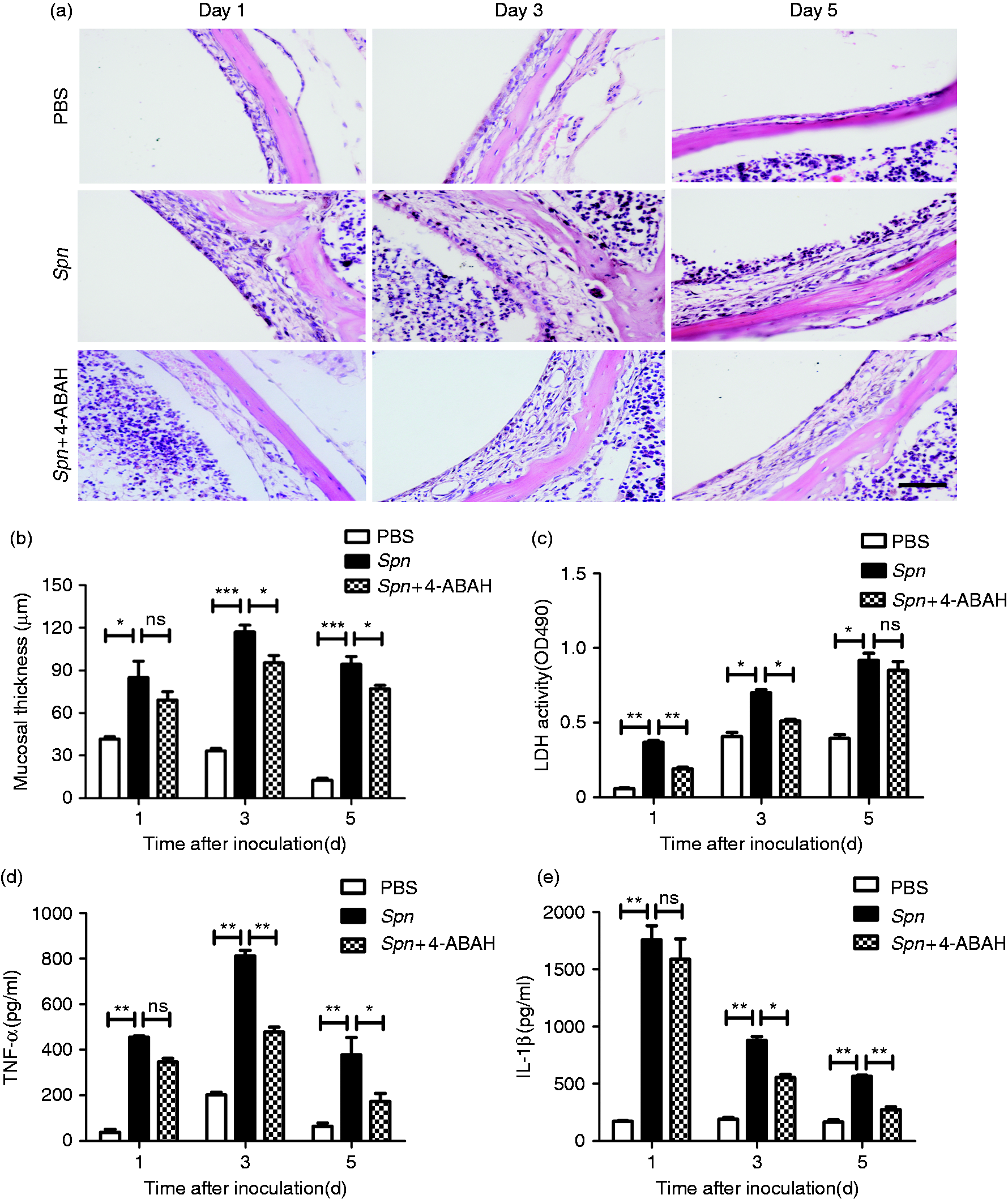
MPO promotes early-stage Spn clearance and worsens tissue damage by enhancing the generation of ROS
To determine the effect of MPO on the production of ROS during AOM, we closely observed the variation of ROS level in 4-ABAH-treated mice. Compared with the Spn group, there was a significant down-regulation of ROS generation in the Spn + 4-ABAH group (Figure 4a, b). DPI was used to inhibit ROS generation to further explore the role of ROS in AOM. The production of ROS was reduced after DPI treatment (Figure 4c, d). To ensure that the difference in bactericidal capacity was caused by the alteration of ROS production rather than variation in the number of neutrophils, the recruitment of neutrophils was closely monitored over time and no statistically significant difference in neutrophil amount was detected between Spn- and Spn + DPI-treated mice (Figure 4e). This result indicated that the bactericidal capacity of mice in the Spn + DPI group, during the entire course of AOM, dropped significantly, mainly on d 1(Figure 4f). Meanwhile, the role of ROS in tissue damage in AOM was also investigated. Compared with the Spn group, the Spn + DPI group suffered epithelial damage to a lesser extent, namely milder mucosal hyperplasia and lower mucosal thickness (Figure 5a, b). Consistently, the activity of LDH was significantly reduced in DPI-treated mice on d 1 and 3, and no significant difference existed between these two groups on d 5 (Figure 5c). The levels of TNF-α and IL-1β decreased in the Spn + DPI group and the Spn group, respectively (Figure 5d, e). These data suggest that MPO in neutrophils facilitates the generation of ROS, and consequently promotes Spn clearance at an early stage and exacerbates tissue injury.
The role of ROS during course of AOM. (a) Neutrophil ROS production in MELF of mice treated with Spn or Spn + 4-ABAH was detected by DCFH-DA (2 μM) staining via FCM on d 3. (b) MFI of ROS production of Spn group and Spn + 4-ABAH group detected by FCM. Values represent mean ± SEM (n = 4–5). *P < 0.05. (c) ROS production of Spn- and Spn + DPI (1 mg/kg)-treated groups detected by FCM on d. 3 (d) MFI of ROS production of the Spn group and Spn + DPI group. Values represent mean ± SEM (n = 4–5). *P < 0.05. (e, f) Quantification of (e) neutrophils and (f) Spn in MELF of mice in Spn group and Spn + DPI group at designated time points. Values in (e) represent mean ± SEM, values in (f) represent individual ear with mean shown by a horizontal bar (n = 4–5). **P < 0.01. ns: not significant. Data are representative of three independent experiments. DPI reduces middle-ear tissue injury. (a) Hematoxylin and eosin staining of middle-ear sections of the PBS group, the Spn group and the Spn + DPI group at designated time points. Scale bar, 100 µm. (b) Mucosal thickness quantification of each group. Values represent mean ± SEM (n = 4–5). *P < 0.05; **P < 0.01; ***P < 0.001. (C–E) Activities of (c) LDH and levels of (d) TNF-α and (e) IL-1β in MELF supernatant of each group at designated time points. Values represent mean ± SEM (n = 4–5). *P < 0.05; **P < 0.01; ***P < 0.001. ns: not significant. Cumulative data from three independent studies are demonstrated in all panels.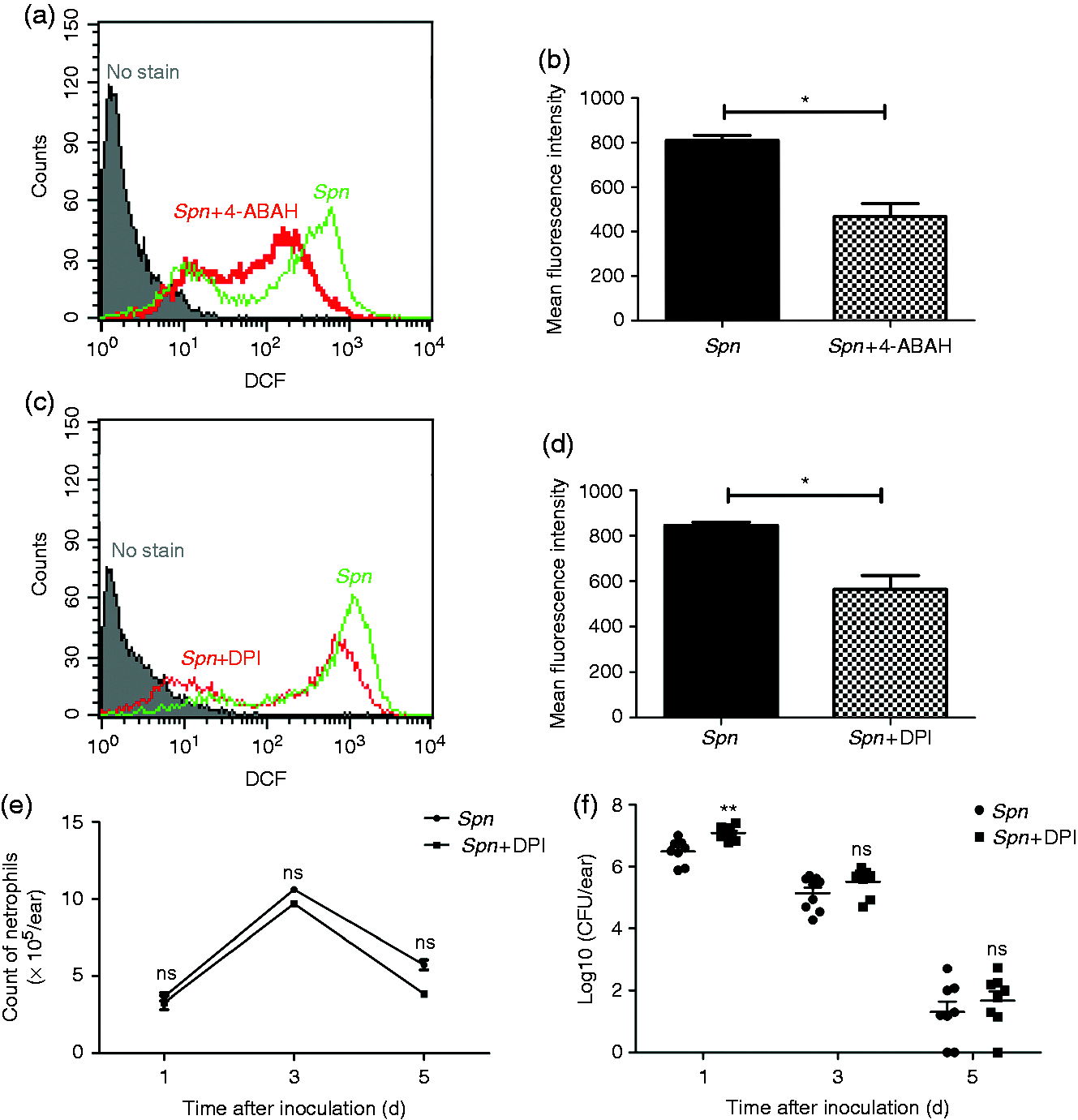
MPO worsens tissue damage by inducing neutrophil apoptosis and necrosis
We examined the kinetics of neutrophil apoptosis by FCM, with Annexin V+/PI- and Annexin V+/PI+ representing apoptotic and necrotic cells, respectively. Results showed that the inhibition of MPO significantly down-regulated the apoptosis and necrosis of neutrophils (Figure 6a–c), indicating that MPO facilitates neutrophil apoptosis and necrosis induced by Spn. In addition, after administrating Z-VAD-FMK, a general caspase inhibitor, we found a significant down-regulation of MPO activity (Figure 6d). Meanwhile, the role of apoptosis in tissue damage during AOM was also investigated. Compared with the Spn group, the Spn +Z-VAD group suffered less epithelial damage, namely milder mucosal hyperplasia and lower mucosal thickness (Figure 6e, f). Consistently, LDH activity and levels of TNF-α and IL-1β decreased in Z-VAD-treated mice (Figure 6g–i). Together, these results suggest that neutrophil-derived MPO can worsen tissue damage by inducing neutrophil apoptosis and necrosis.
MPO worsens tissue damage by inducing apoptosis and necrosis of neutrophils. (a) Representative dot plots to show the percentages of neutrophil apoptosis and necrosis. Cells (1 × 106/ml) in MELF on d 1, 3 and 5 after Spn inoculation in the presence or absence of 4-ABAH were stained with PI and Annexin V. (b, c) Percentages of (b) apoptotic cells and (c) necrotic cells between Spn group and Spn + 4-ABAH group. Values represent mean ± SEM (n = 4–5). *P < 0.05; **P < 0.01. (d) MPO activity of MELF supernatant on d 3 after inoculation with Spn or Spn + zVAD (50 µM). Values represent mean ± SEM (n = 4–5). *P < 0.05; **P < 0.01. Cumulative data from three independent studies are demonstrated. (e) Hematoxylin and eosin staining of middle-ear sections of each group on d 3. Scale bar, 100 µm. (f) Mucosal thickness quantification of each group. Values represent mean ± SEM (n = 4–5). **P < 0.01. (g–i) Activities of (g) LDH and levels of (h) TNF-α and (i) IL-1β in MELF supernatant of each group on d 3. Values represent mean ± SEM (n = 4–5). *P < 0.05; **P < 0.01; ***P < 0.001. Cumulative data from three independent studies are shown in all panels.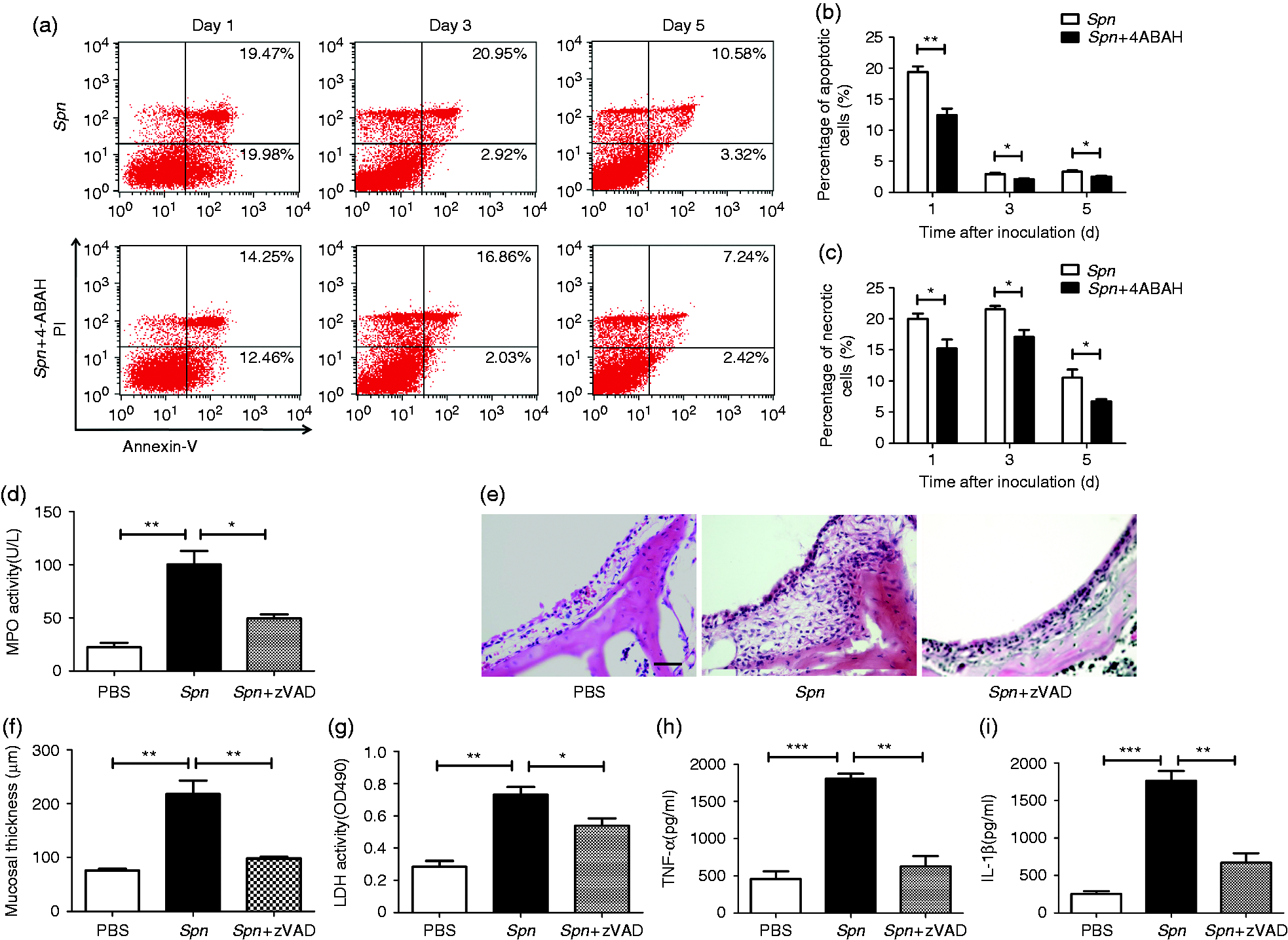
Discussion
Our earlier study found that neutrophils are the major immune cells that first infiltrate the middle-ear cavity, 9 accounting for 98% of the MELF cells in AOM. MPO, the most abundant protein component of neutrophils, reportedly contributes to host defense against multiple pathogenic microorganisms, such as Candida albicans, Candida tropicalis and Trichosporon asahii in lungs. Our previous research has already shown some MPO-independent factors participating in Spn elimination during AOM, such as IL-17A and complements.9,19 However, the mechanism of MPO in combating Spn in AOM has not yet been reported. Studies have shown that the MPO-mediated antimicrobial system mainly includes HOCl, the initial product formed by the oxidation of chloride by MPO and H2O2, and also tyrosyl radical and reactive nitrogen intermediates. 20
In our AOM mouse models, we observed a remarkable up-regulation of MPO. Further study indicated that MPO plays a key role in Spn clearance. However, in a model of pneumococcal pneumonia, compared with the wild type mice, MPO knockout mice had comparable resistance against Spn. 21 These paradoxical results can be attributed to the different responses that neutrophils exert under different pathological states.22,23 An alternate possibility is that neutrophils of MPO knockout mice have adapted to the MPO-lacking situation and thus have enhanced alternative antimicrobial mechanisms independent of MPO. Consequently, these neutrophils manifest themselves with greater sensitivity in combating Spn than 4-ABAH-treated neutrophils. 24 So it is reasonable to speculate that the antimicrobial capacity of MPO in wild type mice neutrophils might be underestimated when compared with that of MPO knockout mice neutrophils.
In addition, our study suggests that MPO facilitates the generation of ROS, and consequently promotes Spn clearance, mainly on d 1. It has been shown that pneumolysin, the potential virulence factor in Spn, will induce intracellular generation of ROS by activating the neutrophil NADPH oxidase system. 21 Nevertheless, with the progress of disease, Spn may up-regulate the expression of the capsule to evade neutrophil phagocytosis; 25 thus, intracellular ROS can hardly wield their ability to kill extracellular Spn. 26 Based on our data, we hypothesize that intracellular ROS is involved in Spn clearance during the early stage of infection. However, during the later stage, in addition to MPO, which generates the effective MPO–H2O2–chloride microbicidal system to kill microbes swallowed into the cells, neutrophils might also kill Spn via serine proteases like elastase, cathepsin G and proteinase 3. 27
It is well established that MPO plays a significant role in defending against invading pathogens. Nonetheless, extensive evidence demonstrates that MPO can be released to extracellular microenvironment, causing damage to a wide variety of targets, such as the cardiovascular system, kidney and lung. 11 Elevated levels of MPO were observed in MELF and middle-ear mucosa of patients with AOM.15,16 Although high expression of MPO in AOM contributes to microbicidal activity, it does worsen middle-ear tissue injury. Our e results showed that the levels of ROS, LDH, TNF-α and IL-1β were reduced following the inhibition of MPO. In addition, inhibition of ROS led to milder tissue damage and reduced LDH activity and pro-inflammatory factor levels. These results demonstrate that MPO induces tissue damage response partly through increasing generation of ROS and modulating production of LDH and pro-inflammatory factors, including TNF-α and IL-1β.
The fate of neutrophils is correlated with the duration and restoration of the AOM pathological process. Filep and El Kebir found that apoptosis could promote the restoration of inflammation. 28 Apoptosis is generally defined as a secure form of cell death characterized by maintaining the integrity of cell membrane for a longer time, as well as inhibiting the release of ROS and degranulation of neutrophils. 29 Caspases are the central regulators of apoptosis. Initiator caspases (including caspases 2, 8, 9, 10, 11 and 12) are closely coupled to pro-apoptotic signals. Once activated, these caspases cleave and activate downstream effector caspases (including caspases 3, 6 and 7), which, in turn, execute apoptosis by cleaving cellular proteins following specific Asp residues. 30 However, follow-up studies have recognized that when the scavenging mechanism fails, secondary necrosis will happen to apoptotic cells, indicating the elimination of cells by disintegration, 31 which is characterized by disruption of cytoplasmic membrane and release of cytotoxic products. 32 Apoptosis and necrosis induced by MPO have been thought to be intimately implicated in tissue destruction.11,33 Our previous work, based on a general caspase inhibitor, Z-VAD-FMK, has shown that apoptosis of neutrophils promotes bacterial clearance in AOM. 9 On this basis, we found that the apoptosis and necrosis of neutrophils were reduced in 4-ABAH-treated mice, particularly necrosis. Taken together, these data confirmed a major effect of MPO on neutrophil death. These necrotic cells will release more MPO and, conversely, that MPO will trigger a new round of apoptosis and necrosis, forming a positive feedback cycle and amplifying tissue inflammation.
In conclusion, MPO serves as a double-edged sword in Spn-induced AOM. It contributes to the removal of bacteria in middle-ear cavity, whereas it causes tissue damage, which is partly associated with its function on increasing ROS generation. Additionally, by provoking neutrophil apoptosis and necrosis, MPO promotes the release of multiple inflammatory substances and, as a result, causes tissue damage. From a holistic perspective, the benefits of MPO in bactericidal activity need to be balanced against the handicap arising from its role in tissue damage. How to make
Yun Xiang, Chunfang Jin and Wei Wang contributed equally to the work
Footnotes
Acknowledgments
We thank Zhiqiang Ding for his help in editing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation grants of China (No. csfc81373151), Scientific and Technological Research Program of Chongqing Municipal Education Commission grants of China (No. KJ130313), and Natural Science Foundation Project of CQCSTC (No. cstc2012jjA10014).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
